Abstract
It is known that women develop alcoholic liver injury more rapidly and have a lower alcohol toxic threshold than men. However, the detailed molecular mechanisms remain unclear. The precise mechanism responsible for the sex difference needs to be determined. Female and male mice were given ethanol by intragastric infusion every day for 4 weeks. The pathological changes were detected by hematoxylin–eosin, Sirius red, oil red O, periodic acid–Schiff, and Hochest33258 staining in the liver of female and male mice. The related gene and protein expression of hepatocytes stress, proliferation and apoptosis, glycogen synthesis, lipid metabolism, and hepatic fibrosis were also systematically analyzed in the female and male mice. Livers from ethanol-treated female mice had more serious hepatocyte necrosis, liver fibrosis (P < 0.01), substantial micro/macrovesicular steatosis (p < 0.01), glycogen consumption (p < 0.05), and hepatocytes apoptosis (p < 0.05) than ethanol-treated male mice. The expression of heat shock protein 27 (HSP27), HSP70, proliferating cell nuclear antigen, B-cell lymphoma/leukemia-2 (Bcl-2), and phosphorylated signal transducer and activators of transcription 3 (p-STAT3) was higher in ethanol-treated male mice than ethanol-treated female mice (P < 0.05 or P < 0.01). But, the expression of Bax (Bcl-2-associated X protein), Caspase 3, CYP2E1 (cytochrome P4502E1), and transforming growth factor βl had the contrary results. Our study suggested that ethanol treatment induced more expression of HSP27 and HSP70, faster hepatocyte proliferation, higher level of glycogen, and interleukin-6 signaling pathway activation, but less hepatocyte apoptosis and CYP2E1 expression in male mice than female mice, which could be helpful to understand the molecular mechanism for the influence of sex difference on alcoholic liver injury.
Introduction
Alcoholic liver disease (ALD) is a major cause of illness and death in China. ALD ranges from fatty liver alone to progressive fibrosis, cirrhosis, and, eventually, hepatocellular cancer. Many of these conditions can be induced by feeding mice and rats with diets that are high in fat or sucrose with ethanol. 1 In humans, women develop alcohol-induced liver injury more rapidly than men. 2 –4 Additionally, the progression of liver injury is greater in women with alcoholic hepatitis who stop or reduce drinking. 5 A number of studies showed sex-related differences in the response of the liver under various types of stress. 6 Differences in the level of circulating sex hormones, 7 hepatic expression of sex hormone receptors, 8 and pattern of growth hormone secretion 9 are postulated to be responsible for the sex dimorphic pathophysiology.
Microarray analysis has been used to analyze the sex differences in hepatic gene expression in a rat model of ethanol-induced liver injury. 10,11 Sharma et al. and Tadic et al. identified some differential expression of hepatic genes in both sexes of rats during ethanol-induced liver injury. 10,11 But they did not systematically analyze the molecular mechanism for the influence of sex difference on alcoholic liver injury.
Stress (heat shock protein 27 (HSP27) and HSP70), proliferation (proliferating cell nuclear antigen (PCNA)), apoptosis (Bax and Bcl-2), metabolism (CYP2E1), collagenous fiber synthesis (transforming growth factor βl (TGF-βl)), and signaling pathway (p-STAT3)-related genes and proteins were systematically detected in the male and female mice during alcoholic liver injury, which will help to better understand the molecular mechanism for the influence of sex difference on alcoholic liver injury.
