Abstract
Cisplatin (CP) is one of the most effective and widely used compounds in the treatment of disease, including cancer, but is known to induce toxicity in patients. Rutin (RUT) is a flavonoid glycoside from Sophora japonica L. that has been shown to possess antioxidative, anti-inflammatory, and antiviral properties. RUT is also known to attenuate cardiotoxicity, isoproterenol-induced cardiac fibrosis, and ischemia/reperfusion-associated hemodynamic alteration, and prevents high glucose-induced renal glomerular endothelial hyperpermeability. In this study, we investigated the effect of RUT on CP-induced nephrotoxicity. CP was used to induce toxicity in human mesangial cells (HMCs), HMCs were pretreated with different concentrations of RUT before being exposed to 10 μg/mL of CP. A positive group was pretreated with antioxidant agent N-acetylcysteine prior to CP administration. At doses between 12.5 and 25 μM, RUT prevented CP-induced reduction in cell viability. Treatment with RUT suppressed intracellular reactive oxygen species and malonic dialdehyde levels and inhibited cell apoptosis. RUT reversed the CP-induced upregulation of p53, cleaved-caspase-3, and increased pro-caspase-3 and pro-caspase-9 levels. In conclusion, the RUT can relieve CP-induced nephrotoxicity by inhibiting the p53/caspase signaling pathway.
Introduction
Cisplatin (CP; PtCl2(NH3)2) is widely used in the treatment of a variety of malignancies. 1,2 However, it can also induce toxicity, including hepatotoxicity, 2 nephrotoxicity, 3 and gonadal toxicity, 4 limiting its clinical use. Approximately 20% of patients who receive an initial dose of CP (50–100 mg/m2) develop nephrotoxicity. 5 Currently, diuretics is the main drug that alleviates CP-induced nephrotoxicity without compromising the antineoplastic effect. Therefore, novel therapeutic agents with better nephroprotective efficacy and safety are required. The mechanism behind CP-induced kidney injury is not yet completely understood. It is thought to induce apoptosis in renal cells as a result of the excessive production of reactive oxygen species (ROS). 6,7 It has been demonstrated that the effect of CP on a variety of intracellular signal transduction pathways, may contribute to their nephrotoxicity. Evidence suggests that apoptosis in renal cells is related to the p53/caspase signaling pathway. 8 Park et al. 9 reported that caspase-9 was upregulated sixfold by CP in a dose-dependent manner, which lead LLC-PK1 cells to apoptosis. Although renal tubular cells have been the cell of choice in most experiments investigating CP-induced renal toxicity, 5 the glomerulus is also an important target for nephrotoxicity. However, there are not enough reports on CP-induced glomerular toxicity, it is hard to fully understand this relationship. 10
Flavonoids are phenolic compounds that are widely found in vegetables and fruits. 11,12 They have been shown to have a variety of biological effects both in vitro and in vivo. Rutin (RUT; C27H30O16) is a dietary flavonoids compound that is extracted from Sophora japonica L. Its immense therapeutic potential can be attributed to its diverse range of properties: antioxidant, anti-inflammatory, anti-allergic, and antiangiogenic. 13,14 Kamel et al. 6 reported that the administration of RUT (30 mg/kg) for 14 days, with a single CP dose on the 10th day, ameliorated CP-induced nephrotoxicity, indicated by the restoration of kidney function and oxidative stress biomarkers. However, the potential nephroprotective role of RUT in glomerulus cells has not yet been demonstrated.
This in vitro study investigated the effects of RUT on CP-induced nephrotoxicity in human mesangial cells (HMCs). In this study, N-acetylcysteine (NAC) was used as a positive drug to explore the antioxidant effect of RUT. 15 NAC has been previously shown to have a good antioxidant effect. The role of ROS generation and the p53/caspase signaling pathway in the nephroprotective effect of RUT was assessed.
Materials and methods
Materials
CP (5 mg/mL; CAS 15663-27-1) was purchased from Jiangsu Hansoh Pharmaceutical Co. Ltd., China, 16 and RUT (purity >98%; CAS 153-18-4) was purchased from Jingzhu Technology Co. Ltd., China. Methyl thiazolyl tetrazolium (MTT; CAS 298-93-1) was purchased from Sigma-Aldrich (Massachusetts, USA), 17 and Hoechst33258 kit, dichlorodihydrofluorescein diacetate (DCFH-DA),NAC, and cleaved-caspase-3 antibody were purchased from the Beyotime Institute of Biotechnology (Nanjing, China). Malonic dialdehyde (MDA) and superoxide dismutase (SOD) kits were purchased from Jiancheng Biological Institute (Nanjing, China). 18 P53, pro-caspase-9, and pro-caspase-3 antibodies were purchased from Santa Cruz Biotechnology, China. All the other chemicals used were of the highest commercially available purity. RUT and NAC were dissolved in dimethyl sulfoxide (DMSO) (CAS 67-68-5; Beijing Hua gong, China) to ensure the concentrations are lower than 0.1%.
