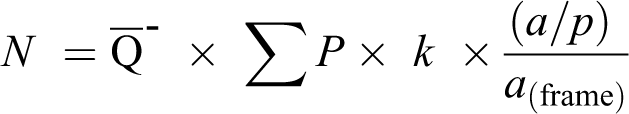Abstract
The objective of the present study was to perform histological and stereological examination of alcohol-induced changes in the optic nerve, considered an extension of white matter of the brain, in rats. This study included 20 male Wistar albino rats aged 60 days and weighing 190–220 g. The rats were divided into three groups: ethanol (n = 7), maltodextrin (n = 7), and control (n = 6) groups. The ethanol group was administered ethanol at a dose of 6.4% (v/v) instead of water for 18 days; the maltodextrin group received maltodextrin for the same time period, and the control group was the sham group. At the end of the experiment, a 0.5-mm long section of the optic nerve starting from the optic chiasma was dissected and examined with routine microscopic histological examination methods. The modified Cavalieri method was used for stereological measurement. Total tissue area ratios were calculated with a point grid provided by the Shtereom 1.5 software package. The statistical comparison of the groups revealed that the ethanol group had a significant reduction in the number of axons and sheath area of the optic nerve compared to the control and maltodextrin groups (p < 0.017, p < 0.022, respectively). These results indicate the toxic effects of ethanol on the optic nerve.
Introduction
Nowadays, alcohol consumption is gradually increasing, and alcohol-related problems pose a serious public health threat. 1 Depending on the total amount and duration of alcohol consumption, many organs, tissues, and systems are damaged. A large body of evidence from experimental animal and human studies has shown the sensibility of the central nervous system to ethanol’s effects and that exposure to ethanol during brain ontogeny can cause irreversible defects in brain function and structure. 2 Some parts of the central nervous system appear to be more sensitive to alcohol’s toxic effects. Alcohol’s adverse effects on brain are generally in the form of volume loss in the white and gray matter as well as ventricular enlargement. 3 –6 These changes are more prominent in the frontal lobes, parietal and medial temporal cortexes, cerebellum, and subcortical/thalamus, pons regions. 7 –9 The optic nerve is the second one among the 12 cranial nerves and originates from a pouch that buds from the diencephalon in the embryonic development. Unlike entire other cranial nerves, the optic nerve (a white matter tract of the central nervous system) is coated by the meninges and surrounded by cerebrospinal fluid (CSF) throughout its length. 10 We found a lot of study on the toxic effects of alcohol on intrauterine optic nerve in our literature review, but we found no studies on male rats of 60 days. This study was aimed at performing histological and stereological examination of the changes in the optic nerve, which is considered an extension of the brain, induced by alcohol that is currently responsible for serious health problems.
Materials and methods
Application of experiment
Our study was approved by Van Yüzüncü Yıl University Animal Studies Local Ethics Committee (2017, No: 06). It included a total of 20 male Wistar Albino rats aged 60 days and weighing 190–220 g. The animals were divided into three groups: the control group (n = 6) that underwent no intervention; the maltodextrin group (n = 7), which underwent oral administration of water + maltose with dextrin (MD, DIXING, China) (to equalize extra calorie added by ethanol consumption); and the ethanol group (n = 7), which underwent oral administration of ethanol (6.4% v/v) (99% Etil Alkol (TEKKIM, Turkey)) diluted with tap water instead of water. Maltose with dextrin was added to the tap water to provide a similar caloric intake to group that did not consume alcohol. 11 The rats were kept in a cage with normal light–dark cycle (12-h/12-h) at an ambient temperature of 21 ± 2°C and were fed standard feed ad libitum. The groups were administered study agents for 18 days. 12 At the end of the experiment, all animals were anesthetized with ketamine hydrochloride (50 mg/kg; Ketalar®, Pfizer, Turkey) and xylazine hydrochloride (15mg/kg; Rompun®, Bayer, Turkey) administered intraperitoneally. The animals were fixed by transcardial perfusion under anesthesia with heparinized saline solution followed by 10% neutral-buffered formalin. 13 Their skull was opened, a 0.5-mm section of the optic nerve starting from the optic chiasma was dissected and examined with routine microscopic histological examination methods (Figure 1). All experimental protocols conducted on rats were performed in accordance with international guidelines.
