Abstract
The receptor megalin plays an important role in the accumulation of polymyxin B (PMB) in renal cells in vitro. This study aimed to examine the effects of cytochrome c (cyto c), a typical megalin ligand, on renal accumulation and nephrotoxicity of PMB in vivo. Thirty Sprague-Dawley rats were randomly divided into the vehicle control group, PMB group, PMB + cyto c 50, 100, or 200 mg/kg group, respectively, and were treated with intravenous cyto c 30 min before the administration of PMB 4.0 mg/kg once a day for consecutive 5 days. On the 4th day after administration, 24 h urine was collected to determine N-acetyl-β-D-glucosaminidase excretion. Six hours after the last injection on the 5th day, kidneys were harvested to assay PMB concentration and observe pathological alterations, and blood samples were collected to assay serum creatinine (SCr), blood urea nitrogen (BUN), and blood β2-microglobulin (β2-MG) levels. Cyto c 50, 100, and 200 mg/kg decreased the accumulation of PMB in the kidney by 18.5%, 39.1% (p < 0.01), and 36.8% (p < 0.01), respectively, and reduced 24 h N-acetyl-β-D- glucosaminidase excretion by 22.5% (p < 0.05), 40.4% (p < 0.01), and 40.4% (p < 0.01), respectively. Kidney pathological damage induced by PMB was markedly reduced by cyto c 100 mg/kg and 200 mg/kg. However, there were no significant differences in SCr, BUN, and blood β2-MG levels among the groups. These results indicated that cyto c may inhibit the renal accumulation and nephrotoxicity of PMB in a rat model, further proving the role of megalin in the accumulation of PMB.
Introduction
Polymyxins, discovered in 1947, are a group of polypeptide antibiotics that consist of five chemically different compounds (polymyxins A–E), 1 of which only polymyxin B (PMB) and polymyxin E (colistin) have been used for clinical practice. However, their intravenous formulations were gradually abandoned in the early 1970s due to a high incidence of nephrotoxicity. 2 With the current emergence of extensively resistant gram-negative bacteria such as Pseudomonas aeruginosa, Acinetobacter baumannii, and Klebsiella pneumonia and the shortage of new active antimicrobial agents, intravenous PMB and colistin are considered valuable therapies again. 3 –7 However, the potential for their application in clinical practice is still limited by their nephrotoxicity. 8 –10 Prevention of nephrotoxicity has important significance in extending the clinical use of the two drugs.
PMB nephrotoxicity was characterized by acute tubular necrosis 11 or tubular epithelial cell apoptosis. 12 It was demonstrated in vitro that PMB uptake into proximal tubular epithelial cells was saturable 13 and exhibited a strong affinity to megalin, 14 a low-density lipoprotein receptor, expressed at the apical membrane of the proximal tubules. Thus, it is postulated that megalin may play an important role in the uptake of PMB into renal tubular cells; the identification of an effective antagonist off megalin will be a new direction in the prevention of PMB nephrotoxicity.
Cytochrome c (cyto c), a typical megalin ligand, is used clinically as adjunctive treatment for tissue hypoxia, such as carbon monoxide poisoning, hypnotic intoxication, severe hypoxia in shock, dyspnea, and myocardial anoxia in China. 15,16 It was reported that cyto c competitively inhibited the binding of colistin, 16 structurally similar to PMB, 17 to megalin in vitro and reduced 24 h urinary N-acetyl-β-D-glucosaminidase (NAG) excretion when cyto c at 100 mg/kg body weight and colistin at 1 mg/kg body weight were concurrently administered to rats only once. 16 To date, little is known about PMB renal accumulation and its nephrotoxicity when PMB is repeatedly administered in an animal model. Therefore, we designed the present study to focus on the effect of cyto c injection on PMB renal accumulation and nephrotoxicity when PMB was repeatedly administered in a rat model in order to observe the role of megalin in PMB renal accumulation in vivo and to determine whether cyto c is an effective antagonist off PMB nephrotoxicity.
Materials and methods
Chemicals and reagents
Cyto c was obtained from Changzhou Fangyuan Pharmaceutical Co. Ltd (Jiangsu, China). Standard PMB sulfate was purchased from Dr. Ehrenstorfer GmbH (Germany). The NAG assay kit was from Beijing DG-technology Co., Ltd (Beijing, China). The serum creatinine (SCr) reagent kit was from Shanghai Sheng Suo You Fu Medical Diagnosis Product Co., Ltd (Shanghai, China). The blood urea nitrogen (BUN) test kit was from Ningbo Rui Bio-technology Co., Ltd (Ningbo, China). The serum β2-microglobulin (β2-MG) test kit was from Prodia Diagnostics (Germany). All other chemicals used in the experiments were of the highest purity available.
