Abstract
Background:
Dexamethasone (DEX) is a common medicine that is capable of causing malformation in the male reproductive system. The aim of this study was to investigate the effect of vitamin C (Vit-C) on spermatogenesis indexes and daily sperm production (DSP) in adult mice treated with DEX.
Methods:
Male Naval Medical Research Institute (NMRI) mice were divided into four groups: Control, DEX (7 mg/kg/day), Vit-C (100 mg/kg/day), and DEX +Vit-C and treated for 7 days with intraperitoneal injection.
Results:
A significant increase in the mean levels of serum and tissue malondialdehyde (MDA) and apoptosis of Leydig cells was found in the DEX group compared to the control group. Sperm motility, DSP, tubular differentiation index, meiotic index, spermatogenesis index, the mean number of spermatocytes, round and long spermatids, and Leydig cells, and also serum testosterone level decreased in the DEX group compared to the control group. The results of this study indicate that Vit-C can significantly prevent the adverse effects of DEX on the mean number of spermatocyte, spermatid, and Leydig cells, tubular differentiation, meiotic and spermatogenesis index, DSP, sperm motility, and the mean levels of serum MDA.
Conclusion:
In conclusion, our results showed that coadministration of Vit-C and DEX prevents the adverse effects of DEX on the spermatogenesis indexes and DSP.
Introduction
Spermatogenesis is an extraordinarily complex process that is sustained by the balance in germ cell proliferation, differentiation, and death in the testes. 1 Several factors such as drug treatment, toxins, and environmental pollutants have adverse effects on spermatogenesis. 2 Dexamethasone (DEX) is a synthetic glucocorticoid, which has been prescribed increasingly in recent years. 3,4 It is commonly used in clinical situations, namely chronic asthma, rheumatoid arthritis, autoimmune diseases, and prevention of graft rejection as a potent anti-inflammatory, immunosuppressive, and analgesic agent. 5,6 DEX produces intracellular reactive oxygen species (ROS) in many tissues and organs including the kidney, liver, 7 vascular endothelial cells, and neural stem cells, which in turn causes lipid peroxidation, 5 decreases daily sperm production (DSP), 8 disturbs sperm motility, 9 and inhibits testosterone production through influencing the anterior pituitary gonadal axis and the testes 4 leading to testicular tissue damage. 10 It also induces Fas/FasL signaling pathway in the Leydig and germ cells causing apoptosis. 11 Conversely, it has been shown that antioxidants can protect against the toxicity created by drugs. Vitamin C (Vit-C), a well-known natural antioxidant, is an essential water-soluble micronutrient required for an array of biological functions and is associated with semen quality and fertility in humans. This vitamin neutralizes free radicals which prevents the adverse effects of ROS on spermatogenesis and protects the sperm. 12 The desired effect of Vit-C on the quantity and quality of spermatozoa 13,14 and also its role in increasing the testosterone level 12 have been reported in previous studies. Therefore, the present study was designed to investigate the protective effects of Vit-C against changes in testicular morphology, undesired alterations in spermatogenic indices, and biochemical parameters in adult mice following exposure to dexamethasone.
