Abstract
During the early part of the past century, hundreds of clinical studies involving more than 37,000 patients were conducted that showed radiotherapy (RT) to be a successful and safe alternative to drug therapy for the treatment of many diverse inflammatory conditions and diseases (e.g. tendonitis, bursitis, arthritis, and serious inflammatory lung conditions). Data from these studies were collected and analyzed with the intent of estimating an optimal dosing range for RT that would induce an efficacious treatment response. RT was reported to be frequently effective after only a single treatment, with a rapid (within 24 h) and often long-lasting (from months to years) relief from symptoms. Over a two-decade span from the 1920s to the 1940s, the therapeutic responses to a single RT treatment consistently improved as the dosing for multiple ailments decreased over time to between 30 roentgen (r) and 100 r. These findings are significant and in agreement with a number of contemporary reports from Germany where RT has been commonly and successfully employed in treating ailments with an inflammatory origin. A proposed mechanism by which RT mitigates inflammation and facilitates healing is via the polarization of macrophages to an anti-inflammatory or M2 phenotype.
Introduction
Various environmental signals have been shown to elicit pleotropic effects in macrophages and to polarize them into one of two distinct phenotypes: (1) a pro-oxidative/pro-inflammatory M1 phenotype or (2) an anti-inflammatory M2 phenotype. 1 This observation spurred considerable interest in using environmental stimuli, such as diet, pharmaceuticals, and lifestyle factors (e.g. exercise), to modulate anti-inflammatory phenotypes and thereby reduce pro-inflammatory symptoms like pain and swelling. Relatively recently, this anti-inflammatory process was shown to be mediated largely by the polarization of macrophages toward an M2 phenotype. 1 –3
Within this context, several studies have indicated that ionizing radiation induces macrophage polarization, with the capacity to polarize macrophages toward either M1 (pro-oxidative) or M2 (anti-inflammatory) phenotypes depending on the radiation dose. 4,5 These observations are potentially significant since they help explain the dual capacity of ionizing radiation to be effective in both treating inflammatory conditions (i.e. M2 mediated) and killing microbes and tumor cells (i.e. M1 mediated).