Cell culture
HMCs were purchased from ATCC (Shanghai, China) and maintained in low glucose dulbecco’s modified eagle medium (DMEM) containing fetal bovine serum (10%), streptomycin (100 μg/mL), and penicillin G (70 μg/mL) at 37°C in a humidified atmosphere of 5% carbon dioxide and 95% air. 19
Pharmacological interference
To induce nephrotoxicity, HMCs were incubated with 10 μg/mL of CP for 24 h. In this experiment, the dosage of RUT was mainly selected based on the previous experiments. We found that 30 μM of RUT reduces the toxicity of pirarubicin-induced cardiomyocytes and improves cell viability in H9C2 cells. 20 The daily administration of 25 mg/kg of RUT reduces isoproterenol (ISO)-induced myocardial fibrosis in rats. 21 Wang et al. 22 demonstrated that 25 μM of RUT prevents high glucose-induced renal glomerular endothelial hyperpermeability. To determine the effect of RUT against CP-induced nephrotoxicity, cells were pretreated with 12.5 μM and 25 μM of RUT for 1 h, followed by incubation with 10 μg/mL of CP for 24 h. NAC was administered as a positive group. According to the previous studies, NAC can have an antioxidant effect at dosages of 1–5 mM. Zuo et al. 23 used NAC (5 mM) as a positive drug in the experiment. Vezir et al. 24 suggested that NAC (2 mM and 5 mM) relaxes vascular smooth muscle cells through a direct effect on KATP channels, by increasing outward K+ flux, partly by increasing mRNA expression of KATP subunit ABCC8, by decreasing intracellular calcium, and by decreasing Na+/K+-ATPase activity. In this study, we used a NAC concentration of 2 mM. In each experiment, divided into five groups: control group, model group (10 μg/mL of CP), RUT low dose group, RUT high dose group, and NAC positive group.
MTT assay
Cell viability was measured using an 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide (MTT) assay; HMCs were seeded in 96-well plates. Once cells had grown to approximately 80% confluence, the respective drugs were administered. After 24 h, 20 μL of MTT (0.5 g/L) was added to each well and incubated for another 4 h. The medium was then removed, and the cells were treated with 100 μL of DMSO and shaken for 10 min. The absorbance of MTT formazan was measured at a wavelength of 490 nm using an ELISA reader (Biopeony, Beijing, China).
Assays for the detection of apoptosis
Cell apoptosis was determined by Hoechst 33258 staining and Annexin V–propidium iodide (PI) staining. For Hoechst 33258 staining, cells were washed with phosphate buffered solution (PBS) and fixed in 4% paraformaldehyde for 10 min after treatment. After PBS washing, cells were stained with 5 mg/L Hoechst 33258 for 30 min at room temperature and examined under a fluorescent microscope (BX50-FLA, Olympus, Japan). HMCs were seeded onto a six-well plate at a density of 1 × 105 cells/well. After the treatment, cells were collected by centrifugation at 1000 r/min for 5 min. The cells were then washed with PBS, and approximately 1 × 105–5 × 105 cells were suspended in 500 μL of Annexin V binding buffer. Subsequently, 5 μL of Annexin V-FITC solution and 5 μL of PI solution were added to the cell suspension, mixed, and incubated for 10 min at room temperature in the dark. 20 The percentage of early and late cell apoptosis was assessed on flow cytometry (BD FACSCalibur; BD Biosciences, Franklin Lakes, NJ, USA).
Determination of intracellular cell ROS level
To determine the intracellular ROS levels, cells were seeded onto glass coverslips. The treatments were carried out once the cells had grown to approximately 80% confluence. The cells were then washed twice with PBS. Finally, cells were incubated with 10 μM DCFH-DA for 10 min. After washing, five nonoverlapping areas were randomly selected and micrographs were captured under a fluorescent microscope (Nikon, Japan). The average fluorescence intensity was calculated from five images using Image J pro-plus software (MEDIA CYBERNETICS Company Rockville, MD, USA). 20
MDA and SOD assays
HMCs were seeded in six-well plates. After the treatment, cells were washed twice with PBS and then homogenized. The homogenate was centrifuged at 5000 r/min for 10 min at 4°C. The supernatants were stored at 80°C for further analyses. The protein concentration of each supernatant sample was determined using the bicinchoninic acid (BCA) method. The levels of MDA and SOD activity and the protein content were determined using specific detection kits according to the manufacturer’s instructions. Concentrations were normalized to the sample protein concentration expressed as a percentage of untreated control cultures.
