Abstract
Diquat (1,1′-ethylene-2,2′-bipyridinium ion; DQ) is a nonselective quick-acting herbicide, which is used as contact and preharvest desiccant to control terrestrial and aquatic vegetation. Several cases of human poisoning were reported worldwide mainly due to intentional ingestion of the liquid formulations. Its toxic potential results from its ability to produce reactive oxygen and nitrogen species through redox cycling processes that can lead to oxidative stress and potentially cell death. Kidney is the main target organ due to DQ toxicokinetics and redox cycling. There is no antidote against DQ intoxications, and the efficacy of treatments currently applied is still unsatisfactory. The aim of this work was to review the most relevant human and experimental findings related to DQ, characterizing its chemistry, activity as herbicide, mechanisms of toxicity, consequences of poisoning, and potential therapeutic approaches taking into account previous experience in developing antidotes for paraquat, a more toxic bipyridinium herbicide.
Introduction
Over the past few decades, the use of pesticides, namely herbicides, has been increasing worldwide. 1 Herbicides are essential in agriculture to improve crop yields, forestry, urban settings, and to control weeds in aquatic systems. However, the inadequate and indiscriminate application of herbicides can generate serious food safety issues and environmental problems, namely due to contamination of aquatic systems by discharge of these substances from manufacturing facilities and/or surface runoffs into natural water resources. 2,3 Herbicides are generally selective to weeds, because they inhibit plant-specific pathways. They may be grouped into several classes, according to their selectivity, activity (i.e. control, suppression, crop safety, and defoliant), timing of application (i.e. preplant, preemergence, and postemergence), method of application (i.e. soil or foliar applied), mechanism of action (i.e. lipid synthesis inhibitors, amino acid synthesis inhibitors, growth regulators, photosynthesis inhibitors, pigment inhibitors, cell membrane disrupters, seedling root/shoot growth inhibitors, nitrogen metabolism inhibitors), or chemical structure (e.g. phenoxyacids, bipyridinium, dinitroaniline, chloroacetamide, carbamate, phenyl acetic acid, benzonitrile, urea, uracil, glyphosate, triazine, and pthalic acid). 4
Diquat (1,1′-ethylene-2,2′-bipyridinium; DQ; Figure 1), is a nonselective, defoliant, preharvest and desiccant herbicide that belongs to the bipyridinium class. It was first synthesized by Dr Fielden in the laboratories of the Dyestuffs Division of the Imperial Chemical Industries (Blackley, England). 5 At that time, it was known that the quaternary ammonium compounds, a class of surface-active agents, had more potent phytotoxic effects than anionic or nonionic substances, which then raised the interest of creating a program to evaluate their herbicidal activity. Hence, in 1955, in a series of investigations at Jealott’s Hill Research Station (Bracknell, England), after observing that quaternary ammonium germicides, as cetyl trimethylammonium bromide, were able to desiccate young seedling plants, Brian et al. 5 discovered the herbicide potential of DQ. This led then Homer et al. 6 to develop a program to synthesize quaternary ammonium salts of isomeric bipyridinium substances and to evaluate their herbicidal activity. Regarding DQ, they noticed that there was a relationship among the herbicidal effect and its specific chemical structure, as the activity was only present when: (i) there was a bridge between the pyridine rings, (ii) that bridge was formed by only two carbon atoms, and (iii) all structures assume a coplanar configuration. Furthermore, they verified that DQ can be easily reduced to a free radical monocation (DQ+●), and this process had a key role in its herbicidal effects, which is more pronounced at daylight than in darkness. Aerobic conditions, high humidity, and temperature were also implicated in the increased herbicidal activity. 6,7 DQ was first introduced in the market by Imperial Chemical Industries in 1958. 8
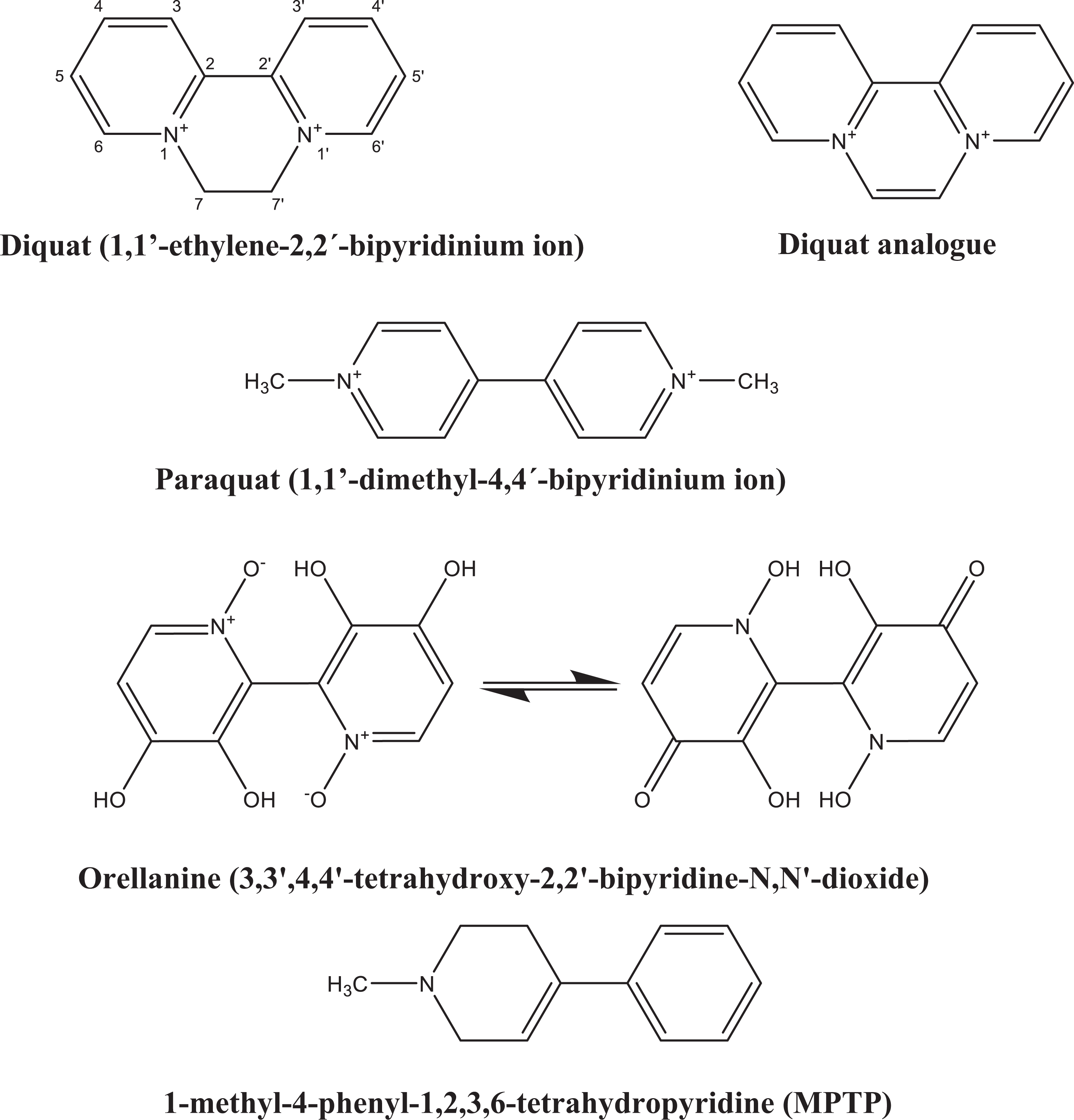
Chemical structures of DQ, PQ, orellanine, and MPTP. DQ: diquat; PQ: paraquat; MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine.
