Abstract
In the present study, the modulatory effect of ursolic acid (UA) on cardiac fibrosis and mitochondrial and lysosomal enzymes activity in isoproterenol-induced myocardial infarction (MI) in rats were examined. Isoproterenol hydrochloride (ISO; 85 mg/kg body weight) was administered subcutaneously for first two consecutive days. ISO-induced MI in rats significantly decreased the activities of mitochondrial tricarboxylic acid cycle enzymes and respiratory chain enzymes while increased the activities of lysosomal glycohydrolases and cathepsins. The expression of matrix metalloproteinase 2 (MMP-2), MMP-9, collagen type I, α-smooth muscle actin (α-SMA), and transforming growth factor-β (TGF-β) were upregulated in ISO-induced MI in rats. UA administration to rats showed increased activities of mitochondrial tricarboxylic acid cycle enzymes and respiratory chain enzymes and decreased activities of lysosomal glycohydrolases and cathepsins in ISO-induced rats. Furthermore, expression of MMP-2, MMP-9, collagen type I, α-SMA, and TGF-β downregulated in UA-administered rats. Thus, our results demonstrate that UA has an anti-fibrotic effect and attenuates the mitochondrial and lysosomal dysfunction in ISO-induced MI in rats.
Introduction
Mortality rate of myocardial infarction (MI), which is the common cause of cardiovascular disease (CVD), is increasing each year. 1 During MI, the ventricle part of the heart ensures significant pathological changes, such as inflammatory reactions, free radical impairment, cardiac apoptosis, extracellular matrix (ECM) deposition, cardiac hypertrophy, and ventricular remodeling. 2 Isoproterenol hydrochloride (ISO) is a synthetic catecholamine and β-adrenergic agonist, which can induce several cardiac dysfunctions in rats. 3 Mitochondria play a major role in the regulation of intracellular calcium ions, particularly during toxic strain and mitochondrial oxidative phosphorylation. 4 So, understanding the molecular and biochemical mechanism(s) of cardio protection and, specifically, the role that mitochondria play, has a major impact on the clinical management of MI. 5 Lysosomal enzymes involved in the acute and chronic inflammatory diseases, which affects the target organ, often results in fibrosis (formation of excess fibrous connective tissue); the activity of lysosomal enzymes alterations were seen in both patients with MI and ISO-induced MI in animal models. 6
Cardiac fibrosis is associated with collagen accumulation resulting in imbalance of synthesis and degradation of matrix metalloproteinases (MMP) -2 and -9, which play an important role in balancing ECM production and degradation in the myocardium. 7 During the upregulated expression of MMP-2 and MMP-9 in various pathophysiological conditions such as left ventricular (LV) remodeling, congestive heart failure, and cardiac fibrosis. 8 β-Adrenergic receptor stimulate collagen type I synthesis and MMP-2 expression in cardiac tissue. 9 In addition, collagen type I is assumed to be the main component of the ECM in the MI heart. 10 Transforming growth factor-β (TGF-β) is involved in pathological conditions of the heart that occurs in response to cardiac hypertrophy through the induction of interstitial fibrosis causing over accumulation of collagen and ECM deposition. 11,12 α-Smooth muscle actin (α-SMA) caused fibrogenesis and scar tissue in various pathological conditions. This condition indicates that fibroblasts differentiate into myofibroblasts, which trigger the ECM signals and accelerate cardiac injury. 13 El Hajj et al. reported that the overexpression of α-SMA, collagen type I and II, and TGF-β1 and MMPs caused fibrosis in ISO-induced MI in rats. 14
A number of dietary phytochemicals have been identified, which protect against experimental MI. The prevention of CVD has been associated with the intake of fresh fruits, vegetables, or plants rich in natural antioxidants. Ursolic acid (UA: 3b-hydroxy-urs-12-en-28-oic acid), a natural pentacyclic triterpenoid, is present in apples, basil, berries, leaves, and flowers; has several biological activities such as antioxidant, anti-inflammatory, antiviral, antitumoral, 15 antihyperlipidemic 16 ; and inhibits MMP-9. 17 However, the effect of UA in cardiac fibrosis, mitochondrial protection, and lysosomal dysfunctions are not yet elucidated. In the present study, we evaluated the protective effect of UA on MMP-2, MMP-9, collagen type I, α-SMA and TGF-β expression; mitochondrial and lysosomal enzymes activities; and histopathological changes in ISO-induced MI in rats.
