Abstract
Vulpinic acid, a lichen compound, has been shown to have many beneficial effects and its medicinal value increases day by day. As in atherosclerosis, endothelial damage is the basis of many diseases. The aim of this study is to investigate the effects of vulpinic acid against oxidative stress damage induced by hydrogen peroxide (H2O2) in endothelial cells. In order to find the IC50 of H2O2 and the protective dose of vulpinic acid, methyl thiazolyldiphenyl tetrazolium bromide (MTT) assays were performed. The amount of reactive oxygen species (ROS) induced by H2O2 and the protective effects of vulpinic acid against ROS were examined by fluorometric DCF-DA kit. The effects of H2O2 and vulpinic acid on actin filaments were determined by tetramethyl rhodamine (TRITC)-phalloidin fluorescence staining. Expression of Tie2 proteins was immunocytochemically analyzed in H2O2- and vulpinic acid-treated cells. After 24 h, the IC50 was found to be 215 μM in HUVECs treated with H2O2. The most effective dose of vulpinic acid against H2O2-associated damage was found to be 15 μM. Vulpinic acid pretreatment was shown to reduce H2O2-induced ROS production significantly (p < 0.05). It was shown that 215 μM of H2O2 caused actin fragmentation, cell shrinkage, and decrease in actin florescence intensity while vulpinic acid protected the cells from these damages. It was found that Tie2 immunoreactivity was decreased in H2O2-treated groups and vulpinic acid pretreatment reduced the expression of this protein. In conclusion, vulpinic acid decreases H2O2-induced oxidative stress and oxidative stress–related damages in HUVECs. It may be drug candidate in the therapy of atherosclerosis.
Introduction
Lichens are a symbiotic association established between fungi and algae. In lichens, vital metabolites are divided into primer and secondary metabolites. Primer metabolites are proteins, fats, carbohydrates, vitamins, and carotenoids. Secondary lichen metabolites are lichen acids, which are present as small crystal structures outside the main body of lichen. Some of the secondary metabolites secreted by lichens are vulpinic acid, usnic acid, atranorin, giroforic acid, lecanoric acid, fisodic acid, protosetraric acid, stictic acid, and salazinic acid. The biological functions of many of these metabolites have not yet been fully characterized. 1 Recent studies have shown that some of these metabolites have antitumoral, 2 antioxidant, 3 and antimicrobial 4,5 effects.
Vulpinic acid, a lichen compound, has been shown to have antiproliferative, antiangiogenic, antimicrobial, and protective effects against ultraviolet rays in cancer cells 6 and its medicinal value increases day by day. 7 –9 The protective effects of this molecule against oxidative stress have never been studied before.
Free radicals can be originated from oxygen and nitrogen. Oxygen-originated ones are named as reactive oxygen species (ROS; superoxide, hydroxyl radicals, and peroxyl radicals) while nitrogen-originated ones are called reactive nitrogen species (RNS). ROS or RNS are mismatched electron-containing molecules resulting from energy metabolism. ROS/RNS cause damage by oxidizing lipids and DNA in the cell and almost all molecules in the tissue randomly. The main component of the intracellular skeletal system is the actin filament. Many studies have shown that ROS affects actin production and formation. Many studies have shown that ROS affects actin production and formation. 10 –12 When the overproduced ROS is not eliminated, oxidative stress occurs in the cell. 13 Oxidative stress is the basis of many diseases such as diabetes, atherosclerosis, cancer, neurodegenerative diseases, liver cirrhosis, and aging. The human body has enzymatic (catalase, superoxide dismutase, and glutathione peroxidase) and nonenzymatic (glutathione) antioxidant defense mechanisms against ROS. 14 The amount of ROS at physiological level is harmless to cells and tissues and it can be removed from cells. In the cases of diseases with overproduced ROS, natural antioxidants (e.g. vulpinic acid) which can be taken as nutrients, since the antioxidant system of the cell is inadequate, can inhibit the progression of disease.
A large number of deaths due to cardiovascular diseases (CVDs) arise from atherosclerosis. Atherosclerosis is a chronic inflammatory disease characterized by the accumulation of lipid and inflammatory cells on the walls of the middle and large arteries. Hypertension, diabetes mellitus, smoking, and dyslipidemia are the risk factors for atherosclerosis and oxidative stress. The first stage of the development of atherosclerosis increased oxidative stress and subsequent endothelial damage. 15 Enzymes such as nicotinamide adenine dinucleotide phosphate oxidase, xanthine oxidase, mitochondrial enzymes, lipoxygenases, myeloperoxidases, and unseparated endothelial nitric oxide synthase are the major ROS generators in the blood vessels. Diseases that have mentioned earlier increase the activity of these enzymes and thus oxidative stress develops. 16 Because of the increase in the amount of ROS in many chronic and acute CVDs, and this increase causes damage primarily to endothelial cells, it is crucial to find and develop new therapeutic agents in this area.