The first report of DQ poisoning dates 1968, when a young boy, aged 18 years, took accidentally a mouthful of undiluted 20% formulation. 9 Since then, at least 60 cases of DQ poisoning worldwide were reported in the scientific literature (Table 1). Most cases derived from suicidal intents by ingestion of concentrated liquid products containing DQ. Accidental ingestion have also occurred, although rarely, as a result of decanting DQ liquid concentrates into soft drink bottles or equipment failure and violations of label requirements during agricultural practice. 50 Treatment usually includes administration of inhibitors of DQ-mediated enzymatic reactions or the use of enzymatic and low-molecular antioxidants. Other therapeutic strategies include either decreasing absorption or enhancing excretion. However, these are mainly ineffective in reducing the DQ toxicity and the development of an antidote formulation is of paramount importance to prevent morbidity and mortality.
Reported cases of DQ poisoning.
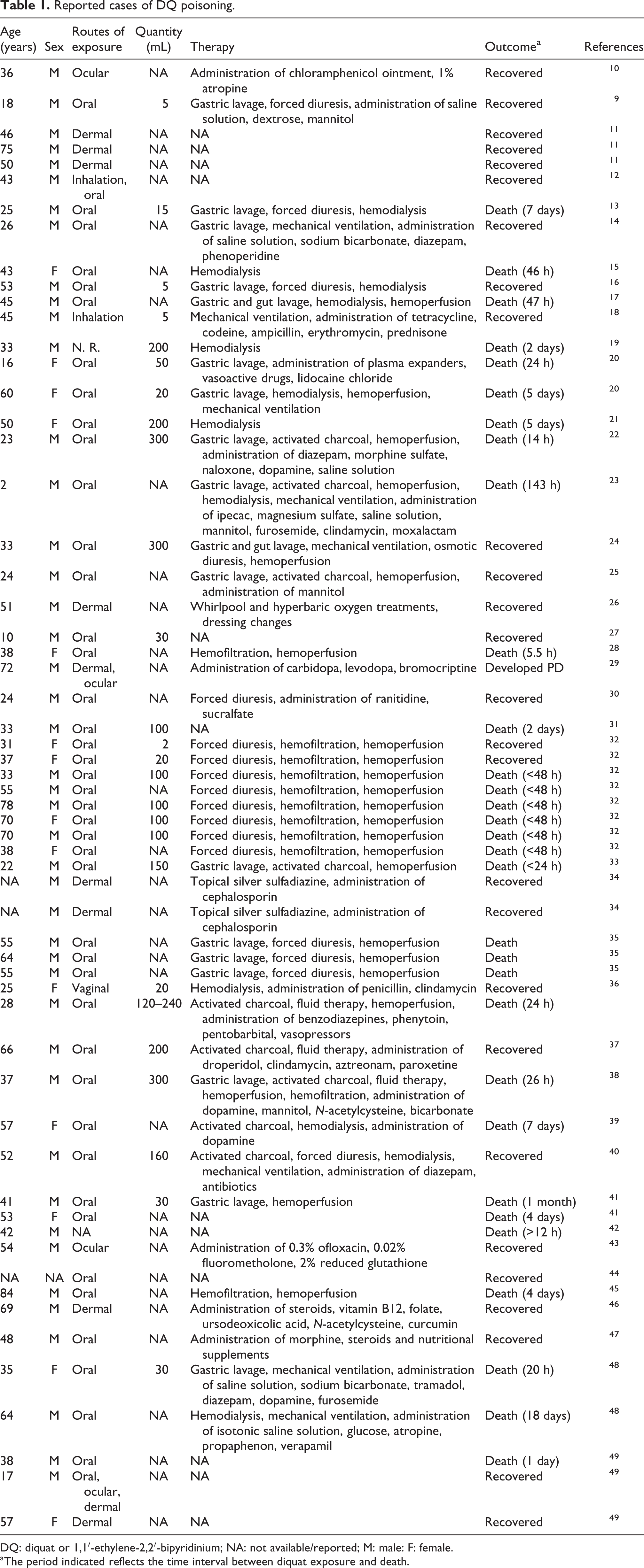
DQ: diquat or 1,1′-ethylene-2,2′-bipyridinium; NA: not available/reported; M: male: F: female.
aThe period indicated reflects the time interval between diquat exposure and death.
The aim of this work was to provide a comprehensive review of the published literature related to DQ, namely regarding its chemistry, activity as herbicide, mechanisms of toxicity, poisoning outcomes, and possible therapeutic approaches. Indeed, despite all treatments already described, frequently, it is not possible to prevent a fatal outcome caused by DQ poisoning due to the absence of an antidote or effective treatment. It is expected that our previous experience focusing the toxicology of paraquat (PQ; another bipyridinium), namely the development of efficient treatments, may offer further insights into and discussions about the toxicology of DQ aiming to reduce morbidity and mortality.
Methods
An English exhaustive literature search was performed in PubMed (US National Library of Medicine) and patents to identify all articles containing relevant information about preclinical, clinical, and experimental toxicology of DQ, without a limiting period. Furthermore, from the retrieved journal articles, electronic copies of the full papers were obtained and reviewed. Also, books on pesticides were reviewed to find additional information.
Physical and chemical properties and formulations
DQ belongs to the bipyridinium chemical class. It is composed by pyridine rings (i.e. two aromatic rings in which one carbon atom is replaced by a nitrogen atom coupled by a saturated (CH2) n bridge; in this case, n = 2 (i.e. ethylene group; Figure 1). This makes it a highly polar divalent cationic compound that is easily reduced to an unstable DQ+●. 51 Further reduction yields polyhydrobipyridyl derivatives and atmospheric oxygen regenerates DQ2+. The distance between the two nitrogen atoms is about 3.5 Å. 52 If n = 3 or 4, the compounds would be triquat or tetraquat, respectively. Also, there is another analogue of DQ which contains an unsaturated (CH)2 bridge. 53 All of these belong to a quaternary ammonium salts (“quats”) group, together with other herbicides such as PQ and difenzoquat, and some growth regulators such as chlormequat and mepiquat. The physical and chemical properties of DQ2+ are summarized in Table 2.
Physical and chemical properties of DQ.

DQ: diquat or 1,1′-ethylene-2,2′-bipyridinium.
The herbicide occurs mainly in the form of highly hygroscopic salts, supplied mainly as aqueous solutions of dibromide salt monohydrate, although it can also be associated with chlorine, iodine, methylsulfate, or sulfate anions (the active part of the herbicide is the DQ2+ itself, whereas the anion does not have any rule). 54,55 It is sold either alone or in combination with other herbicidal agents, namely with PQ, amitrole, terbutylazine, or simazine. The most common trade name is Aquacide®.