Western blot analysis
Cells were seeded in six-well plates. After treatment, cells were washed twice with PBS and then lysed with cell lysis buffer at 4°C for 30 min. Samples were centrifuged at 12,000 r/min for 10 min, and the supernatant was collected. Protein quantification, SDS-PAGE separation, and blotting on polyvinylidene fluoride (PVDF) membrane were carried out. Membranes were blocked in 5% milk in PBS with 0.1% Tween-20 at room temperature for 1 h, then incubated with primary specific antibodies against pro-caspase-3, p53, pro-caspase-9 (1:500), cleaved-caspase-3 (1:300), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (1:1000) antibodies overnight at 4°C. Signals were revealed using the appropriate secondary peroxidase-conjugated antibodies, and the bands were visualized by chemiluminescence using a ChemiDoc XRS system (Bio-Rad, Hercules, CA, USA). Image J software was used to analyze the quantifiedwestern blot. GAPDH was used as an internal control. 20
Statistical analysis
We used Student’s t-tests with the program GraphPad Prism (version 5.0; GraphPad Software, San Diego, California, USA) and one-way analysis of variance for multiple comparisons to determine the significance of the differences between the two groups. The data were represented as mean ± standard deviation, and each experiment was repeated three times. A value of p < 0.05 was considered significant.
Results
RUT protects HMCs against CP-induced toxicity
The effect of different concentrations of RUT on the survival of HMCs measured by MTT assay is shown in Figure 1(a). As indicated, treatment with RUT for 24 h did not significantly affect the cell viability of HMCs.