Materials and methods
Animals and alcoholic liver injury induction
Male and age-matched female BALB/c mice (approximately 6- to 8-week old and weighing 22 ± 2 g) were purchased from the experimental animal center of Henan province and maintained in an air-conditioned animal room at 25°C with free access to water and food under 12-h light/dark cycles. All animals were acclimatized to the environment for 1 week prior to the experiment. Three groups of female mice (10 mice/group) and three groups of male mice (10 mice/group) were designated as follows: normal feeding (NF), normal feeding plus water intake by intragastric infusion (NFW), or normal feeding plus 56% (v/v) ethanol (the red star erguotou white wine, 56°C, Beijing, China) intake (0.15 ml/10 g weight) by intragastric infusion (NFE) every day for 4 weeks. The given dose of ethanol could significantly induce liver injury for 4 weeks in mice. 12 All animals were killed at the end of the experiment (nine in the morning after 4 weeks) by ether inhalation and exsanguination via the abdominal aorta. After euthanization, blood samples were collected from each animal and livers harvested. Part of the liver was used for histopathological analysis, while the remaining part was immediately frozen in liquid nitrogen for subsequent analysis. The study was conducted in accordance with the Guide for the Care and Use of Laboratory Animals (Eighth Edition) 13 and approved by the Committee of Animal Experimentation of Henan University of Science and Technology (Reference number, HUST13Q0518).
Serum AST and ALT activity detection
Blood samples were collected from the mice at the end of the experiment (4 weeks). Instead of harvesting the liver and later collecting blood from already dead mice, the blood was left to clot at room temperature for approximately 15–30 min. After complete clotting, it was rimmed using an applicator stick and then centrifuged for 10 min at 2500 r/min. Then, serum was separated. Serum aspartate transaminase (AST) and alanine transaminase (ALT) activities were determined with a commercial assay kit (Nanjing Jiancheng Biological Technology, Inc., China) by colorimetric method. Enzyme activities were expressed as an international unit per liter (IU/L).
Histopathological examination
Liver specimens were obtained from the mice at the end of fourth week after the experiment. Samples of liver were fixed in 10% formaldehyde for 24 h and then dehydrated and embedded in paraffin. Then, 6-µm-thick sections were cut from each paraffin-embedded tissue and stained with hematoxylin–eosin (H&E). Sections were examined microscopically for specific staining, and photographs were taken using a digital image-capture system (MoticamPro Motic, Xiamen, China). Necrosis was graded 1 if there was one focus or two and 2 if more than two foci per low-power field were found. Focal necrosis was graded as 3. Ten low-power fields were examined per liver. 14,15
Sirius red staining
The histopathological sections of each group were stained with Sirius red to observe changes of collagen fibers. The 6-µm-thick sections of liver tissues were stained with Celestine blue dye (Amresco, Shanghai, China) for 5–10 min and washed with distilled water 5 times for 2 min. Sirius red saturated solution was treated for 10–20 min after conventional dewaxing and hydration. The changes of collagen content in liver slices were observed under a microscope (MoticamPro Motic) and taken pictures according to a unified parameter. Integrated optical density of collagen fibers in the liver was measured for each mouse by ImagePro Plus 6.0 software (Media Cybernetics, Rockville, Maryland, USA). We examined 10 low-power fields in the liver of each mouse. 16
Oil red O staining
The liver tissues of each group were fixed in liquid nitrogen and freeze-dried. Then, 10-µm-thick sections were cut from each frozen tissue and stained with oil red (Amresco). The changes of lipid droplets in liver tissues were observed under a microscope (MoticamPro Motic) and taken pictures according to a unified parameter. Steatosis score in the liver was measured for each mouse. Ten low-power fields were examined per liver, and the degree of lipid infiltration was graded from 0 to 4 according to Järveläinen et al., 15 with 0 depicting no fat present and 4 depicting that >75% of cells contain fat.
PAS staining
Glycogen was stained within the liver with periodic acid–Schiff (PAS) staining as described by Li et al. 17 Shortly, tissue sections were incubated in 0.8% periodic acid (Sigma, Shanghai, China) followed by incubation in Schiff’s reagent (Sigma). Then, sections were counterstained with hematoxylin (Amresco). Firstly, the pictures received uniform white balance processing and double color segmentation and, secondly, received optical density analysis using Motic Images Advanced 3.2 software (MoticamPro Motic). 16 We examined 10 low-power fields in the liver of each mouse.