DQ is classically synthetized in two steps: (i) pyridine is oxidatively coupled to 2,2′-bipyridine over a heated Raney nickel catalyst and (ii) quaternization of 2,2′-bipyridinium with ethylene dibromide (1,2-dibromoethane) to form the ethylene bridge, as shown in Figure 2. 56 The carcinogen ethylene dibromide is an essential starting substance for the DQ dibromide production and remains as a manufacturing impurity in DQ-containing liquid formulations. Notwithstanding, it is not considered to be a threat to human health because it dissipates from products with time. 8,10,50,57,58

Synthesis of DQ. DQ: diquat or 1,1′-ethylene-2,2′-bipyridinium.
DQ dibromide, in particular, has an appearance of crystalline yellow powder with no characteristic odor and is completely dissociated in aqueous solution over the full pH range. 59 Aqueous solutions of DQ are nonvolatile, nonflammable and nonexplosive. Moreover, it is very stable under neutral and acidic conditions but readily degrades in alkali solutions above pH 9 due to the uptake of a single OH− molecule, which ruptures one of the pyridine rings. 59
Formulations of 20% (m/v) are particularly common. More concentrated formulations need to be diluted before use mainly due to the corrosive effects. 60 When dealing with these products, inadvertent spills and splashes can expose the handler to the herbicide. Wearing personal protective equipment is recommended 60 : (i) chemical-resistant and waterproof rubber gloves, headgear, footwear, and socks; (ii) long-sleeved shirts and long impermeable pants; (iii) chemical-resistant apron; (iv) eye protection; and (v) filter respirator.
Mode of action as herbicide
Easiness of handling (crystalline salts), high solubility in water (for preparation of spraying solutions), low vapor pressure (minimal fumes), high binding capacity (inactivation after soil binding), and quick desiccation/action (once photosynthesis starts), all make DQ an appropriate herbicide for agricultural needs. It is particularly useful for pasture renovation and to protect fruit orchards and diverse plantation crops (e.g. potato, vine, banana, rice and sunflower), presenting a weed and grass species-dependent effectiveness. It can also act as defoliant for cotton, soybeans and hops and diminish the moisture content of grain, saving costs of seed drying and advancing the rice harvest season without influencing seed germination. 50,55,61 After application, DQ penetrates rapidly through the leaf surface. This absorption may increase with high light intensity, humidity, and the specific adjuvant that is incorporated in que liquid formulation, usually the surfactant Agral [nonionic liquid containing 90% (v/v) alkyl phenol ethylene oxide], which ensures a good penetration of DQ in the foliage. 55,62 DQ is referred as a desiccant because it can quickly dry out an entire plant, due to the rupture of cell membranes that allows water to escape, and it is mainly used for preharvest desiccation, aiding the harvesting of seed and fodder crops. 50,55,61 Because it is strongly adsorbed to soil, DQ is not taken up by plant roots.
DQ is also applied in various aquatic reservoirs, such as lakes, ponds, drainage ditches, among others, to control floating, emerging, and submersed weeds. This is of great importance, as aquatic weeds can impair the recreational value of those aquatic systems, block irrigation channels, hinder navigation, and damage pumping stations and hydroelectric turbines. The efficacy of DQ as herbicide may be potentiated by the association of copper sources, as was demonstrated by Garlich et al. 63 in some algae, although the application of chemical products needs to be cautious and moderate, concerning environmental safety and health. The same is true for the use of DQ, as it was proved that it can exert toxic effects in some nontarget species like Lymnaea stagnalis freshwater snails, 64 Stizostedion vitreum walleye, Micropterus salmoides largemouth bass and Micropterus dolomieu smallmouth bass fish, 65 and Hyalella azteca amphipods, 66 having an average threshold toxicity to fish of about 20 ppm, which means that the toxicity is reduced by a factor of two comparatively to PQ. 67 Eastern spiny softshell turtles (Apalone spinifera spinifera) are not sensitive to DQ. 68 Moreover, the efficacy of DQ is reduced at lower water temperatures 69 and higher levels of turbidity. 70 Young et al. 71 determined taste and odor thresholds in drinking water for DQ dibromide of at least 56 µg L−1 and 8900 µg L−1, respectively, although these values cannot be associated with the potential toxicity of the herbicide.
After application, penetration through the leaf surface occurs almost immediately. This absorption is increased by high light intensity and humidity and by added nonionic adjuvants in the formulation, which ensure good spray retention and wetting of target foliage. 55
Photosystem II is inhibited by a great variety of herbicides, and the photosystem I electron pathway is diverted by bipyridinium herbicides. 72 Herbicidal activity, as well as DQ-induced toxicity to mammals, was found to be linked to DQ redox potential. 73 Indeed, once it is applied as an herbicide to manage terrestrial and aquatic vegetation, DQ2+ is reduced from the primary electron acceptor of photosystem I inside chloroplasts, thus generating reactive oxygen species (ROS) and inhibiting photosynthesis through chlorophyll breakdown, ion leakage, and DNA fragmentation. Initial work on the mode of action of bipyridinium herbicides by Mees 74 indicated that their ability to cause rapid kill is dependent not only on the photosynthetic activity of plants (sunlight) and on O2 but also on humidity and temperature. 6 Zweig et al. 75 further found that bipyridinium herbicides cause a deviation of electron flow from photosystem I (which normally transfers its electron to ferredoxin), leading to an inhibition of oxidized nicotinamide adenine dinucleotide phosphate (NADP+) reduction during photosynthesis (Figure 3). During this process, DQ●+ is produced in the cell at the expense of nicotinamide adenine dinucleotide phosphate (NADPH). 77 Thus, DQ is only toxic to the green parts of the plant, where the photosynthesis occurs. 78 DQ2+ is then rapidly reoxidized by the O2 produced in chloroplasts. 78 In the reoxidization, superoxide radical (O2 ●−) is generated, with the subsequent oxidative deleterious effects and consequent cell death. This redox cycle occurs until the supply of free electrons ceases. 79 Therefore, the mechanism of toxic action of DQ involves cyclic reduction–oxidation reactions, which produce ROS and depletion of reduced NADPH.

Herbicide mechanism of DQ. In PSI, PC transfers its electron (e−) through a series of steps (P700, A0, A1, Fe-S, FA, FB) to FRD and finally to NADP+. DQ2+ binds near the FRD binding site in PSI and accepts an e −, becoming diquat monocation free radical (DQ+●), which initiates a series of reactions leading to cell membrane disruption and plant death. The formation of such free radicals stops electron transport to NADP+ and effectively inhibits normal functioning of PSI. A0: modified chlorophyll a—electron acceptor chlorophyll; A1: phylloquinone; Fe-S: iron-sulfur proteins; PsaA, PsaB, PsaC, PsaD, PsaE: polypeptides subunits of PSI; A: ferrodoxin-NADP+ reductase; granum: stack of thylakoids. DQ: diquat or 1,1′-ethylene-2,2′-bipyridinium; PSI: photosystem I; PC: plastocyanin; FRD: ferrodoxin; NADP+: oxidized nicotinamide adenine dinucleotide phosphate. Adapted from Dinis-Oliveira et al. 76
Other applications
It was reported that DQ was effective in the treatment of columnaris, a disease that affects a diversity of fish species all over the world. 80 The etiological agent of this illness, the Flavobacterium columnare, is a bacterium that damages fish fins, gills, and skin by external and systemic infection. Affected fish can present frayed necrotic fins with gray to white margins, necrotic and depigmented skin, and also white to brown necrotic areas in the gills. The efficacy of the treatment was possibly due to the antibacterial effect of DQ on F. columnare that have colonized on external surfaces. As DQ is a divalent cationic compound, it has low permeability and absorption in gill epithelia and skin of infected fish. Notwithstanding, further studies should be done to optimize dose concentration and to evaluate repeated treatments and duration. 80,81
Inactivation in contact with soil
Some studies have reported that DQ is more environment-friendly than PQ with comparable herbicide use rate and efficacy, proving to be a valid alternative. 61 Due to its cationic nature, after the release into the environment, DQ residues may strongly adhere to clay minerals and organic matter present in the soil, such as montmorillonite 82 and sepiolite, 83 forming stable electrostatic interactions through base exchange between their cationic group and the carboxyl groups on those substances. 84 Although this prevents DQ from microbial degradation, it also limits its biological activity, as it becomes inactivated and diminishes its environmental mobility. In addition, they are not taken up by plant roots due to their strong adsorption and deactivation by soil colloids. DQ can, however, be taken up by the roots of plants growing in places without adsorbing colloids, such as pure sand, leading consequently to phytotoxicity. 7,40,85,86 On the other hand, up to 5% of DQ residues can be transported within soils, mainly through leaching, surface runoff, and drain-flow pathways. After spraying the herbicide into terrestrial areas, the reentry interval is about 24 h.