Methods
The study was performed on 24 healthy adult male Naval Medical Research Institute (NMRI) mice (6 weeks old) weighting 35 ± 2 g. Mice were obtained from animal facility of Pasture Institute of Iran. All animals were kept under the same environmental conditions regarding diet and water, light (12 h), and temperature (21–23 °C) in accordance with the Principles of Laboratory Animal Care. The animals were randomly divided into four groups (n = 6): Control (received 2 ml normal saline), DEX (7 mg/kg/day i.p. Sigma-Aldrich St. Louis, Missouri, Germany; for each mice, 0.25 mg of DEX powder was solved in 2 ml normal saline), Vit-C (100 mg/kg/day i.p. Sigma; for each mice, 3.5 mg of Vit-C powder was solved in 2 ml normal saline), and DEX + Vit-C group. Treatment was carried out for 7 days in all groups. The DEX dose was selected on the basis of Orazizade et al. study that demonstrated 7 mg/kg/day of DEX treatment for 7 days induces toxicity in the testis tissue. 6 The given dose of vitamin C was also selected based on the study by Ayyildiz et al. who surveyed the effect of six different doses of ascorbic acid (25, 50, 100, 200, 400, or 800 mg/kg) and found that 100 mg/kg was the most effective dose. 15
Surgical procedure
About 24 h after the last injection, mice were weighed and killed following ether anesthesia. Their left caudal epididymis was cut to evaluate sperm parameters. Blood samples were also collected from the animal’s hearts to assess the serum levels of malondialdehyde (MDA) and testosterone. Testes were carefully removed from each animal and weighed. 16 The right testes were used for histopathological study, for this purpose, the immersion volume was calculated using the immersion method and then the testes were fixed in 10% formalin for 1 week. 17 The left testes were stored at −80°C to evaluate the tissue level of MDA and DSP. 17
Estimation of sperm motility and sperm tail length
Excised caudal epididymis was placed in a small clean petri dish containing 1/5 ml of Ham’s F10 culture medium. Then epididymis was cut by a sharp sterile blade into pieces to release the sperms, the petri dish was then incubated at 37°C for 5 min. The released sperms were used to assay sperm motility and tail length of sperms. For the analysis of sperm motility, 10 μl of the sperm suspension was placed on a Neubauer Chamber and examined under light microscope (Olympus BH-2, Japan) with 200× magnification. Each sperm was classified either as progressive motile sperm (PMS), nonprogressive motile sperm (NPMS), or nonmotile sperm (NMS) and also the percentage of motile and immotile sperms was calculated. 18
To estimate sperm tail length, the sperm suspension was stained with eosin-nigrosin, one drop of it was smeared on a glass slide and then observed under light microscopy (Olympus BX41TE, Japan) equipped with camera (Olympus DP12, Japan)with 400× magnification, and 200 sperms were counted using a test grid.
The test grid containing two elements was superimposed on the monitor image. The first element is the unbiased counting frame. If a sperm’s head lay inside the frame and did not touch the forbidden lines (left and inferior borders of the frame), it was sampled. Another element is Merz grid that is a curve consisting of two equal semicircles.
To estimate the average sperm tail length, the following formula was used. 19
Where “a/l” is the Merz grid constant, which is obtained by multiplying the area of each basic tile of the grid by the length of semicircles. Within this tile, there are two semicircles of length of π.d (perimeter of a circle); where “d” is the diameter of the semicircle, “asf” is the area of the basic tile divided by the area of the counting frame, and “ΣI” is the assembled intersections of the tails with the semicircles. “ΣN” is the total number of the counted sperm in the unbiased counting frame in all fields.
Stereological study
The orientator method was used to obtain isotropic uniform random (IUR) sections. 19 For this purpose, the testis tissue was randomly placed on the ϕ-clock which each half of it was divided into nine equal parts. A random number between 0 and 9 was selected; an appropriate cut was made along the selected number. One of the cut surfaces was then placed vertically on the θ-clock which each half of it was divided into nine unequal parts. Then the second cut was made by selecting a random number and the cut was made along that number. The cut surface of the other half of the testis was also placed parallel to the 0–0 direction of the θ-clock and then again, the random number was selected and the cut was made along the selected number. Indeed, the tissues were cut into 9–11 slices. Three circle segments were randomly prepared from the testis tissue slabs using a trocar. The two vertical diameters of each segment were measured and their mean radius was estimated and considered as prefixing radius (before). After tissue processing and embedding, 5 and 20 µm thick sections were prepared on a rotary microtome (Leitz 1512, Germany) and mounted on slides, then stained using Heidenhain Azan method.
Estimating the shrinkage and the total volume of the testis
After tissue processing and staining, the trocar radiuses were again measured and the obtained mean radius was considered as the post-fixing radius (r after), and the amount of shrinkage in each testis was estimated using the following equation. 16
To obtain the total volume of the testis, the amount of shrinkage was subtracted from the testis volume estimated by the immersion method.
Estimating the number of spermatogenic, Sertoli, and Leydig cells
To estimate the number of cells, the optical dissector method and an unbiased counting frame were used. 20
Twenty micrometer thick sections were observed with 100× oil immersion objective on a microscope (Olympus BX41TE) that was equipped with a microcator (ND 221 B, Heidenhain, Germany) and camera (Olympus DP12). At each microscopic field, 5 µm from the top and bottom of the sections was ignored as a guard area against the cutting artifacts (depth of counting field was 10 µm).