Apoptosis analysis
Apoptosis in the hepatocytes of mice in different groups was further analyzed using Hochest33258 staining (Amresco). We use the test lattice to count the rate of apoptosis. Briefly, the test lattice has 121 landing points. We counted the number of apoptosis-positive cells in each field of vision and divided them by 121, which was the rate of the apoptosis. Ten low-power fields were examined per liver under a fluorescence microscope (Olympus, Tokyo, Japan), and apoptosis rates were counted for each mouse. 17
Western blot of HSP27, HSP70, Bax, Bcl-2, Caspase 3, CYP2E1, TGF-βl, and p-STAT3 in the liver of mice
Protein samples of 70 μg from the mice in different groups were adjusted to the composition of the electrophoresis sample buffer (50 mM Tris, pH 6.8, 10% glycerol, 5% beta-mercaptoethanol, 2% sodium dodecyl sulfate (SDS), 0.1% bromphenol blue) and boiled for 5 min prior to analysis. SDS-polyacrylamide gel electrophoresis (PAGE; 10% polyacrylamide gels) in 1 mm slab gel was performed as described by Li et al. 18 The proteins were transferred from the gel to the nitrocellulose membranes. Then, the membrane was probed with a monoclonal antibody to mouse PCNA, Bcl-2, Bax, Caspase 3, HSP70, HSP27, VEGF, and CYP2E1 (Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA)). The signal was detected by horseradish peroxidase detection system using 3,3′-diaminobenzidine (Sigma). Protein bands were quantified with Gel Pro Analyzer software 4.0 (Media Cybernetics Inc., Bethesda, Maryland, USA), and the intensities of the bands were normalized against beta-actin (protein value = intensity of target protein/intensity of beta-actin). Each experiment was repeated for three times.
Statistical analysis
All data were presented as the mean ± standard deviation. Statistical comparisons were made using two-way analysis of variance with the Tukey–Kramer test for multiple comparisons. All statistical analyses were performed using SPSS 13.0 (SPSS Inc., Chicago, Illinois, USA).
Results
Effect of water or ethanol treatment on hepatic enzyme markers
The levels of AST and ALT were significantly upregulated in the liver of mice that received ethanol treatment when compared with the normal or control group (p < 0.05 or p < 0.01) (Figure 1). The levels of AST and ALT were significantly higher in the female group than in the male group after ethanol treatment (p < 0.01) (Figure 1).