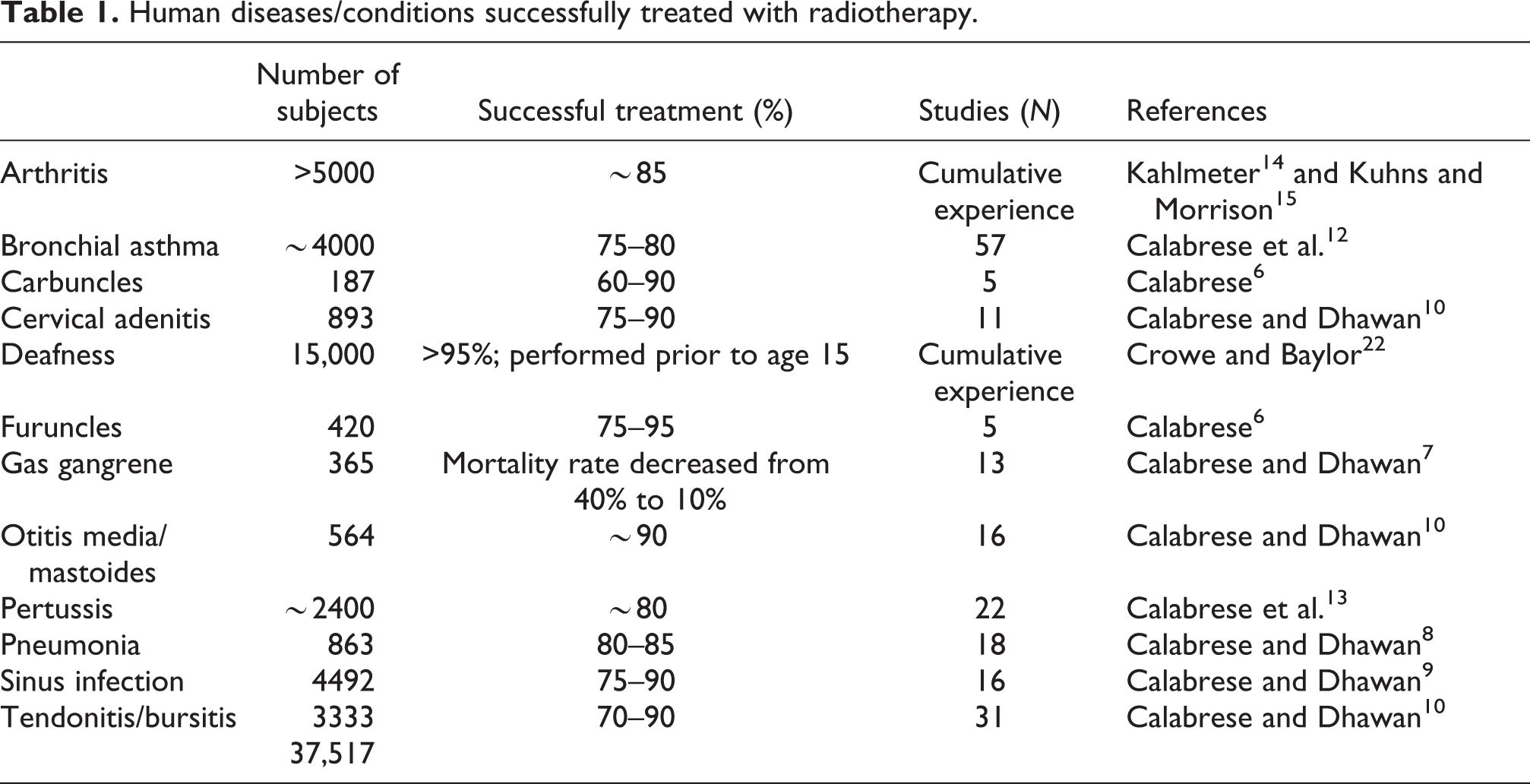
To identify the most effective range of dosing used in the radiotherapy (RT) of many ailments possessing significant inflammatory components, it was necessary to retrieve and reevaluate as much historical data as possible. Herein, RT studies are identified 6 –13 that were conducted on more than 37,000 patients in the first part of the last century, ranging in time from about 1910 to the early 1950s. Each ailment had its own historical foundation as was described in prominent medical journals of the era. The strengths and limitations of the studies were assessed in each review publication according to the specific ailment, as cited above. In general, the clinical successes of RT were not only substantial for each type of ailment but also repeatedly confirmed by independent studies.
Despite differing from each other extensively in terms of etiology, symptomatology, and conventional therapy, all 13 ailments were resolved using the same treatment modality, RT (Tables 1 and 2). The remarkable success of RT in the treatment of multiple diverse ailments is likely related to the view that inflammation is a central feature of each ailment and that RT somehow counteracts the inflammatory process, affording protection and enabling repair of tissue. Since each of these ailments displays the central pathological feature of marked inflammation, it is not unreasonable to deduce that RT at the right dose can act as a potent anti-inflammatory agent.
Human diseases/conditions successfully treated with radiotherapy.
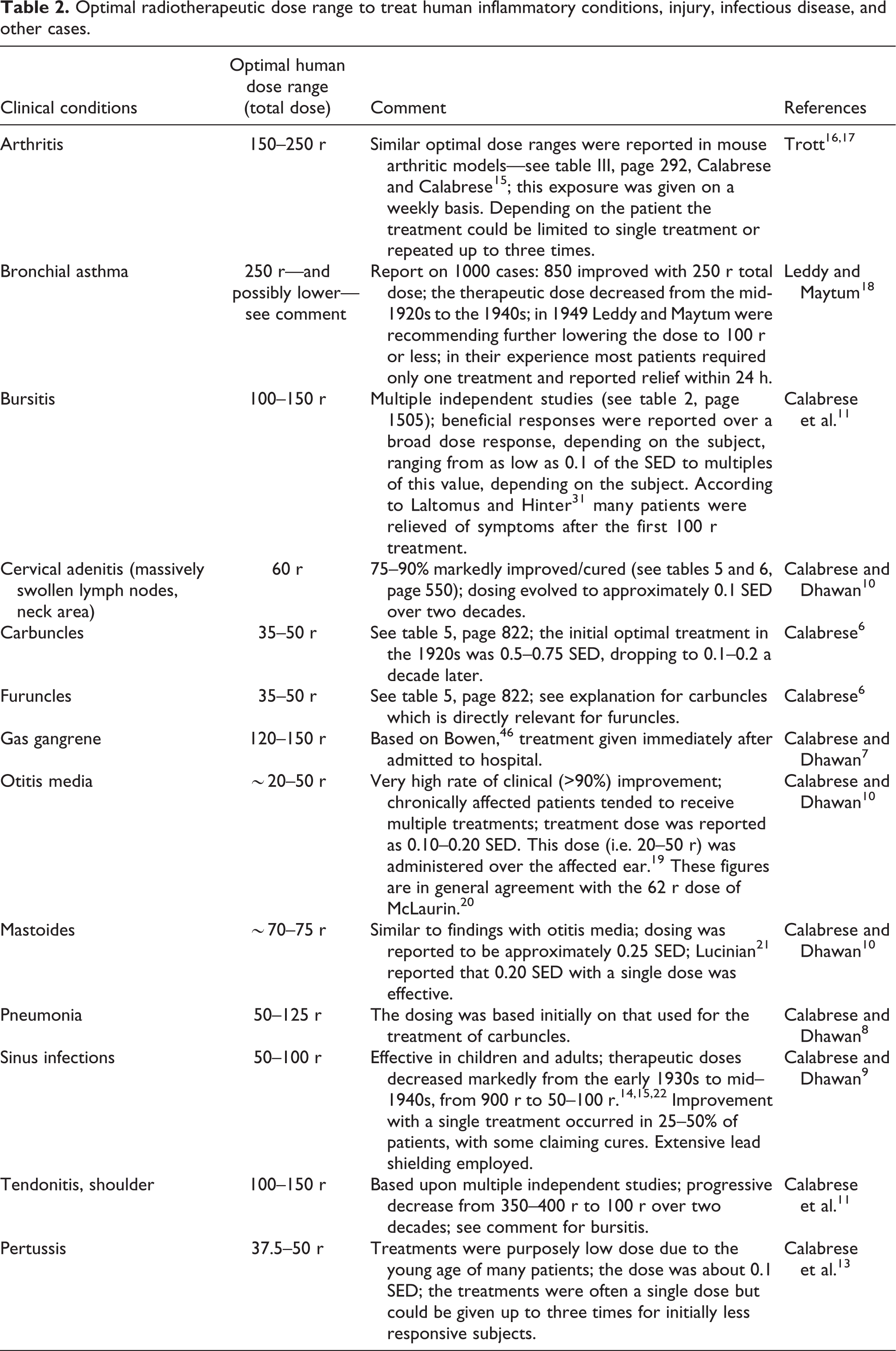
Optimal radiotherapeutic dose range to treat human inflammatory conditions, injury, infectious disease, and other cases.
Anti-inflammatory response: Mediated by macrophage polarization
A mechanistic understanding of how RT could enhance the healing and recovery of so many diverse ailments was not known in the early decades of the 20th century. Recent findings, however, have indicated that relatively low doses of X-rays/γ-rays can induce the development of a highly integrated and systemic response involving the polarization of macrophages toward an anti-inflammatory M2-like phenotype. 