Animals
Healthy adult male-specific pathogen-free grade Sprague-Dawley rats, weighing 200 ± 10 g, were purchased from the Laboratory Animal Center of the Academy of Military Medical Sciences, PLA of China (certificate no. SCXK (Army) 2012-004). The animals were housed in clean plastic cages and maintained under standard conditions (12 h light and 12 h dark, 25 ± 5°C, and 35–60% humidity) with free access to tap water and standard rat diet throughout the experiment. An acclimatization period of 3 days was allowed before experimentation. The Institutional Animal Care and Use Committee of Air Force General Hospital approved the use of animals, and the animal experimental protocol was designed in accordance with the Guide for the Care and Use of Laboratory Animals of the Research Council.
Treatment of animals
The rats were randomly divided into the following five groups: (i) the saline group (control group), which received 0.9% sodium chloride (NaCl) 3 mL/kg; (ii) the PMB group, which received PMB 4.0 mg/kg body weight/day alone; (iii) the PMB + cyto c 50 mg/kg body weight/day group; (iv) the PMB + cyto c 100 mg/ body weight/day group; (v) and the PMB + cyto c 200 mg/ body weight/day group. With the exception of the PMB group and control group, cyto c was administered to all animals 30 min before administration of PMB at 4.0 mg/kg body weight/day. All drugs including 0.9% NaCl were administered via the caudal vein for five consecutive days. Six hours after the last injection on the 5th day, the animals were killed with sodium thiopental (30–40 mg/kg body weight) to harvest the kidneys and obtain blood samples.
Determination of PMB concentration in kidneys
The left kidneys which were stored at −20°C were thawed at room temperature, then minced and homogenized accordingly in a 1:6 (g: mL) ratio of renal tissue to normal saline. The renal tissue homogenate (200 µL) was transferred to a microcentrifuge tube, into which 40 µL of sulfosalicylic acid (100%, w/v) was added. The microcentrifuge tube was oscillated for 30 s and centrifuged at 6640 × g for 10 min, and the upper supernatant was retained for high-performance liquid chromatography tandem mass spectrometry analysis. 18 A MSLAB-C18 (50 mm × 4.6 mm) column was used for the analysis. Gradient chromatography was performed with 0.1% formic acid in water (A) and 0.1% formic acid in acetonitrile (B) at a flow rate of 0.8 mL/min. The gradient used was 0 min, 5% B; 1 min, 25% B; 1–4 min, 95% B; 4-7 min, 5% B; 7.1 min, stop. The injection volume was 10 µL.
Positive ion electrospray tandem mass spectra were recorded with the multiple reaction monitoring detection mode. The ion spray voltage was set at 5000 V, and the probe temperature was set at 450°C. Nitrogen was used as the collision gas. The nebulizer (GS1), curtain, and turbo gas (GS2) were set at 55, 10, and 60 psi, respectively. The declustering potential, collision energy, collision cell exit potential, and entrance potential were set at 50, 25, 10, and 10 V, respectively. The molecular ion and daughter ion for polymyxin B1 were 602.5 and 241.2, respectively. It should be noted that the content of polymyxin B1 determined represented that of PMB in the present experiment as polymyxin B1 as the main component of PMB which consisted of polymyxin B1, B2, B3, and Ile B1. 19
The lower limit of quantification was found to be 30 ng/mL for PMB based on a 10:1 ratio of the signal to noise. The precision of the method was within the acceptance range of 0–15%.
Functional evaluation of nephrotoxicity
After the injection on the 4th day, rats were immediately placed in metabolic cages to collect urine for 24 h. The volume of urine was then measured and aliquots were stored at −80°C to analyze NAG excretion. NAG in the urine was measured by a DNM-9602 microplate reader (Perlong Medical, Beijing, China) using the NAG assay kit.
Blood samples were collected following puncture of the abdominal aorta. The blood samples were transferred into pro-coagulation tubes and centrifuged for 10 min at 7968 × g and then stored at 4°C to determine SCr, BUN, and serum β2-MG with a Mode 7600 Automatic Analyzer (HITACHI, 7600-010, Japan) using the creatinine reagent kit, urea nitrogen test kit, and β2-MG test kit the following day.
Renal histological examination
The right kidneys which were fixed in 4% neutral formalin solution were processed conventionally for fluorescent microscopic observation according to a standard pathological procedure. 18 A semiquantitative evaluation of renal tissue pathology was performed using previously published criteria. 18 The following six indices: tubular brush border loss, renal tubular edema, tubule epithelial cell vacuolar degeneration, renal tubular epithelial cell necrosis, renal tubular dilatation, and interstitial inflammation were used to evaluate renal tubular toxicity. Scores obtained were defined as follows: 0 = no pathological change and 1 = appearance of any one of the above six indices. Five representative and non-overlapping fields of view were chosen from the center and four corners of a tissue slice to evaluate the scores according to these six indices by one pathologist in a blinded fashion. The total scores in each field of view were obtained by adding the scores of these six indices. The higher the total score, the greater the renal insult.