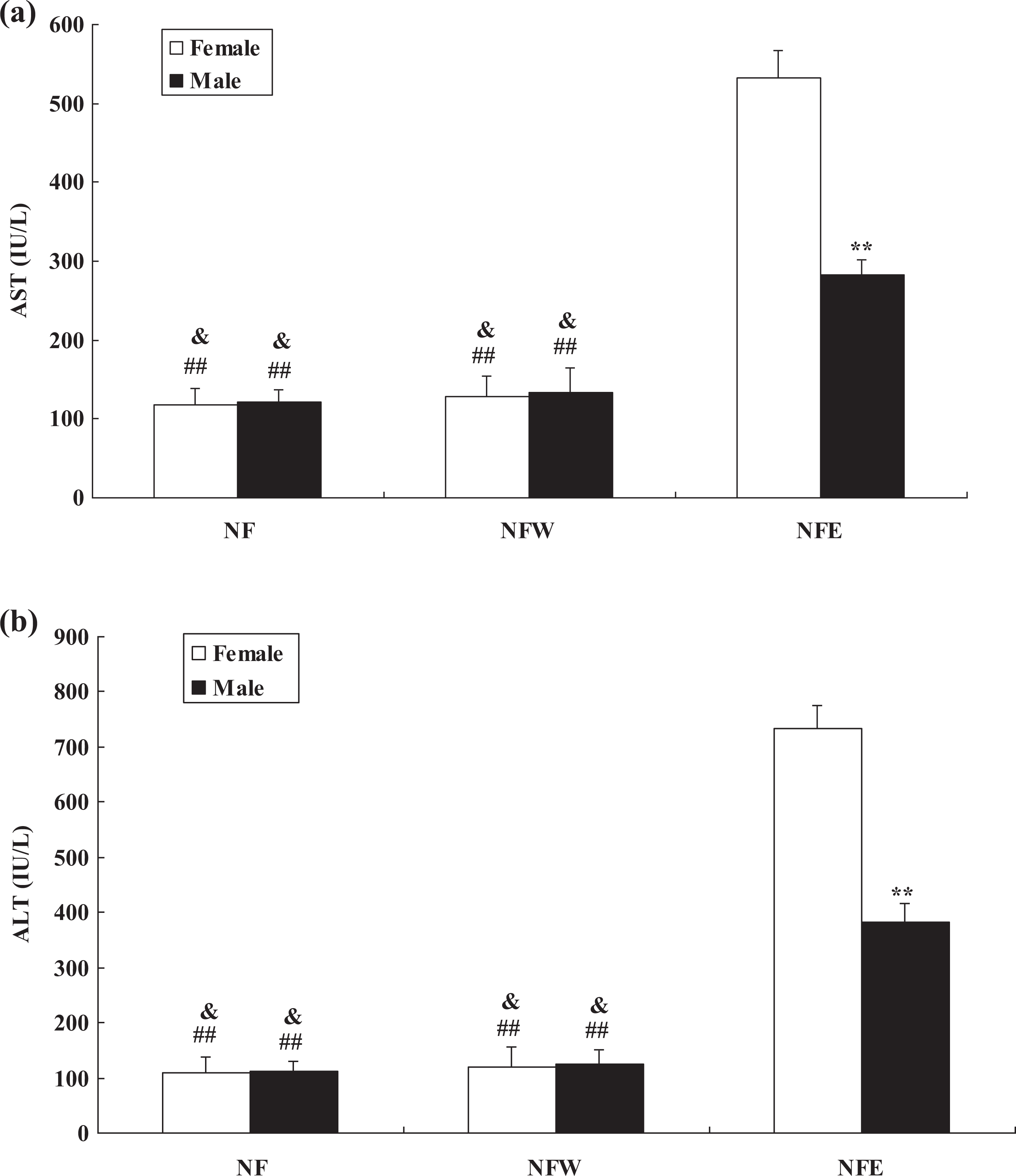
Serum AST (a) and ALT (b) levels in the mice at the end of 4 weeks after water or ethanol treatment. **p < 0.01: significant difference in the female mice when compared with the male mice. ##p < 0.01: significant difference in the NFE female group when compared with the NF group or NFW group. &p < 0.05: significant difference in the NFE male group when compared with the NF group or NFW group.
Histopathological studies
Ethanol treatment significantly induced liver injury in the mice compared with the normal and control group (p < 0.05 or p < 0.01) (Figure 2(a) to (f)). The necrosis score was significantly higher in the liver of female mice than male mice after ethanol treatment (p < 0.01) (Figure 2(g)). Livers from ethanol-treated female mice had more serious liver fibrosis (p < 0.01) (Figure 3) and substantial micro/macrovesicular steatosis (p < 0.01) (Figure 4) than ethanol-treated male mice.

Histological examination of liver injury in the mice by hematoxylin–eosin staining at the end of 4 weeks after water or ethanol treatment (a to f). (g) Necrotic score. The arrow indicates the site of pathological change. **p < 0.01: significant difference in the female mice when compared with the male mice. ##p < 0.01: significant difference in the NFE female group when compared with the NF group or NFW group. &p < 0.05: significant difference in the NFE male group when compared with the NF group or NFW group (scale bar: 50 µm).

The changes of collagen fibers in the liver of mice at the end of 4 weeks after water or ethanol treatment (a to f). The arrow indicates the site of collagen fibers. g: IOD of collagen fibers in the liver was measured for each mouse by ImagePro Plus 6.0 software (Media Cybernetics, Maryland, USA). **p < 0.01: significant difference in the female mice when compared with the male mice. ##p < 0.01: significant difference in the NFE female group when compared with the NF group or NFW group. &p < 0.05: significant difference in the NFE male group as compared with the NF group or NFW group (scale bar: 50 µm).

