Abstract
The effects of X-ray radiation on spermatogenesis, sperm motility, and PIWI-interacting RNAs (piRNAs) in mice were analyzed. Male C57BL/6 J mice were divided into control and two irradiation groups (n = 9 mice/group). After irradiation of their reproductive regions, the mice were fed for 3 days (irradiation group 1) or 7 days (control and irradiation group 2). The sperm viability, motility, velocity, and motion curve were analyzed. After piRNA expression profiling, quantitative reverse-transcription polymerase chain reaction was conducted for validation. Ionizing radiation led to vessel dilation and congestion, fewer spermatogenic cells, and reduced sperm production compared to the control. At 3 and 7 days postirradiation, the sperm count (grade d) increased while sperm viability and sperm lateral head displacement decreased. At 7 days, the sperm abnormality rate was higher compared to the control. Many piRNAs were differentially expressed after irradiation, including decreased and increased expression of mmu_piR_009082 and mmu_piR_020217, respectively. Downregulated piRNAs were involved in Rap1 signaling, non-homologous end-joining, hedgehog signaling, oxytocin signaling, and cholinergic synapse. Upregulated piRNAs participated in pathways including proteoglycans in cancer, phosphatidylinositol signaling, cGMP-PKG signaling, and stem cell pluripotency regulation. X-ray irradiation inhibited spermatogenesis and increased abnormal sperm rate in mice. piRNA-related signaling pathways may be involved in this process.
Keywords
Introduction
Ionizing irradiation can induce somatic and genetic effects in exposed parents and their offspring. 1 The gonads are radiosensitive, and an overdose of radiation causes reproductive dysfunctions such as infertility and chromosomal aberrations. 2 Ionizing radiation greatly alters the count, morphology, and chromosomal structure of sperm. In healthy humans, in vitro X-ray, β-ray, and γ-ray irradiation may cause chromosomal aberrations including exchange-type, chromatid-type, and haplotype changes. The frequencies of these abnormalities are positively correlated with the radiation dose. Additionally, ionizing radiation may induce abnormal expression of sperm motility-related proteins and decrease sperm viability. 3 Kumar et al. found that long-term exposure to low-dose radiation decreased the viability and increased the rates of head abnormalities and vacuoles in sperm, despite not significantly changing the concentration of sperm. 4
Although studies have demonstrated radiation-induced abnormalities in the motility, morphology, and count of sperm, the underlying mechanism remains unclear. Recent studies of reproductive system development have highlighted the importance of PIWI-interacting RNAs (piRNAs), which are mainly expressed in germ cells and have gained attention because of their unexplained role in radiation-induced sperm injury. 5,6
In the present study, we examined the effects of X-ray radiation on spermatogenesis and sperm motility and determined the expression profile of piRNAs in male mice. Our results showed that X-ray radiation negatively affected sperm development and that piRNAs may be involved in this process. This study provides insight into the roles of piRNAs in radiation-induced sperm injury.
Methods
Study approval
All animal experiments were approved and conducted in accordance with the guidelines of the ethics committee of Soochow University (number ECSU-201800075).
Animals and radiation
All mice were housed in the animal facility of the Experimental Animal Center in Suzhou University which was maintained at 18–24°C with a 12-h light/12-h dark cycle. Each animal was fed standard mice food and provided tap water ad libitum. Twenty-seven male adult C57BL/6 J mice weighing 20 ± 2 g were randomly divided into a control group and two different irradiation groups (n = 9 mice per group). After 6-MV X-ray irradiation (total dose of 4 Gy) localized to the reproductive regions, the mice were fed for 3 days (irradiation group 1) or 7 days (control group and irradiation group 2). Mice in the control group and radiation groups were executed by euthanasia. The testes and epididymis were collected from six mice in each group. Organs from one side were subjected to pathology analyses, while those from the other side were used for sperm viability and motility assays. The testes and epididymis were collected from the other three mice in each group and used for piRNA profiling.