The numerical density (defined as the number of cells per unit volume of the testis tissue, Nv ) of different types of cells was estimated as:
in which ΣQ denotes the number of counted cells, “h” is the tissue thickness considered for counting (the disector height), ΣP is the total number of points superimposed on the selected fields, and a/f is the area of the frame in the true tissue scale.
The total number of each kind of cells per testis (N total) was calculated by multiplying the total volume of the testis (V) in the numerical density of these cells (N v):
Spermatogenesis indexes
In total, 100 round seminiferous tubule cross sections in each testis tissue were randomly selected under a light microscope. All spermatogenic and Sertoli cells were counted and spermatogenesis indexes including tubule differentiation index (TDI), spermiogenesis index (SPI), meiotic index(MI), and Sertoli cell index(SCI) were calculated. TDI is the percentage of seminiferous tubules containing at least three differentiated germ cells from the B spermatogonia stage, 21 SPI is the percentage of tubules that obtain the overall rate of spermatogenesis, 22 MI is the ratio of the number of round spermatids to spermatocytes, 23 and SCI is the ratio of the total number of spermatogonia and spermatocytes to the number of Sertoli cells. 24
DSP estimation
The left testis was decapsulated and the parenchyma was homogenized in 2 ml buffer (0.9% NaCl and 0.05% tritonX-100) for 4 min. Spermatid cells in each sample were counted. The testicular spermatid number (TSN) per gram testis was calculated. DSP was determined by dividing the TSN by 4.84 (the duration of steps 14–16 spermatids in the mouse seminiferous epithelial cycle). 25
Assessment of the apoptosis rate using Terminal deoxynucleotidyl transferase (TdT) dUTP Nick-End Labeling (TUNEL) assay
Fragmentation of the DNA in histological sections was assessed using TUNEL assay, according to the kit instructions (in situ Cell Death Detection Kit, Roche Pharm AG, Grenzach-Wyhlen, Germany).
Five micrometer thick sections were deparaffinized and rehydrated. Endogenous peroxidase activity was neutralized with 0.3% hydrogen peroxide (H2O2; Merck KGaA, Darmstadt, Germany) in methanol for 10 min. Digestion with 20 μg/ml proteinase K for 30 min at 37°C was carried out and the sections were then incubated with the reaction factor at 37°C for 60 min in a humidified dark chamber, followed by incubation with Converter-POD for 30 min at 37°C. For the negative control, a series of tissue sections were processed in an identical manner, except that they were incubated in the reaction buffer without the presence of TDT. The samples were stained with diaminobenzidine substrate (Roche) for 10 min, and washed with distilled water and PBS, and then counterstained with Harris’ hematoxylin and dehydrated in 95% and 100% ethanol, cleared in xylene, and mounted with Permount Entellan (Merck).
Measurement of serum MDA level
To determine the level of MDA, a TCA-TBA-HCL solution (2 ml, 12% w/v trichloroacetic acid, 0.375% w/v thiobarbituric acid, and 0.25 mol/l hydrochloric acid) was prepared. Then, one volume of the sample was mixed with two volumes of the TCA-TBA-HCL solution and the samples were placed in bain-marie for 15 min. After removing the samples from the bain-marie, they were quickly cooled using cold water and centrifuged for 10 min. The supernatant was carefully removed and its absorbance was measured at 532 nm. The concentration of MDA was calculated using its extinction coefficient, which was 1.57 × 10 M−1 cm−1, expressed in nmol/ml. 26
Evaluation of testes MDA level
The testes tissue was lysed in KCl and added to the stock solution (2 ml, 12% w/v trichloroacetic acid, 0.375% w/v thiobarbituric acid, and 0.25 mol/l hydrochloric acid) and the tissue MDA was quantified using the thiobarbituric acid reactive substances (TBARS) assay and expressed as micromole per grams of tissue samples. 16
Serum testosterone determination
The serum testosterone level was evaluated with the testosterone ELISA kit (Monobind Testosterone ELISA kit, EIA-37K5J5 Monobind Inc, Lake Forest, California, USA) according to the manufacturer’s instructions.
Statistical analysis
The results were analyzed by one-way analysis of variance and Tukey’s test, using the Statistical Package for Social Sciences (SPSS) version 16/0 (SPSS Inc., Chicago, Illinois, USA), and the means were considered significantly different at p < 0.05.