The changes of steatosis in the liver of mice at the end of 4 weeks after water or ethanol treatment (a to f). (g) Steatosis score in the liver was measured for each mouse. The arrow indicates the site of steatosis. All data were presented as the mean ± standard deviation. **p < 0.01: significant difference in the female mice when compared with the male mice.##p < 0.01: significant difference in the NFE female group when compared with the NF group or NFW group. &p < 0.05: significant difference in the NFE male group when compared with the NF group or NFW group (scale bar: 50 µm).
Detection of glycogen by PAS staining
Our results indicated that ethanol exposure led to glycogen consumption in the liver of female and male mice (Figure 5). The content of glycogen was higher in the liver of male mice than female mice before and after ethanol treatment (p < 0.05) (Figure 5).

Glycogen content assessment of liver sections from the mice at the end of 4 weeks after water or ethanol treatment by PAS staining assay (a to f). (g) The pictures received uniform white balance processing, double color segmentation, and optical density analysis using Motic Images Advanced 3.2 software (Motic China Group Co., Ltd, Xiamen, China). The arrow indicates the site of glycogen. *p < 0.05: significant difference in the female mice when compared with the male mice. ##p < 0.01: significant difference in the NFE female group when compared with the NF group or NFW group. &&p < 0.01: significant difference in the NFE male group when compared with the NF group or NFW group (scale bar: 50 µm). PAS: periodic acid–Schiff.
Apoptotic studies
Our results showed that ethanol treatment remarkably induced hepatocytes apoptosis compared with the normal and control group (p < 0.05 or p < 0.01) by Hochest33258 staining (Figure 6). Livers from ethanol-treated female mice had more hepatocytes apoptosis than ethanol-treated male mice (p < 0.05) (Figure 6).

Apoptosis analysis of liver sections prepared from the mice at the end of 4 weeks after water or ethanol treatment by Hochest33258 staining (a to f). (g) Apoptosis rate of apoptosis-positive cells among total hepatocytes. The arrow indicates the site of apoptotic hepatocytes. **p < 0.01: significant difference in the female mice when compared with the male mice. ##p < 0.01: significant difference in the NFE female group when compared with the NF group or NFW group. &p < 0.05: significant difference in the NFE male group when compared with the NF group or NFW group. Each experiment was repeated three times (scale bar: 50 µm).
Western blot of HSP27, HSP70, PCNA, Bcl-2, Bax, Caspase 3, CYP2E1, TGF-βl, and p-STAT3 in the liver of mice
The expression of HSP27, HSP70, Bax, Caspase 3, CYP2E1, TGF-βl, and p-STAT3 was significantly upregulated, but the expression of PCNA and Bcl-2 was significantly downregulated in the liver of ethanol-treated mice compared with the normal and control group (p < 0.05 or p < 0.01) (Figure 7). At the same time, HSP27, HSP70, PCNA, Bcl-2, and p-STAT3 were highly expressed in ethanol-treated male mice than ethanol-treated female mice (p < 0.05 or p < 0.01) (Figure 7). But the expression of Bax, Caspase 3, CYP2E1, and TGF-βl had the contrary results (Figure 7).

The influence of ethanol on the expression of HSP27, HSP70, PCNA, Bcl-2, Bax, Caspase 3, CYP2E1, TGF-βl, and p-STAT3 in the liver of mice at the end of 4 weeks. The expression of these molecules was detected by Western blot (a). The protein bands were quantified for HSP27 (b), HSP70 (c), PCNA (d), Bcl-2 (e), Bax (f) Caspase 3 (g), CYP2E1 (h), TGF-βl (i), and p-STAT3 (j) with the Gel-Pro Analyzer 4.0 software (Media Cybernetics Inc.), and the intensities of the bands were normalized against β-actin. AU represents arbitrary unit. *p < 0.05 or **p < 0.01: significant difference in the female mice when compared with the male mice. #p < 0.05 or ##p < 0.01: significant difference in the NFE female group when compared with the NF group or NFW group. &p < 0.05 or &&p < 0.01: significant difference in the NFE male group when compared with the NF group or NFW group.
