Abstract
Lipopolysaccharide (LPS) as an endotoxin forms part of the cell wall of gram-negative bacteria and is responsible for initiating an acute inflammation after entering the living tissue. In this study, male rats were divided into eight groups: control group, vitamin E (VE) treatment group (200 mg/kg body weight (b.w.)), sodium selenite (SS) treatment (0.35 mg/kg b.w.) group, VE + SS treatment group (200 + 0.35 mg/kg b.w.), LPS treatment group (10 mg/kg b.w.), LPS + VE (10+200 mg/kg b.w.), LPS + SS treatment (10 + 0.35 mg/kg b.w.), and LPS + SS + VE treatment (10 + 0.35 + 200 mg/kg b.w.) group. Oxidative stress parameters, pathological changes, immunohistochemical analyses, terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end-labeling (TUNEL) assay, and changes in DNA structure with comet assay of the kidney were investigated at the end 6 h comparatively with the control group. When LPS-treated group was compared with the control group, antioxidant enzyme activities were decreased and malondialdehyde (MDA) levels, changes in histological and DNA structure and apoptosis were increased significantly at the end of 6 h. However, when LPS + SS and/or VE-treated group were compared with the LPS-treated group, superoxide dismutase, catalase, glutathione peroxidase, and glutathione-S-transferase activities were increased and MDA levels were decreased significantly at the end of the treatment period. Light investigations figured out pathological changes in kidneys of LPS- and LPS + SS and/or VE-treated groups. There was a decrease in the number of proliferating cell nuclear antigen-positive cells and an increase in the number of TUNEL-positive apoptotic cells in the wall of the distal and proximal tubules. As a result, it was observed that the combined use of antioxidants was more protective than their use alone against LPS.
Introduction
Endotoxin is part of the cell walls of gram-negative bacteria and responsible for launching an acute inflammation after entering the living tissue. 1 The endotoxin molecule is found in the lipopolysaccharide (LPS) layer as long as it remains on its membrane. 2 It is the key molecule that triggers the sequence of events of endotoxemia, which is evolved during the rapid growth of the cell or cell destruction. Sepsis is released by breakdown of the bacteria and causes endotoxemia. 3
Experimental studies have shown that LPS applications can be performed to induce sepsis/endotoxemia in animals. LPS has been used by many investigators to produce experimental endotoxemia in animals such as rabbit, rat, mouse, dog, goat, horse, buffalo, or cattle. 4 –8 Metabolic, immunological, physiological, toxicological, and pharmacological studies are being carried out using LPS with septic shock. 9
Septicemia can activate different cells such as macrophages, neutrophils, endothelial, and epithelial cells further resulting in a number of regulatory abilities such as cytokines, chemokines, platelet activation factor, interferon γ, prostaglandins, leukotrienes, and proteases. These events bring immune cell activation together with the sequence reactive oxygen species (ROS) emerging. 10 Cells can tolerate light oxidative stress through the antioxidant defense system under physiological conditions. That is, all biological cells have normal conditions under oxidative stress at the tolerable level. However, where the defensive systems are not sufficient, the balance between ROS and antioxidants is compromised, and thus, macromolecules are damaged in the cells. Free radicals are caused by oxidative stress, and endotoxic shock with oxidative stress results in high mortality. 11
Selenium (Se) is required for the growth of humans and animals at a low concentration. The biological importance of Se is a cofactor of glutathione peroxidase (GPx).
12,13
Each subunit of the GSH-Px enzyme, which contains a Se atom in the form of selenocysteine, plays an important role in the reduction of hydrogen peroxide (H2O2) to water in the cell. The function of GSH-Px is complementary to the function of vitamin E (VE). VE, as a good free radical scavenger, is a lipophilic vitamin that is located within the biological membranes.
14
VE has a tocopherol structure. Naturally, there are various tocopherols such as alpha, beta, gamma, delta, eta, and zeta.
Sepsis drew attention to modern medicine according to the high rate of sepsis mortality, the increase in the number of cases every year, and the failure of preclinical/clinical research results. The aim of this study is to evaluate the acute toxicity of LPS on rat renal cells as histology, formation of apoptosis, changes in antioxidant enzyme acivities, malondialdehyde (MDA) levels, and DNA structure and to investigate the protective effect either alone or as a combination of SS and VE on kidney damage caused by LPS.