Results
Histopathological findings
Normal structure of the testis tissue and germinal epithelium was observed in the control group (Figure 1(a)). In the DEX group, no abnormal appearance was seen in the testis after treatment but irregular and vacuolated seminiferous tubules and a less compact arrangement of spermatogenic cells was observed in the structure of the testis (Figure 1(b)). Meanwhile, the DEX + Vit-C group presented similar tissue structure to the control group with an increase in the height of the germinal epithelium (Figure 1(c)). Moreover, the Vit-C group showed regular seminiferous tubules similar to the control group.

Micrographs of testis tissue in different groups of mice (5 μm-thick sections, stained with Heidenhain Azan method and magnification of 200×). (a) Control group: Normal structure of the testis tissue. (b) DEX group: Featuring irregular (circle) and vacuolated seminiferous tubules (arrows) and a less compact arrangement of spermatogenic cells. (c) DEX + Vit-C group: Presenting similar tissue structure to the control group with an increase in the height of germinal epithelium. Vit-C group (not shown): Normal structure of the testis tissue. DEX: dexamethasone; Vit-C: vitamin C.
Conversely, apoptotic Leydig cells were observed in the DEX group (Figure 2(b)), but in the other groups, apoptotic cells were not observed (Figure 2(a) and (c)).

Micrographs of testis tissue in different groups of mice (5-μm thick sections, stained with TUNEL staining, magnification of ×400). (a) Control group: Apoptotic cells were not observed. (b) DEX group: Apoptotic Leydig cells were apparent. TUNEL-positive cells (dark brown nucleus) are marked (arrows). (c) DEX + Vit-C group: apoptotic cells were not observed. Vitamin C group (not shown): Apoptotic cells were not observed. DEX: dexamethasone; Vit-C: vitamin C
Number of spermatogenic, Sertoli, and Leydig cells
A significant reduction in the mean number of spermatocyte, long and round spermatid, and Leydig cells was observed in the DEX group when compared to the control group (p < 0.001). Meanwhile, the number of long and round spermatid cells in the DEX + Vit-C treated group was similar to the control group (Table 1).
Comparing the number of the spermatogonia, spermatocyte, spermatid, sertoli, and Leydig cells in different groups of mice after 7 days of treatment.a
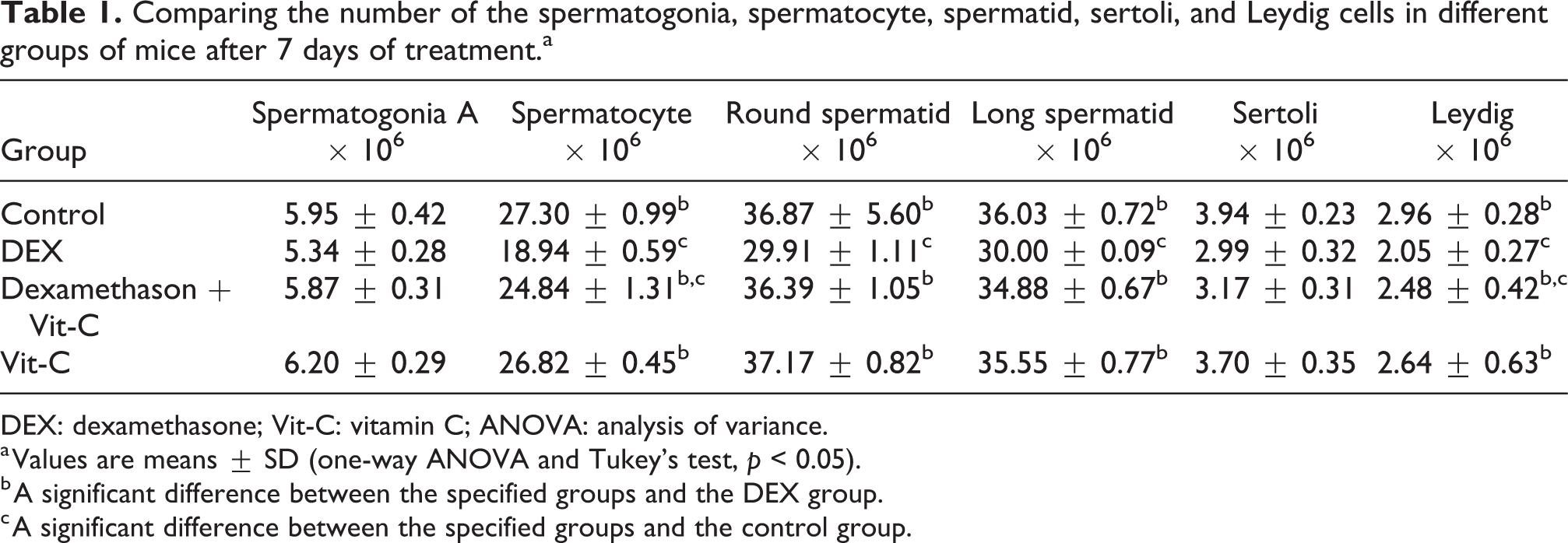
DEX: dexamethasone; Vit-C: vitamin C; ANOVA: analysis of variance.