23 –25 The polarization response of macrophages to physical stimuli, such as nonionizing radiation, represents a rather recent and novel observation. Chemical agents, on the other hand, have routinely been used as polarization stimuli and have therefore played a fundamental role in identifying typical biomolecular events involved in mediating an M2 as well as M1 polarization response in macrophages. The following events have been identified as being involved in mediating an M2 polarization response: decreases in nitric oxide levels and in inducible nitric oxide synthase, decreases in reactive oxygen species, increases in heme oxygenase, suppression of tumor necrosis factor α, increases in tumor growth factor β, and the activation of several transcription factors, including nuclear factor κβ and activating factor protein 1. The fact that this chemically elicited biomolecular response is similar to the biomolecular response induced specifically by low-dose X-rays lends support to the idea that—like certain chemicals—RT mediates its anti-inflammatory effects via the polarization of macrophages toward an anti-inflammatory M2-like phenotype. 1
Results
Tables 1 and 2 indicate that RT, at a relatively narrow dose range of between 30 roentgen (r) and 150 r (0.3–1.5 Gy), was effective in treating a spectrum of inflammatory ailments (some with concomitant infectious components). The skin erythema (i.e. skin reddening) dose (SED) is an upper bound exposure point of reference and was used in the estimation and selection of most doses for all end points. The SED had long been established as a biological exposure standard to judge excessive exposure to ionizing radiation; it was a type of visual radiation badge. This visual biomarker was used before Muller 26 who reported that X-rays could induce gene mutations in the fruit fly. 27 A detailed review of the historical foundations of the SED indicates that its recommended value of 350–500 r 28 varied to some extent during the 1920s and 1930s. The SED estimate could be affected by X-ray filtration techniques and multiple characteristics of the patient, such as age, gender, degree of skin pigmentation and vascularization, among other factors. This historical context indicates that the SED represents a value (and thus fractional values) 6 possessing a fair degree of uncertainty and some inaccuracy, even though it was a widely recognized frame of reference at the time. If safe and effective doses of RT were based on uncertain and inaccurate estimates of SED, it stands to reason that the optimal dosing range of RT, as reported in the historical literature, would also be less than accurate.