Materials and methods
Chemicals
LPS (Escherichia coli LPS, serotype 0127: B8) and sodium selenite (Na2SeO3, SS; 99% pure) were obtained from Sigma Aldrich, Germany; VE (
Animal grouping and treatment
This study was carried out with the approval of the ethics committee from Erciyes University Laboratory Animal Breeding and Experimental Research Center (HADYEK) (16/133). Male Wistar rats were used in the study (approximately weighing 300–320 g). Rats had been quarantined 10 days before application. The rats were housed in special cages, standard laboratory diet, and were fed with water. Rats were irradiated at room temperature of 22 ± 30°C, with 12-hlight/12-h dark photoperiod exposure. Rats were divided into eight groups, each consisting of seven rats. Group 1: control rats, which received no chemical (n = 7); group 2: VE-treated rats (n = 7); group 3: SS-treated rats (n = 7); group 4: VE + SS-treated simultaneously (n = 7); group 5: LPS-treated rats (n = 7); group 6: rats received VE, 60 min before administration of LPS (n = 7); group 7: rats received SS, 60 min before administration of LPS (n = 7); and group 8: VE + SS were administered simultaneously, 60 min before LPS treatment (n = 7). A single dose of LPS intraperioneally (i.p.; 10 mg/kg b.w.; n = 7) was given to the experimental groups. VE and SS of 200 mg/kg b.w. and 0.35 mg/kg b.w. (n = 7), respectively, were given orally. After 6 h of treatment, the rats were killed and dissected, and the kidneys were taken for light microscope investigations and biochemical and molecular analyses.
Determination of MDA levels
To determine the amount of MDA, kidney from each rat in each group tissues was used. Based on the method used by Ohkawa lipid peroxidation, which reacts with thiobarbituric acid (TBA) at 532 nm, the amount of MDA, the end product, was measured. 20 Absorbance was read at 532 nm in the spectrophotometer to the TBA added mixture. The amount of MDA is expressed as nanomoles per milligram protein.
Determination of antioxidant enzyme activities
Increasing pyrogallol absorbance was measured at autoxidation in an alkaline medium at 440 nm for 3 min using the Marklund method for total superoxide dismutase (SOD) determination. 21 One unit of total SOD activity indicates the autoxidation of pyrogallol. It was calculated as the amount of protein causing 50% inhibition. Later, total SOD activity was given for per milligram of protein as units per milligram protein.
Catalase (CAT) enzyme activity was assayed by the method described by Aebi. 22 Reduced absorbance was measured at 240 nm for 3 min showing the breakdown of H2O2. Changes in absorbance per unit time using a constant number (ε 240: 0.0394 mM/cm) were taken as a measure of CAT activity. Enzyme activity was given in units per milligram protein.
The glutathione peroxidase (GPx) assay is described by Paglia and Valentine. 23 Oxidized NADPH to nicotinamide-adenine-dinucleotide phosphate causes reduction absorbance at 340 nm. It was indirectly used to determine the activity of GPx at 340 nm for 3 min. GPx activity was measured in units per milligram protein.
The glutathione-S-transferase (GST) assay developed by Habig et al. was utilized. 24 1-Chloro-2,4-dinitrobenzene (CDNB) was used as substrate for all isozymes of GST. CDNB was conjugated with reduced glutathione (GSH) by the GST enzyme and the absorbance increased at 340 nm due to the oxidation of glutathione. The rising absorbances were read at 340 nm for 3 min for the determination of enzyme activity. The specific activity of the enzyme was given in units per milligram protein.
Assessment of histological changes
Kidney tissues were obtained from rats for light microscopy studies. The tissues were identified in formaldehyde fixator for light microscopic examinations of kidney tissue. After washing and dehydration, the tissues were turned into paraffin blocks. Microtome was used for the preparation of paraffin blocks, and sections of 6–7 μm thickness were cut. The obtained sections were stained with hematoxylin–eosin staining and photographed on an attachable microscope (Olympus E-330, Tokyo, Japan). Ten preparates of kidney tissue samples taken from each animal were examined. Each preparate was evaluated for histopathology. The preparates in all groups were plotted as none (−), mild (+), moderate (++), and severe (+++) damage.