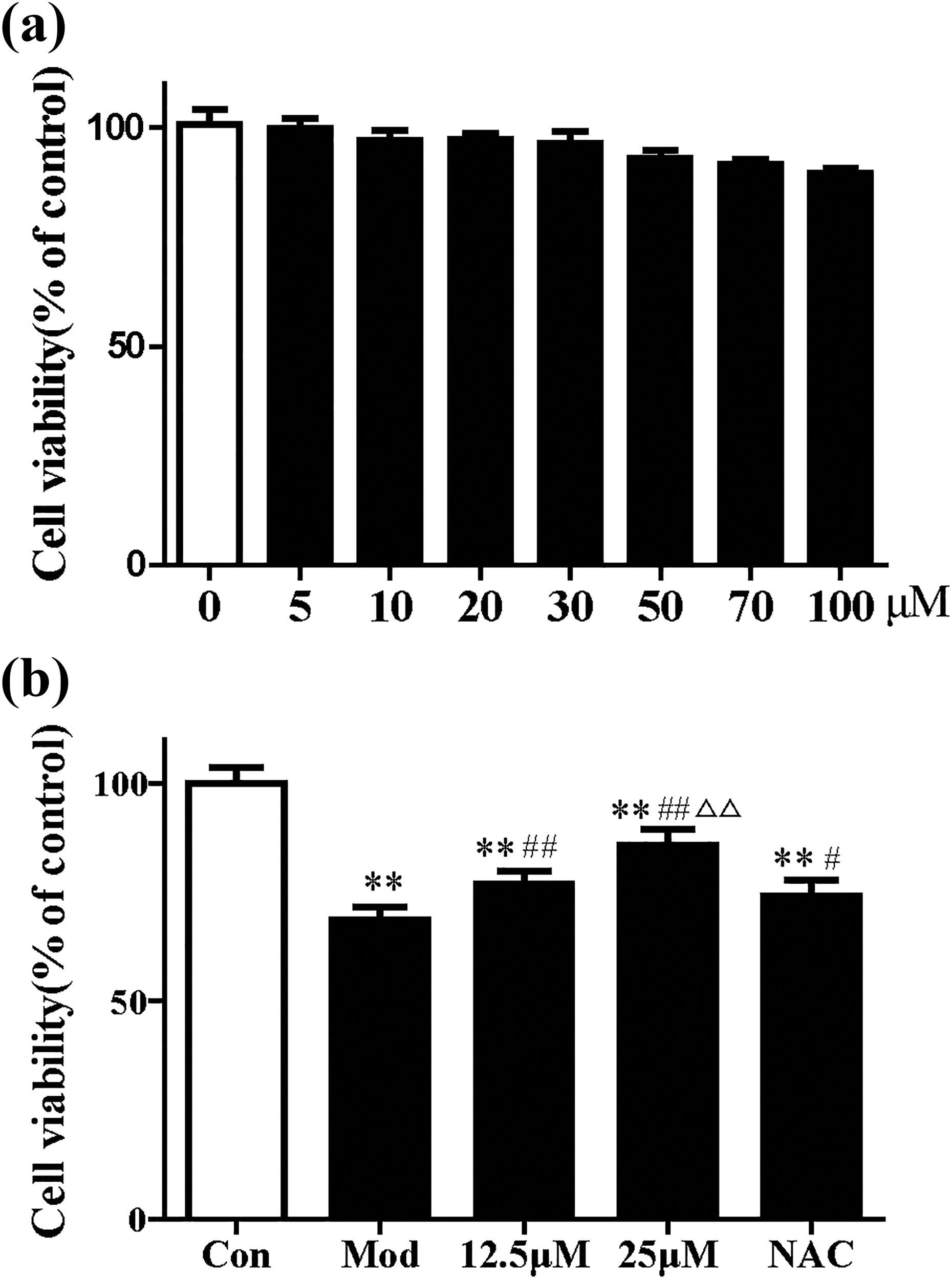
Cell viability assessment using MTT assay. (a) MTT assay showing the effect of RUT on HMC viability. (b) RUT protects HMCs against CP-induced cytotoxicity. The data are expressed as mean ± SD. **p < 0.01 was compared with the control group; #p < 0.05 and ##p < 0.01 were compared with the model group; and △p < 0.05 and △△p < 0.01 were compared with NAC. HMC: human mesangial cell; RUT: rutin; CP: cisplatin; NAC: N-acetylcysteine SD: standard deviation.
As indicated in Figure 1(b), treatment of CP significantly reduced cell viability compared to the control group (p < 0.01). Pretreatment with NAC, an antioxidant agent, attenuated the decline in cell viability induced by CP compared to treatment with CP alone (p < 0.05). Moreover, treatment with RUT at concentrations of 12.5 μM and 25 μM reversed the reduction of cell viability in CP-treated cells (p < 0.05).
RUT protects HMCs against CP-induced apoptosis
To investigate the effect of RUT on cell apoptosis, Hoechst 33258 staining was carried out (Figure 2(a)). CP exposure remarkably increased the proportion of apoptotic cells (p < 0.05), as is evident from the presence of condensed or fragmented nuclei, whereas control cells presented round, uniformly colored nuclei. Administration of RUT significantly suppressed CP-induced cell apoptosis (p < 0.05). Most of the nuclei were evenly stained, showing a normal cell status, and apoptotic bodies were reduced. CP