Materials and methods
Experimental animals
Male albino Wistar rats (160–180 g) were procured from the Central Animal House, Department of Experimental Medicine, Rajah Muthiah Medical College and Hospital, Annamalai University and maintained in an air-conditioned room (25 ± 3°C) with a 12-h light/12-h dark cycle. Feed and water were provided ad libitum to all the animals. The study protocols were approved by the Institutional Animal Ethics Committee of Rajah Muthiah Medical College and Hospital (Reg No.160/1999/CPCSEA, Proposal number: 560), Annamalainagar.
Chemicals
ISO and UA were purchased from Sigma-Aldrich (St Louis, Missouri, USA). MMP-2, MMP-9, TGF-β1, collagen type I, and α-SMA were purchased from Santa-Cruz Biotechnology, Inc., (Dallas, Texas, USA), and a goat anti-rabbit, the secondary antibody was purchased from Genei (Bangalore, Karnataka, India). Enhanced chemiluminescence (ECL)-kit was purchased from GenScript ECL kit (Piscataway, NJ, USA). All other chemicals used in this study were of analytical grade obtained from E. Merck (Vikroli, Mumbai) and HIMEDIA (Mumbai, India).
Experimental induction of MI
MI was induced by subcutaneous (s.c.) injection of ISO (85 mg/kg body weight (BW)) in the right thigh of the rat, dissolved in physiological saline, for two consecutive days. 18 The ISO first injection was given at 6.00 p.m. and the second injection at 6.00 p.m. of the next day.
Experimental design
The animals were randomly divided into four groups with six rats in each group. UA (40 mg/kg BW) was completely dissolved in 5% dimethyl sulfoxide (DMSO) and diluted with physiological saline. Group I: Control (5% DMSO s.c., from 3rd day to 9th day) Group II: Control + UA (40 mg/kg BW, s.c., from 3rd day to 9th day) Group III: ISO control (85 mg/kg BW, s.c., for 1st and 2nd day) Group IV: ISO (85 mg/kg BW, s.c., for 1st and 2nd day) + UA (40 mg/kg BW, s.c., from 3rd day to 9th day)
The total duration of the study was 9 days. On the 10th day, the rats were euthanized by cervical dislocation. Blood samples were collected in a dry test tube and allowed to coagulate at ambient temperature for 40 min. Serum was separated by centrifugation at 2000 r/min for 10 min. The heart tissue was excised immediately, washed with chilled isotonic saline, and used for analysis of various parameters.
Isolation of mitochondria from the heart tissue
The heart mitochondria were isolated from the heart tissue using differential centrifugation by the method of Johnson and Lardy. 19 Then, 20% (w/v) homogenate was prepared in 0.25 M sucrose containing 0.05 M Tris-HCl buffer and 5.0 mM ethylenediaminetetraacetic acid (EDTA). The homogenate was centrifuged at 600 × g for 10 min. The supernatant fraction was centrifuged (Himac SCP 70G, Hitachi, Japan) in a refrigerated centrifuge at 10,000 × g for 5 min at 4°C to bring down the mitochondrial pellet. After centrifugation, the supernatant was poured off, while the loose upper part of the mitochondrial pellet may come off as well. Most of the pellet, containing healthy mitochondria, was dense enough to remain behind. The white foamy material, near the top of the tube, consisting of lipids, was removed by wiping the inside of the tube with cotton. Because any mixing of lipids with the mitochondria suspension will cause them to uncouple (lose their ability to maintain a chemiosmotic gradient). After using a Pasteur pipette to remove the last bit of liquid, the remaining mitochondrial pellet was resuspended in potassium chloride (KCl) and used for the estimation of various parameters. The activities of heart mitochondrial enzymes such as isocitrate dehydrogenase (ICDH), 20 α-ketoglutarate dehydrogenase (α-KGDH), 21 succinate dehydrogenase (SDH), 22 malate dehydrogenase (MDH), 23 cytochrome c oxidase, 24 and nicotinamide adenine dinucleotide (NADH) dehydrogenase 25 were analyzed.
Isolation of lysosomal fraction from the heart tissue
The lysosomal fraction of the heart tissue was isolated by the method of Arumugham and Bose. 26 Fresh heart tissue was homogenized in ice-cold 0.25 M sucrose solution. The homogenate was filtered and centrifuged at 3000 × g for 10 min. The pellet was removed and homogenized and resuspended as before. The supernatants were combined and centrifuged again at 15,000 × g for 20 min. The lysosomal pellet was suspended in 1.15% KCl and homogenized and used for the assay of enzymes. The activities of lysosomal enzymes such as α-glucosidase and α-galactosidase, 27 β-glucosidase and β-galactosidase, 28 β-glucuronidase and β-N-acetyl glucosaminidase, 29,30 cathepsin-B 31 and cathepsin-D 32 were analyzed.