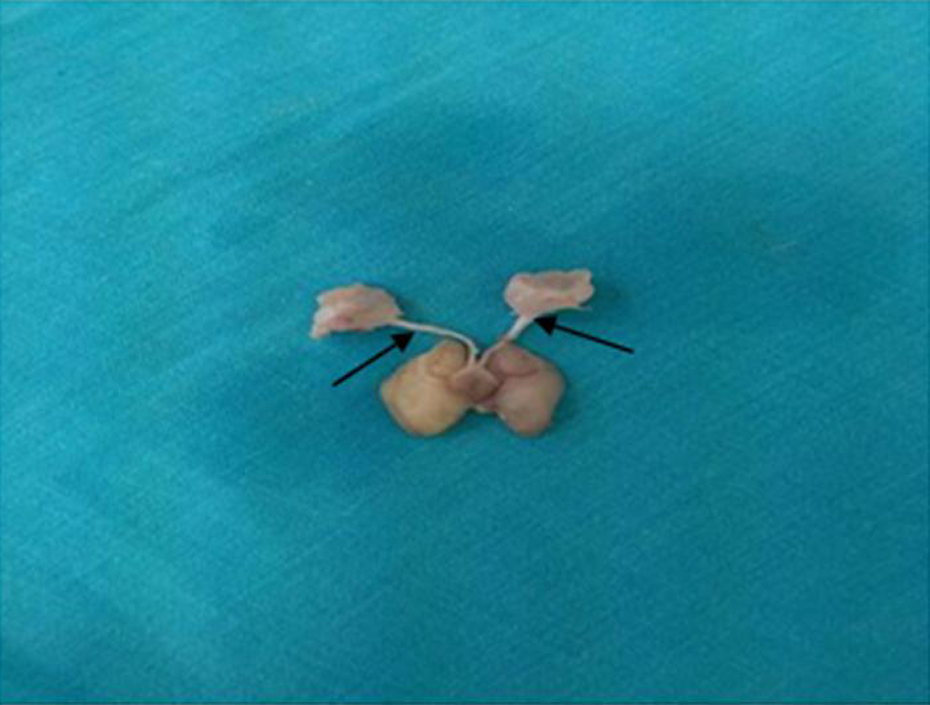
Macroscopic image of the rat brain and optic nerve (black arrows).
Histological staining
The sampled optic nerves remained in a 2.5%-glutaraldehyde and then post-fixed with 2% osmium tetroxide (OsO4), were dehydrated in graded concentrations of ethanol, and were embedded in resin (Epon-812, Sigma, Missouri, USA). For light microscopy, 0.5 µm (semi-thin section) transverse sections were obtained using an ultramicrotome (Leica RM 2125, Leica instrument, Nussloch, Germany). The sections were then stained with 1% Toluidine Blue in 1% Borax. 14 The sections were then examined under light microscope (Nikon, Eclipse Japan) and photographed.
Stereological analysis
In stereology, a systematic and random sampling is utilized to obtain quantitative and unbiased information.
15,16
The groups were compared with the stereological method with respect to changes in the=cross-sectional area of the optic nerve and the axonal number. An unbiased stereological dissector–Cavalieri combination method known as the physical dissector combination was used.
16
–18
Then, total optic nerve area ratios were measured with a point grid provided by the Shtereom v. 1.5 software package. Total optic nerve area ratios estimation using formula:
All axons in the optic nerve were counted at a magnification of 200× using light microscopy (Nikon (Y-IM) Eclipse, Japan). The total number of axons in the optic nerve was estimated using the formula
where N is the total axon number,
Statistical analysis
Statistical analysis of study groups was done using the Kruskal–Wallis test. The significance level was set at p < 0.05. All statistical analyses were performed using SPSS (version 13) software package.
Results
At the end of the experiment, we found no change in rat weights (Table 1). As for the light microscopic examination of the groups the sample of control group appeared to optic nerve sheath normal appearance. The samples of the ethanol group had a thinned, undulating optic nerve sheath with patchy areas of detachment. In the samples of the maltodextrin group, optic nerve sheath thickness was increased, but its margins were not significantly altered (Figure 2).