Statistical analysis
Values are expressed as means ± standard deviations (SD). The statistical significance of differences between mean values was analyzed by the nonpaired t-test. Multiple comparisons were performed using analysis of variance. Differences were considered significant at p < 0.05 or p < 0.01.
Results
Animal response to PMB
Following the administration of PMB 0.2 mL (0.2 mg), the rats in all groups showed auricle redness and slightly dispirited consciousness, which quickly recovered within 10 s. The rats were subsequently administered the remaining dose of PMB and behaved normally.
Renal tissue concentration of PMB
As shown in Table 1, PMB concentration in renal tissue in the PMB group (36.38 ± 3.81 μg/g renal tissue) were significantly higher than those in the PMB + cyto c 100 mg/kg body weight group (22.16 ± 2.24 μg/g renal tissue) and the PMB + cyto c 200 mg/kg body weight group (22.94 ± 3.48 μg/g renal tissue) 6 h after the last injection on the 5th day (p < 0.01). Cyto c at 50, 100, and 200 mg/kg body weight decreased the accumulation of PMB in the kidney by 18.5%, 39.1%, and 36.8%, respectively.
The amount of PMB accumulated in rat kidneysa (n = 6).
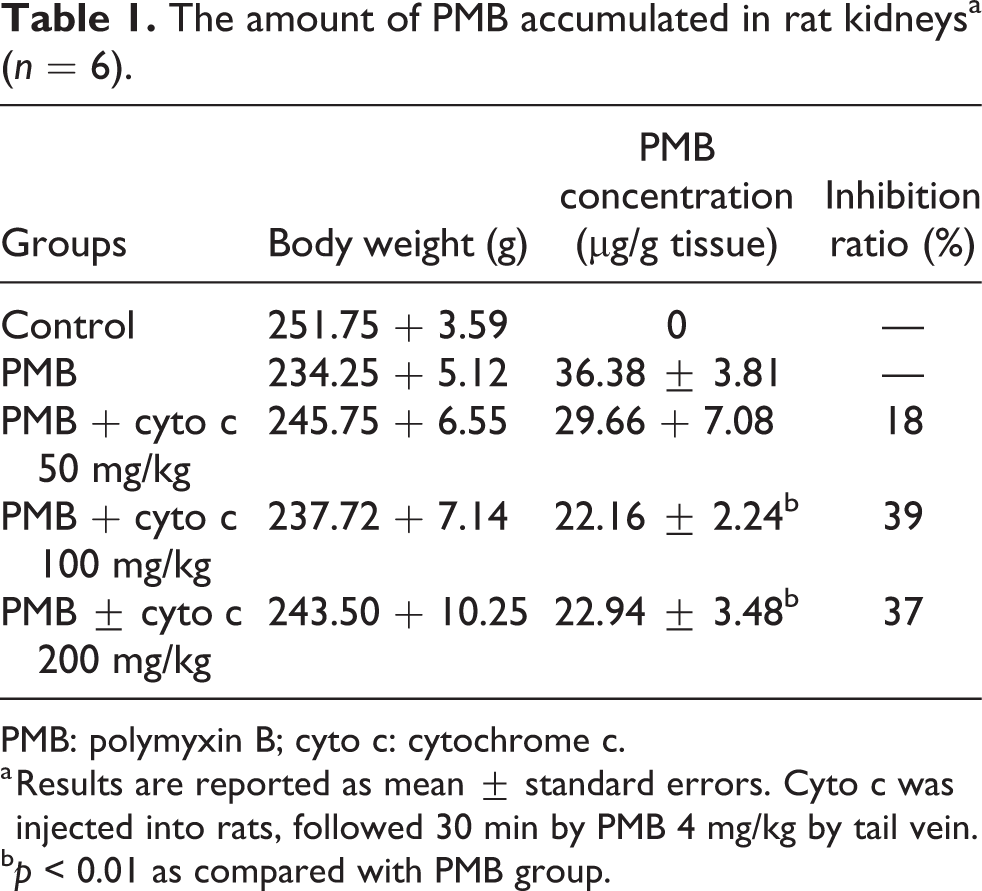
PMB: polymyxin B; cyto c: cytochrome c.
a Results are reported as mean ± standard errors. Cyto c was injected into rats, followed 30 min by PMB 4 mg/kg by tail vein.
b p < 0.01 as compared with PMB group.