apoptosis was further evaluated by Annexin V–PI staining followed by flow cytometric analysis (Figure 2(b)). Similar to the Hoechst 33258 staining results, pretreatment with RUT caused a greater reduction in the percentage of apoptotic cells compared to the model group (p < 0.05).
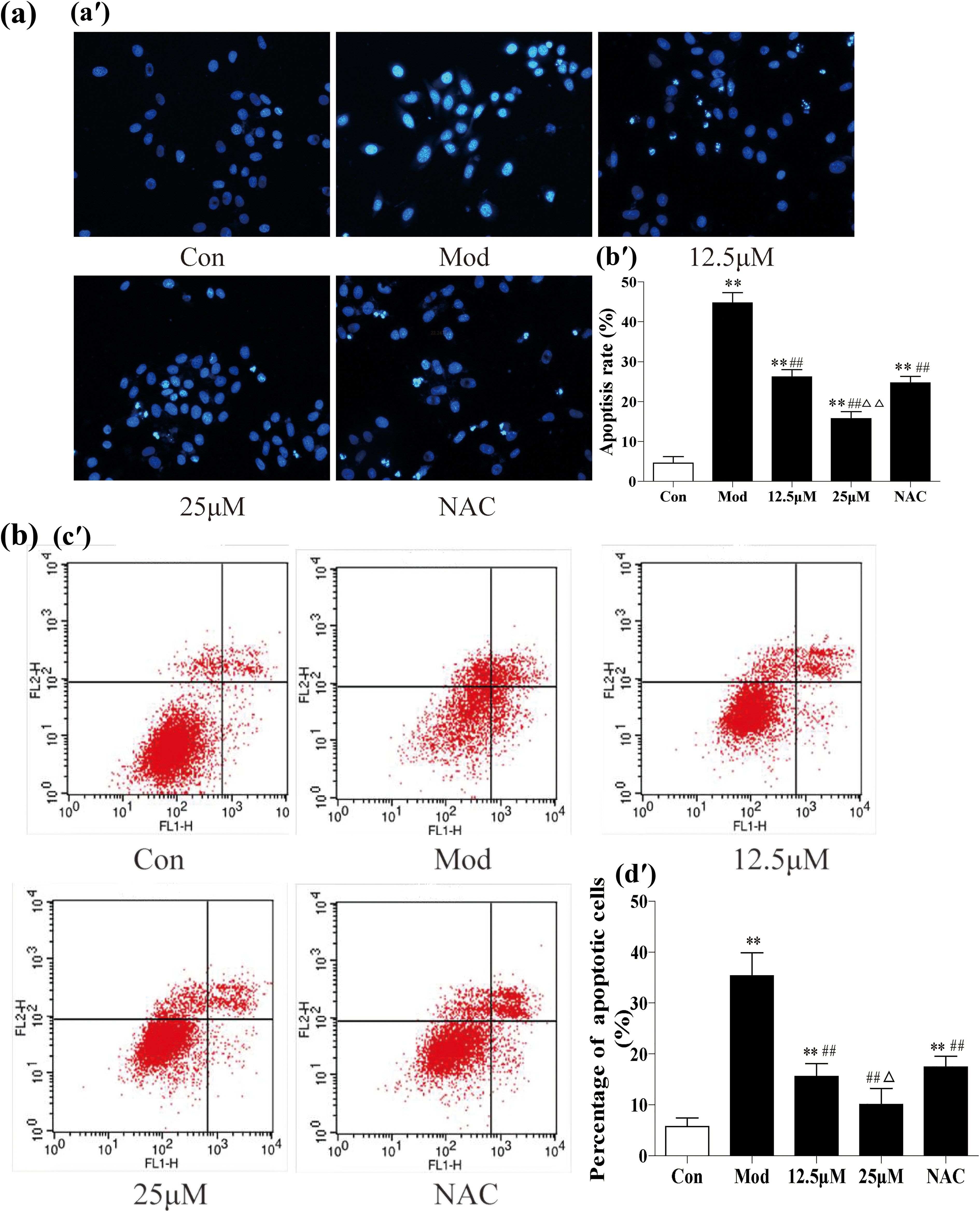
RUT protects HMCs against CP-induced apoptosis. (a) Determination of cell apoptosis by Hoechst 33258 staining; (a′) representative images of Hoechst 33258 staining (original magnification ×200) and (b′) average percentage of apoptotic cells. (b) Determination of cell apoptosis by Annexin V–PI staining followed by flow cytometric analysis; (c′) representative data of flow cytometric analysis and (d′) average percentage of early and late apoptotic cells. The data are expressed as mean ± SD. *p < 0.05 and **p < 0.01 were compared with the control group; #p < 0.05 and ##p < 0.01 were compared with the model group; and △p < 0.05 and △△p < 0.01 were compared with NAC. HMC: human mesangial cell; RUT: rutin; CP: cisplatin; NAC: N-acetylcysteine PI: propidium iodide; SD: standard deviation.
RUT reduces ROS concentration in HMCs
The intracellular ROS level was low in the control group (Figure 3). After exposure to CP, the concentration of intracellular ROS increased significantly compared to the control group (p < 0.05). Pretreatment with RUT significantly inhibited ROS level in HMCs (p < 0.05). The most evident antioxidation effect was observed in cells that had been pretreated with 25 μM of RUT compared with the NAC group (p < 0.01).