Discussion
Women develop alcoholic liver injury more rapidly and have a lower alcohol toxic threshold than men. The mechanism for this difference is poorly characterized, and differences in first-pass metabolism in the stomach, or elimination rate, or alcohol distribution volume in the body have been suggested. 19 Our study systematically analyzed the molecular mechanism for the influence of sex difference on alcoholic liver injury in mice.
Our results showed that the expression of HSP27 and HSP70 was remarkably induced in the male mice than the female mice after ethanol treatment. It has been suggested that the primary function of HSPs is to serve as molecular chaperones in which they recognize and bind nascent polypeptide chains and partially folded intermediates of proteins, preventing their aggregation and misfolding, or as chaperonins that facilitate protein folding directly. 20,21 Molecular chaperones are necessary not only for the folding of newly synthesized proteins in the cell but also for the protection of proteins during exposure to stressful situations such as heat shock, which causes proteins folded previously to unfold. 22,23 Therefore, the expression of HSP27 and HSP70 was significantly induced in the male mice, which help them to resist the liver injury induced by ethanol treatment than female mice. This may be an important reason to interpret that women develop alcoholic liver injury more rapidly than men.
Our study further indicated that there was a significant decrease in the level of expression of PCNA in the liver of female and male mice after ethanol treatment. The expression of PCNA was higher in the male mice than the female mice after ethanol treatment. PCNA is a subunit of the mammalian DNA polymerase delta and is synthesized primarily during the S phase of the cell cycle. 24 PCNA is a relay or an anchoring molecule that functions as a molecular integrator for proteins involved in the control of the cell cycle, DNA replication, DNA repair, and cell death. 25,26 PCNA has been shown to be a good marker to distinguish proliferating cells. 27,28 Our results proved that the proliferation of hepatocytes in male mice was better than female mice, which led to accelerating the liver repair in male mice than female mice. This is another reason that women develop alcoholic liver injury more rapidly than men.
Hepatocytes apoptosis is an important influence factor during liver injury. Our results showed that ethanol treatment remarkably induced hepatocytes apoptosis compared with the normal and control group. Livers from ethanol-treated female mice had more hepatocytes apoptosis than ethanol-treated male mice. These explained that the liver injury in female mice was more serious than male mice due to more hepatocytes apoptosis after ethanol treatment. In addition, apoptosis-related factors were differently expressed in female and male mice after ethanol treatment. The expression of Bax and Caspase 3 was significantly upregulated in the liver of ethanol-treated mice compared with the normal and control group. At the same time, these molecules were highly expressed in ethanol-treated female mice than ethanol-treated male mice. But the expression of Bcl-2 had the contrary results compared with abovementioned molecules in the liver of mice after ethanol treatment. Caspase 3 is a member of the interleukin (IL)-1 beta-converting enzyme or cell death effector-3 family, which is involved in the induction of apoptosis and has been considered to be correlated with apoptosis because of the most downstream enzyme in their apoptosis-inducing pathway. 29,30 Bax is an important proapoptotic marker and Bcl-2 is an important antiapoptotic marker, which play important roles in regulating apoptosis. 31 The higher expression of Bax and the lower expression of Bcl-2 in female mice than male mice were important reasons that led to difference of hepatocyte apoptosis in alcohol-injured mice. So, ethanol treatment influenced the apoptosis of hepatocytes by regulating the expression of Caspase 3, Bax, and Bcl-2 in female and male mice, which further influenced the difference of liver injury during ethanol intake in female and male mice.