Assessment of immunohistochemistry
The mouse monoclonal antibody (proliferating cell nuclear antigen (PCNA); Thermo) reacts with PCNA a cell cycle-related nuclear protein that accumulates in the late G1 and S phases of proliferating cell. 25 Paraffin sections were passed through the xylem, followed by alcohol series for removal of paraffin and then rehydrated. The slides were incubated with 3% H2O2 prepared in absolute methanol for 30 min to inhibit the endogenous peroxidase activity of the tissues. To remove the antigenic structures, the sections were boiled in citrate buffer solution (pH 6.0) in a 600-W microwave oven for 10 min. Following this, the tissues were covered with PCNA using commercial monoclonal primary antibodies and ready-to-use biotinylated secondary antibody and then incubated at room temperature for 15 min. Subsequently, sections were incubated with horseradish streptavidin peroxidase conjugate for 15 min at room temperature and 3,3′-diaminobenzidine tetrahydrochloride-H2O2 substrate was applied and then the process was terminated after 15 min. After contrast dyeing with Harris hematoxylin, the tissues were dehydrated in the alcohol series. The transparent sections in the xyloid were covered with an adhesive (Entellan; Merck). The proliferative index was assessed by counting the number of PCNA-positive cells at 200× magnification in five randomly chosen fields per slide in seven animals. Images of tissue were obtained from Olympus light microscope (Olympus BX51, Tokyo, Japan). 26
TUNEL (apoptosis) assay
The terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end-labeling (TUNEL) method was used to demonstrate apoptosis in kidney tissue. For this, the In situ Cell Death Detection Kit, Fluorescein kit from Roche, Switzerland was used, and the staining procedure was performed according to the kit procedure. To summarize, kidney tissues of 5–6 µm in thickness were washed with phosphate-buffered saline (PBS) and then rehydrated. The antigen was then placed in a microwave oven in 0.01 M sodium citrate buffer for 5 min at 350 W for antigen recovery and then allowed to cool for 20 min in room heat. The tissues were washed 3 times for 5 min with PBS and then incubated with the TUNEL reaction mixture in the kit for 60 min at 37°C in a humid and dark environment. Antibody staining with 4′,6-diamidino-2-phenylindole was performed on the washed tissues 3 times for 5 min with PBS. The tissues covered with glycerol closure solution were imaged at 450–500 nm wavelength on an fluorescence microscope. 27
Assessment of DNA structure with comet assay
The tissues were separated by 1 g thin slices with a scalpel. Then, 5 ml of cold phosphate buffer was transferred to the beaker and mixed for 1 min at 500 r/min. The slides were coated with a thin agarose gel (0.5%) and frozen (+4°C) overnight to ensure that the cells were better retained. The supernatant was used as the cell suspension. The cell suspension was mixed with 100 ml low-melting agarose (0.8% in phosphate buffered salt solution (FFT)). Then, 100 μl of this mixture was covered with slides so that it was spread over the coated slides with no air bubbles. The cell suspension was stored in the refrigerator for 20 min and allowed to freeze. The coverslip was then carefully separated from the slides. The coated slides placed in lysis buffer for 2–9 min (0.045 M Tris-borate-EDTA (TBE), pH: 8.4, 2.5% sodium dodecyl sulfate). The slides from here were placed for 2 min in an electrophoresis solution. After electrophoresis, the slides were stained with 60 μl (20 μg/ml) of ethidium bromide solution. 28
Data analysis
The statistical data used in this study were the one-way variance analysis (ANOVA) and the Tukey’s test in the Windows SPSS 11.0 computer program. A value of p < 0.05 was considered statistically significant.
Results
Evaluation of biochemical results
Given SS and/or VE, LPS, LPS + SS + VE to rats compared with the control group after 6 h. In addition, LPS + SS and/or VE-treated group was compared with LPS-treated group for MDA level and SOD, CAT, GPx, and GST activities. Biochemical changes were shown in Figure 1.

(a) MDA and (b–e) antioxidant enzymes values in the kidney of rats treated with LPS (10 mg/kg b.w.), VE (200 mg/kg b.w.), and/or SS (0.35 mg/kg b.w.), with n = 7 in each group. Significance at p < 0.05. MDA: malondialdehyde; LPS: lipopolysaccharide; VE: vitamin E; SS: sodium selenite; SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase; GST: glutathione-S-transferase; b.w.: body weight.