Some studies regarding the analysis of treated water show that DQ residues, after sprayed into the water, rapidly disappear in a few days due to their rapid absorption and concentration in the aquatic plants, remaining attached until the decaying weeds disintegrate into the bottom mud. Then, instead of returning to water, the residues adhere strongly to the mud. 81 Submerged weeds generally decay in situ after herbicide treatment, not being removed from the water. In an area where weed growth has been substantial, the uptake of the oxygen from the water in the decay process may diminish its content to such low levels that could be lethal to fish. Therefore, it is necessary to apply DQ as far as possible before weed grows in a great extent and in some parts of the reservoir at different times, not over the entire area at once, so that fish have time to move to untreated areas when the dissolved oxygen significantly drops. DQ is more effective in slow-moving or still water rather than in streams or moderate-to-rapid flow canals. 8,81 Of note, various ecotoxicological studies have shown that DQ, in subacute concentrations, did not cause severe effects in some fish species, such as piauçu (Leporinus microcephalus), Nile tilapia (Orechromis niloticus), and guppy (Phallocerus caudimaculatus), being a proper product for the control of aquatic macrophytes. 87 –89 The treated water can be used for domestic purposes, crop irrigation, and animal consumption only after 14 days of herbicide application and for swimming after 24 h. 22,90,91
UV-mediated degradation on plant surfaces and in water
DQ it is not metabolized in plants. Indeed, when several plants are exposed to DQ and maintained in the dark, no breakdown of the herbicide occurred. 92 However, when exposed to sunlight, it can be extensively decomposed photochemically by ultraviolet (UV) radiation on plant surfaces and in water at a rate of about 10% per year. 93 Although slow, the degradation in soil is significant over a long period and represents the most important process for removing or negating the effects of DQ on the environment. The photooxidative breakdown of one phenyl ring leads to the formation of the major degradation product 1,2,3,4-tetrahydro-1-oxopyrido[1,2-a]-5-pyrazinium ion (TOPPS), which is then degraded to picolinamide and via picolinic acid to volatile compounds. The photochemical degradation products of DQ (including those formed in water) are shown in Figure 4. The adsorption and photodegradation processes are thus important for removing the effects of DQ on the environment. 91,94

Mammals’ metabolism and photochemical UV degradation on plant surfaces and in water of DQ. UV: ultraviolet; DQ: diquat or 1,1′-ethylene-2,2′-bipyridinium.
DQ is also degraded by several soil common microorganisms when incubated in culture solution with bacteria, fungi, and yeast. 95 However, since it is firmly adsorbed to clay colloids, degradation is very slow and claimed to be 5–7% per year. 91 Although slow, it has been postulated that this rate of degradation is sufficient to ensure that DQ residues do not accumulate in the soil. 81,96
Toxicokinetics
The toxicokinetics of DQ has been studied in a variety of animal species, especially rats, 54 goat, 97 cows, 98 hens, and rabbits. 99 Figure 5 presents the toxicokinetics of DQ and respective percentages of the administered dose.

Toxicokinetics of DQ and respective percentages of the administered dose. DQ: diquat or 1,1′-ethylene-2,2′-bipyridinium.
Absorption
DQ can be absorbed by oral, pulmonary, ocular, or dermal routes. Poisoning by ingestion of concentrated formulations is more common than other routes. 100 Volumes of 5–50 mL are typically ingested, but fatalities usually occur for volumes greater than 15 mL of a 20% concentrated formulation. 100 DQ is poorly absorbed (<10%) from the gastrointestinal tract (GIT) and excreted largely unchanged in the feces. Also, the absorption may significantly decrease with the presence of food and intestinal microorganisms that can degrade the herbicide. DQ can cross the cell membranes, although slowly due to its hydrophilic nature, mostly by diffusion and, in lesser extent, by active transport via cation pumps. 91,101,102
Absorption from the lung after inhalation and by skin seems also to be very low due to its hydrophilic nature, meaning that systemic toxicity is unlikely through these routes. The penetration of acidic and basic pesticides through skin can be influenced by the skin surface pH, which is weakly acidic (pH 4.2–5.6). DQ exist as fixed charged cations and remain dissociated at all pH values. A study from Feldmann and Maibach 103 showed that after the application of DQ on skin at a dose of 4 µg cm−2 in acetone, only 0.3% was absorbed, which demonstrates that skin is a strong barrier against DQ. DQ absorption increased to 1.4% with occlusion and to 3.8% if the skin was damaged.
Dermal exposure to foliar residues is less significant than during handling with concentrated formulations of DQ. Notwithstanding, prolonged contact with these products leads to skin irritation, which facilitates the penetration of the herbicide through the skin, rising to 3.8% in 24 h. Moreover, Charles et al. 104 proved that DQ was absorbed 26 to 37 times more slowly through the lungs than other quaternary ammonium compounds, with the absorption limited to the porous regions of the membranes. Fatalities resulting from intramuscular injection, vaginal, and percutaneous exposure were also reported. 91,100
Distribution
Bipyridyls have large volumes of distribution. After absorption, DQ is rapidly distributed throughout the body in the aqueous phase, except for brain and spinal cord, a fact supported by its hydrophilic nature and slow passage across membranes. 100 It achieves peak and blood tissue levels after 2 h, followed by rapid decline and elimination through the urine. 105 As observed earlier, unlike PQ, it is not selectively concentrated in the lungs by the polyamine uptake system because it does not fulfill the strict structural requirements; its half-life in the lungs is five times shorter than PQ. 99,106 –112 Indeed, in animal studies, DQ causes mild, reversible injury to type I pneumatocytes but does not injure the type II cells. The rapid efflux of the accumulated [14C]DQ indicates that the lungs would not be a major site for storage of this compound for any significant period in vivo. 104 No progressive pulmonary fibrosis has been noted in DQ poisoning. 113,114 The slow passage across membranes can result in an apparent sequestration in various tissues as systemic levels rise and fall. 23 Liver, kidney, GIT, and lung had the highest residues immediately following dosing; at 96 h, significant residues were seen primarily in the eye lens.