Another factor reducing confidence in the accuracy of the optimal dosing range for RT, as reported in the literature, was the progressive lowering of RT doses (fractions of the SED) that occurred over time, as clinical outcomes noticeably improved with the use of lower doses of RT. For example, in the 1920s, X-ray doses used in the treatment of carbuncles ranged from 0.5 to 0.75 SED (i.e. 175–375 r), dropping to 0.1 to 0.2 SED (i.e. 35–100 r) during the 1930s. 6 Similarly, shoulder tendonitis was successfully treated with X-rays at 350–400 r in the 1930s, 29,30 but by the late 1940s, the treatment dose had declined to 100 r. 31 Furthermore, clinicians from one subgroup specialty learned from another, copying the more effective treatment protocol irrespective of the designated ailment. For example, in selecting an RT dose to treat pneumonia, clinicians were swayed by publications describing the successful treatment of carbuncles with X-rays. 6
This array of different starting points, protocols, end points, and inter-clinician collaborations created a somewhat confusing clinical picture for this novel treatment modality, especially in the 1920s and early 1930s. However, from the 1930s to the mid-1940s, clinicians tended to select the most effective treatment protocols within disease categories, gradually leading to a convergence and reduction in the treatment dosing range across ailments. Table 2 presents the optimal RT doses/dose ranges administered in clinical practices to responsive subjects toward the end of the historical RT era. The continuation of this declining trend in RT dosing values from the late 1930s to well into the 1940s without an apparent leveling off suggests that the doses used in the treatment of inflammatory ailments may still not have been optimized. This view is consistent with extensive data from animal studies showing that optimized X-ray-induced anti-inflammatory phenotypes were induced with approximately 0.5 Gy. 23,24 More recent studies have reported an accelerated healing of bone fractures with single doses (0.5 Gy) of X-rays in male Sprague-Dawley rats. 32 Similarly, Jabbari et al. 33 reported that a single dose of γ-rays (0.75 Gy) immediately after surgery significantly accelerated the healing process for a full-thickness wound in male Sprague-Dawley rats. Supporting this possibility was an experimental study by Kataoka et al. 34 in which a single 0.25 Gy exposure significantly enhanced liver catalase activity in adult mice 24 h after exposure. These diverse findings raise the question of whether lowering the doses and/or reducing the number of treatments could improve the healing response and, thus, the efficacy of treatment.
Clinicians, however, were sensitive to the possibility that any defined dose of RT might not only elicit varying therapeutic efficacies among different patients (depending on their genetic variability as well as developmental and age-related differences) but might also elevate risks of cancer. For example, for safety reasons clinicians tended to further reduce exposures to children 13 while still reporting successful treatment outcomes. In further support of the efficacy and safety of lower doses, recent human studies showed that simulating cumulative radon spa exposures over about 5 months (with a 40-min duration plus a radio-nebulizer exposure) was equivalent to a dose rate of approximately 0.005 mSv/day and could significantly alleviate symptoms of rheumatoid arthritis. 20 This represents a total cumulative RT dose that approximates the optimal dose given at one time in the cited historical studies. These observations further suggest that optimal dosing could be achieved at still lower doses/dose rates for adults.
The above findings are consistent with the reports of Seegenschmiedt et al. 35,36 showing that RT treatment of shoulder pain and bursitis in Germany today is quantitatively comparable to the treatment of similar ailments in the historical literature. However, most of the ailments in the present review of the historical literature are not currently being treated with RT in Germany. These ailments include gas gangrene, sinusitis, otitis media, cervical adenitis, mastoiditis, pneumonia, bronchial asthma, carbuncles, and furuncles. Thus, only in the clinical treatment of shoulder tendonitis and bursitis does the historical clinical literature reviewed herein overlap with RT treatment in contemporary Germany.