Change weight in groups during experimental period.a

aLetters a, b, c before values denote intergroup differences.
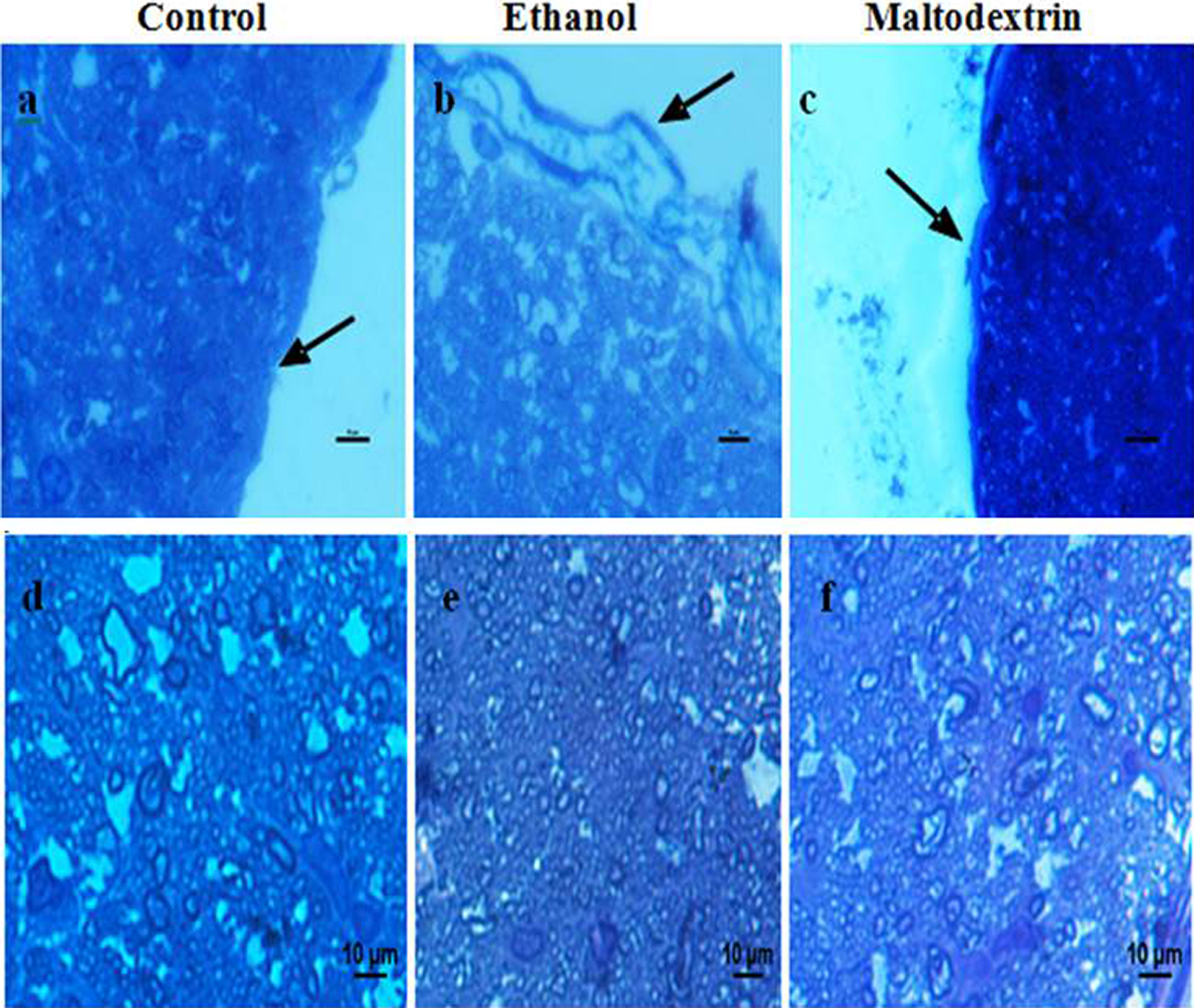
(a) to (f): Light microscopic view of the optic nerve (×2); (a) to (c): the optic nerve sheath (black arrows); (d) to (f): the axons of the optic nerve (Toluidine Blue in Borax, scale bars, 10 μm).