Metabolism
Since DQ is relatively resistant to metabolism, adsorption to clay soil particles, and photodegration in water and plant surfaces are the major dissipation routes. In animals, including humans, DQ is minimally metabolized by oxidation of the pyridine rings to less toxic mono- and dipyridone derivatives (Figure 4). Cleavage of one of the pyridine rings either from parent DQ or DQ monopyridone produces TOPPS, which is subsequently decomposed to picolinamide and picolinic acid, possibly via pyridine-2-carboxamide as an intermediary metabolite, although this was not identified. Further volatile products are produced but not yet identified. 92 These metabolic pathways occur mainly in the liver via cytochrome P450 enzymes, and all metabolites are more easily excreted through the urine. 51,91,94 Fuke et al. 35 identified both DQ dipyridone and monopyridone in the serum of three DQ-poisoned patients, with dipyridone reaching up to 20% of DQ in one patient. In vitro studies suggested that the cecal microflora of the rat can metabolize DQ to the monopyridone. 91,115 As mentioned earlier, there is no reported metabolism of DQ in plants. The rapid desiccation of green parts of weeds after application may contribute to the observed results.
Elimination
Due to low GIT absorption, almost all of the herbicides is excreted unchanged in the feces (about 94% in the first 24 h) with slight or no biliary excretion. 100 Absorbed DQ ion and metabolites are mainly eliminated via urine within 48 h, irrespective of the route of administration. 51,54 In an in vivo study performed with rats, after DQ gavage administration (45 mg DQ ion/kg body weight [bw]), the herbicide was excreted mostly unchanged in urine and feces (6% and 89% of dose, respectively), and its metabolite DQ monopyridone was present mainly in feces (5% of dose) and in a lesser extent in urine.
Furthermore, urianalysis of rats after subcutaneous route of 10 mg DQ ion/kg bw revealed the presence of DQ (75%), DQ monopyridone (3%), and DQ dipyridone (6%). 91,115 Similar results were obtained with Williams and collaborators 91,116 after oral administration of DQ in rats, although there were up to 80% of DQ unchanged and only 1% of picolinic acid, DQ monopyridone, and DQ dipyridone in urine.
Mechanisms of toxicity
Although DQ does not bind covalently to macromolecules (i.e. lipids, proteins, and nucleic acids), it may lead to oxidative stress due to the ability of DQ to generate ROS intracellularly through reduction–oxidation (redox) cycling processes (Figure 6). This capacity is greater with regard to other herbicides because of its high redox potential (E 0) value, which is a measure of electron affinity (see Table 2). 101

Schematic representation of the redox cycling of DQ and ROS and RNS generation. Cellular reductases such as cytochrome P450 reductase catalyze one electron reduction of DQ2+, forming the DQ monocation free radical (DQ+●). This, in the presence of oxygen, forms superoxide radical (O2 ●−), regenerating the parent compound. DQ redox cycling activity requires NADPH that is produced from glucose via glucose-6-phosphate dehydrogenase, the first enzyme of the HMP. The mechanism of cellular uptake of DQ is unknown. DQ: diquat or 1,1′-ethylene-2,2′-bipyridinium; ROS: reactive oxygen species; RNS: reactive nitrogen species; SOD, superoxide dismutase; NADPH: nicotinamide adenine dinucleotide phosphate; HMP: hexose monophosphate pathway; CAT: catalase; GPx: glutathione peroxidase; Gred: glutathione reductase; FR: Fenton reaction; HWR: Haber–Weiss reaction.
DQ is reduced by receiving a single electron from NADPH, the main source of reducing equivalents in cells, in a pH-independent reaction catalyzed by cytochrome P450 reductase, forming NADP+ and a highly unstable DQ+●, which, in turn, transfers an electron to molecular oxygen (O2) to generate O2 ●−. DQ+● returns then to its original form and can, even in small amounts, undergo this continuous process to generate large quantities of O2 ●−, which is neutralized spontaneously or enzymatically via superoxide dismutase (SOD) to produce hydrogen peroxide (H2O2) and O2. 117 Under normal conditions, H2O2 is converted into water by catalase (CAT) and glutathione peroxidase (GPx). Otherwise, with the large increase in ROS production, the cellular protective mechanisms, either nonenzymatic components [e.g. glutathione (GSH), thioredoxin, selenium, vitamins C and E] or antioxidant enzymes (e.g. SOD, GPx, GR, CAT), become overwhelmed, resulting in oxidative stress and consequent cellular dysfunction and injury. 118 –120 Oxidized glutathione (GSSG) increases and is excreted through the bile. It was proved that DQ significantly increases biliary excretion of GSSG, which could therefore be used as a biomarker of hepatic oxidative stress. 50,118,121
Both DQ+● and O2 ●− reductively release iron from ferritin, converting the ferric ions (Fe3+) into ferrous ions (Fe2+). This is an important process for the toxic effects of DQ, as Abe et al. 122 found in experiments with Fischer-344 rats that higher body iron stores result in higher DQ toxicity. These Fe2+ ions catalyze the Fenton reaction, forming a more powerful oxidant hydroxyl radical (HO●) from H2O2. 50,123,124 All these ROS, as well as the released iron, can subsequently damage biological molecules, whose effects will be described subsequently. Because it is a potent prooxidant, DQ is used in various in vivo and in vitro studies in toxicology and cellular biology fields to induce oxidative stress.
ROS can damage the allylic hydrogens of polyunsaturated fatty acids (LH) present in the cell membranes, generating peroxyl radicals (L●) that remove allylic hydrogens from the adjacent fatty acids to create lipid hydroperoxides (LOOH), leading to lipid peroxidation (LPO).
125
This phenomenon decreases the fluidity and increases the permeability of the membrane, as it becomes easier for phospholipids to switch between the two monolayers, and practically, there is no restriction for compounds to cross the membrane. It may also lead to membrane rupture and, consequently, to necrosis of tissues. LPO can inactivate membrane-bound receptors or enzymes, which, in turn, may disrupt normal cellular function.
119,126
The LPO products can induce mutations and modified patterns of gene expression through formation of DNA adducts.
125
Using a sensitive and specific index of lipid peroxidation, it was shown in vivo that the DQ hepatoxicity is, at least partially, a consequence of ROS-induced lipid peroxidation and that the powerful antioxidant and free radical scavenger melatonin has protective effects.
126
Liver damage and lipid peroxidation induced by DQ were also significantly reduced in transgenic mice overexpressing glutathione peroxidase 4.
127
Moreover, besides ROS, DQ-induced oxidative stress was also evidenced by reactive nitrogen species (RNS) generation, namely the decreased serum nitric oxide (NO) in piglets
128
and peroxynitrite (ONOO−) generation in hepatocytes.
129
ONOO− is formed by the reaction of O2
●− and ●NO at a diffusion-limited rate.
130
Some data suggest that ●NO can strongly protect plants from DQ-related damage due to its scavenging ROS potential.