a Values are means ± SD (one-way ANOVA and Tukey’s test, p < 0.05).
b A significant difference between the specified groups and the DEX group.
c A significant difference between the specified groups and the control group.
Spermatogenesis indexes
A significant reduction in the TDI, SPI, and MI in the DEX group was found when compared to the control group. The DEX + Vit-C group did not show a significant difference in the mentioned parameters when compared to the control group (p > 0.05) (Table 2).
The TDI, SPI, SCI, MI, and the motility of sperm in different groups of mice after 7 days of treatment.a

TDI: tubule differentiation index; SPI: spermatogenesis index; SCI: sertoli cell index; MI: meiotic index; DEX: dexamethasone; Vit-C: vitamin C; ANOVA: analysis of variance; PMS: progressive motile sperm; NPMS: nonprogressive motile sperm; NMS: nonmotile sperm.
a Values are means ± SD (one-way ANOVA and Tukey’s test, p < 0.05).
b A significant difference between the specified groups and the DEX group.
c A significant difference between the specified groups and the control group.
Sperm motility
A significant decrease in the mean percentage of PMS (p < 0.001) and a significant increase in the percentage of NPMS (p < 0.01) and NMS (p < 0.001) was found in the DEX group when compared to the control group. The mean number of PMS and NMS was similar in the DEX + Vit-C group when compared to the control group (Table 2).
Daily sperm production
The DSP in the DEX group was significantly lower than the control (p < 0.02), but in the DEX + Vit-C group, no significant difference was found when compared to the control (Table 3).
Comparison of the daily sperm production, sperm tail length, serum testosterone level, and MDA level in different groups of mice after 7 days of treatment.a

DEX: dexamethasone; Vit-C: vitamin C; ANOVA: analysis of variance; MDA: malondialdehyde.
a Values are means ± SD (one-way ANOVA and Tukey’s test, p < 0.05).
b A significant difference between the specified groups and the DEX group.
c A significant difference between the specified groups and the control group.
Sperm tail length
The mean sperm tail length showed no significant difference in all four groups (Table 3).
Serum testosterone level
Serum testosterone level reduced significantly in the DEX group (p < 0.02) compared to the control, whereas it showed an increase in the DEX + Vit-C group when compared to the DEX group, but this increase was not significant (Table 3).
Malondialdehyde concentration
The concentrations of serum and testes MDA were significantly higher in the DEX group compared to the control group, while in the DEX + Vit-C group, it reduced significantly compared to the DEX group (Table 3).
Body and testis weight
No significant difference (p > 0.05) was observed in the mean body and testis weights of mice in all experimental groups (Table 4).
The mean body and testis weight (g) in different groups of mice after 7 days of treatment.a

DEX: dexamethasone; Vit-C: vitamin C; ANOVA: analysis of variance.
a Values are means ± SD (one-way ANOVA and Tukey’s test, p < 0.05).