Sperm viability and motility assay
Semen collection
Mice were killed and the testes were exposed. The testis from one side was dissected and cut into pieces in a pre-warmed centrifuge tube. The supernatant was collected for microscopic examination.
Methods and parameters
The count, viability, motility, velocity, and motion curve of the sperm were analyzed using a WIJY-9000 sperm analysis system (Beijing Weili New Century Science and Tech. Deve. Corp., Ltd., Beijing, China).
Sperm viability and related parameters were as follows: a, b, c, and d represent rapid forward movement, slow forward movement, non-forward movement, and extremely slow or no movement, respectively. The survival rate was calculated as the percentage of viable sperm among all sperm.
Viability parameters were as follows: (1) average path velocity (VAP) was the average velocity of sperm along the trace curve (μm s−1); (2) curvilinear velocity (VCL) was the velocity of the sperm head in the actual path (μm s−1); (3) straight line velocity (VSL) was the linear movement velocity of the sperm head (μm s−1); and (4) beat/cross-frequency (BCF) was the frequency at which the sperm head crossed the movement curve (Hz).
Motion parameters were as follows: (1) linearity (LIN) was the degree of curvature of the sperm movement path (%); (2) straightness (STR) was the straightness of the sperm movement path (%); (3) wobble (WOB) was the degree of wobble in the actual path as compared to the average path (%); (4) amplitude of lateral head displacement (ALH) was the ratio of the amplitude of lateral head displacement between the actual and average motion curves (μm); (5) mean angle of deviation (MAD) was the time average of the instantaneous turning angle of the sperm head along its motion curve (°).
Sperm abnormality analysis
Mice were killed and the testes were exposed. The epididymis from one side was dissected and heated in a 4-mL incubator at 37°C. Tissues were cut into pieces and filtered. The precipitates were fixed in methanol and stained with 2% eosin. A total of 500 sperm were observed by microscopy at 400× magnification and abnormal sperm were counted. The rate of sperm abnormality was calculated as abnormal sperm/total sperm × l00%.
Pathologic analysis of reproductive system
The reproductive organs (testis, epididymis, and seminal vesicle) were fixed in 4% formalin, embedded in paraffin, and stained with hematoxylin–eosin.
piRNA expression profile analysis
piRNA expression analysis was performed by Yunxu Biotech, Inc. (Shanghai, China).
RNA extraction and quality control
RNA was extracted from tissue samples (50–100 mg) using TRIzol® reagent (Life Technologies, Carlsbad, California, USA) according to the manufacturer’s instructions.
The concentration of extracted RNA was measured using a NanoDrop™ ND-1000 spectrometer (Thermo Fisher Scientific, Waltham, Massachusetts, USA). The optical density (OD)260/OD280 ratio was used to measure RNA purity. An OD260/OD280 ratio of 1.8–2.1 was considered to indicate that sample was pure. RNA samples were separated by denaturing agarose gel electrophoresis to detect sample degradation.
Library construction and sequencing
High-throughput sequencing was conducted by CloudSeq Biotech (Shanghai, China). Briefly, the total RNA extracted from each sample was used to prepare a piRNA sequencing library, which involved steps for 3′-adaptor ligation, 5′-adaptor ligation, cDNA synthesis, polymerase chain reaction (PCR) amplification, and size selection of approximately 150-base pair PCR amplicons (corresponding to approximately 30-nt piRNAs).
The libraries were denatured into single-stranded DNA molecules, captured using Illumina flow cells, amplified in situ as clusters, and sequenced for 50 cycles using an Illumina HiSeq sequencer following the manufacturer’s instructions (San Diego, California, USA).