In this study, it is aimed to investigate the effects of vulpinic acid, a lichen secondary metabolite, against oxidative stress damage induced by hydrogen peroxide (H2O2) in endothelial cells.
Material and methods
Lichen material, Letharia vulpina, was collected from Çataçık Forest at Sundiken Mountain in Mihalıçık-Eskisehir province in Turkey at the altitude of 1500 m. Letharia vulpina samples were stored at the Herbarium of Anadolu University in the Department of Biology (ANES).
Isolation and characterization of vulpinic acid
The air-dried lichen samples were first ground, and then 10-g portion of sample was added to 100-mL acetone. The mixture was first treated in an ultrasonic bath for 1 h, then left at room temperature overnight and filtered. The crude extract residue in filtrate was obtained after removal of solvent using a rotary evaporator. Vulpinic acid (Figure 1) was isolated from the extract residues using silica packed column chromatography (Hexane-dichloromethane mixture [1:2]). Vulpinic acid was obtained as bright yellow microcrystals. The purity of vulpinic acid was confirmed by analytical high performance liquid chromatography (HPLC) analysis using an Agilent 1100 HPLC system with an Inertsil OD-3 column (GL Sciences, Tokyo, Japan) (5 µm, 4.6 × 150 mm2) and methanol/water (70%) as an eluent system (flow rate 1.0 mL/min, detection at 254 nm). Vulpinic acid provides a single retention-time peak in HPLC at 11.71 min. The characterization of vulpinic acid was done based on melting point and comparison of nuclear magnetic resonance (NMR) spectra and Rf values in A, C, and G eluent systems with those in literature. 17,18 The melting point was determined using a Mettler Toledo (Greifensee, Switzerland) MP90 apparatus and was uncorrected. Proton NMR (1H NMR; 400 MHz) and carbon-13 NMR (13C NMR; 100 MHz) spectra were recorded on an Agilent (Santa Clara, USA) DD2 400-MHz spectrometer in deuterated chloroform (CDCl3) with tetramethylsilane (TMS) as an internal standard. The melting point of vulpinic acid was recorded as 149–151°C (lit 148°C). The NMR spectrum recorded in CDCl3 showed accordance with previous reports. 19,20 1H NMR (400 MHz, CDCl3) δ 3.87 (s, 3H), 7.22–7.28 (m, 2H), 7.30–7.36 (m, 1H), 7.32–7.48 (m, 5H), 8.12 (m, 2H), and 13.77 (s, 1H). 13C NMR (100 MHz, CDCl3) δ 54.7, 105.4, 116.0, 128.0, 128.4, 128. 6 (2C), 128.8, 129.1, 130.1, 132.1, 155.1, 160.4, 166.1, and 171.9.
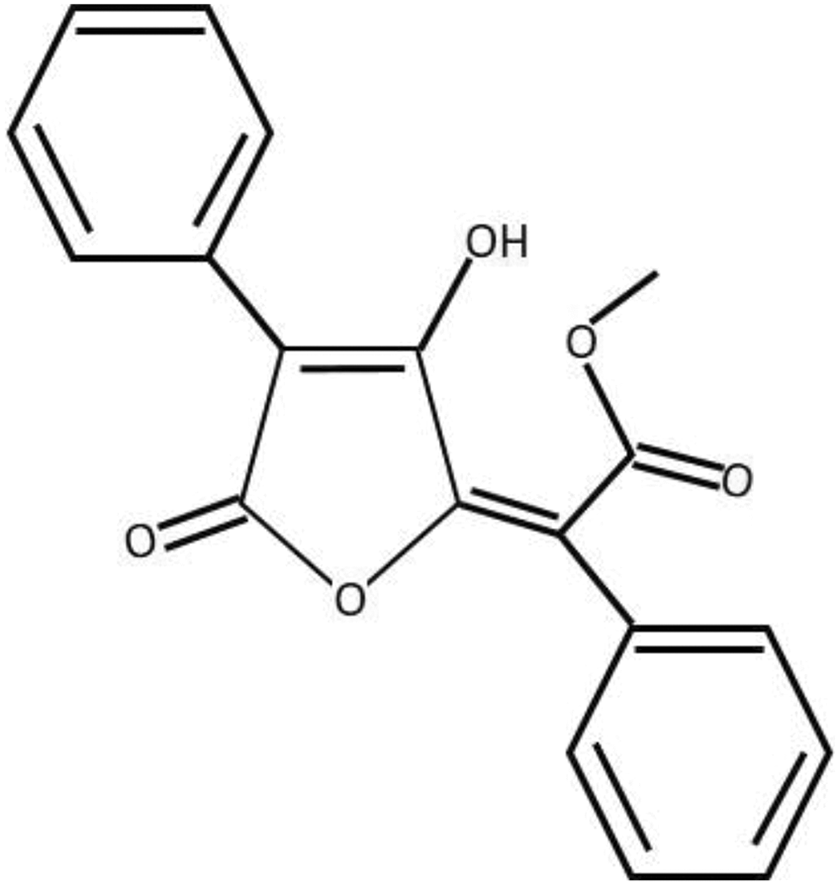
Chemical structure of vulpinic acid. 8
Cell cultures
Human umbilical vein endothelial cell line (HUVEC—CRL-1730) used in this study was purchased from American Type Culture Collection (Manassas, Virginia, USA) cell bank. Cells were incubated at 37°C with 95% air and 5% carbon dioxide (CO2) in flasks containing 20% inactivated fetal bovine serum, 0.1-mg/mL heparin, 0.03-mg/mL endothelial cell growth supplement (Sigma Aldrich, E0760, Darmstadt, Germany), 1% penicillin/streptomycin, and Ham’s F-12 K media with 1.176-g/L sodium bicarbonate according to instructions of the cell bank.