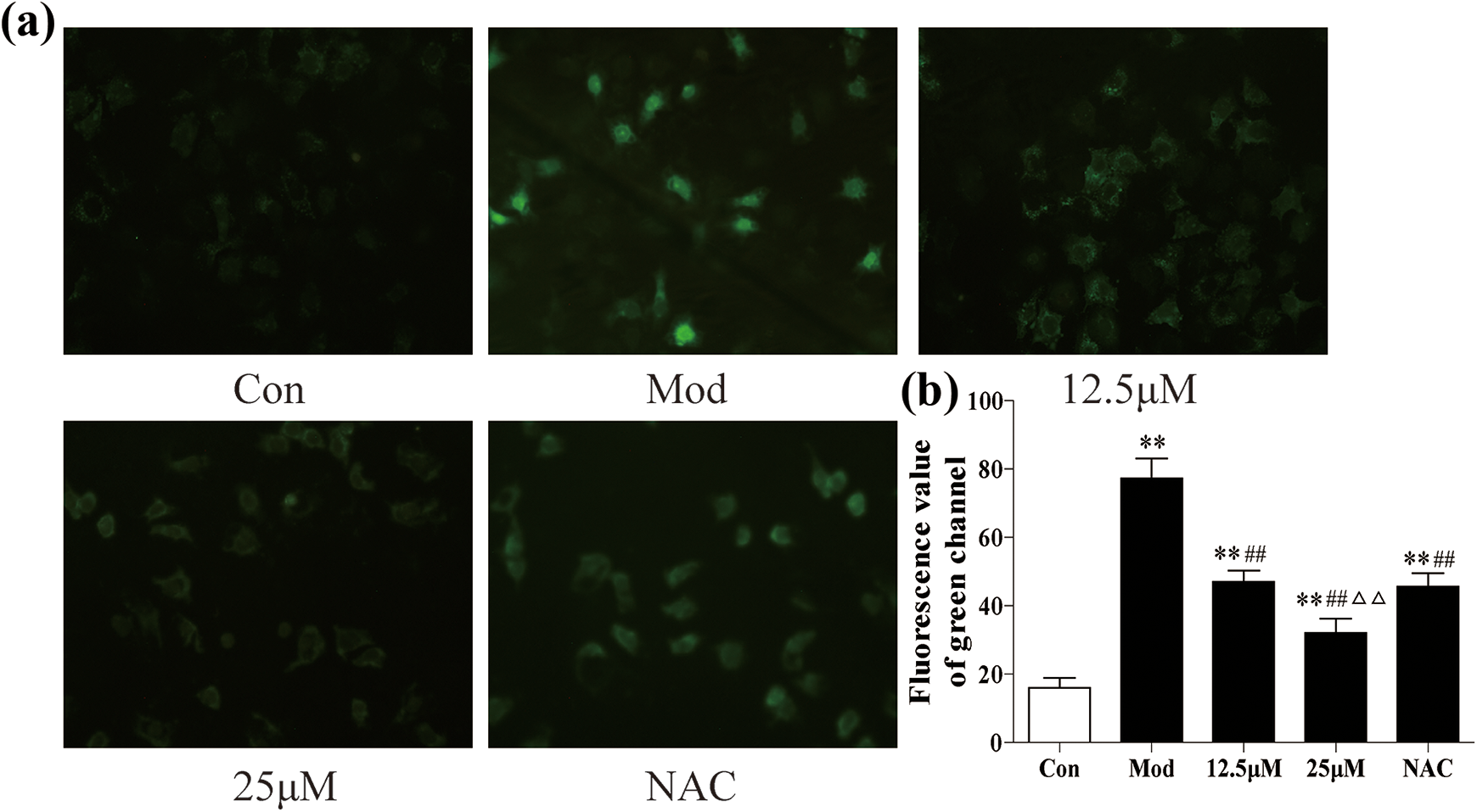
Intracellular ROS concentration. (a) Representative images of DCFH-DA staining (original magnification ×200). (b) Average fluorescence intensity was quantified. The data are expressed as mean ± SD. *p < 0.05 and **p < 0.01 were compared with the control group; #p < 0.05 and ##p < 0.01 were compared with the model group; and △p < 0.05 and △△p < 0.01 were compared with NAC. DCFH-DA: dichlorodihydrofluorescein diacetate; RUT: rutin; CP: cisplatin; NAC: N-acetylcysteine ROS: reactive oxygen species; SD: standard deviation.
SOD and MDA levels in HMCs
We examined the effects of RUT on SOD and MDA levels as the principal parameters in evaluating free radical metabolism (Table 1). After exposure to CP, the level of SOD was significantly lower, and the level of MDA increased significantly compared to the control group (p < 0.05). However pretreatment with RUT significantly inhibited the level of MDA and increased SOD levels in HMCs (p < 0.05). The most evident increase of SOD was observed in cells that had been pretreated with 25 μM of RUT compared to the NAC group (p < 0.01).
SOD and MDA levels in HMCs.a

HMC: human mesangial cell; MDA: malonic dialdehyde; SOD: superoxide dismutase; NAC: N-acetylcysteine; RUT: rutin; SD: standard deviation.
a The data are expressed as mean ± SD.
bp < 0.01 and cp < 0.05 were compared with the control group.
dp < 0.05 and ep < 0.01 were compared with the model group.
fp <0.01 and p < 0.05 were compared with NAC.
p53 protein expression in HMCs
As illustrated in Figure 4, the expression of p53 increased significantly in CP-treated cells compared to the control (p < 0.05). Similarly, after pretreatment with RUT, the expression of p53 decreased significantly compared to cells treated with CP only. The most evident suppression of p53 expression was observed in cells that had been pretreated with 25 μM of RUT compared to the NAC group (p < 0.05).