Data analysis
Raw data were generated after sequencing, image analysis, base calling, and quality filtering on an Illumina sequencer. First, Q30 was used to perform quality control analysis. The adaptor sequences were trimmed using cutadapt software (v1.9.3) and the adaptor-trimmed-reads (≥15 nucleotide) were retained. Next, the trimmed reads from all samples were pooled, and Piano software was used to predict novel piRNAs. The trimmed reads were aligned to the merged rat piRNA databases (known piRNAs from piRNABank plus the newly predicted piRNAs) using Novoalign software (v3.02.12) with at most one mismatch. The number of mature piRNA-mapped tags for each piRNA was defined as the raw expression level of that piRNA. Read counts were normalized by the “tag counts per million aligned piRNAs” approach. piRNAs that were differentially expressed between two samples were filtered by their fold-change values. piRNAs that were differentially expressed between two treatment groups were filtered by their fold-change values and p values. To compare two samples, standardized reads were used to calculate the fold-change, p value, and false discovery rate. Differentially expressed piRNAs were selected based on a fold-change ≥ 2.0 and a p value ≤ 0.05.
piRNA-binding transposons and targets were predicted by Miranda software (v3.3a). piRNA-target networks were plotted using Cytoscape software (v2.8.0) and pathway analyses (Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG)) were performed.
Validation of differentially expressed piRNAs
Primers for piRNA
Real-time PCR was performed to detect the expression of 10 piRNAs (mmu_piR_015386, mmu_piR_009082, mmu_piR_038671, mmu_piR_029800, mmu_piR_004455, mmu_piR_020217, mmu_piR_021962, mmu_piR_026790, mmu_piR_017444, and mmu_piR_001851). The primers used are shown in Table 1.
Primers for real-time PCR.

RNA extraction and quality control
The procedures were the same as those described in piRNA expression profile analysis section.
cDNA synthesis
The reverse transcription mixture contained 2-µL deoxynucleotides (2.5 mM each), 2 µL 10× reverse transcription buffer, 0.3-µL specific primers (1 µM), 800-ng total RNA, 0.2-µL MMLV reverse transcriptase (200 U μL−1), 0.3-µL RNase inhibitor (40 U μL−1), and RNase-free water to a total reaction volume of 20 µL. The thermal cycling conditions for reverse transcription were 16°C for 30 min, 42°C for 40 min, and 85°C for 5 min. After the reactions, the samples were placed on ice or preserved at −20°C.
Real-time PCR
The real-time PCR reaction mixtures contained 5 µL 2× master mix, 0.5-µL PCR forward primer (10 µM), 0.5-µL PCR reverse primer (10 µM), and 2-µL RNase-free water. After centrifugation at 5000 r min−1, the reaction mixtures (8 µL) were added into a 384-well PCR plate and cDNA samples (2 µL) were added. The PCR plate was sealed and placed on ice. The thermal cycling parameters were as follows: initial denaturation at 95°C for 10 min, followed by 40 cycles of 95°C for 10 s and 60°C for 60 s. A melting curve was generated by heating in increments of 0.05°C s−1 to 95°C which was held for 10 s, followed by 60°C for 60 s and 95°C for 15 s. The relative expression of piRNA was calculated using the 2−ΔΔCT method.
Statistical analysis
Data were analyzed using SPSS 17.0 software (SPSS, Inc., Chicago, Illinois, USA). The enumeration data were analyzed using the χ2 test with an α value of 0.05 and an α′ value of 0.008. Student’s t test was used to analyze other differences between groups. A value of p < 0.05 was considered statistically significant.
Results
Pathological analysis of the reproductive system
Spermatogenic cells and sperm were observed in the seminiferous tubules of control mice (Figure 1(a) and (b)). At 3 days after irradiation (4 Gy), we observed dilated and congested vessels and decreased spermatogenic cell levels and sperm production (Figure 1(c) and (d)). At 7 days after irradiation, the seminiferous epithelium was thinner along and showed decreased spermatogenic cell levels and sperm production in the seminiferous tubules (Figure 1(e) and (f)).

Hematoxylin-eosin staining of the reproductive system. (a) and (b): Spermatogenic cells and sperm in the seminiferous tubules in control mice were observed at ×100 (a) and ×200 (b) magnification. (c) and (d): Dilated vessels at 3 days post-irradiation (×100); decreased number of spermatogenic cells and sperm production; (e) and (f): decreases in the number of spermatogenic cells and sperm production at 7 days after irradiation were observed under ×100 (e) and ×200 (f) magnification.