Stereological findings
The comparison of the mean total optic nerve areas revealed no significant inter-group differences (p > 0.271). The comparison of the mean optic nerve sheath area showed that the ethanol group had a significantly reduced level, while the maltodextrin group had a significantly increased level (p < 0.022; Table 2).
The descriptive statistics and comparison of study parameters between the study groups.a
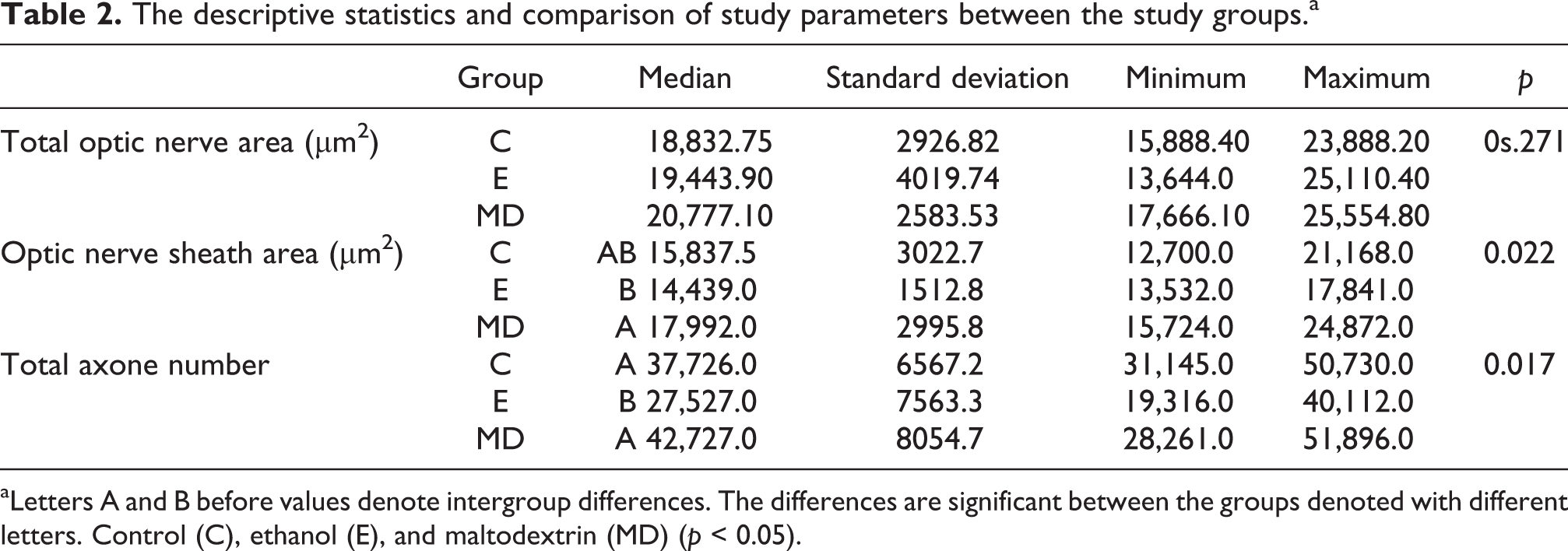
aLetters A and B before values denote intergroup differences. The differences are significant between the groups denoted with different letters. Control (C), ethanol (E), and maltodextrin (MD) (p < 0.05).
The intergroup comparison of the mean total axonal number showed that the ethanol group had a reduced total axon number compared to the control group and the maltodextrin group (p < 0.017), although the maltodextrin group had a similar total axonal number with the control group (Table 2).