There was no statistically significant change in SS, VE, and SS + VE-treated groups compared with the control group at the end of 6 h.
LPS caused a significant increase in the MDA level and decrease in SOD, CAT, GPx, and GST activities of kidney tissue for the LPS treatment group. A significant decrease in the MDA level and increase in antioxidant enzyme activities were observed in the LPS + VE, LPS + SS, LPS + VE + SS treatment groups. A significant (p < 0.05) alteration in biochemical results was observed in the LPS-treated groups when the LPS group and the LPS + VE, LPS + SS, LPS + VE + SS group were compared with kidney cells of rats.
Histopathology and immunohistochemistry
No pathological findings were found in the histological sections of the control group kidneys and SS and/or VE-treated rats. Glomerulus and Bowman’s capsule, where the blood is filtered, were seen in the normal structure. No degeneration was found in the proximal and distal tubules in kidney of rats (Figure 2).

Kidney sections of rats in the control group (n = 7), ×200, H & E. G: glomerulus, P: proximal tubule, D: distal tubule; H & E: hematoxylin–eosin.
Six hours after LPS treatment, hemorrhage, tubular degeneration, inflammatory cell infiltration, glomerular atrophy, glomerular lobulation, and dilatation of Bowman’s space were detected in the kidneys of the rats according to the light microscopic examination (Figure 3). However, LPS + VE and/or SS-treated groups displayed hemorrhage, necrosis, and tubular degeneration. The LPS + VE + SS-treated groups showed histopathological change such as tubular degeneration (Figure 4). All of these histopathological changes were scored and summarized in Table 1.

Kidney sections of rats in the LPS treatment groups (a and b), ×200, H & E, with n = 7 in each group. Hemorrhage (↓), tubular degeneration (♦), inflammatory cell infiltration (▀), glomerular atrophy (•), glomerular lobulation (→), dilatation of Bowman’s space (▴). LPS: lipopolysaccharide; H & E: hematoxylin–eosin.
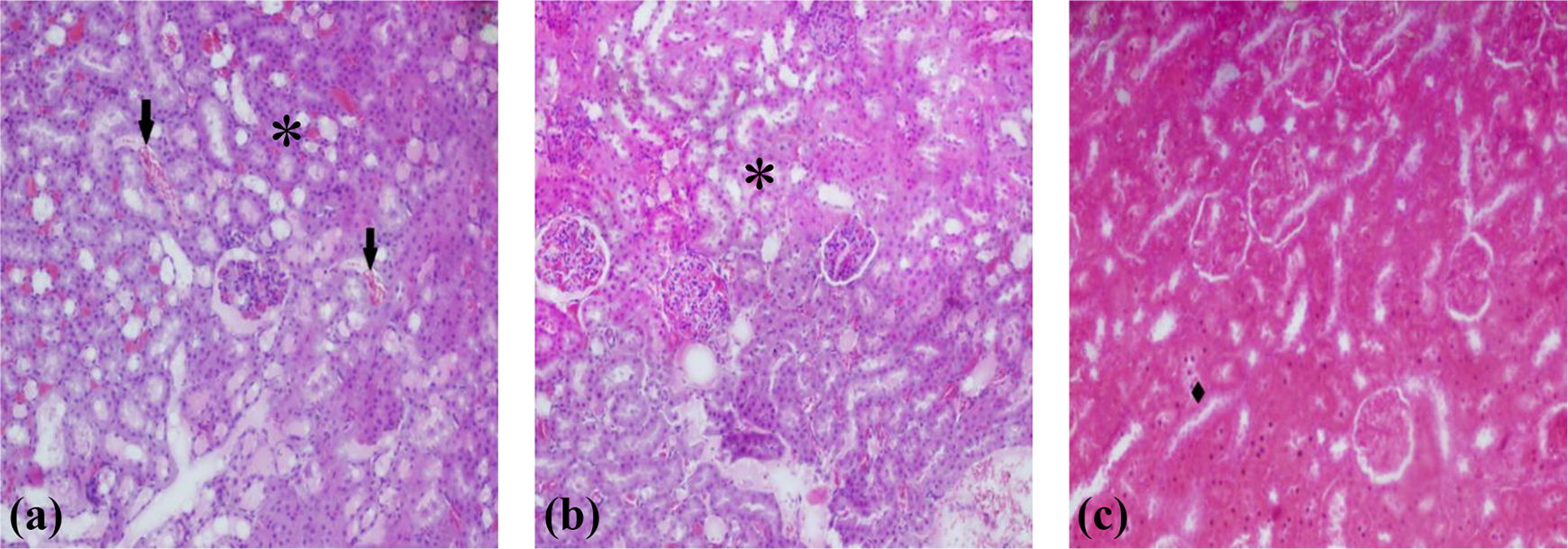
Kidney sections of LPS + SS-treated rats (a), LPS + VE-treated rats (b), LPS + SS + VE-treated rats (c) showing hemorrhage (⇢), necrosis (*), and tubular degeneration (♦), ×200, with n = 7 in each group. LPS: lipopolysaccharide; VE: vitamin E; SS: sodium selenite.