Determination of sperm viability and motility
The sperm viability, motility, and count in each treatment group are shown in Figure 2. Compared to the control group, irradiation of the other two groups did not affect sperm of grades a, b, c, and a + b + c; however, the sperm count at grade d was significantly increased while sperm viability was decreased at 3 and 7 days after irradiation (Figure 2(a)). Moreover, irradiation had no effects on the VAP, VCL, VSL, BCF, and MAD at 3 and 7 days after irradiation (Figure 2(b)). The ALH was clearly decreased, while LIN, STR, and WOB remained unchanged (Figure 2(c)). However, the rate of sperm abnormality was increased in mice at 7 days after irradiation (Figure 2(d)).

Effects of ionizing irradiation on sperm viability and motility. (a) Sperm degrees and viability; the sperm count at grade d was significantly increased while sperm viability was decreased at 3 and 7 days following irradiation. (b) Sperm motility; (c) sperm viability; amplitude of lateral head displacement obviously decreased while linearity, straightness, and wobble remained unchanged. (d) Sperm abnormality rate; the rate of sperm abnormality was increased in mice at 7 days after irradiation.
piRNAs profile
Significant changes in the expression profile of piRNAs were observed at 3 and 7 days postirradiation. Cluster analyses of the piRNA profiles at 3 and 7 days postirradiation are shown in Figure 3(a) and 3(b), respectively. The top 20 differentially expressed piRNAs are listed in Table 2 (3 days after irradiation) and Table 3 (7 days after irradiation).

Hierarchical clustering of differentially expressed piRNAs. Hierarchical clustering of piRNAs at 3 days (a) and 7 days (b) after irradiation. piRNA: PIWI-interacting RNA.
Differentially expressed piRNAs in sperm at 3 days after irradiation.

Differentially expressed piRNAs in sperm at 7 days after irradiation.