Metabolism was also differently influenced in female and male mice during ethanol treatment. The expression of CYP2E1 was more significantly induced in the liver of female mice than male mice. CYP2E1, a microsomal enzyme involved in xenobiotic metabolism and generation of oxidative stress, has been implicated in promoting liver injury. 32 Wong et al. 33 have demonstrated that CYP2E1 plays a major role in carbon tetrachloride (CCl4) toxicity based on a previous study with Cyp2e1-null mice. Dey and Cederbaum 34 reported that induction of CYP2E1 promoted liver injury in obese mice. Dey and Kumar 32 believe that changes in regulation of CYP2E1 under hyperglycemic conditions are closely linked with increased oxidative stress and injury in liver. So, ethanol treatment induced more CYP2E1 expression with oxidative stress increasing in female mice, which led to aggravating liver injury. Lipid metabolism and glycogen synthesis were also affected by ethanol treatment in female and male mice. Livers from ethanol-treated female mice had more serious substantial micro/macrovesicular steatosis than ethanol-treated male mice, which indicated that ethanol treatment could influence liver injury by regulating lipid metabolism in female and male mice. Our results also indicated that ethanol exposure led to glycogen consumption in the liver of female and male mice. The content of glycogen was still higher in the liver of male mice than female mice before or after ethanol treatment, which suggested that the high level of glycogen in male mice may be helpful in accelerating liver repair after ethanol treatment.
Livers from ethanol-treated female mice had more serious liver fibrosis than ethanol-treated male mice. The expression of collagen content and TGF-β1 in female mice was significantly higher than those in male mice. The deposition of collagen fibers is an important process of hepatic fibrosis. The increase of collagen fibers in liver tissue and hepatic fibrosis was mainly promoted by hepatic stellate cell (HSC). 35 In the process of liver injury, HSC is the target of cytokines released by inflammatory cells and regenerated liver parenchyma cells. In many media that promote the increase of collagen fiber, TGF-β1 has the strongest effect. 35 TGF-β1 could stimulate HSC to produce the extracellular matrix, which accelerates the production and accumulation of collagen fibers. 36 So, ethanol treatment induced more TGF-β1 expression to stimulate HSC to produce the extracellular matrix in female mice, thereby accelerating the production and accumulation of collagen fibers and promoting liver fibrosis in female mice compared with male mice.
In addition, the expression of p-STAT3 was significantly induced in the liver of ethanol-treated mice compared with the normal and control group. At the same time, p-STAT3 was highly expressed in ethanol-treated male mice than ethanol-treated female mice. The expression of IL-6 was significantly increased in liver diseases, such as hepatitis, alcoholic liver, and liver cancer and was closely related to the occurrence and development of liver diseases. 37 –39 Studies have shown that the degree of liver damage was significantly increased in IL-6 knockout mice during CCl4-induced chemical liver injury, 40 which suggests that IL-6 plays an important role in the protection of liver in CCl4-induced liver injury in mice. It is currently believed that IL-6 can stimulate the proliferation of hepatocytes through the activation of related signal pathways and play a role of protective factor for liver cells. 41 In addition, IL-6 could also reduce the apoptosis of hepatocytes by maintaining the expression of several antiapoptotic factors. 42 p-STAT3 is an important marker for activation of IL-6 signaling pathway. 40 Our results suggested that IL-6 signaling pathway played more important protective roles in ethanol-induced liver injury in male mice than female mice. Besides these, p-STAT3 is involved in the regulation of hepatocyte regeneration after liver injury and promoting the regeneration of hepatocyte. 43,44 Our results indicated that p-STAT3 promoted the regeneration of hepatocyte more quickly in ethanol-treated male mice than ethanol-treated female mice, which suggested that the high level of p-STAT3 in male mice may be helpful in accelerating liver repair after ethanol treatment.
In conclusion, our study suggested that ethanol treatment induced more expression of HSP27 and HSP70, faster hepatocyte proliferation, higher level of glycogen, and IL-6 signaling pathway activation but less hepatocyte apoptosis and CYP2E1 expression in male mice than female mice. These could be helpful to understand the molecular mechanism for the influence of sex difference on alcoholic liver injury. Women should pay more attention to the liver injury caused by alcohol.
Footnotes
Acknowledgments
This work was supported by the Science & Technology Innovation Program in Universities of Henan Province (#18IRTSTHN026), Outstanding Youth of Science and Technology Innovation in Henan Province (#184100510006), the innovation team of Henan University of Science and Technology (#2015XTD003), and Henan province’s Key Project of tackle key problems of science and technology (#172102310693). The authors thank all the members who assisted in the laboratory to carry out the experiments. The experiments comply with the current laws of China.
Authors’ contributions
San-Qiang Li and Ping Wang contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