The effect of PMB on the level of urinary NAG
The 24-h urinary NAG excretion after day 4 was significant higher in the PMB group (1.78 ± 0.27 U/day) than in the control group (0.87 ± 0.24 U/day) (p < 0.01, Figure 1). Cyto c (50, 100, and 200 mg/kg/day) resulted in a significant decrease in urinary NAG excretion (1.38 ± 0.22, 1.06 ± 0.13, and 1.06 ± 0.07 U/day, respectively, Figure 1). Cyto c (50, 100, and 200 mg/kg/day) decreased urinary NAG excretion by 22.5% (p < 0.05), 40.4% (p < 0.01), and 40.4% (p < 0.01), respectively, compared with the PMB group. There was no significant difference between PMB + cyto c 100 mg/kg body weight group or PMB + cyto c 200 mg/kg body weight group and control group (p > 0.05), respectively.
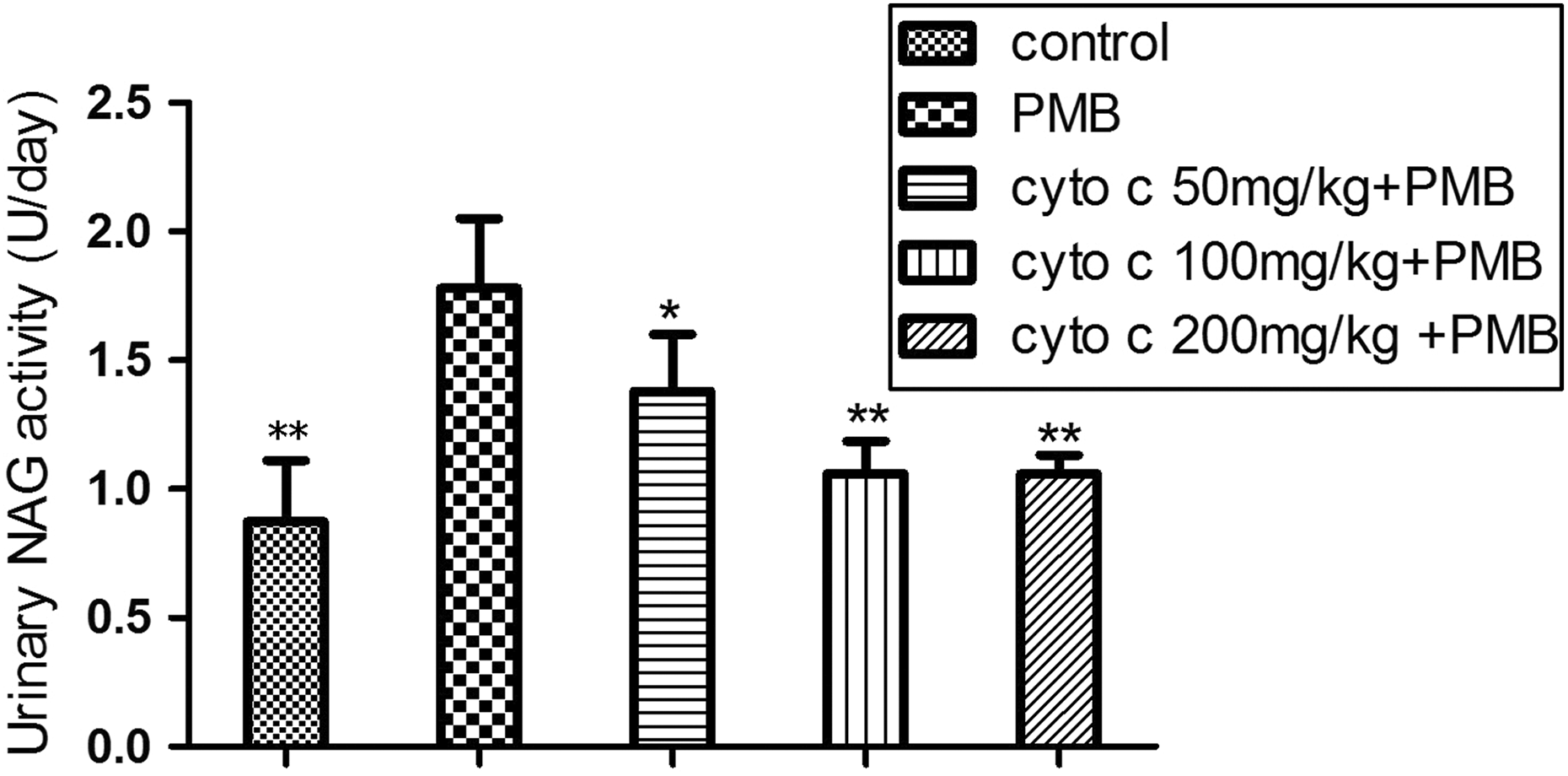
Effects of cyto c on urinary NAG excretion induced by PMB 4 mg/kg. Results were expressed as mean ± SD for six animals in each group. *p < 0.05 (versus PMB group); **p < 0.01(versus PMB group). PMB: polymyxin B; cyto c: cytochrome c; NAG: N-acetyl-β-D-glucosaminidase; SD: standard deviation.