Pathway analysis of differentially expressed piRNAs
We performed GO and KEGG pathway analyses of piRNAs that were downregulated in sperm at 3 days after irradiation and found that these piRNAs (mainly mmu_piR_015386, mmu_piR_009082, mmu_piR_009083, and mmu_piR_009084) were involved in several pathways including Rap1 signaling, non-homologous end-joining, hedgehog signaling, oxytocin signaling, and cholinergic synapse (Figure 4).
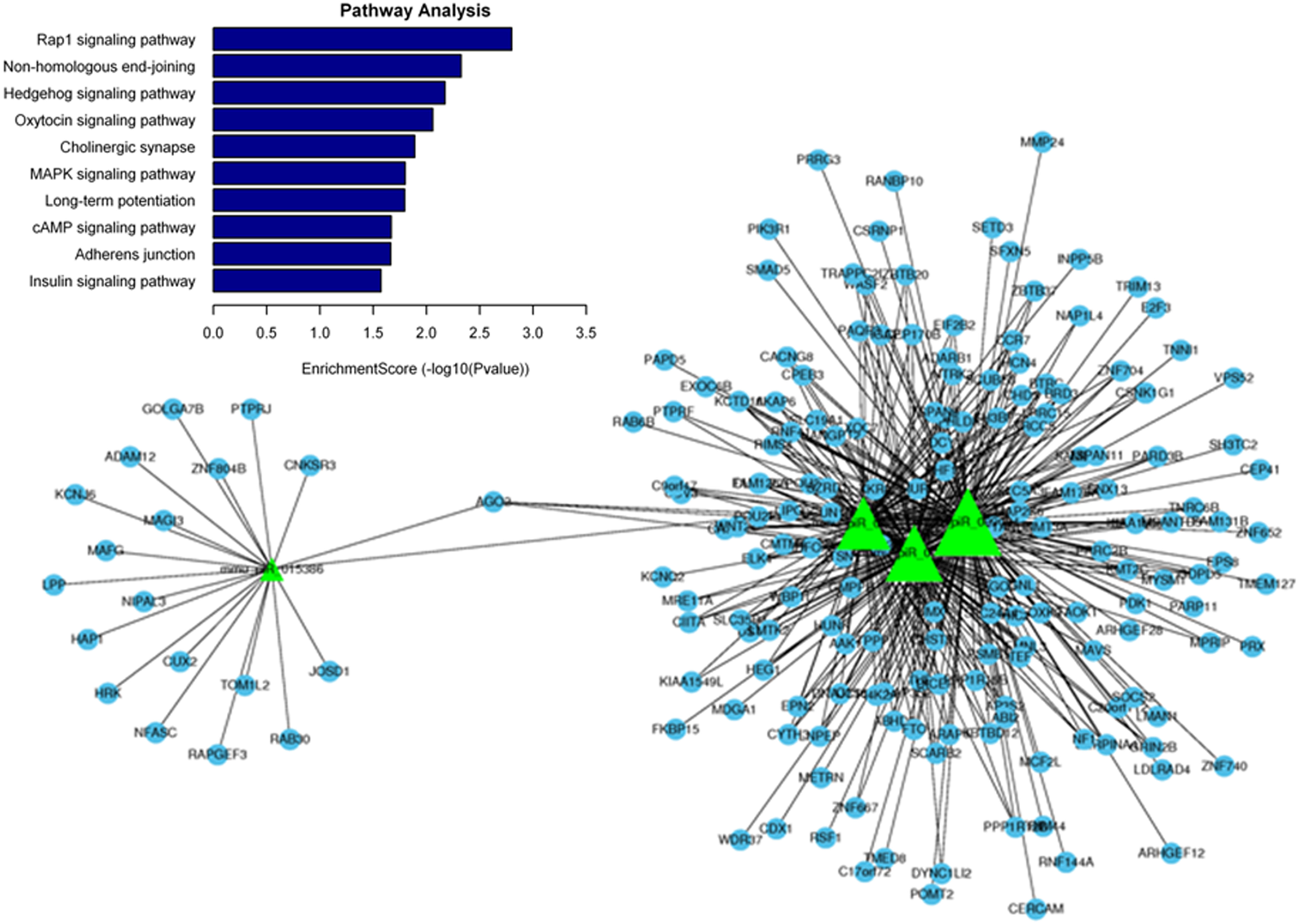
Pathway analysis of downregulated piRNAs at 3 days post-irradiation. Downregulated piRNAs are presented as green triangles and their predicted target genes are shown as blue circles.
We performed GO and KEGG pathway analyses of piRNAs that were upregulated in sperm at 3 days after irradiation and found that these piRNAs (mainly mmu_piR_029800, mmu_piR_004455, mmu_piR_001851, mmu_piR_036956, and mmu_piR_019801) were involved in signaling pathways including that of proteoglycans in cancer, phosphatidylinositol signaling, cGMP-PKG signaling, stem cell pluripotency regulation, and insulin secretion (Figure 5).

Pathway analysis of upregulated piRNAs at 3 days post-irradiation. Upregulated piRNAs are presented as red triangles and their predicted target genes are shown as blue circles. piRNA: PIWI-interacting RNA.
We performed GO and KEGG pathway analyses of piRNAs that were downregulated in sperm at 7 days after irradiation and found that these piRNAs (mmu_piR_000268, mmu_piR_020932, mmu_piR_017444, and mmu_piR_038671) were involved in several pathways including hepatitis B, prolactin signaling, cholinergic synapse, oxytocin signaling, and insulin secretion (Figure 6).

Pathway analysis of downregulated piRNAs at 7 days post-irradiation. Downregulated piRNAs are presented as green triangles and their predicted target genes are shown as blue circles. piRNA: PIWI-interacting RNA.