Western blot analysis of p53 signaling pathway protein expression. GAPDH was used as an internal control. (a) p53 protein expression. (b) p53 protein and GAPDH ratio. The data are expressed as mean ± SD. *p < 0.05 and **p < 0.01 were compared with the control group; #p < 0.05 and ##p < 0.01 were compared with the model group; and △p < 0.05 and △△p < 0.01 were compared with NAC. RUT: rutin; CP: cisplatin; NAC: N-acetylcysteine; SD: standard deviation.
Protein expression of caspase regulators
We assessed the protein expression of several crucial regulators of caspase expression (Figure 5). When cells were exposed to CP, the expression levels of pro-caspase-9 and pro-caspase-3 decreased, and the expression of cleaved-caspase-3 increased compared to the control group (p < 0.05). Cleaved caspase-3 decreased significantly after RUT treatment compared to that in the model group (p < 0.05). The most evident suppression of caspase expression was observed in cells that had been pretreated with 25 μM of RUT compared to the NAC group (p < 0.05).

Western blot analysis of caspase signaling pathway protein expression. GAPDH was used as an internal control. (a) Representative data of pro-caspase-9, pro-caspase-3, and cleaved-caspase-3. (b) The proteins and GAPDH ratio. The data are expressed as mean ± SD. *p < 0.05 and **p < 0.01 were compared with the control group; #p < 0.05 and ##p < 0.01 were compared with the model group; and △p < 0.05 and △△p < 0.01 were compared with NAC. RUT: rutin; CP: cisplatin NAC: N-acetylcysteine; SD: standard deviation.
Discussion
CP is widely used in the treatment of many types of cancer, including head and neck, lung, testis, ovary, and breast cancers. 2,25 Nephrotoxicity (occurring in about 20–30% of patients receiving CP 26 ) is the dose-limiting side effect of CP. 27 Other side effects can include hypomagnesemia (40–100% of patients), 28 Fanconi-like syndrome, distal renal tubular acidosis, hypocalcemia, renal salt wasting, and hyperuricemia. 29 Although the mechanism behind CP-induced nephrotoxicity is not well understood, previous studies suggested that CP interacts with DNA through the formation of covalent adducts between certain DNA bases and the platinum compound, leading to cell cytotoxicity. 30 Other studies have suggested that CP-induced ROS and immune response are the mediators of nephrotoxicity. 31 –33
RUT has various pharmacological properties, 34 but its potential role in nephroprotection is yet to be proven. Alhoshani et al. 35 demonstrated that the antioxidant and anti-inflammatory effects of RUT may prevent CP-induced nephrotoxicity in rats by reducing oxidative stress, inhibiting the interconnected ROS/c-Jun N-terminal kinase (JNK)/Tumor Necrosis Factor (TNF)/P38 Mitogen-activated protein kinase (MAPK) signaling pathways, and repairing the histopathological changes against CP administration. These experiments confirmed that RUT can play a protective role both in vivo and in vitro. Although renal tubular cells are normally used in vitro experiments investigating CP-induced renal toxicity, the glomerulus is also an important target in nephrotoxicity. However, there are no reports on CP-induced glomerular toxicity and its mechanism yet. Khafaga and Bayad 36 showed that renal tissues from rats had extensive glomerular and vascular congestion, vacuolar degeneration, proximal tubular epithelial necrosis and desquamation, intraluminal cast formation, glomerulus atrophy, and epithelial cast formation in Bowman’s space after a single injection of CP. According to their quantitative morphometric analysis, both glomerular area and glomerular volume were significantly reduced. It is also known that p38 MAPK can activate p53 protein and induce apoptosis. 37 In the present study, HMCs were used to investigate the pathogenesis of CP nephrotoxicity in the p53/caspase signaling pathway and the protective effect of RUT. The results suggest that the protective effect of RUT appears to be associated with its ability to scavenge intracellular ROS and inhibit cell apoptosis by modulating the p53/caspase signaling pathway.