Grading of the histopathological changes in the kidney sections.a

LPS: lipopolysaccharide; VE: vitamin E; SS: sodium selenite.
a The features are scored as follows: none (−), mild (+), moderate (++) and severe (+++), with n = 7 in each group.
In our study, immunohistochemical PCNA method was used to evaluate cell proliferation in kidney tissues of control, LPS, and LPS + SS and/or VE-treated groups. In the kidney of the control group, numerous PCNA-positive cells were observed in the glomerulus Bowman’s capsule and tubule cells. Examination of the kidney tissues of the LPS group revealed that the number of PCNA-positive cells in the kidney’s tubule cells was significantly reduced. Examination of the kidney tissues of the LPS + SS and/or VE-treated group revealed that the number of PCNA-positive cells in the kidney cells was higher than in the LPS-treated group (Figure 5).

Control and all treatment groups showing PCNA protein expression with immunohistochemical analysis in kidneys of male rats: (a) control group, SS-treatment group, VE-treatment group, SS + VE treatment group are similar with each other; (b and c) LPS treatment group; (d) LPS + SS treatment group; (e) LPS + VE treatment group; and (f) LPS + VE + SS treatment group. Single arrow shows PCNA expression with multiple apoptotic cells in the group, ×200, with n = 7 in each group. LPS: lipopolysaccharide; VE: vitamin E; SS: sodium selenite; PCNA: proliferating cell nuclear antigen.
Also PCNA-positive markers were counted, and PCNA index values of the groups were calculated. In our study, PCNA index value decreased in the LPS group compared with the control group, and PCNA index value increased in the LPS + SS and/or VE-treated group, which is statistically significant compared with the LPS group values (Table 2).
Results of the PCNA index and TUNEL assay in control and treatment groups in rats’ kidney.a

LPS: lipopolysaccharide; VE: vitamin E; SS: sodium selenite; PCNA: proliferating cell nuclear antigen.
a Values are expressed as mean ± SD (n = 7 in each group). Superscript letters indicate significant differences among the exposed ones to control and different treatments in kidney tissues of rats. Significance at p < 0.05.
Determination of apoptosis with TUNEL assay
TUNEL staining was performed to determine the number of apoptotic cells in the kidney tissue. It was determined that the apoptotic cells were counted by counting TUNEL-positive cells, and the apoptotic cells in the LPS-treated group increased statistically compared with the control group. It was also determined that the number of apoptotic cells decreased statistically significantly in all three groups when SS, VE, and SS + VE were given for protective purposes after LPS treatment compared with the experimental group (Figure 6; Table 2).

Histological features of apoptotic areas in the kidney of rats. A, B, C → control; D, E, F → SS; G, H, I → VE; J, K, L → SS + VE; M, N, O → LPS; P, R, S → LPS + SS; T, U, V → LPS + VE; X, Y, Z → LPS + SS + VE (n = 7 in each group). LPS: lipopolysaccharide; VE: vitamin E; SS: sodium selenite.