The levels of SCr, BUN, and serum β2-MG
The levels of SCr, BUN, and serum β2-MG were not significantly different among control group, PMB group, and PMB + cyto c 50, 100, or 200 mg/kg group (p > 0.05), respectively, 6 h after the last administration on the 5th day (Table 2).
Comparison of BUN, SCr, and β2-MG in rats of all groups.a
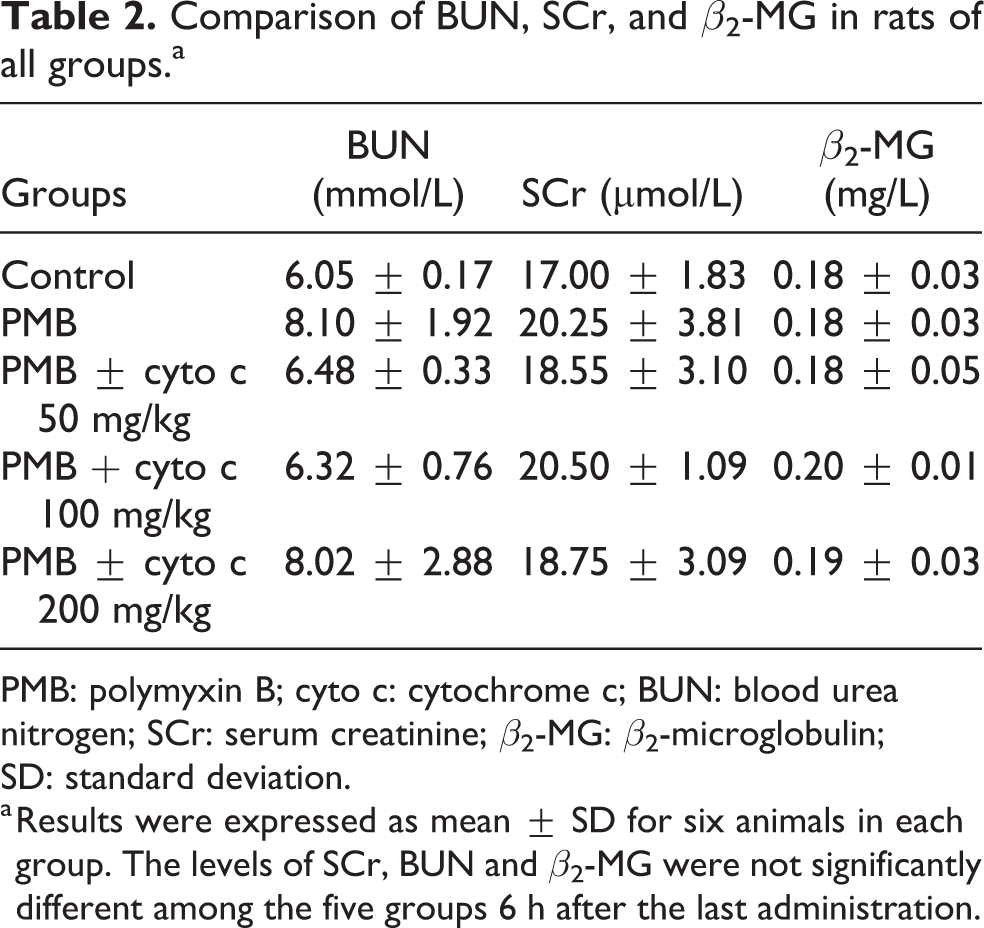
PMB: polymyxin B; cyto c: cytochrome c; BUN: blood urea nitrogen; SCr: serum creatinine; β2-MG: β2-microglobulin; SD: standard deviation.
a Results were expressed as mean ± SD for six animals in each group. The levels of SCr, BUN and β2-MG were not significantly different among the five groups 6 h after the last administration.
Renal histological examination
The histopathological changes in kidneys were evaluated as described in the “Materials and methods” section. As shown in Figure 2, renal glomeruli and renal tubules in rats receiving saline injection showed to be normal under fluorescent microscope (Figure 2(a)). However, after PMB treatment, histopathological changes included tubule epithelial cell vacuolar degeneration, renal tubular edema, tubular brush border loss, renal tubular dilatation, interstitial inflammation, or renal tubular epithelial cell necrosis (Figure 2(b)). There was a trend of histopathological changes in the PMB + cyto c 50 mg/kg body weight group for reduction compared with the PMB group, but moderate renal tubular dilatation and tubular brush border loss were still present (Figure 2(c)). In the PMB + cyto c 100 mg/kg body weight and PMB + cyto c 200 mg/kg body weight groups, kidney damage induced by PMB was markedly reduced as kidney morphology appeared almost normal except for mild renal tubular dilatation and edema (Figure 2(d) and (e)).