Cytotoxicity assays
Nontoxic and protective doses of vulpinic acid and IC50 dose for H2O2 were determined via MTT assay. Vulpinic acid was dissolved in dimethyl sulfoxide (DMSO) at a final concentration of 0.1%. The working solutions were freshly prepared before each experiment by diluting the stock solution with the supplemented media to obtain the final concentrations of 5, 10, 15, 20, 40, 60, 80, 100, 200, and 300 μM of vulpinic acid. H2O2 was freshly prepared from stock solution at the concentrations of 100, 125, 150, 175, 200, 250, 275, 300, 350, 375, and 400 μM diluted in the media, before the experiment. In order to find the protective dose of vulpinic acid against H2O2, HUVECs were seeded at 1 × 105 per well in 96-well cell culture plates. The cells were incubated at 37°C for 24 h for adhesion. At the end of the 24-h incubation period, the media of the adherent cells was removed. Cells were incubated with the nontoxic and protective doses of vulpinic acid for 24 h. At the end of the incubation period, media with vulpinic acid was removed. The cells were maintained in a CO2 incubator at 37°C for 24 h with fresh media containing IC50 dose of H2O2. At the end of the incubation period, the media was removed. Cells were allowed to incubate for 2 h with 0.5-mg/mL MTT solution. Then, the MTT stain was removed from the cells. In order to dissolve the formazan salts formed by the living cells, 0.1 mL of DMSO was added to each well. The optical density of the cells in the plates was read at 570 nm in the ELISA microplate reader. Untreated cell viability rate was assumed 100%, and the survival rate of the test cells was expressed as a percentage. The MTT assay was repeated three times independently.
Cellular ROS detection assay
2’,7’–dichlorofluorescin diacetate (DCF-DA) method and ready-to-use measurement kit (Cell Biolabs, San Diego, California, USA) were used to determine the amount of intracellular ROS. HUVECs were plated at 1 × 105 cells per well in 96-well plates and incubated for 24 h in an incubator containing 5% CO2 at 37°C for adhesion. After the incubation period, the media of the cells was removed and the plates were washed three times with phosphate buffered saline (PBS) for 5 min; 100 μL of 1X DCFH-DA/media solution was added to wells and then the plates were incubated at 37°C for 60 min. At the end of this period, the media of the cells was removed. The plates were washed three times with PBS for 5 min. The following procedure was carried out on cells: No treatment was administered to the control group; 15-μM vulpinic acid was applied to the vulpinic acid group for 24 h; 215-μM H2O2 was applied to the H2O2 group for 24 h. Vulpinic acid and the IC50 dose of H2O2 were applied to the vulpinic acid + H2O2 group for 24 h, respectively. At the end of the experiment, the fluorescence in wells was measured at 480 nm (excitation) and 530 nm (emission) using a fluorometric plate reader (SpectraMax M2 Microplate Reader, San Jose, CA, USA).
Fluorescent staining of F-actin
HUVECs plated on sterile coverslips in six-well plates were cultured in an incubator containing 5% CO2 at 37°C for 24 h. At the end of this period, the media of cells was removed. The cells were then incubated with the protective dose of vulpinic acid for 24 h. At the end of the incubation period, media with vulpinic acid was removed. Then, the fresh media containing IC50 dose of H2O2 was applied to the cells, and the plate was incubated for 24 h at 37°C in a CO2 incubator. After the incubation period, the media was removed. At the end of the experiment, cells that adhered to the coverslips were fixed at 37°C using a 3.7% paraformaldehyde in PBS for 15 min. After fixation, the coverslips were washed three times with PBS. The cells were treated with 0.2% Triton X-100 (Sigma, Taufkirchen, Germany, T8787) for 5 min and then rinsed three times with PBS. Afterward, cells were stained with 5-μg/mL TRITC-phalloidin dissolved in PBS for 60 min at 37°C. The coverslips were washed three times with PBS and incubated in the dark with 1-mg/mL 4’,6-diamidino-2-phenylindole, dihydrochloride (DAPI) for 30 min at 37°C. The coverslips were washed with PBS and examined under a confocal microscope (Zeiss, Oberkochen, Germany, LSM 800) to obtain the images.