We performed GO and KEGG pathway analyses of piRNAs that were upregulated in sperm at 7 days after irradiation and found that these piRNAs (mainly mmu_piR_002198, mmu_piR_020217, mmu_piR_030405, mmu_piR_001851, and mmu_piR_033064) were involved in pathways including morphine addiction, pathways in cancer, stem cell pluripotency regulation, the role of proteoglycans in cancer, and chronic myeloid leukemia (Figure 7).

Pathway analysis of upregulated piRNAs at 7 days post-irradiation. Upregulated piRNAs are presented as red triangles and their predicted target genes are shown as blue circles. piRNA: PIWI-interacting RNA.
Validation of differentially expressed piRNAs
Real-time PCR was performed to validate the expression of several piRNAs (mmu_piR_015386, mmu_piR_009082, mmu_piR_038671, mmu_piR_029800, mmu_piR_004455, mmu_piR_020217, mmu_piR_021962, mmu_piR_026790, mmu_piR_017444, and mmu_piR_001851).
As shown in Figure 8, the expression of mmu_piR_009082 was reduced at 3 and 7 days after irradiation. Expression of mmu_piR_038671 and mmu_piR_029800 remained unchanged at 3 days after irradiation but decreased at 7 days. Expression of mmu_piR_020217 was elevated at 3 and 7 days after irradiation, while that of mmu_piR_021962 remained unchanged at both 3 and 7 days, and that of mmu_piR_026790 was significantly elevated at 7 days after irradiation.
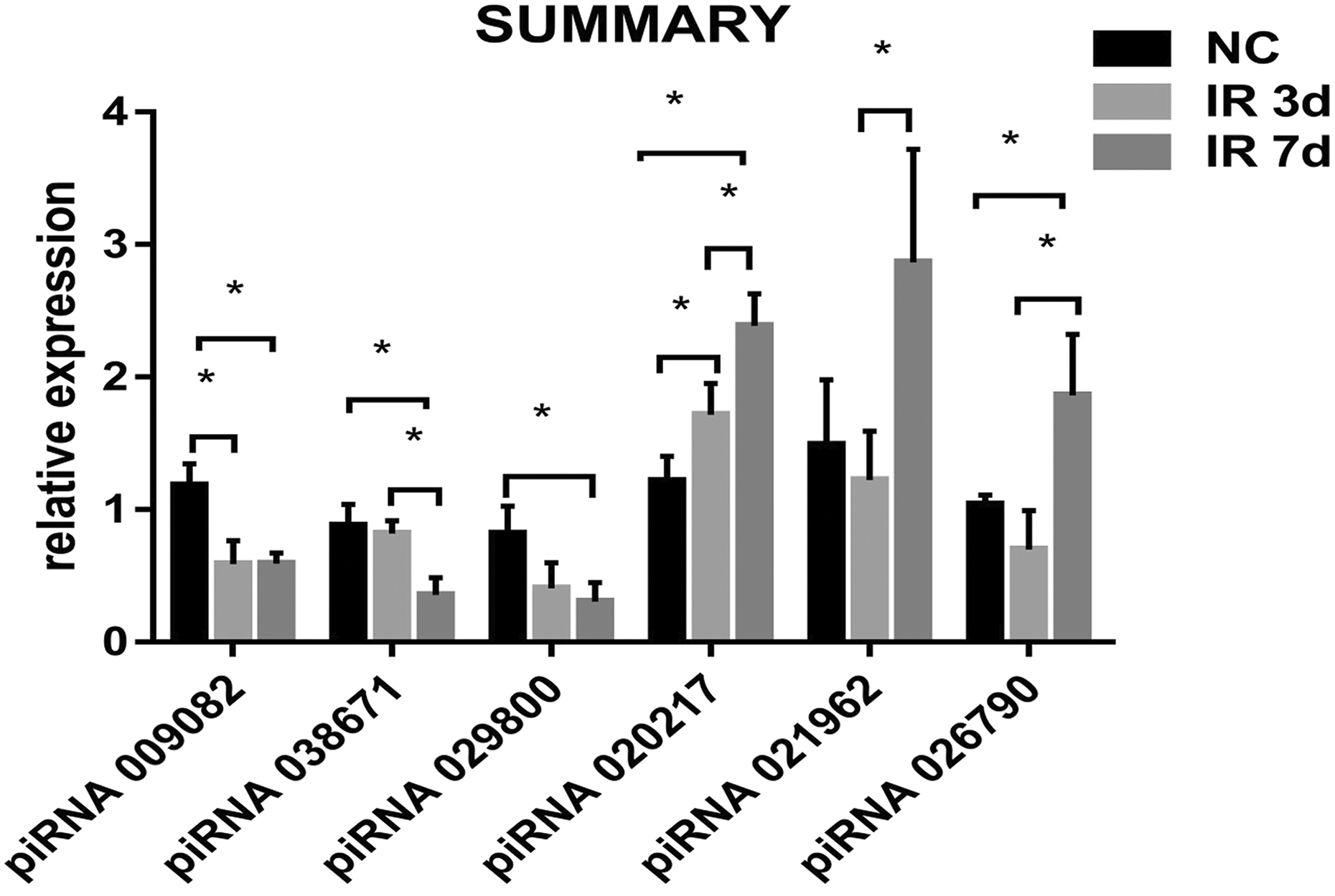
Validation of differentially expressed piRNAs by real-time PCR. The expression of mmu_piR_009082 was reduced at 3 and 7 days after irradiation; that of mmu_piR_020217 was elevated. piRNA: PIWI-interacting RNA; PCR: polymerase chain reaction.
Discussion
Male reproduction is a complex process involving cell proliferation and differentiation. Spermatogenesis is the process by which spermatogonial cells produce spermatocytes after mitotic division. During this process, the spermatocytes divide meiotically into haploid spermatids, which finally transform into mature spermatozoa. 7,8 The male genital gland, the testis, can produce sperm and androgens, while the epididymis is a tube in which sperm maturation occurs. Both organs are targeted by exogenous reproductive hazards. The sensitivity of germ cells to various hazardous substances varies among developmental stages; thus, changes in sperm quality reflect an early reproductive injury. Irradiation alone or in combination with other chemical substances can greatly alter gene expression and induce reproductive dysfunctions in the male reproductive system. 9 The count, viability, and morphology of sperm are key elements affecting fertility and function as sensitive indices for evaluating the effects of physical and chemical factors on sperm cells. Sperm viability and the rate of sperm abnormality are clinically used parameters for evaluating male infertility. Many hazardous factors can induce sperm injury and abnormality. Therefore, in the present study, the viability, motility, and abnormality rate of sperm were used to evaluate the effects of irradiation on reproductive function in male mice.