131
On the other hand, the NG-nitro-
Regarding proteins, the misfolding and oxidation of proteins, especially through carbonylation that can occur through lipid-dependent and lipid-independent pathways, result in disrupted functions that are crucial for the organism. It can lead, for example, to dysfunctional enzymes or disturbance of blood protein carriers present in the circulatory system. 125 Several studies implicate DQ exposure in protein oxidation. 133,134
Metal cations can infiltrate through the membranes and adhere to DNA because of its polyanionic structure, allowing the formation of HO●, which could then cause DNA strand breakage, chromosomal abnormalities, and cell cycle perturbations. 125 It was also found that ROS may have a great impact at a pretranslational level of mRNAs from a rat liver, because the expression profile of certain phase I, phase II, and antioxidant enzymes were decreased after exposure of DQ. 135 Moreover, in vitro studies with hamster cells showed that low concentrations of DQ can raise the cell cycle rate, while higher concentrations may reduce the number of mitotic cells. 136 –138
DQ administration to the rat produced an increased synthesis of liver glycogen and an increase in blood glucose that appeared to be mediated by the adrenal, because adrenalectomy prevented these changes. 100 These effects are thought to be due to catecholamine release and high circulating concentrations of corticosteroids.
Some reported median lethal dose (LD50) values are presented in Table 3. As can be observed for rat, the value for oral administration is about 10-fold higher than by subcutaneous injection, suggesting that the oral toxicity is moderate, which is consistent with the information provided of DQ being poorly absorbed from the GIT. 99,139 Furthermore, the oral toxicity is greater than the dermal toxicity by two to four times when comparing the LD50 values of rat and rabbit, in consonance with the fact that the herbicide cannot penetrate easily through the skin. Comparing to other species, DQ is highly toxic to cattle by oral route and has slight ecological impact on invertebrates and fish, demonstrated by exposure and toxicity data, probabilistic risk assessment, and ecological procedures modeling, as it rapidly dissipates in water. 64,81 Despite the chemical similarity to PQ, acute toxicity is somewhat lower for DQ. 72
LD50 and NOAEL values of DQ (mg ion/kg body weight) in some animal species considering various exposure routes. Estimate of acceptable daily intake for humans is 0–0.002.

LD50: median lethal dose; NOAEL: no-observed-adverse-effect level. DQ: diquat or 1,1′-ethylene-2,2′-bipyridinium.
The major metabolite, DQ monopyridone, is significantly less toxic than DQ by oral route (LD50 higher than 400 mg ion/mg ion/kg bw).
Putative link between diquat exposure and neurodegenerative diseases
Several compounds such as 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) and the pesticides DQ, PQ and rotenone have been implicated in Parkinson’s disease (PD), an age-related multifactorial neurodegenerative disorder. 110,140 –142 PD is an idiopathic disease of the nervous system categorized by a significant loss of pigmented dopaminergic neurons, mostly of the substantia nigra pars compacta 143 which are fundamental for the control of motor function, 144 as well as the occurrence within surviving neurons of huge eosinophilic inclusions known as Lewy bodies, which contain α-synuclein fibrils, neurofilaments, ubiquitin, and other proteins. 110,142,145 These features clinically result in various symptoms, namely rigidity, resting tremor, postural instability, and bradykinesia (slowness of voluntary movement). 144,145 Since DQ is structurally similar to PQ and MPTP (Figure 1), it was speculated that this herbicide may also play a role in the development of PD, and some reports support that there is a link. 29,109,110,146 –149
In a study in Rana temporaria frogs, it was observed that 14C-labelled DQ accumulates in neuromelanin-containing nerve cells after intraperitoneal injection. 148 Some years later, in an in vitro study performed in primary mesencephalic cultures from Sprague-Dawley rats 146 found that DQ induced significant changes in the morphology and number of dopaminergic neurons. Moreover, DQ also reduced the dopamine uptake by more than 80%. Drechsel and Patel 143 demonstrated that DQ may generate H2O2 aplenty in a concentration-dependent way either in brain mitochondria isolated from Sprague-Dawley rats or in primary midbrain cell cultures obtained from embryonic rats. These studies suggest that redox cycling of the herbicide, with ROS production that leads consequently to oxidative stress, has a role on dopaminergic cell degeneration and that should be taken in account when evaluating the environmental risk factors for PD. Because DQ has higher reduction potential than PQ (E 0 = −0.33 V for DQ and E 0 = −0.35 V for PQ), it can accept an electron and undergo a redox cycling more rapidly, which means that it may be more neurotoxic than PQ. 146
Nisar and et al. 149 performed an in vitro study where they treated SH-SY5Y neuroblastoma cells and human neural precursor stem cells line N1997 with DQ at different concentrations. They found that DQ reduced significantly cell viability in a dose-dependent manner. They also observed that DQ neither modified α-synuclein expression nor impaired mitochondrial activity, both features implicated in PD etiology, specifically on the inclusion of Lewy bodies. Authors then suggested that this herbicide may have a role in PD in a rarer way where Lewy bodies do not constitute a feature, promoting α-synuclein aggregation in a different mode and independently of mitochondria. The mechanisms of cell influx of DQ is not clear, although some results had refuted the hypothesis of an entry mechanism through the system L carrier (LAT-1) that is used by PQ for entering into the brain. 150
Despite of a case report dated from 1989, where a 72-year-old farmer developed symptoms of PD several days after exposure of an aqueous solution containing 10% DQ dibromide, 29 much more has to be found to prove the link between exposure and the development of PD, as data are inconsistent mainly due to statistical analysis, study design, modifications in exposure routes, and methodological differences. 144,151 Moreover, there have been relatively consistent pathologic brain changes noted in reported fatal cases of DQ poisoning. These consist of brain stem infarction, particularly involving the pons. 20 It is not clear whether these postmortem changes represent direct toxicity or secondary effects related to the systemic illness and therapy.
Also, other neurodegenerative diseases need to be evaluated, as it was verified that DQ can lead to axonal degeneration, a pathologic feature involved not only in PD but also in Alzheimer’s and Huntington’s diseases. 147 Authors evidenced that DQ exposure and the deficiency of SOD in dorsal root ganglion neurons culture obtained from C57BL/6 strain mice may predispose to axonal degeneration. Nevertheless, axonal degeneration in an in vivo study remains to be established.
Developmental and reproductive toxicology
Adverse systemic effects were noted in the rat, both in parents and offspring (cataracts and eye pathologies in both sexes of F0 and F1 at > 240 ppm; an increase of hypertrophy and hyperplasia of collecting duct epithelium and tubular dilatation in the renal papilla in both sexes of F1 at 240 ppm; F1 and F2 pups showed hydronephrosis at >240 ppm). In rats, a developmental no-observed-adverse-effect-level (NOAEL) of 12 mg kg−1 day−1 for intrauterine growth retardation was proposed (Table 3). 72 NOAEL of 3.6 mg kg−1 day−1 for both maternal (clinical signs, death) and developmental toxicity (skeletal anomalies, exencephaly, and umbilical hernia) was calculated for mouse. 72 The rabbit appears to be a highly sensitive to DQ in developmental toxicity studies. 72 The NOAEL for maternal toxicity (histopathological changes in the liver, intestine, and vasculature; mortality) was 1.0 mg kg−1 day−1 and the developmental NOAEL was below 3.0 mg kg−1 day−1. 72 Delayed ossification of the ventral tubercle of the cervical vertebrae was noted in all treatment groups compared with the controls. Zhang and collaborators, 152,153 demonstrated that chronic exposure to DQ at 8 or 12 mg kg−1 DQ by intraperitoneal injection twice a week for four consecutive weeks induced reproductive toxicity in female imprinting control region (ICR) mice. The formation of ROS through redox cycling processes in granulosa cells and oocytes resulted in follicle development impairment (with reduction of the ovary weights), apoptosis induction, and oocyte quality reduction, leading to fewer fetal mice as well as a significant decrease of litter sizes comparing to control.