Discussion
Alcohol (ethanol) affects central nervous system in various ways. In addition to its direct toxic effects on neurons, it may injure brain through its metabolic products (acetaldehyde, etc.), nutritional deficits secondary to heavy alcohol intake, and hepatic disorders. 19 Ethanol is a well-known optic nerve toxin.It is usually one of the few substances believed to cause toxic optic neuropathy. 20 The occurrence mechanism of alcohol-induced neuropathology has not been fully explained. Generally accepted that the widespread pathway, is mitochondrial injury and unbalance of extracellular and intracellular-free radical homeostasis. 21 Neuroimaging studies has demonstrated alcoholism-related abnormalities in white matter macrostructure and microstructure. At the same time, neuroimaging has shown a brain volume reduction related to alcoholism. 22 Human alcoholics typically show important volume loss in subcortical and cortical brain structures, including both white and gray substance shrinkage. 23 It was reported that cessation of alcohol consumption led to partial improvement of brain shrinkage 24 and an increase in the cerebral white matter volume. 25 However, our study did not detect any difference in the optic nerve area despite a reduction in the axonal number in rats. The same size of the total area of the optical nerves among the groups suggests that the small-diameter axons are relatively lost in ethanol-exposed rats. It was shown that neurons were formed from neuronal stem cells in at least two regions of human brain. 23 Animal studies have shown that alcohol prevents neuronal stem cell proliferation and reduces neuronal survival in a dose-dependent manner. 26 Prenatal and postnatal alcohol exposure causes various effects and clinical symptoms in the central nervous system. Mothers who had taken a single dose of alcohol throughout pregnancy are known to bear children with the specific findings of the fetal alcohol syndrome. 27 Optic nerve from prenatal versus postnatal alcohol-exposed rats demonstrated a decrease in size. Ultrastructural changes and retarded development in macroglial cells, myelin sheaths, and optic axons were also demonstrated. 28 Especially three principal process emerge during both transition from juvenileness/adolescence to adulthood and brain maturation phases. 29 The advancing myelination of axons results in developmental increases in cortical white substance through adolescence and into adulthood serves to speed up the knowledge flow throughout axons and is supposed to increase the overall speed of knowledge processing within the brain. 30 Alcohol exposure in this period causes impaired brain maturation. Although the underlying mechanisms of alcohol damage caused by alcohol during the adolescence period are still unclear, different mechanisms have been proposed. These mechanisms suggest that ethanol drinking might induce excitotoxic neuronal damage and induce adolescent brain damage by mechanisms involved in neuroinflammation. 31 In agreement with these literature data, our study stereologically demonstrated a reduction in the axonal number of the optic nerve, which is considered an extension of the white matter, in ethanol-exposed rats compared to the control group.
Another study suggests that the use of ethanol in the development of an optic neuropathy has no direct effect on the optic nerve, probably due to malnutrition. 32 In our study, we did not find any serious weight loss indicating that the ethanol group was undernourished.
Optic nerve sheath provides important information about intracranial pressure and CSF shifts. 10 Ethanol is a simple molecule and spreads into all fluid compartments by simple diffusion. Its CSF concentration is almost identical to its serum concentration. 33 Ding et al. have shown that as the amount of alcohol consumed increases CSF-filled areas of the brain become larger. 34 Consistent with this information, neuropathological study showed that CSF-filled spaces lining the brain and spinal cord fluid (pericerebral area) is distinctly in an increasing trend in men drinking more than eight standard drinks per day compared to men drinking five to eight standard drinks per day. 35 We showed disruption of optic nerve sheath among rats subjected to ethanol. When compared with the control group stereologically, the ethanol group had a significant reduction in the optic nerve sheath area. These findings suggest that a low ethanol dose was administered, so that it did not produce any increment in the CSF volume, and it caused a reduction in the nerve sheath area by disrupting the structure of connective tissue elements found in nerve sheath. Frontal cortex myelination occurs between the ages of 13 and 21 years. The vulnerable period of ethanol is not a single short period but is continuous for years in adolescents. 36
Conclusions
In the light of these data, more effective alcohol control strategies during the adolescence and late puberty periods should be established as ethanol’s adverse effects cause more serious health effects during those periods. By this way, it is possible to have more healthy generations and to reduce alcohol’s significant financial burden brought about by its huge mortality and morbidity risk.
Footnotes
Acknowledgments
The authors are grateful to Van Yuzuncu Yil University Department of Scientific Research Projects (TYL-2017-6300) for funding this study and Sıddık Keskin for statistical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Van Yuzuncu Yil University Department of Scientific Research Projects (TYL-2017-6300). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.