During the past several years, researchers have studied the effects of ionizing irradiation on male reproductive function, focusing on spermatogenic cells and chromosomal aberrations of sperm upon exposure to radiation. Animal experiments showed that high dose rates (3.3–166.6 mGy min−1) of 60Co irradiation at day 15 post-coitum (p.c.) had a short-term effect on the survival of germ cells. A large proportion of germ cells rapidly underwent p53-independent apoptosis following radiation treatment. Apoptotic cells were strongly clustered and the remaining germ cells divided and differentiated normally, with tubules in the adult testis showing a normal phenotype. However, a low dose rate (0.6 mGy min−1) induced testicular atrophy and decreased seminiferous tubules at day 19 p.c. Irradiation (250 mGy min−1) also showed a delayed effect on germ cells; irradiation at day 9 p.c. decreased the number of spermatocytes and induced apoptosis with clustered apoptotic cells sterility occurred at day 15 p.c. 10 Ionizing irradiation can induce germ cell apoptosis with varying potencies according to the cell type in the following order: spermatogonia, primary spermatocytes, secondary spermatocytes, and spermatids. Irradiated sperm with a chromosomal aberration retains some fertilization ability but can lead to the production of abnormal embryos. Kamiguchi et al. investigated chromosomal aberrations in sperm irradiated with three types of ionizing irradiation (X-ray, β-ray, and 137Cs γ-ray). They found that the rates of exchange-type, chromatid-type, and haplotype chromosomal aberrations were positively correlated with the irradiation dose. 11 Additionally, prostatic brachytherapy can decrease the viability and concentration of sperm and increase the DNA fragmentation index to have long-term effects on fertility. 12 Moreover, earthworms exposed to X-ray radiation exhibited morphological abnormalities in sperm. 13 In the present study, we found that irradiation localized to the reproductive system decreased the thickness of the seminiferous epithelium and reduced spermatogenesis, sperm viability, and motility. At 7 days after irradiation, an elevated rate of sperm abnormality was observed. These results show that 6-MV-X-ray radiation decreased the reproductive capability of male mice.