Immunohistochemical staining and scoring of Tie2
HUVECs plated on sterile coverslips in six-well plates were cultured in an incubator containing 5% CO2 at 37°C for 24 h. At the end of this period, the media of cells was removed. The cells were then incubated with the protective dose of vulpinic acid for 24 h. At the end of the incubation period, vulpinic acid media was removed. Then, the fresh media containing IC50 dose of H2O2 was applied to the cells and the plate was incubated for 24 h at 37°C in a CO2 incubator. After the incubation period, the media was removed. At the end of the experiment, cells that were adhered to the coverslips were fixed at 37°C using a 3.7% paraformaldehyde in PBS for 15 min. After fixation, the coverslips were washed three times with PBS. The cells were treated with 0.2% Triton X-100 (Sigma, T8787) for 5 min and then rinsed three times with PBS. Endogenous peroxide activity was blocked by applying 3% H2O2 to the cells on coverslips for 15 min and the coverslips were washed three times for 3 min with PBS. After the application of Ultra V blocking solution (Thermo, Waltham, Massachusetts, USA, TP-125-BN) to coverslips for 10 min, Tie2 primer antibodies (Santa Cruz Biotechnology, sc-9026, Santa Cruz, USA) were added to the solution without washing and the cells were incubated overnight at +4°C. Subsequently, the samples washed with PBS were incubated with biotinylated secondary antibody (Thermo, TP-125-BN) for 10 min, and then, the samples were washed again with PBS and incubated with the streptavidin-peroxidase enzyme complex (Thermo, TP-125-BN) for 10 min. Visible immunoreactivity was obtained via applying the chromogenic 3-amino-9-ethylcarbazole (Thermo, TA-125-SA) after washing the sections with PBS. After the staining of nuclei with hematoxylin, the coverslips were closed and cells were examined under a binocular light microscope (Olympus, Shinjuku, Tokyo, Japan, BX51) to obtain appropriate images.
An immunohistochemistry based scoring system was utilized for the semiquantitative analyses of Tie2 as the percentage of positive cells in a blind fashion and 10 randomly chosen 40X objective fields. The positively stained cells for Tie2 were graded on a five-point scale: 0 = no stain, 1 = <15%; 2 = 15–25%; 3 = 25–50%; 4 = 50–75%; 5 = >75–100%. 21 Finally, the scores from all groups were calculated and statistical analysis was performed.
Statistical analysis
All experiments were repeated three times. SPSS software (Statistical Package for the Social Sciences, version 21.0, Armonk, New York, USA) was used to calculate the data. p Values less than 0.05 were considered statistically significant. Shapiro–Wilk test was performed to examine whether the data were normally distributed. One-way analysis of variance followed by Tukey’s tests was performed in normal distributed data.
Results
Cytotoxicity assay results
Vulpinic acid was found to be noncytotoxic below the dose of 20 μM. The cell viability reached a maximum at 15 μM, and after 15 μM, it was found to fall in a dose-dependent manner. At 300 μM, the viability decreased to 37% (Figure 2). The IC50 of H2O2 was found to be 215 μM (Figure 3).

Effects of vulpinic acid on cell viability after 24-h incubation in HUVECs. Vulpinic acid was found to be noncytotoxic between the doses of 1–20 μM, and the cell viability was highest at 15 μM.