In our previous experiment, we found that administration of RUT at the concentrations ranging from 12.5 to 50 μM, reversed the reduction of cell viability in CP-treated cells. The most evident protective effect was observed in cells that were pretreated with 25 and 50 μM. However, there was no significant difference between the two groups. There were also some experiments using one or two doses. Li et al. 21 demonstrated that RUT (25 and 50 μM) exhibits cardioprotective effects on cardiac fibrosis induced by ISO in the rat heart. Wang et al. 22 showed that RUT (25 μM) prevents high glucose-induced renal glomerular endothelial hyperpermeability by inhibiting the ROS/Rhoa/Rho Associated protein kinase (ROCK) signaling pathway. Mladenka et al. 38 indicated that RUT at a dose of 46 mg/kg could aggravate isoprenaline-induced cardiotoxicity and in vitro experiment, they found that this significant aggravation of isoprenaline toxicity was seen with 30−300 μM RUT concentrations. Therefore, whether high dose of RUT aggravates CP-induced nephrotoxicity needs further investigation. The dosages of 12.5 and 25 μM were used in this experiment. We first determined the effect of RUT in the cells treated with CP. MTT assay results revealed that RUT reversed CP-induced loss of cell viability. After Hoechst 33258 staining and Annexin V–PI staining, we found that most of the nuclei showed a normal cell status, and apoptotic cells were reduced; the most evident effect was observed at the dose of 25 μM. In accordance with our findings, Park et al. 9 demonstrated that pretreatment with 100 μM of RUT greatly alleviates the loss of cell viability in human dopaminergic cells exposed to rotenone. Others have also reported on the anti-inflammatory effects of RUT at concentrations between 25 μM and 50 μM on cultured human umbilical vein endothelial cells. 39 This discrepancy in results may be attributed to differences in cell cultures and experimental paradigms used.
We then found that pretreatment with RUT dramatically inhibited CP-induced elevation in intracellular ROS levels, increased SOD, and reduced MDA levels. These results suggest that the nephroprotective action of RUT is likely to be associated with its ability to eliminate ROS. Persistent generation of intracellular ROS may lead to oxidative stress and induce mitochondrial-associated cell apoptosis in the kidney. 40 Similar antioxidation effects have been observed using other plant-derived compounds, such as quercetin which prevents CP-induced ROS generation and suppresses apoptosis of tubular epithelial cells. 41 Losartan was also shown to attenuate ROS production, reduce oxidative stress, and inhibit cell apoptosis in HMCs treated with angiotensin II. 42
The p53 protein is an important element in many intra and extracellular processes. This protein regulates the repair of cellular DNA and induces apoptosis. It is also responsible for the regulation of senescence and of cell entering the subsequent stages of the cellular cycle. p53 is also involved in inhibiting angiogenesis and the induction of oxidative shock. 43 Caspases, a unique and closely related family of cysteine proteases, play a central role in the development and regulation of cell death, 44 divided into initiator and effectors’ caspases. Once activated by the signaling pathway, caspases can degrade intracellular proteins and trigger apoptosis. 45 It has also been demonstrated that, as a result of stress factors, p53 can cross into the mitochondria and activate the expression of proapoptotic genes, including Puma, Bax, apoptotic protease activating factor-1 (Apaf-1), and Noxa, and can also inhibit the expression of antiapoptotic genes, such as those of the family Bcl-2. These proapoptotic proteins, together with p53, are transported into the mitochondria where they induce an increase in the permeability of mitochondrial membranes and the release of cytochrome c, which connects with the Apaf-1 and with the caspase-9 proenzyme, to create a complex called the “apoptosome.” The apoptosome, in turn, brings about caspase-9 activation, which consequently promotes the caspase-3 proenzyme to the active protease stage, which then adheres to the group of effectors’ caspases. These caspases are responsible for intracellular protein lysis as well as for the morphological characteristic changes of apoptosis. 46,47 The generation of ROS, accompanied by the activation of the p53/caspase signaling pathway, contributes to apoptosis in HMCs. 48 In the present study, a significant upregulation of p53 and cleaved-caspase-3 was detected in cells treated with CP, which suggests that activation of the p53/caspase signaling pathway may contribute to renal apoptosis. Pretreatment with RUT efficiently inhibited the elevated expressions of p53 and caspase proteins. RUT reversed the CP-induced upregulation of p53 and cleaved-caspase-3, and increased pro-caspase-3 and pro-caspase-9 levels. It seems that RUT itself was sufficient to block the p53/caspase pathway. RUT is a multifunctional natural product with multiple pharmacological properties. 49 As such, it is possible that RUT has multiple intracellular targets to inhibit caspase signaling pathway. Consistent with our findings, Zhou et al. 50 reported that RUT prevented ROS production and apoptosis in human lens epithelial cells by inhibiting the NF-κB/caspase signaling pathway.
In conclusion, pretreatment with RUT prevented CP-induced ROS generation and cell apoptosis in cultured HMCs. The effect was mediated by inhibiting the p53/caspase signaling pathway. Our findings provide a fundamental elucidation of the nephroprotective effects of RUT. Nevertheless, our study has several limitations. First, the relationship between ROS production and cell apoptosis remains unclear. Second, although RUT and CP altered the expression of caspase pathway-related proteins, it is not clear whether Bax is an upstream mediator of caspase family. Third, the nephroprotective effect of RUT should be tested in a rodent model of nephrotoxicity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Science and Technology Development Project of Jilin Province (No. 20150204015YY).