Western blot analysis for cardiac fibrotic markers
Protein lysates were separated by SDS-PAGE and transferred to polyvinylidene difluoride (PVDF) membranes (Millipore) for the analysis of MMP-2, MMP-9, collagen type I, and α-SMA. 33 The heart tissue samples were homogenized in an ice-cold Radioimmunoprecipitation assay buffer (RIPA) buffer (1% Triton, 0.1% SDS, 0.5% deoxycholate, 1 mmol/L EDTA, 20 mmol/L Tris (pH 7.4), 150 mmol/L NaCl, 10 mmol/L NaF, and 0.1 mmol/L phenylmethylsulfonyl fluoride (PMSF)). The homogenate was centrifuged at 12,000 r/min for 15 min at 4°C to remove debris. Protein concentration was measured by the method of Lowry et al. 34 Samples containing 50 μg of total cellular proteins were loaded and separated using 10% SDS polyacrylamide gel electrophoresis. The resolved proteins were blot transferred onto a polyvinylidene difluoride (PVDF) membrane (Millipore). Membranes were washed with Tris-buffered saline (TBS) containing 0.05% Tween 20 (TBST) and were blocked using TBS containing 5% bovine serum albumin (BSA). The membranes were then washed with TBST and then kept overnight at 4°C with respective primary antibodies. After this, membranes were incubated with their corresponding secondary antibodies (IgG conjugated to horseradish peroxidase (HRP)) for 2 h at room temperature. The PVDF membranes were then washed thrice with 10-min interval, and protein bands were visualized by an ECL method using ECL-kit (GenScript ECL kit). Bands were scanned using a scanner and quantitated by Image J, a public domain Java image processing software, Wayne Rasband, NIH, Bethesda, Maryland, USA.
Immunohistochemistry
Paraffin-embedded tissue was cut to obtain sections of about 4 µm thicknesses. The mounted paraffin-embedded slices were deparaffinized in xylene and rehydrated using an ethanol/H2O gradient. Heat-mediated antigen retrieval step was carried out for 10 min, and then the slides were allowed to cool to room temperature for another 20 min. This was followed by peroxidase block treatment (to block endogenous peroxidase enzyme activity) for 10–15 min and then power block treatment (to block non-specific binding of antibodies to highly charged sites) for another 15 min. The sections were incubated with the concerned diluted primary antibody (TGF-β) solution (for 2 h, 1:200), followed by treatment with the super enhancer solution (for 30 min) and supersensitive poly-HRP solution (for 30 min). After color development with 3,3′-diaminobenzidine tetrahydrochloride (DAB) and counterstaining with hematoxylin, the sections were observed under the light microscope and photographs were taken.
Statistical analysis
Values are expressed as means ± standard deviation. The data were statistically analyzed by one-way analysis of variance (ANOVA), followed by Duncan’s multiple range test (DMRT) using a statistical package program (SPSS 17.0 for Windows). The values of p < 0.05 were considered as statistically significant.
Results
Mitochondrial TCA cycle enzymes and respiratory chain enzymes
Table 1 presents the activities of mitochondrial TCA cycle enzymes, ICDH, α-KGDH, SDH, MDH, cytochrome c oxidase and NADH dehydrogenase in control and ISO-induced MI in rats. ISO-induced rats showed significantly decreased activities of mitochondrial TCA cycle enzymes and respiratory chain enzymes when compared to control rats. UA treatment significantly increased these enzymes and were observed in ISO-induced rats.