Analytical toxicology
Several methods allow an effective extraction and subsequent precisely and unequivocally identification of the herbicide in various samples, including water, soils and plant materials and biological fluids, for environmental, clinical, and forensic purposes.
Samples
The nature of the sample often does not allow direct analysis of DQ due to contaminants present in the matrix that interfere with the identification of the herbicide. In most situations, the DQ concentration may be so low that it is not detectable by the analysis method under consideration, which could lead to false negatives. Hereupon, extraction techniques are important to remove the interferences and to concentrate DQ. These procedures enable the reduction of detection limits, improving the sensitivity of analytical methods.
Water samples from irrigation channels, lakes, rivers, or other water reservoirs usually do not require an extensive clean-up. Because the presence of common contaminants, such as humic acids, surfactants, particulates, inorganic salts, among others, can compromise the efficiency of the extraction procedures or posterior quantification, a filtration step using membrane filters should be performed. If the extraction is not immediately executed, samples should be stored at 4°C. Also, it is extremely important to collect water samples in plastic bottles rather than glass, where DQ may be tightly adsorbed. 154
Due to the high sorption characteristics of DQ and bipyridinium herbicides in general to soil and plants, extraction have been generally achieved through refluxing or heating with strong hydrochloric or sulfuric acids to free DQ from their adsorbed state. Although this procedure is widely used, it is drastic and time-consuming. Hence, alternative methods involving sample homogenization with an organic solvent (e.g. acetone, methanol, acetonitrile, or ethyl acetate), alone or in mixture with pH-adjusted water, using a homogenizer, blender, or sonicator, and then heating the homogenized sample in acidic solution have been proposed. 4,154
Biological samples including blood, plasma, urine, milk, among others, contain a wide range of compounds that could lead, for example, to the clogging on the solid-phase extraction (SPE) column if that technique is applied. Therefore, one of the steps to be performed before DQ recovery is deproteinization with distinct acids, such as perchloric, sulfosalicylic, and trichloroacetic acid. Other useful step is adding ethylenediaminetetraacetic acid (EDTA) to remove interferences by numerous metal ions. 154
Extraction
DQ extraction from samples is not an easy task, however, given its high solubility in water and high binding capacity. Liquid–liquid extraction, for instance, is not a suitable technique for this herbicide. 155 On the other hand, chemical separations based on polyurethane foam (PUF) revealed to be a good alternative. 85 PUF is a low-cost sorbent material with the ability to separate organic and inorganic substances from aqueous medium due to its high sorption capacity, easily modifiable surface or structure, chemical and thermal resistance, selectivity and wide-range retention capacity properties. Specifically, the PUF-sodium dodecylsulfate (SDS) sorbent gave great results for extracting cationic herbicides as is the case of DQ. 85,156
SPE techniques proved to be very useful for extracting herbicide residues with high efficiency. Various sorbents have been applied, mainly silica, 157 graphitized carbon, 158,159 cation exchange, 160,161 and nonpolar phases like C8 and C18. 154,162,163 It was shown by Agostini de Moraes and collaborators 164 that chitosan and alginate are suitable biopolymers for the adsorption and removal of DQ from water sources. Microwave-assisted extraction requires sophisticated equipment but proved to be a fast and specific method for the extraction of DQ with excellent recoveries. 8
Detection procedures
Various methods are applied for the detection of DQ, all of them differing in the principles of detection, type of data provided, selectivity, and sensitivity. Therefore, the selection of the method is crucial in any analytical study, according to its objective and the type of results to be obtained. The most commonly applied methods for detecting DQ are described below.
Figure 7 presents the spectrophotometric spectrum DQ and colored derivatives obtained after reaction with different compounds, which are useful to toxicological analysis. In emergency situations, when a DQ poisoning is suspected and a quantitative assessment is not readily available, a rapid and simple colorimetric test can be done by analyzing urine, adding sodium bicarbonate or hydroxide, followed by sodium dithionite powder, which gives a green color in the presence of DQ (Figure 8). A strong green color obtained with a urine sample taken more than 4 h after ingestion suggests a poor prognosis. However, this assay is not sufficiently specific, as PQ may undergo a similar reaction producing a blue color, interfering with the analysis. 76,50,111,170 –173

Absorption spectrophotometric spectrum of DQ2+ (blue), DQ radical monocation (DQ+●) after reduction with sodium dithionite (green), complex after reduction with 2-mercaptoethanol,

Representative qualitative colorimetric test (intensity of the green color) by adding sodium bicarbonate and sodium dithionite to urine samples containing different DQ concentrations (μg/mL). DQ: diquat or 1,1′-ethylene-2,2′-bipyridinium. Adapted from the studies of Dinis-Oliveira et al. and Vohra et al. 76,170
Spectrophotometry is considered the standard procedure for DQ analysis by the Official Methods of Analysis of AOAC International. 90,174 It takes in consideration the absorbance measure of DQ2+ at 310 nm or the formation of a green radical at 430 nm (DQ+●) by reduction with sodium dithionite or other reducing agents, as mentioned before. This technique does not distinguish between the parent compound and its metabolites. 59 Some methods based on second-derivative spectroscopy were developed, 35,175,176 which are advantageous over classical spectrophotometric techniques due to the enhancement of the DQ+● peak, as well as the supression of larger absorption bands as a result of nonspecific matrix absorption. Fuke et al. 35 developed a method where DQ2+ was reduced with sodium hydroxyde–dithionite solution in urine and centrifuged, while serum was firstly deproteinized with sulfosalicylic acid, centrifuged and then DQ2+ was reduced with the same solution (Figure 9). The second-derivative spectrum of DQ+● showed four main peaks at 464, 445,430, and 380 nm and six satellite maxima at 482, 454, 437, 423, 387, and 373 nm, which were derived from the zero-order peaks at 461, 444, 428, and 380 nm, respectively. The highest amplitude of 380–387 nm cannot be used, because the amplitudes below 390 nm overlapped with those of PQ. Therefore, both supernatants were analyzed by second-derivative spectrophotometry at the amplitude peaks of 454 to 464 nm, obtaining the spectra of Figure 9. This method is particularly useful for diagnosis and prognosis of DQ intoxication.

(a) Pretreatment procedures for DQ analysis in urine and plasma (10 µg mL−1) before the second-derivative spectrophotometric analysis. (b) Zero-order and second-derivative spectra. The qualitative analysis is made by observing the presence of inflection points at about 437, 445, 454, and 464 nm. The quantification is made with amplitudes measurable between 380 nm and 387 nm only if DQ is present, and between 454 nm and 464 nm if PQ is also present. DQ: diquat or 1,1′-ethylene-2,2′-bipyridinium; PQ: paraquat or bipyridinium.