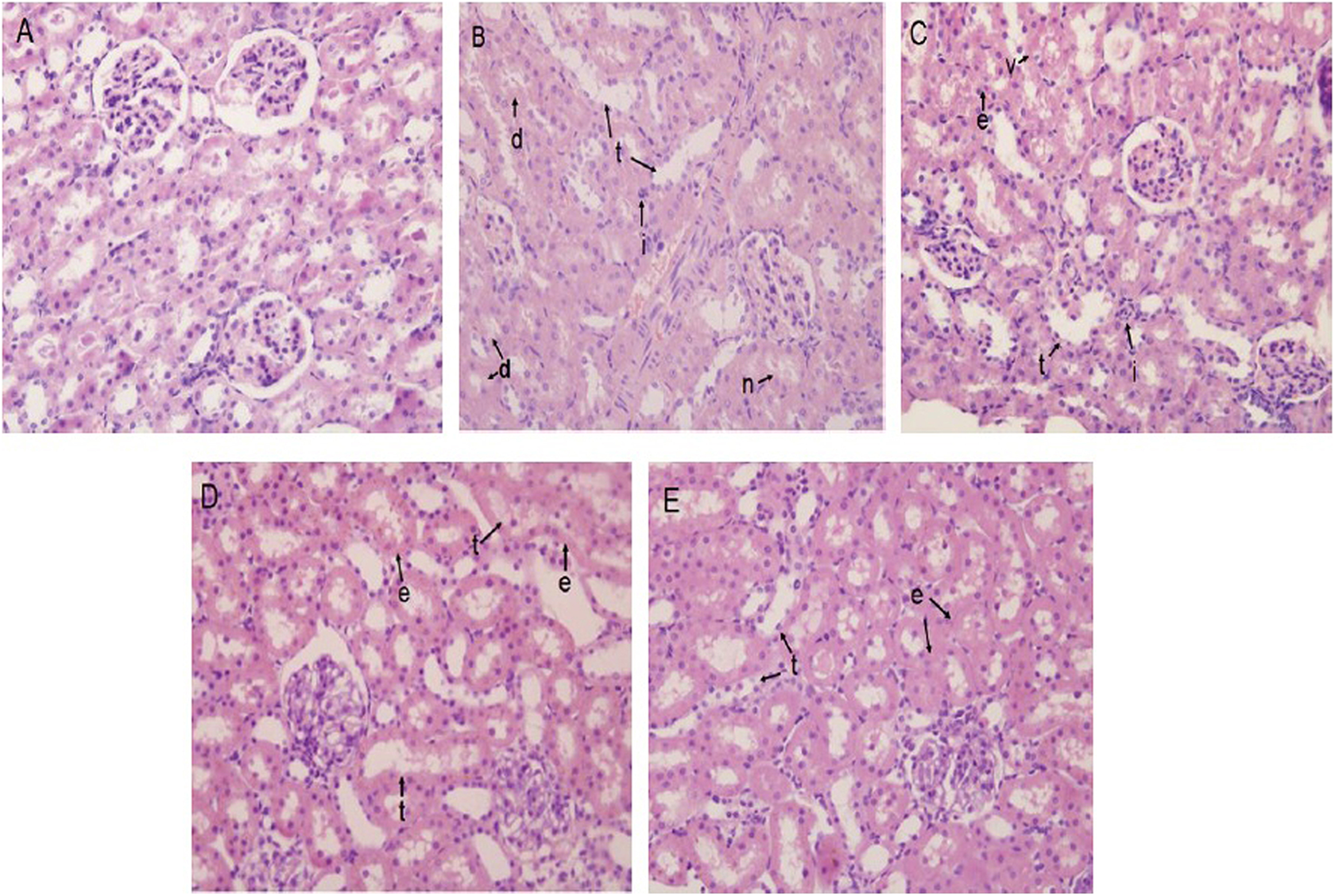
Histopathological observation of renal sections (H&E staining, ×400). (a) Control group, (b) PMB group, (c) PMB + cyto c 50 mg/kg group, (d) PMB + cyto c 100 mg/kg group, (e) PMB + cyto c 200 mg/kg group. e: renal tubular edema; d: tubular brush border desquamation; i: interstitial inflammation; n: renal tubular epithelial cell necrosis; t: renal tubular dilatation; v: tubule epithelial cell vacuolar degeneration; PMB: polymyxin B; cyto c: cytochrome c.
Semiquantitative analysis showed that the scores for renal tissue damage in the PMB + cyto c 100 mg/kg body weight and the PMB + cyto c 200 mg/kg body weight groups were significantly lower than that in the PMB group (p < 0.01) (Table 3).