Effect of UA on the activities of mitochondrial TCA Cycle enzymes and respiratory chain enzymes in the heart of control and ISO-induced rats.a
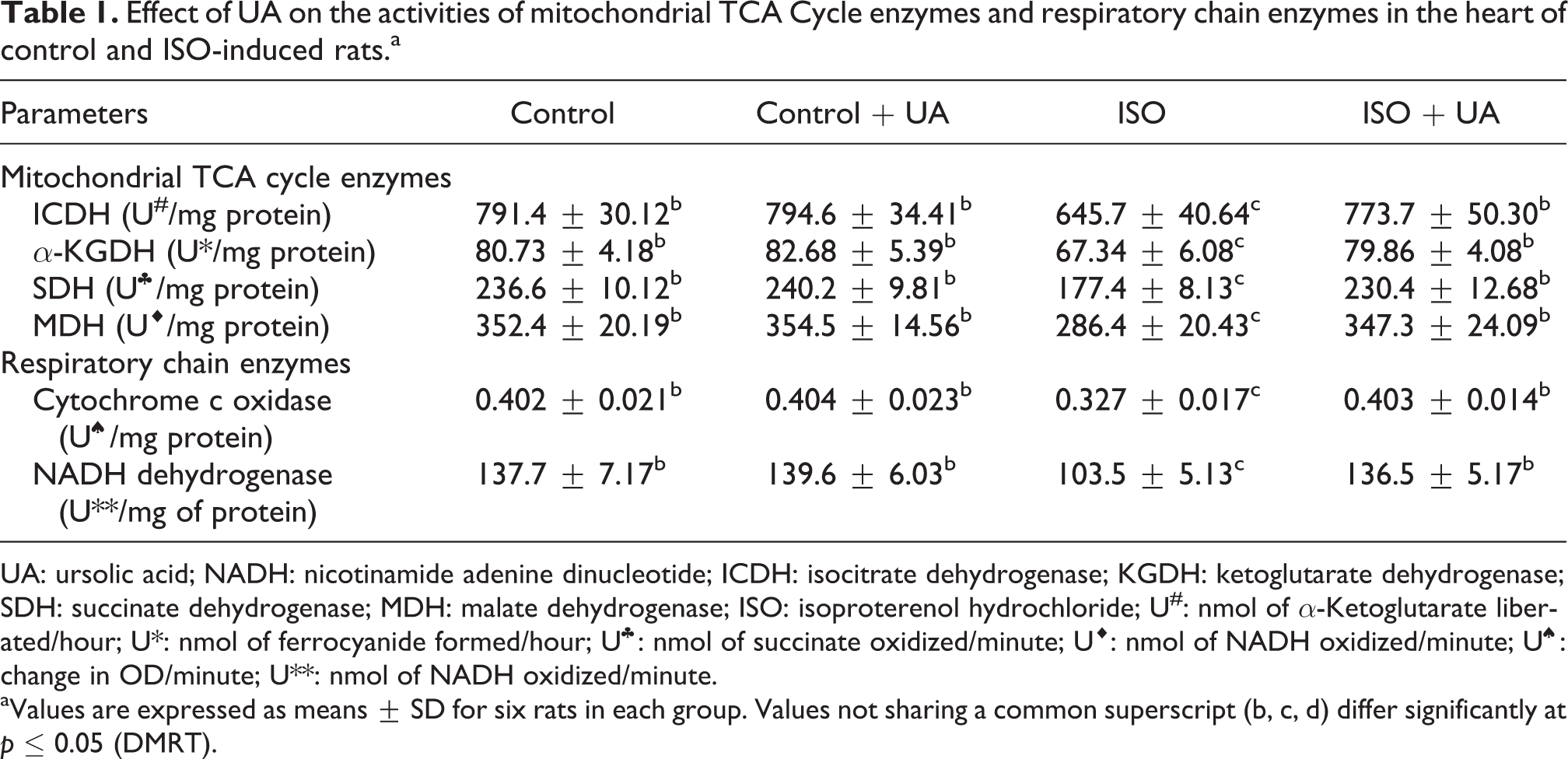
UA: ursolic acid; NADH: nicotinamide adenine dinucleotide; ICDH: isocitrate dehydrogenase; KGDH: ketoglutarate dehydrogenase; SDH: succinate dehydrogenase; MDH: malate dehydrogenase; ISO: isoproterenol hydrochloride; U#: nmol of α-Ketoglutarate liberated/hour; U*: nmol of ferrocyanide formed/hour; U♣: nmol of succinate oxidized/minute; U♦: nmol of NADH oxidized/minute; U♠: change in OD/minute; U**: nmol of NADH oxidized/minute.
aValues are expressed as means ± SD for six rats in each group. Values not sharing a common superscript (b, c, d) differ significantly at p ≤ 0.05 (DMRT).
Lysosomal enzymes
The activities of serum and heart tissue lysosomal glycohydrolases (α-glucosidase, β-glucosidase, α-galactosidase, β-galactosidase, β-glucuronidase, and β-N-acetylglucosaminidase) and cathepsins (B and D) are observed in Tables 2 and 3. Serum and heart glycohydrolase and cathepsins activities were significantly elevated in ISO-induced rats. UA treatment showed the activities of glycohydrolases and cathepsins toward normalcy in ISO-induced rats.