Based on these findings, a RT dose ranging between 0.3 Gy and 1.0 Gy is likely to produce an anti-inflammatory phenotype that can reduce inflammation and pain as well as promote healing. The interindividual variation, however, that inherently characterizes a response to any given treatment dose of RT remains an issue. If one treatment dose fails to elicit adequate relief, then the degree of interindividual variation may affect the decision both to administer another treatment dose and to determine its size. Furthermore, it would be neither unexpected nor surprising if a certain proportion of patients, due to interindividual variation, were nonresponsive, even after several treatments. The heterogeneity of the human population would suggest that a broad therapeutic window exists for RT and that the determination of an optimal treatment dose for each individual may be challenging.
Another important consideration regarding the clinical application, dosing, and efficacy of RT is the duration or time of relief from pain and inflammation following treatment. RT studies from the historical literature on the treatment of bronchial asthma and bursitis/tendonitis 11 explicitly address the duration of relief. In the case of bronchial asthma, the duration of relief displayed considerable interindividual variation but was typically in the period from 1 year to 2.5 years. 37 These findings by Scott 37 were similar to the earlier results of Schilling 38 who found that 25% of patients remained free of asthma symptoms more than 2 years after treatment. Maytum and Leddy 39 reported similar findings with patients that were followed for 16 months. In the case of Hull et al., 40 almost all patients (over 90%) were nearly completely relieved of symptoms 18 months after treatment. When the study by Hull et al. was expanded to include more than 1500 patients, it essentially replicated the success of the original study. In another study by Waldbott, 41 similar findings were reported in patients who were followed out to 3 years (twice as long as Hull’s study). Together, these findings lend support to the premise that limited treatment with RT can result in prolonged periods of relief, lasting many months to several years in the case of bronchial asthma. Furthermore, the interindividual variations appear to be considerable and the differences in treatment protocols appear to affect the total dose delivered per patient as well as the efficacy and, ultimately, the success of the treatment.
The duration and efficacy of RT in the treatment of bursitis and tendonitis were investigated in six studies that included more than 600 patients whose treatment results were followed for extended periods of up to 6 years. 29,42 –46 The general consensus of these studies was that the treatment benefit was virtually permanent for the vast majority of patients and was unrelated to the acute, subacute, or chronic classification of the inflammatory condition. The observation that RT can frequently induce nearly permanent cures of serious inflammatory conditions raises important theoretical and practical questions with broad medical and public health implications.
Evidence mined from the historical literature and presented in this study indicates that relatively low doses of radiation can suppress inflammation and essentially “cure” many ailments whose underlying etiologies possess strong inflammatory components. Data from this study suggest that macrophages are polarized more toward M2 and the anti-inflammatory state when doses descend below 1.0 Gy and, conversely, more toward M1 and the pro-inflammatory state when doses ascend above 1.0 Gy. Knowledge of the optimal RT dosing ranges required to modulate the polarization of M1 and M2 phenotypes in patients could enable the clinical induction and regulation of pro- and anti-inflammatory immune states for the targeting, treating, and curing of a variety of inflammatory, infectious, and tumorigenic diseases (Figure 1).

It has been suggested that macrophages do not really exist as 100% pure M1 or M2 phenotypes but rather as varying mixes of M1 and M2 phenotypes at both the single cell and cell population levels. 1,47 Because radiation-induced polarization induces changes in the M1 and M2 phenotypes, the “phenotypic potential” for M1 and M2 to mediate pro- and anti-inflammatory states would also change accordingly. The identification by flow cytometry, for example, of one or more molecular biomarkers that are specific to the expression of M1 and M2 responses may possibly serve as qualitative and quantitative measures of M1 and M2 “phenotypic potential.” Assuming that it was possible to measure the “phenotypic potential” of M1 and M2 for individual macrophages in a cell population, it would then be possible to electronically calculate the aggregate “phenotypic potential” for the entire cell population, that is, total M1 (M1T) and total M2 (M2T). The “phenotypic potential” having the larger value (M1T or M2T) would qualitatively denote the overall “phenotypic potential” (M1 or M2) of the cell population, while the numeric difference between total “phenotypic potentials” (M1T − M2T or M2T − M1T) would be a quantitative measure of the net “phenotypic potential” of the entire cell population. Theoretically, data from such an experimental approach would be important because it could be used to predict and modulate, for any given dose of RT, the type and intensity of inflammatory response, that is, pro-inflammatory (M1) or anti-inflammatory (M2). The predictive and modulatory aspects of such an experimental approach for optimal dose determination may have valuable clinical appeal.