Discussion
As our data showed, treatment with DEX in adult mice leads to apoptosis in Leydig cells and reduction in the mean number of spermatocytes, spermatids (long and round) and Leydig cells, TDI, SPI, MI, and DSP, which is in accordance with other studies. 4,9,27,28
DEX induces cell apoptosis 6, 11 through the activation of the Fas/FasL pathway, activation of the caspases, mitochondrial depolarization of the cells, and also the activation of pre-apoptotic proteins such as Bax. On the other hand, DEX increases the formation of free radicals such as ROS inducing a cellular redox imbalance and oxidative stress, which in turn disturbs cell membrane through lipid peroxidation leading to cell damage and disrupted cell–cell interactions among testicular cells. 29
Our data revealed a significant decrease in sperm motility following treatment with DEX compared to the control group. Other studies have also indicated that treatment with DEX decreases sperm motility, 4,9 which is mainly related to the sperm tail length and to the ATP concentration in the mid-piece. As, in this study, no significant decrease in the sperm tail length was found in the DEX group; therefore, this decrease in sperm motility may be due to the interruption in ATP production as a result of oxidative stress, thus, affecting sperm motility. 30 Previous reports have also shown that the main target of DEX is the mitochondria, in fact, Mutsaers and Tofighi reported that DEX treatment downregulates 72% of the genes involved in the mitochondrial respiratory chain, causing a reduction in ATP concentration and therefore decreasing sperm motility. 31 We also showed that DEX reduces serum levels of testosterone, which has been reported previously by other studies 9,28 which demonstrated that DEX causes a significant decrease in the transcription of genes encoding enzymes responsible for the biosynthesis of testosterone such as P450SCC (p450 side-chain cleavage), 17B-HSD (17B-hydroxyl steroid dehydrogenase), 3B-HSD (3B- hydroxyl steroid dehydrogenase), and P45017α (cytochrome P450-450-dependent 17α-hydroxylase). 32
DEX has also been shown to repress cholesterol transporting protein steroidogenic acute regulatory protein (StAR) gene 28, 33 in the Leydig cells decreasing testosterone biosynthesis. Meanwhile, other studies have reported that DEX can inhibit the secretion of luteinizing hormone (LH), 28 which consequently leads to the suppression of testosterone production. This reduction in the testosterone level can lead to the inactivation of kinases responsible for the regulation of processes required to maintain spermatogenesis and can also prevent cell division in the androgen-dependent stage of spermatogenesis causing a reduction in the number of cells and the DSP. 34 Furthermore, as mentioned earlier, DEX decreased the mean number of Leydig cells, which can in turn lead to a reduction in the serum testosterone level and spermatogenesis suppression.
MDA is a lipid peroxidation product which is usually used to evaluate oxidative stress. The ROS-induced injury normally causes an increase in the MDA level. 35 In the present study, the MDA level in the serum and the testis were significantly higher in the DEX group compared to the control group, which is consistent with previous studies. This is probably due to an increase in lipid peroxidation level as a result of ROS production and the reduction in the level of antioxidant enzymes following treatment with DEX. 27
There were no significant differences in the mean body and testis weight of mice in the DEX group compared to the control. These results are in agreement with previous studies. 8,9 However, some studies have indicated that DEX can reduce body weight. 7,36 This conflict can possibly be due to the duration of treatment or the selected dose of DEX.
In this study, we demonstrated that co-treatment of mice with DEX and Vit-C could prevent the adverse effects of DEX and compensate for the reduction in the mean number of spermatocytes, round and long spermatids, Leydig cells, DSP, TDI, MI, SPI, sperm motility, and serum testosterone levels and could reduce the mean levels of serum and tissue malondialdehyde. Vit-C can increase glutathione (GSH) concentration and the activity of testicular antioxidant enzymes such as catalase and superoxide dismutase that are essential for sperm survival by reducing the injuries resulting from oxidative stress. 13,14 On the other hand, Vit-C can increase the activities of testicular 3b-HSD and 17b-HSD enzymes, 37 which increase testosterone production and thus the number of spermatogenic cells and the DSP. Vitamin C is also an important cofactor for the hydroxylation of collagens that cause cell–cell interactions between testicular cells, which plays a key role in spermatogenesis, 12 improving the differentiation of these cells and finally increasing the number of cells and DSP.
Conclusion
The results of the present study indicate that DEX treatment in mice induces testicular injury as is evident from the undesired changes in the number of spermatogenic cells, spermatogenesis indexes, DSP, sperm motility, and MDA and testosterone levels. Vit-C, as a strong antioxidant, can ameliorate all the adverse effects of DEX treatment on the testis structure and therefore its application in therapeutic regimens including DEX is suggested.
Footnotes
Acknowledgments
The authors wish to thank Miss Samira Naderi and Mr Mehdi Farahani for their Technical assistance and also the Arak University for its financial support during this investigation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Arak University provided financial support for this research.