Effect of UA on the activities of lysosomal enzymes in the serum of control and ISO-induced rats.a

DMRT: Duncan’s multiple range test; UA: ursolic acid; ISO: isoproterenol hydrochloride; U♣: μmol of p-nitrophenol liberated/hour; U*: μmol of p-nitroaniline liberated/hour; U♦: μmol of tyrosine liberated/hour.
aValues are expressed as means ± SD for six rats in each group. Values not sharing a common superscript (b, c, d) differ significantly at p ≤ 0.05 (DMRT).
Effect of UA on the activities of lysosomal enzymes in the heart tissue of control and ISO-induced rats.a

DMRT: Duncan’s multiple range test; UA: ursolic acid; ISO: isoproterenol hydrochloride; U♣: μmol of p-nitrophenol liberated/hour; U*: μmol of p-nitroaniline liberated/hour; U♦: μmol of tyrosine liberated/hour.
aValues are expressed as means ± SD for six rats in each group. Values not sharing a common superscript (b, c, d) differ significantly at p ≤ 0.05 (DMRT).
MMP-2, MMP-9, TGF-
β
, collagen type I, and
α
-SMA protein expressions
Figure 1(a) and (b) represents the expression of MMP-2, MMP-9, collagen type I, and α-SMA by Western blot analysis in the heart tissue of ISO-induced rats. The protein expression of MMP-2, MMP-9, collagen type I, and α-SMA were significantly upregulated expression in ISO-induced MI in rats. UA administration significantly downregulated the expression of MMP-2, MMP-9, collagen type I, and α-SMA in heart tissue of ISO-induced rats.

Effect of UA on MMP-2, MMP-9, TGF-β, collagen type I, and α-SMA protein expressions in the heart tissue of control and ISO-induced rats. (a) MMP-2, MMP-9, TGF-β, collagen type I and α-SMA protein expressions by Western blot analysis. (b) Band intensity scanned by the densitometer. Histogram depicts quantitation of three independent experiments (means ± SD), with data normalized by defining the control group with MMP-2, MMP-9, TGF-β, collagen type I, and α-SMA as 1 unit. Values not sharing a common superscript (a, b, c) differ significantly at p ≤ 0.05 (DMRT). DMRT: Duncan’s multiple range test; MMP: matrix metalloproteinase; TGF: transforming growth factor; UA: ursolic acid; ISO: isoproterenol hydrochloride; SMA: smooth muscle actin.
Figure 2 shows the immunohistochemistry expression of TGF-β in heart tissue of ISO-induced rats. Intense cytoplasmic expression of TGF-β was observed in capillary vessels around cardiomyocyte in ISO-induced rats. UA administration to rats significantly decreased the expression of TGF-β.

Effect of UA on TGF-β protein expression in the heart tissue of control and ISO-induced rats by immunohistochemistry. (a) Control rat shows normal architecture and there was no TGF-β expression. (b) Control + UA rat shows normal architecture and there was no TGF-β expression. (c) ISO-control rat shows intense cytoplasmic expression of TGF-β in capillary vessels around cardiomyocyte. (d). ISO + UA: decreased expression of TGF-β. TGF: transforming growth factor; UA: ursolic acid; ISO: isoproterenol hydrochloride.
Discussion
Our study demonstrated that UA protected against ISO by changing the activities of dehydrogenases in TCA cycle, respiratory enzymes, and lysosomal enzymes. Furthermore, UA inhibited the expression of MMP-2, MMP-9, collagen type I, α-SMA, and TGF-β in ISO-induced MI in rats. Ischemia is associated with an impairment of myocardial energy production, mitochondrial respiration, and diminished oxygen uptake. ISO has been reported to cause tissue hypoxia where there is an oxygen demand. 35 In hypoxia condition, TCA cycle enzyme activities are expected to be low. 36 These enzymes are located in the inner mitochondrial membrane and affected by the excessive production of free radicals induced by ISO. The reduction in these enzymes proved the defect in aerobic oxidation of pyruvate, which might result in the low production of ATP molecules. 37
Electron transport and oxidative phosphorylation require the coordinated action of five enzyme complexes, which together are composed of different structural proteins. A reduction in enzyme content could arise in a failure of an assembly of electron transfer chain complexes or enhanced rates of degradation of complexes. 38 Decreased activities of the respiratory marker enzymes NADH-dehydrogenase and cytochrome c oxidase were observed in ISO-induced MI in rats, which is in agreement with the previous reports. 39 Cytochrome c is normally bound to the inner mitochondrial membrane by association with cardiolipin. Peroxidation of cardiolipin leads to dissociation of cytochrome c and its release through the outer mitochondrial membrane into the cytosol. 40,41 Cytochrome c in the cytoplasm triggers the activation of caspase-9, which triggers the caspase cascade and ultimately leads to apoptosis. 42 In the present study, UA treatment showed increased activities of ICDH, α-KGDH, SDH, MDH, NADH dehydrogenases, and cytochrome c oxidase. UA could be protecting “SH” groups of dehydrogenases from free radical attack and thus lowered the accumulation of reducing equivalents; thereby, oxygen consumption could be maintained to normalize the TCA cycle.