All of the ailments considered in this study have a significant inflammatory component and should benefit preferentially from doses of radiation that polarize macrophages to an M2-like anti-inflammatory phenotype, that is, at less than 1.0 Gy. However, many of the ailments cited in this study, such as pertussis, gangrene, pneumonia, otitis media, and sinus infection, may also include an infectious component that would benefit preferentially from doses of radiation that polarize macrophages to an M1-like pro-inflammatory phenotype, that is, at more than 1.0 Gy. In such an instance where both inflammation and infection are present to varying degrees, benefits may be derived from doses either above (i.e. to treat infection) or below (i.e. to treat inflammation) 1.0 Gy. The assumption here is that both pro- and anti-inflammatory activities are induced simultaneously to promote unequally microbial killing (M1) and tissue healing (M2), depending on the dose being above or below 1.0 Gy, respectively. The historical trend toward lower doses producing the most effective treatments for inflammation suggests that inflammation can have a dominant effect on patient perceptions of health and wellness following therapy.
During the period of the 1920s to 1940s, the assessment of cancer risk for RT treatments of the various ailments reviewed in this study was not discussed. In his retrospective historical review of successful RT treatments of bronchial asthma, Waldbott 48 stated that despite high rates of clinical success he “relinquished this treatment, however, because of the possibility of long-term adverse effects.” Webber 49 provided a similar perspective and raised the issue that children who were administered RT to treat pertussis may experience an increased cancer risk. Webber 49 noted that “it is generally unappreciated by physicians that during the two decades from 1920-1940, hundreds of children received potential carcinogenic doses of radiation therapy to the thorax for whooping cough” (see p. 449). Webber 49 then concluded by raising the issue that as of the 1970s hundreds of adults may have a potentially enhanced risk of thyroid cancer due to radiation treatments for pertussis during infancy.
Since the use of RT for the treatment of benign diseases is commonly employed in Germany, the question of cancer risks due to treatment has been the object of considerable discussion since the early 1990s. 16,50 Trott and Kamprad 17 argued that the effective dose method of the International Commission of Radiation Protection (ICRP) to estimate risk to the general population from occupational or environmental low-dose radiation exposure was not adequate for estimating the risk of cancer induction by the RT of malignant or nonmalignant diseases. This judgment was based on findings that the types of second cancers following RT are different from those caused by low-dose total-body irradiation as reported for Japanese A-bomb survivors. These RT researchers claimed that cancer risk due to the RT of benign diseases therefore should be empirically based on epidemiologic data from studies of patients who have received RT for nonmalignant diseases in the past. They further claimed that RT-induced tumors do not follow the linear non-threshold (LNT) model as used in radiation protection risk assessment. 51 In most cases, they claimed that cancer risks estimated by the standard LNT method overestimated the true risks by one order of magnitude.