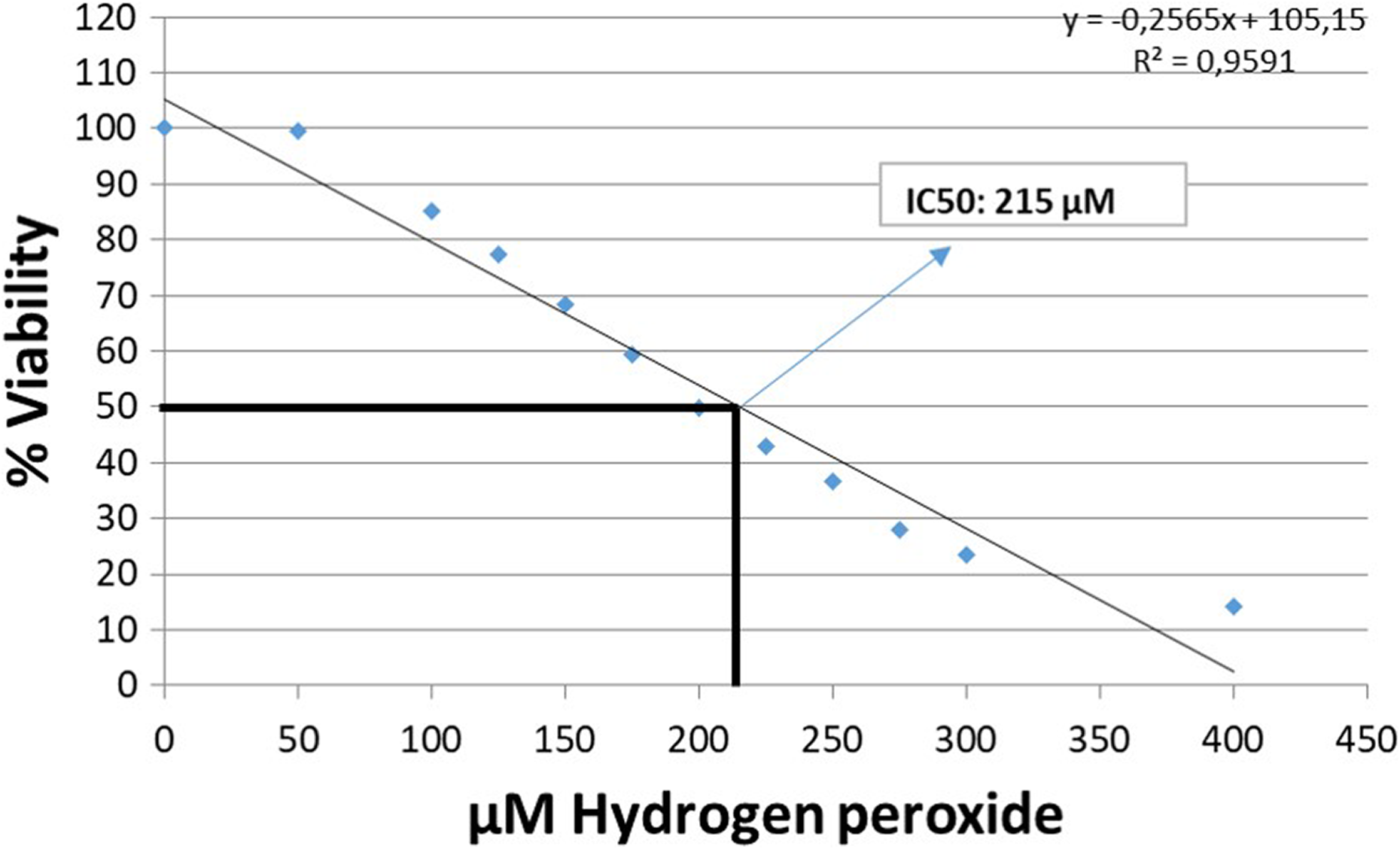
Effects of H2O2 on cell viability after 24 h incubation in HUVECs. The IC50 dose of H2O2 was found to be 215 μM. H2O2: hydrogen peroxide.
When the protective effects of vulpinic acid against the effects of H2O2 was examined, it was found that 15 μM of vulpinic acid significantly increased the viability by 41% (p < 0.001; Figure 4) in HUVECs treated with vulpinic acid for 24 h followed by H2O2 treatment for 24 h; 15-μM vulpinic acid was used as an effective concentration in all other experiments.

The protective effect of vulpinic acid against IC50 dose of H2O2. It was observed that 15 μM of vulpinic acid significantly increased the cell viability by 41% (p < 0.001). H2O2: hydrogen peroxide.
Cellular ROS detection assay
Vulpinic acid decreased the amount of ROS below the control group level (p > 0.05). In the H2O2-treated group, the ROS production was significantly increased (p < 0.05), and the vulpinic acid decreased this increase to the control levels (p < 0.05; Figure 5).

ROS measurements; 15-μM vulpinic acid reduced ROS production in HUVECs treated with 215-μM H2O2 (*p < 0.05). ROS: Reactive oxygen species; H2O2: hydrogen peroxide.
Confocal immunofluorescence results of actin
The cytoskeleton plays a major role in the physiology of endothelial cells including adhesion, permeability, vasculogenesis, and motility. The actin filaments were stained with TRITC-phalloidin. In the control and vulpinic acid groups, intracellular actin proteins were found to be homogeneous and filamentous. In the group treated with H2O2 alone, it was observed that cells became rounded and shrunk, actin filaments were disrupted forming clusters in the cytoplasm, and fluorescence intensity was decreased. In the group treated with vulpinic acid followed by H2O2 treatment, it was observed that the intracellular actin damage was decreased, actin proteins were homogeneously distributed in the cell in a filamentous manner, and cells had normal size and morphology (Figure 6).