ISO-induced MI results in increased lysosomal hydrolases activity that may be responsible for tissue damage and infarcted heart. Intracellular release of lysosomal enzymes following myocardial ischemia may directly or through activation of the complement pathway result in cell injury and death. 43 In this study, we observed increased levels of serum and heart glycohydrolases and cathepsins in MI rats. Cytosolic acid hydrolases released from lysosomes and from the sarcoplasmic reticulum induce dysfunction and distribution of mitochondria, sarcoma and other organelles. 44 An increased total activity of glycohydrolases and cathepsins studied indicate the possible infiltration of inflammatory cells at the location of infarction. We observed that UA administration to the rats reduces the extent of lysosomal damage and/or increases lysosomal stability by their membrane stabilizing actions. 45
MMP-2 and MMP-9 play a major role in extracellular membrane remodeling because of their ability to initiate and continue the degradation of fibrillar collagen. 46 Increased MMP expression during heart failure may induce cardiac myocyte loss due to apoptosis 47 and ultimately lead to MI and heart failure. UA has been shown to inhibit the MMP expression and its mechanism of action against MI might be inhibiting the expression of MMP-2 and MMP-9, which further attenuates myocardial injury and damage, and thereby, it retains the normal functioning of the heart. 48 TGF-β is a locally generated cytokine with a significant function for healing and tissue fibrosis. TGF-β stimulates fibroblast proliferation and differentiation as well as the release of collagen to the ECM. 49 TGF-β expression is increased in the ischemic as well as a hypertrophied heart. 50 UA administration decreased the expression of TGF-β in ISO-induced rats. Kanzler et al. reported that the preventive effects of UA in cirrhosis and fibrosis of the liver may in part be attributed to antagonism against TGF-β. 51
Fibrosis is another integral feature of cardiac hypertrophy, characterized by the abnormal expression of ECM and accumulation of collagen, which is evidenced by the activation of α-SMA and collagen type I. 7 Generally, types I and III account for >90% of the total collagen content of the heart and are predominantly responsible for interstitial fibrosis. 52 Parthasarathy et al. reported that collagen I is increased in the heart after MI and dysregulates the expression of these protein contributes to collagen remodeling and subsequent cardiac fibrosis following MI. 53 ISO-induced cardiac fibroblast activation; cardiac fibroblasts into activated α-SMA-positive myofibroblasts is consistently noted following MI, both in human patients and in experimental models. 54 Hence, in our study, we found a suppressed expression of α-SMA and collagen I following UA treatment in ISO-induced fibrosis in heart tissue.
In conclusion, the results of the present study suggested that UA modulated the cardiac mitochondrial and lysosomal dysfunction in ISO-induced MI in rats. Our study demonstrated that UA downregulates the cardiac fibrosis by inactivating MMPs and TGF-β signaling pathways, which further contributes to the reduction of collagen-I and α-SMA in ISO-induced rats. These results indicate that UA may be a novel therapeutic agent for cardiovascular diseases by protecting the cardiac fibrosis and may attenuate MI condition.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of following financial support for the research, authorship, and/or publication of this article: This work was supported through a major research project (SR/SO/BB-36-2008 Dt.27.07.2009) from the Department of Science and Technology, New Delhi, India to the author KV Pugalendi.
References
 B activation induced by carcinogenic agents through suppression of IκBα kinase and p65 phosphorylation: correlation with down-regulation of cyclooxygenase 2, matrix metalloproteinase 9, and cyclin D1
B activation induced by carcinogenic agents through suppression of IκBα kinase and p65 phosphorylation: correlation with down-regulation of cyclooxygenase 2, matrix metalloproteinase 9, and cyclin D1