Changes in DNA structure
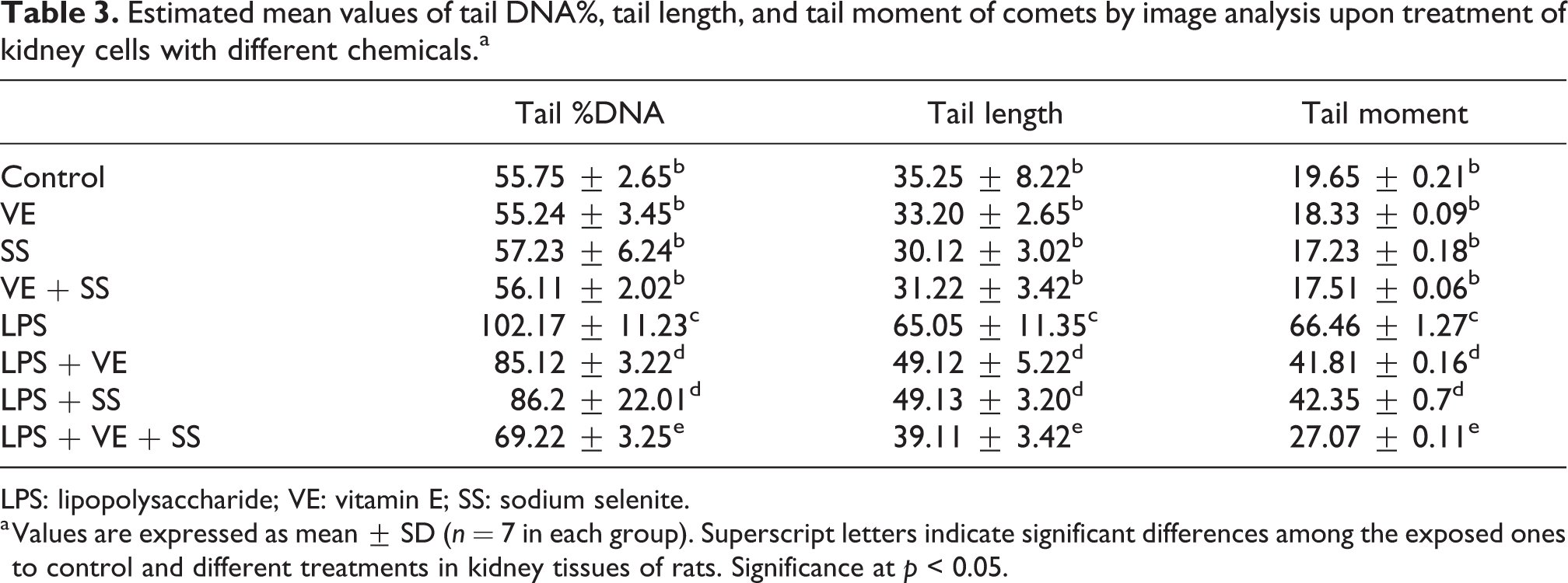
DNA damage was determined with tail %DNA, tail length, and tail moment in the kidney tissue. In the comet assay test, it was determined that no DNA damage occurred in VE, SS, and VE + SS treatment when compared with the control groups (Table 3; Figure 7).
Estimated mean values of tail DNA%, tail length, and tail moment of comets by image analysis upon treatment of kidney cells with different chemicals.a
LPS: lipopolysaccharide; VE: vitamin E; SS: sodium selenite.
a Values are expressed as mean ± SD (n = 7 in each group). Superscript letters indicate significant differences among the exposed ones to control and different treatments in kidney tissues of rats. Significance at p < 0.05.

DNA damage in rat kidney exposed to LPS plus VE and/or SS (a) control, SS, VE and SS + VE treatment groups are similar to each other, (b) LPS treatment group, (c) LPS + SS treatment group, (d) LPS + VE treatment group, and (e) LPS + VE + SS treatment group (n = 7 in each group). LPS: lipopolysaccharide; VE: vitamin E; SS: sodium selenite.
As a result of the application of LPS, it was observed that primary DNA damage occurred and detected with the comet test. DNA damage is statistically significant in all applications compared with the control. LPS application significantly increased tail %DNA and tail length in the comet tail in kidney cells. Especially a twofold increase was observed compared with the control group at a dose of 10 mg/kg (Table 3; Figure 7).