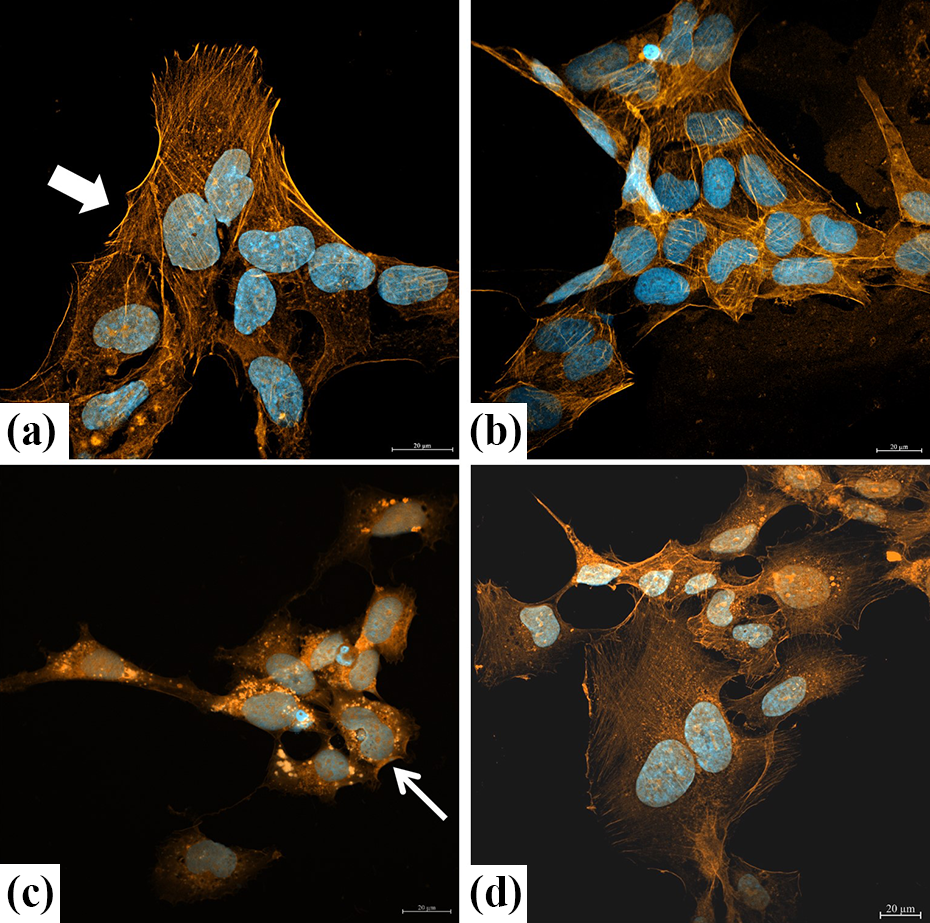
Actin immunofluorescence staining of HUVECs. (a) HUVECs with homogeneous and filamentous actin filaments (red) and normal shape nuclei (blue). Also, cells had uniform boundaries, cytoplasm, and size (arrow) in the control group. (b) HUVECs with normal shape, cytoplasm, and actin filament (red) structure in the vulpinic acid alone group. (c) In the group treated with H2O2 alone, it was observed that cells became rounded and shrunk (thin arrow), actin filaments were disrupted forming clusters in the cytoplasm, and fluorescence intensity was decreased. (d) In the group treated with vulpinic acid followed by H2O2 treatment, it was observed that the intracellular actin damage was decreased, actin proteins were homogeneously distributed in the cell in a filamentous manner, and cells had normal size and morphology. Scale bars are 20 μm in all figures. H2O2: hydrogen peroxide.
Immunocytochemistry results
Tie2, a receptor tyrosine kinase, that is found primarily on endothelial cells and plays an essential role in the development and remodeling of the vasculature. The control group and vulpinic acid-treated groups showed similar Tie2 immunoreactivity (immunohistochemically scored as 4.5). The immune reaction was intensely cytoplasmic and vesicular. In the group treated with IC50 dose of H2O2 alone, immunoreactivity was reduced (immunohistochemically scored as 2), and vesicle staining was decreased compared to the control and to the group treated with vulpinic acid alone. In the pretreatment group, it was observed that vulpinic acid increased Tie2 immunoreactivity (immunohistochemically scored as 3.5; Figure 7).