Semiquantitative analysis of renal tissue insult of rats in each group.a
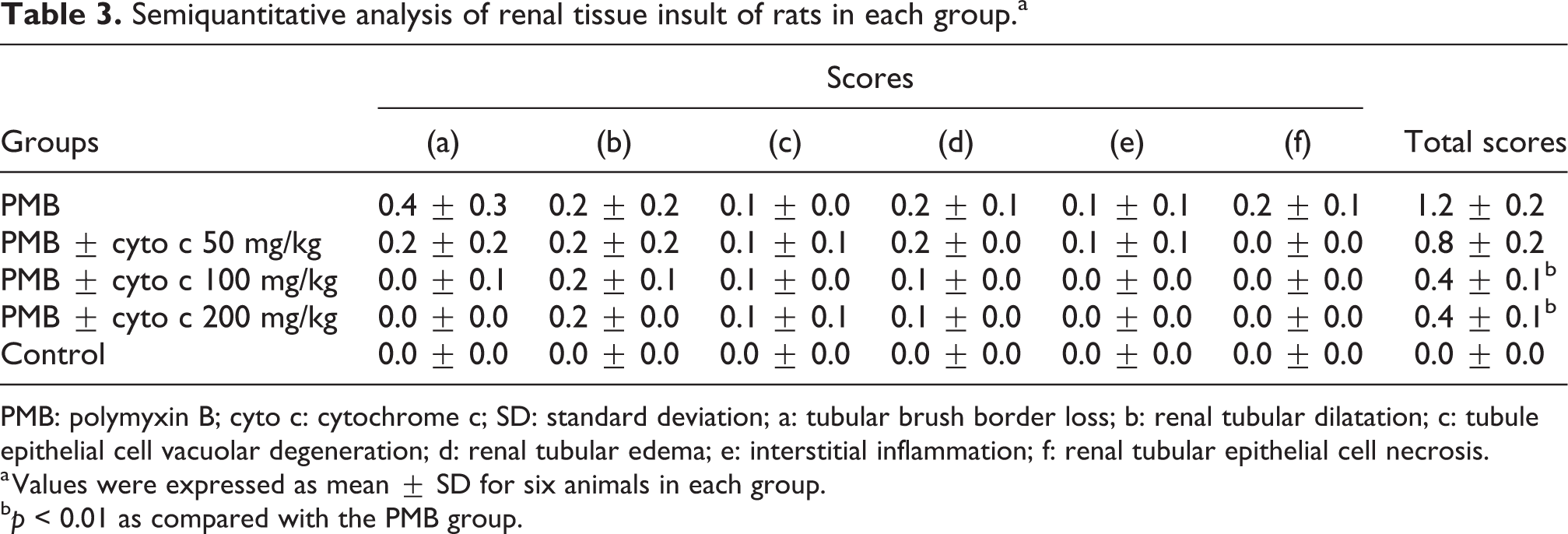
PMB: polymyxin B; cyto c: cytochrome c; SD: standard deviation; a: tubular brush border loss; b: renal tubular dilatation; c: tubule epithelial cell vacuolar degeneration; d: renal tubular edema; e: interstitial inflammation; f: renal tubular epithelial cell necrosis.
a Values were expressed as mean ± SD for six animals in each group.
b p < 0.01 as compared with the PMB group.
Association between urinary NAG or renal pathology scores and renal PMB concentrations
As shown in Figure 3, the amount of NAG excretion or renal pathology scores decreased with reduced accumulation of PMB in the kidney, with linear equations as follows: y = 0.0512x − 0.102 (R 2 = 0.9927) or y = 0.0575x − 0.8977 (R 2 = 0.9979), respectively.

Association of urinary NAG value or pathological insult scores with PMB concentrations in kidney. NAG: N-acetyl-β-D-glucosaminidase; PMB: polymyxin B.
Discussion
It is unclear whether megalin may play an important role in the renal accumulation of PMB and whether exogenous cyto c can effectively prevent PMB nephrotoxicity. Therefore, the effects of exogenous cyto c on the renal accumulation and nephrotoxicity of PMB were determined in a rat model in the present study.