DNA damage was decreased in the kidney tissue of SS and/or VE treatment. LPS + SS-, LPS + VE-, LPS + SS + VE treatment groups in the kidney tissue caused a significant decrease in DNA damage for 6 h as indicated by a greater migration of DNA fragments on the agarose gel (Figure 7; Table 3).
The results of the comet assay showed an increase of 85.12 ± 3.22 and 49.12 ± 5.22 μm in the mean tail %DNA and tail length, respectively, after LPS + VE and an increase of 86.2 ± 22.01 and 49.13 ± 3.20 μm, respectively, after LPS + SS treatment when compared with the LPS treatment group (Table 3), whereas the mean tail %DNA and tail length values ranged from 69.22 ± 3.25 to 39.11 ± 3.42 in the LPS + VE + SS treatment group (p < 0.05; Table 3).
Discussion
LPS, a component of the cell wall of gram-negative bacteria, is widely used to produce experimental endotoxemia in laboratory animals. Although LPS has been obtained from many gram-negative bacteria such as Escherichia coli, Salmonella typhimurium, Klebsiella pneumoniae, and Pseudomonas aeruginosa, septic shock studies concentrated on LPS from E. coli. LPS types available include O26: B6, O55: B5, O111: B4 in different serotypes. Metabolic, immunological, physiological, toxicological, and pharmacological studies are carried out concerning the septic shock with LPS. When the literature is examined, it can be observed that the dose to be administered has a wide range of doses between 1 mg/kg and 80–100 mg/kg. 16,18 In this study, LPS was used from E. coli with O55: B5 serotypes and was i.p. given 10 mg/kg b.w. SS and/or VE; then, pathological changes, MDA level, antioxidant enzyme activities, apoptosis, and DNA structure were evaluated in the rat kidney after 6 h; however, none of the rats died during the experimental period.
Redox imbalance and oxidative stress observed during sepsis which thus leading to organ damage and endothelial dysfunction act as important therapeutic targets in sepsis research. In a previous study on patients, at critical level, the results indicated that increased lipid peroxidation was relative to the control group. 29 Antioxidant conditions in patients with septic shock decreased plasma concentrations of retinol (vitamin A), VE, beta-carotene, and lycopene; the decreased levels were reported to be detected in these patients compared to the healthy subjects. 30 Galley et al. reported increased concentrations of redox-reactive iron in patients with septic patients and reported that when vitamin C levels decreased in plasma levels, lipid peroxidation was associated with this increase. 31 MDA is the end product of lipid peroxidation resulting from the interaction between cells and intracellular membranes and ROS. 32 MDA formed by peroxidation causes cross-linking and polymerization of membrane components. This changes intrinsic membrane properties such as deformation, ion transport, enzyme activity, and aggregation of cell surface components. 15 Because of this property, MDA is genotoxic and carcinogenic for mutagenic cell cultures. 33 SOD is an antioxidant enzyme that has great regulation for the regulation of free radical-mediated processes in biological systems. 34 SOD reduces H2O2 to a superoxide by giving electron from the ROS, while CAT and Se-dependent GPx degrade H2O2. 35 Antioxidants capture both ROS and provide protection in the cell provides protection by blocking lipid peroxidation in the cell membrane. At the same time, it blocks signal transduction of inflammatory cytokines and endotoxin or modifies it in the early period of the inflammatory response, thereby regulating cellular activation. 36 In this study, the rats were treated with LPS for 6 h. When LPS was given, LPS-administered group showed a statistically significant increase in MDA level and decrease in SOD, CAT, GPx, and GST enzyme activities compared with the control group at the end of the treatment period. When the antioxidant enzyme activities and MDA levels of LPS + SS + VE treatment group were examined, it was found that there was a statistically significant increase in SOD, CAT, GPx, and GST activities and a significant decrease in MDA level in this group compared with the LPS, LPS + SS, and/or VE applied groups.