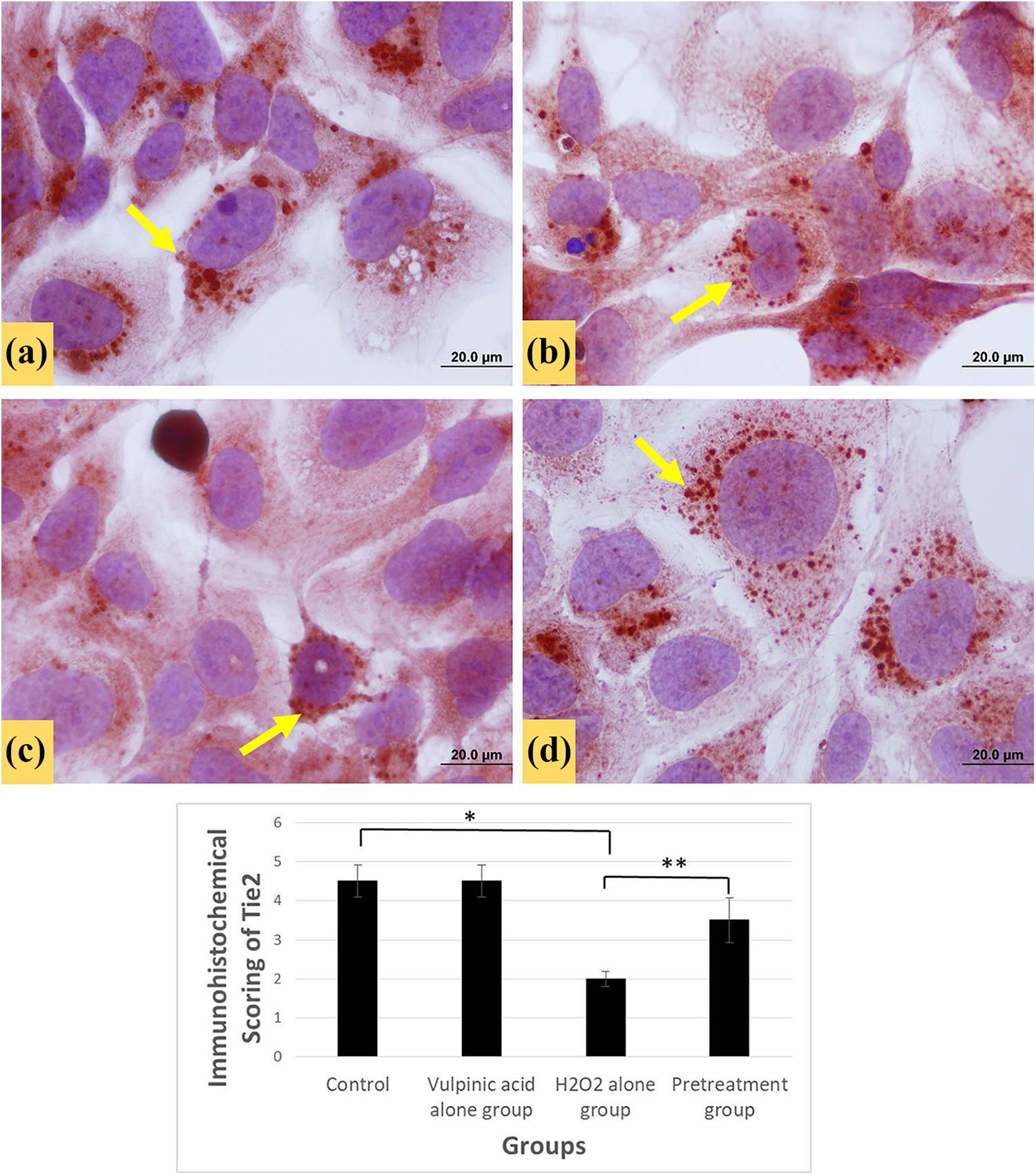
Images of HUVECs stained with Tie2. (a) HUVECs with uniform boundaries and normal size in the control group. Immunoreactivity (arrow) scored as 4,5. (b) Similar Tie2 staining with control group in the vulpinic acid alone group. Immunoreactivity scored as 4,42. (c) Decreased immunoreactivity in the H2O2 alone group and scored as 2. (d) In the group treated with vulpinic acid for 24 h followed by H2O2 treatment for 24 h, Tie2 immunoreactivity was increased and scored as 3,5. Scale bars are 20.0 µm in all figures. *Control and H2O2 groups are significantly different from each other p < 0.05; **H2O2 and pretreatment groups are significantly different from each other p < 0.05. H2O2: hydrogen peroxide.
Discussion
In this study, it was shown that vulpinic acid had protective effects against H2O2-induced oxidative stress in human umbilical vein endothelial cells (HUVEC).
The cytotoxic effects of vulpinic acid on HUVECs were examined. The doses below 20 μM of vulpinic acid were found to be noncytotoxic. The cell viability reached a maximum at 15 μM, and after 15 μM, it fell in a dose-dependent manner. Koparal also has examined the effects of vulpinic acid on HUVECs for 24 h and has found similar results. 8 There are not many studies in the literature that have examined the noncytotoxic and toxic doses of vulpinic acid on normal cells. The limitations of the studies highlight the need for further evidence in this regard.