We found that the kidney concentration of PMB at 4.0 mg/kg body weight/day administered for consecutive 5 days into the tail vein was 36.4 ± 3.8 μg/g kidney tissue and was higher than that (13.04 ± 3.09 μg/g kidney tissue) reported by Abdelraouf et al., 11 who administered PMB at 4.0 mg/kg body weight intravenously only once, indicating that PMB may accumulate in the kidney. When cyto c and PMB were sequentially administered, cyto c at 50, 100, and 200 mg/kg body weight was found to decrease the accumulation of PMB in the kidney by 18.5%, 39.1%, and 36.8%, exhibiting a dose-dependent relation. These results showed that megalin may contribute to kidney accumulation of PMB, and cyto c may competitively block the binding of PMB to megalin. The remaining PMB that was not accumulated in the kidney was believed to be eliminated in the urine. 16
In the present study, it was also found that the effectiveness of cyto c at 100 mg/kg body weight was similar to that of cyto c at 200 mg/kg body weight, showing that the antagonistic effect of cyto c at more than 100 mg/kg body weight on the binding of PMB to megalin reached saturation. Based on the higher dose of cyto c used in the present study, we believe that the affinity of PMB to the receptor appeared to be much higher than that of cyto c, consistent with the findings reported by Suzuki et al. 16 In addition, cyto c at 100 or 200 mg/kg body weight, a dose with maximal antagonistic effect, inhibited PMB accumulation in kidney only by 39.1% or 36.8%, respectively, indicating that besides megalin, other transporter routes such as human oligopeptide transporter 2 (PEPT2) may also mediate renal uptake of the drug. 20
We then examined the effect of cyto c on PMB-induced nephrotoxicity by measuring urinary NAG excretion, an early and sensitive biomarker for proximal tubular cell damage. 16,21 –23 Rats were injected intravenously with PMB at 4.0 mg/kg body weight/day for consecutive 4 days (cumulative dose, 16 mg/kg body weight), and the 24 h urinary NAG excretion was twice as high as that in control rats, which was similar to the results reported by Yousef et al., 23 who found that the 24 h urinary NAG excretion in rats treated with colistin (cumulative dose, 32.5 mg/kg body weight) was 3.8-fold higher than that in control rats on day 6, 23 but was different from the results reported by Suzuki et al, 16 who found that when the rats were intravenously injected with colistin at 1.0 mg/kg body weight only once, the 24 h urinary NAG excretion was three times higher than that in control rats. These differences may be related to different observation time, different experimental conditions, and different operators. Sequential administration of cyto c at 50, 100, and 200 mg/kg body weight and PMB significantly attenuated PMB-induced NAG content by 22.4%, 40.5%, and 40.6%, respectively, which were slightly higher than the results reported by Suzuki et al, 16 who found that coadministration of cyto c at 100 mg/kg and colistin at 1.0 mg/kg body weight significantly decreased 24 h urinary NAG excretion by 30.2%. These results indicated that cyto c at higher dose may reduce renal tubular toxicity of PMB.
SCr, BUN, and serum β2-MG are common biochemical markers reflecting glomerular damage. 24 We found that there were no significant difference in SCr, BUN, and serum β2-MG among the groups, which was similar to the results reported by Abdelraouf et al., 21 who found no significant fluctuation in SCr in any of the rats treated with PMB at 5 mg/kg body weight daily for up to 10 days. These findings indicated that PMB administered at a lower dose such as 4 or 5 mg/kg body weight daily may not result in injury to renal glomeruli.
In this study, kidney pathological changes in PMB-treated rats included severe tubular brush border loss, renal tubular dilatation, moderate tubule epithelial cell vacuolar degeneration, renal tubular edema, mild interstitial inflammation, and renal tubular epithelial cell necrosis, but the glomeruli remained almost intact. These findings were similar to those reported by Abdelraouf et al., 21 indicating that renal tubules are the main target sites of PMB-induced renal damage. In addition, we found that cyto c at 100 and 200 mg/kg body weight administered 30 min prior to PMB administration, resulted in mild renal tubular dilatation and edema (Table 3 and Figure 2), indicating that cyto c at a higher dose can reduce the above pathological insults.
It is noteworthy that the extent of decreased NAG excretion or renal pathological insults was found to be consistent with that of reduced PMB accumulation in the kidney when cyto c at 50, 100, and 200 mg/kg body weight administered 30 min prior to PMB administration in the present study (Figure 3), proving that megalin was crucial for PMB accumulation in kidney and indicating that the prevention of PMB nephrotoxicity may be realized by blocking the receptor megalin, although PEPT2 might also mediate renal uptake of the drug. 20
In summary, we found that cyto c administration at higher dose may reduce PMB-induced nephrotoxicity such as urinary NAG or renal tubular pathological insults by decreasing PMB renal accumulation when PMB was repeatedly administered in vivo. These results further proved that PMB uptake in the kidney was via megalin. In China, the highest dose of exogenous cyto c used in clinical practice is approximately 1 mg/kg daily according to its product information leaflets, significantly lower than that (100 mg/kg daily) used in the present study, showing that cyto c may not be an effective antagonist for PMB nephrotoxicity clinically. Thus, our future study will include the following aims: (1) to screen different megalin ligands for effective inhibition of PMB uptake with drugs used clinically to prevent nephrotoxicity in vivo; 2) to test for gene polymorphisms expressing megalin to personalize the choice of PMB, that is, to use PMB clinically if the genotype shows low expression of megalin.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