piRNAs are small non-coding RNAs that were first reported in 2006 and form RNA-protein complexes through interactions with PIWI proteins. 5,14 –16 PIWI/piRNA complexes play critical roles in germ stem cell maintenance, gametogenesis, and embryonic development. 16 –19 Recent studies showed that the cyanobacterial toxin microcystin–leucine–arginine induces reproductive toxicity by affecting the piRNA signaling pathway. 20,21 Thus, piRNA signals play an important role in exogenous substance-induced reproductive toxicity. In our study, many piRNAs were found to be differentially expressed in sperm exposed to radiation. These changes included a decrease in mmu_piR_009082 expression and an increase in mmu_piR_020217 expression, which may be critical in X-ray-induced reproductive toxicity. Downregulated piRNAs were mainly involved in several signaling pathways including Rap1 signaling, non-homologous end-joining, hedgehog signaling, oxytocin signaling, cholinergic synapse, hepatitis B, prolactin signaling, cholinergic synapse, oxytocin signaling, and insulin secretion.
Although piRNA was first identified 12 years ago, little is known about how PIWI/piRNA regulates gene expression. Reuter et al. showed that Miwi slicer activity directly cleaves transposon messenger RNAs, suggesting an explanation for the continued maintenance of repeat-derived piRNAs long after transposon silencing is established in germline stem cells. 22 Additionally, PIWI proteins appear to be involved in a two-pronged mammalian transposon silencing strategy: one promotes transcriptional repression in the embryo, while the other reinforces silencing at the posttranscriptional level after birth. However, Vourekas et al. demonstrated that Miwi directly binds to spermiogenic mRNAs without using piRNAs as guides, and independent biochemical analyses of testis mRNA ribonucleoproteins showed that Miwi functions in the formation of mRNA ribonucleoprotein complexes that stabilize mRNAs essential for spermiogenesis. 19 Thus, PIWI/piRNA may suppress the expression of some genes and positively regulate the expression of other genes. We predicted that PIWI/piRNA induced by X-ray irradiation would suppress non-homologous end-joining and activate Rap1 signaling after X-ray exposure. We found that spermatogenesis was inhibited and the sperm abnormality rate was increased; a schematic diagram of these results is shown in Figure 9. Thus, these piRNA-related signaling pathways may play important roles in radiation-induced reproductive toxicity. Further studies are needed to determine their functions and underlying mechanisms.

Schematic diagram. PIWI/piRNA induced by X-ray irradiation suppressed non-homologous end-joining and activated Rap1 signaling after X-ray exposure. As a result, spermatogenesis was inhibited and sperm abnormality was increased.
Conclusions
In summary, localized gonadal irradiation with 6-MV-X-rays can induce a decrease in the reproductive capability of male mice and this process may involve many piRNA-related signaling pathways.
Footnotes
Author Contributions
Xiaolong Liu and Qiu Chen contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of following financial support for the research, authorship, and/or publication of this article: This work was supported by ITER (2014GB112006) and Introduction project of “team of clinical medical experts” in Suzhou city (SZYJTD201705) and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