Today, among all causes of death, death from CVDs takes the leading place. One of the most important causes of CVD is atherosclerosis. The World Health Organization reports that 16.7 million people die from atherosclerotic CVDs annually. 22 Atherogenesis is a complex process involving some mechanisms such as endothelial dysfunction, neovascularization, vascular proliferation, apoptosis, matrix degradation, oxidative stress, inflammation, and thrombosis. 23 Although the pathophysiological mechanisms of atherosclerosis have not yet been elucidated, most hypotheses regarding the pathogenesis and progression are related to the impairment of normal homeostatic mechanisms that lead to oxidative stress in endothelial cells. Studies have shown that oxidative stress in endothelial cells is an important cause of atherogenesis. Oxidative stress is defined as a redox state that occurs as a result of impairment of the balance between antioxidant and oxidant (ROS and RNS) activities. 15 Endothelial cell dysfunction results in the earliest detectable changes in the atherosclerotic lesions. 24 In this study, we investigated the protective effects of a lichen acid, vulpinic acid, against oxidative stress. When we look at other lichen acids used in oxidative stress other than vulpinic acid, Polat et al. found that a lichen compound usnic acid has antioxidant features. 25 Emsen et al. studied, with livetoric acid and physodic acid, two lichen acids isolated from Pseudevernia furfuracea and found both have antioxidant capacity. 26
In order to generate oxidative stress in HUVECs, 215 μM (IC50 dose) of H2O2 was used, while the IC50 dose of H2O2 was 300 μM for Park, 27 180 μM for Memory and Arrays, and 250 μM for Amorn et al. 28,29 We think that this difference has arised from the fact that the HUVECs that were used in the experiments had had different passage numbers or that the manufacturers had used different cell isolation protocols. In order to examine the protective effect of vulpinic acid, the cells were first treated with vulpinic acid for 24 h and then with H2O2 for 24. At the end of the experiment, the changes in the amount of ROS were examined and it was observed that vulpinic acid significantly suppressed ROS production at 15 μM. This is the first study to show the antioxidant properties of vulpinic acid. Fernandez-Moriano et al. found that fumarprotocetraric acid, a lichen metabolite, emerges as a potential drug candidate in the therapy of oxidative stress–related diseases, such as the neurodegenerative disorders via intracellular redox modulation at 1–25 μM. 30 Lichen acids are promising in the treatment of oxidative stress–based diseases.
Endothelium is a semipermeable barrier between blood and underlying tissue. The continuity and vital physiology of this barrier depends on the strength of the cytoskeleton and cell–cell linkage units. Actin protein has important roles in both the cytoskeleton and the lateral cell junctions. 31 Cells constantly change the filamentous actin network according to their environmental and intracellular biochemical changes. Like many intracellular macro and microstructures, actin proteins are also affected by oxidative stress and change their structures. 32 Lu and his colleagues have shown that smoking results in endothelial damage through increased oxidative stress in the lungs and impaired F-actin structure in endothelial cells in rats. 33 Simoneau et al. have shown that increased oxidative stress leads to actin fragmentation, impaired endothelial cell layer, and increased endothelial permeability. 34 In this study, we also have shown that H2O2, which was administered to the cells to create an oxidative stress model, causes shrinkage in endothelial cells, fragmentation in actin filaments, and cell shape deformities. These findings are similar to those in the literature. When we pretreated the cells with vulpinic acid as a protective agent, unlike in the literature, it was shown that vulpinic acid prevented these degenerations occur in actin proteins and cell morphology.
The receptor tyrosine kinase Tie2 is vastly expressed in endothelial cells. It regulates angiogenesis, endothelial cell survival, proliferation, migration, adhesion and cell spreading, and reorganization of the actin cytoskeleton. 35 In our study, it was observed that the expression of Tie2 protein was decreased by oxidative stress, and when the cells were treated with vulpinic acid before H2O2, Tie2 expression in cells was found to be similar to control levels. Balogh et al. have shown that expression of pro-angiogenic cytokines and receptors; VEGF, ANG2, and Tie2 proteins have been decreased, when they generate oxidative stress via 4-hydroxy-2-nonenal in HUVECs. 36 This finding is parallel to our findings. Tie2 protein has also been shown to be downregulated in endoplasmic reticulum stress associated with oxidative stress. 37
In conclusion, it is shown that vulpinic acid obtained from lichens protects endothelial cells against oxidative stress damage. Endothelial dysfunction is the basis of many diseases. One of the most important causes of this endothelial damage is the deterioration of the oxidative balance. The fact that vulpinic acid, which can be isolated from abundant lichens in the nature, changes this balance positively increases the medicinal importance of this plant. An in-depth investigation of the positive impacts of vulpinic acid on oxidative stress may help to develop new treatment agents and methods in the treatment of many oxidative stress–based diseases such as atherosclerosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