Ottolenghi et al. 51 indicated that the most significant factor affecting cancer risk is the site of treatment. They noted that for all peripheral area treatments, such as Dupuytren’s contracture, tennis elbow, or heel spur, radiation cancer risks are very low and similar to those from common diagnostic X-ray examinations. However, for treatments of benign diseases involving sites with significant amounts of red bone marrow (i.e. head, shoulder, thorax, hip, and pelvis), the principal risk is the potential induction of leukemia. Therefore, the treatment optimization criterion should also include the reduction of the mean bone marrow dose. A recent paper by Cuttler has suggested a relatively high threshold (i.e., 500 mSv) for ionizing radiation-induced leukemia in humans, adding further debate and uncertainty to this issue. 52
Sautter-Bihl et al. 50 provided a quantitative estimate of cancer risk based on RT for the treatment of inflammatory joint conditions using an LNT approach. Their approach used expected average exposures that were adjusted for multiple factors, such as differential distances to the irradiated areas. The assumed exposure was 6× 1 Gy dose used for joint treatments and was converted into a whole-body dose based on measured doses and a standard whole-body conversion formula. Based on this exposure and the LNT model, the authors estimated an additional 20–40 malignancies per million people over a lifetime. Since the average age of the patients was 54 years, they argued that the risks of inducing malignances were not of any practical relevance. Their reasoning was based on the expected prolonged latency of tumor growth and their estimate of low risk that was derived from a very conservative risk estimation method (i.e. LNT). In Germany, RT is recommended only after standard non-RT treatments have failed. Due to concerns over possible cancer risks, patients under 40 years of age would only be eligible for RT in exceptional cases and then only after being followed up with a determination of all possible risks versus expected benefits. 36
A final consideration concerns a potentially novel treatment option for RT. It has been reported that a protective anti-inflammatory phenotype can be induced in mice with equal efficacy when a standard X-ray dose targets a small area of inflammation (e.g. an arthritic joint) or when it is administered more broadly as a whole-body exposure. 2,23,28 –31,35 Unfortunately, this RT phenomenon is not known to have been examined in human clinical studies involving any inflammatory ailments, although such a study could have potentially significant implications. However, in the field of photobiomodulation (PBM), a similar phenomenon has been widely reported to occur in humans. Irradiation via PBM at a remote location from the site of injury has been shown to be very effective in enhancing healing and preventing tissue damage (e.g. healing skin wounds and crushed nerves as well as preventing brain and heart damage) (see Stone et al. 53 for a review). These findings are of considerable practical importance as they suggest that RT of an ailment located in a sensitive area (e.g. near bone marrow) could be minimized or possibly avoided. In addition, the application of RT for the treatment of degenerative neurological diseases with significant inflammatory components, such as Alzheimer’s and Parkinson’s disease, is now being explored 54 –59 in several case studies and a phase 1 clinical trial. Likewise, recent papers suggest the utility of RT for different types of cancers, several autoimmune diseases, and diabetes. 19 –21
RT can be a very useful tool but, like alternative agents and technologies, its utility should be assessed within the context of other therapeutic considerations, that is, severity of ailment, alternative treatments, and patient characteristics, such as age, gender, health status, treatment risks versus benefits, among others.
Conclusions
The annals of medical history report that tens of thousands of patients inflicted with more than a dozen serious inflammatory ailments during the first half of the 20th century were successfully and safely treated with low doses of radiation. The dose range of therapeutic effectiveness for RT was observed to be broad, generally extending from as low as 20 r (e.g. approximate upper limit of common single computed tomography scans) to 200 r, approximately a 10-fold range. The magnitude of the beneficial response was consistently greater as the treatment dose was reduced across a spectrum of clinical conditions studied over several decades. The available data did not preclude the possibility that the efficacy of treatment might be enhanced even further at lower doses. Considerable interindividual variation in response was reported, typically leading to the use of follow-up treatments for those patients not responding to a single treatment. The duration of the beneficial responses to RT was assessed for bronchial asthma and bursitis and tendonitis. In each case, multiple studies reported that “relief” of symptoms extended from months to years depending on the patient. In the case of bursitis and tendonitis, several research groups considered the relief as permanent. The pervasive anxiety and fear of radiation-induced cancers following the detonation of atomic bombs in 1945 as well as the proven efficacy and safety of novel antibiotics and other pharmaceuticals being developed around the same time with anti-inflammatory properties combined to elicit the rapid demise of low-dose RT. Despite the near worldwide elimination of low-dose RT over 70 years ago, Germany has continued to study and use it clinically as an effective and highly valuable alternative treatment for a host of inflammatory diseases. Low-dose radiation treatments are well-documented and have been proven as safe and effective; they therefore should be seriously considered as viable clinical options and studied further. The judicious application of RT by the medical community has the potential to be an effective complement to drug therapy, offering low-cost, low-risk, and often enduring relief for many symptoms of serious and potentially life-threatening inflammatory ailments.
Footnotes
Authors’ note
The views and conclusions contained herein are those of the authors and should not be interpreted as necessarily representing policies or endorsement, either expressed or implied. Sponsors had no involvement in study design, collection, analysis, interpretation, writing and decision to and where to submit for publication consideration.
Acknowledgements
EJC acknowledges longtime support from the US Air Force (AFOSR FA9550-13-1-0047) and ExxonMobil Foundation (S18200000000256).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