LPS causes histopathological and cytopathological changes in mammals and other living tissues. The target organs affected by LPS are the organs such as kidney, 37 liver, 33 and lungs. 38 Many studies have shown that LPS affect the kidney. 37,39 In one study, it was reported renal tubular vacuolar changes, including degeneration and necrosis of tubular epithelial cell and diaphanous tubular cast, were observed in the kidney of rats treated with LPS, as a result of the examination under light microscopy. 37 In this work, LPS was administered i.p. to rats for 6 h, and the effects of this sepsis material on kidney tissue were investigated. When kidney tissues of LPS-treated rats were examined under light microscopy, hemorrhage, inflammatory cell infiltration, tubular degeneration, glomerular atrophy, glomerular lobulation, and dilatation of Bowman’s space kidney cells were determined. Histopathologically, less pathological changes were observed in LPS + SS, LPS + VE, and LPS + SS + VE treatment groups compared with the LPS group.
Although the cause of the sepsis event is the microorganisms or toxins, the immune response of the organism results in widespread inflammation, multiple organ dysfunction, and organ failure. LPS entering the organism is first bound to the LPS-binding protein. There is an extreme increase in the production of inflammatory cytokines, Hsp proteins, ROS, and reactive nitrogen species (NO·, O2·, ONOO·, OH·, and H2O2) in many cell types. 40,41 Sepsis is responsible for organ dysfunction and death, cell hypoxia, and apoptosis. 33 Apoptosis plays an important role in the level of sepsis parenchymal tissue. Apoptosis was detected in lung, liver, kidney, and intestinal structures. 42 Immunohistochemical detection of PCNA demonstrates DNA damage resulting in both active and carcinogenesis. Its expression can be used as a marker of cell proliferation. However, when compared to other proliferation markers, they give higher results due to their properties. 43 In this study, PCNA expression was evaluated in the control and treatment groups. PCNA expression in the LPS treatment group was found lower than in the non-treatment group. LPS + VE, LPS + SS, and LPS + VE + SS groups have a stronger PCNA expression than the LPS group.
Apoptosis is demonstrated by routine histological techniques. In addition, TUNEL dyeing technique, which is more sensitive than detecting apoptosis, has been used. TUNEL staining technique mainly shows DNA fragmentation as an indicator of apoptosis. The free 3′-OH ends of the DNA fragments are labeled with diaminobenzidine. The marked areas are counted, and the amount of apoptosis is determined. In vitro studies and animal models have shown that upon LPS treatment, LPS-induced kidney cell apoptosis is increasing. 39 TUNEL painting images detected in this study are compatible with the findings of Wen and Wan who used the same technique. 44,45 However, renal cell apoptosis was significantly suppressed with SS and/or VE. The results showed that treatment of SS and/or VE had effects to protect kidney cell apoptosis induced by sepsis.
In general terms, free radicals are molecules that contain an oxygen atom that has lost an electron. This causes these molecules to become unstable and reactive. That is, these molecules show a high affinity to the electrons of other molecules around them. For example, these molecules interact with DNA, leading to dysfunction, mutation, and cancer. 46 Free oxygen groups act on DNA, causing structural changes in nucleic acids, resulting in mutations with changes in chromosomes. DNA damage and mutations come into play with this event. 47 –49 ROS-induced DNA damage causes single- or double-stranded DNA damage; purine, pyrimidine, or deoxyribose modifications; and DNA cross-linking. 50 In addition, DNA damage may result in the arrest or induction of transcription, induction of signal transduction pathways, replication defects, and genomic instability associated with carcinogenesis. 51,52 In this work, when the LPS group and the control group were compared at the end of 6 h, it was observed that the rats in the LPS-treated group had a significant increase in the tail %DNA, tail length, and tail moment in the kidney tissues. Protective effects of nonenzymatic antioxidants, SS and VE, against LPS-induced toxic effects on the kidney were investigated.
Conclusions
The high rate of sepsis mortality, the increase in the number of cases every year, and the failure results of preclinical/clinical research continue to challenge modern medicine. There are few proven agents in addition to antimicrobial and supportive treatments in sepsis. There is an urgent need for research about sepsis, which is a systemic inflammatory response syndrome to decrease the mortality rates over the available borders. Since the physiopathology of sepsis is rather complex, the extraordinary developments in molecular biology have also been reflected in the area of sepsis, and many points in this complex process have become more understandable. In this study, VE and/or SS were reduced significantly with LPS-induced nephrotoxicity in rats, but not completely protected. Despite this, more studies should be done to find the most preventive effect of antioxidants or their combinations for septic infections and its damages, so that the deaths should be reduced.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