The formation of red complexes by reducing DQ either with 2-mercaptoethanol (Figure 7), dithiothreitol or

Colorimetric analysis through reduction of DQ with 2-mercaptoethanol. DQ: diquat or 1,1′-ethylene-2,2′-bipyridinium. Adapted from the work of Maiti et al. 165
The ion-selective electrodes of potentiometric methodologies are not selective for herbicides, which may lead to misinterpretations in identification and quantification of DQ, mainly in complex matrices. 178
Luminescence techniques, especially chemiluminescence, are widely employed due to their speed, wide dynamic range, great sensitivity, and ease of implementation. Similarly, flow injection analysis (FIA) methods are simple, inexpensive, reproducible, and give faster results. After that, some analysis methods coupled FIA with chemiluminescence were performed for the detection of DQ in water. This hyphenated technique provides lower detection limits and higher sample throughputs and proved to be simple, sensitive, and fast enough for use in routine analysis. 90
Quantum dots (QDs), previously optimized in oat grains, can also be used as a reducing agent, presenting some advantages in relation to other reduction procedures, such as reaction rapidity and stability of the reduction product (reduction with sodium dithionite would take 5–6 min to reach equilibrium, and the product is very unstable). 179 Thus, given their unique properties of wide absorption, slight emission, size-tunable optical feature, good photostability, and intense brightness, QDs are useful for analytical and biomedical purposes, allowing rapid, simple, and sensitive methods that may achieve lower detection limits. 180
Analysis of DQ by gas chromatography (GC) is difficult due to the low volatility and high polarity of the molecule and hence would require a derivatization step to increase the selectivity or sensitivity of the detection. Notwithstanding, this derivatization step, as well as low concentration levels of the analyte, decrease the method reproducibility. 125,155 A GC method coupled with mass spectrometry (MS) was previously optimized for urine and plasma samples by converting DQ and PQ into lipophilic and thermally stable and volatile compounds using sodium borohydride. 181 This GC-MS method was fast, specific, selective, and sensitive for the analysis of these substances in cases of acute intoxication and did not require prior deproteinization procedure. 181 Nitrogen–phosphorous detection (NPD), alkali-flame ionization detection (AFID), or thermionic detection, which is a flame ionization detection (FID) slightly modified by adding a bead covered by an alkaline salt, are also commonly used detection methods for the analysis of bipyridinium herbicides. 125
The low volatility and high solubility in water make DQ compatible for liquid chromatography (LC) analysis. LC is useful due to its high sensitivity and selectivity, with detection limits generally higher than those achieved by GC, although it requires expertise and expensive reagents and instrumentation. 125 Reversed-phase columns and reagents that form volatile ionic pairs, for example, heptafluorobutyric (HFBA), pentafluoropropionic (PFPA), or trifluoroacetic (TFA) acids, are generally applied when performing LC analysis to improve separation and resolution. 178 Moreover, in experiments with different mobile phase compositions, Evans et al. 182 found that DQ do not form observable ion–pair complexes or clusters with chloride or acetate anions, possibly due to steric influences, not affecting the reproducibility of the method. Besides DQ, the concentrations of its metabolites (DQ monopyridone and DQ dipyridone) could also be determined in human biological samples using a high-performance liquid chromatography (HPLC) with UV and fluorescence detection, which proved to be a sensitive, selective, and reliable method. 42 A simple, modified quick, easy, cheap, effective, rugged, and safe procedure was developed for the determination of diquat in potato using reversed-phase LC coupled with tandem MS (MS/MS) in a total run time of 10 min. 183
A supercritical fluid chromatography (SFC) was also developed for the detection and separation of DQ and others pesticides. The mobile phase in SFC, being carbon dioxide the most commonly used, is nonreactive, economic, nontoxic, and easy to handle, allowing a high resolution at high flow rates and increased length columns, as well as a wide range of separation modes. Ishibashi et al. 184 developed a method that proved to be robust, rapid, high-throughput, reproducible, and very efficient for the analysis of various pesticides, including DQ, with diverse polarities and molecular weights in some agricultural products.
MS is a very powerful technique, as it can provide information about molecular structures of the compounds of interest, and can be highly selective and sensitive when used in the single ion-monitoring mode. The Environmental Protection Agency (EPA) and the US Food and Drug Administration thus recommend that, for DQ identification purposes, methods involving MS should be used, using three ions to confirm the presence of the substance. 4,178,185 Many mass analyzers have been employed for the analysis of DQ residues, such as single quadrupole, 186 triple quadrupole, 178 magnetic sector, 187 ion trap, 188 and time-of-flight, 189 as well as numerous ionization interfaces, namely particle beam, 160 fast atom bombardment, 190 atmospheric pressure chemical ionization, 191 and electrospray ionization. 192
Capillary electrophoresis (CE) is an adequate analytical technique for the analysis of ions, based on differences in their electrophoretic mobility, and has the advantages of high separation efficiency, short optical path length defined by the column diameter for on-capillary UV detection and low consumption of reagents that are toxic to the operator, although the small volumes typically injected compromise its sensitivity. It has been proposed for the analysis of UV-absorbing “quats” such as DQ, not only in water, but also in other matrices such as soil, serum, and urine. 193 The most commonly used mode in CE is capillary zone electrophoresis due to the ionic nature of the herbicide. 185,194 –196 Various studies were made coupling CE with MS, and this combination proved to be a very useful and powerful tool for the separation and identification of “quats” in water samples, as it provides the high-resolution potential of CE and the detection and identification properties of MS. 193
Experimental observations and clinical history
Typically, diagnosis is based on the history of ingestion and the presence of oral burns. If not handled properly or when protective equipment is not used, exposure to aerosols containing DQ can cause mouth irritation, nose with inflammation and bleeding of the nasal mucosa. There are reports that concentrated solutions of DQ can influence the nail growth and cause yellow nail discoloration, dimpling and shedding, and delay the healing of superficial cuts on the hands (Figure 11). 11,197 Clark and Hurst described the appearance of discolored transverse lines, then cracking, and, finally, shedding in the nail following contact with 20% DQ. 99 Toxic effects seems to occur due to local exposure and not by ingestion, as the toenails and asymmetric lesions have been reported. 11,197

Local effects related to DQ. (a) and (b) Ulcerated lips and tongue after accidental exposure during manufacture. (c) Conjunctival congestion in the right eye after being splashed with a liquid formulation containing 5% PQ and 7% DQ. (d) Proximal nail shedding. (e) Mees’ lines. (f) Yellow nail discoloration. DQ: diquat; PQ: paraquat. Figure 11(a) reproduced from the work of Adams et al.47, Figure 11(c) from the work of Kitagawa et al.43, Figure 11(d) and (e) from the work of Kibby and Ring 197, and Figure 11(f) from http://www.dermatorium.net/kategoriler.asp?id=209&katid=4, with permission.
Also, splashes directly into the eye can lead to ocular burn, conjunctivitis, conjunctival congestion (Figure 11), and corneal scarring. Moreover, a chronic diet administration of DQ can lead to bilateral cataract as was demonstrated for rats and dogs. 193,198 The process of redox cycling that occurs in the eye is believed to be the likely mechanism o cataract formation.
When ingested, corrosive damage to the oral mucosa may occur, resulting in mouth hemorrhagic ulceration (Figure 11) and mucosal edema of the tongue and oropharynx. 50 In a study performed in rats, even repeated ingestion of low doses of the herbicide that may be found in food (0.1 mg kg−1 day−1 per os) may induce a mild GIT inflammation allied with mast cell hyperplasia. 199 DQ is also associated with occupational voice disturbances, immune suppression, and hormone disruption. 164,200 In this matter, Sechi et al. 29 had described a DQ intoxication case in a 72-year-old Italian farmer, who developed skin reddening with hyperkeratosis, akathisia, and dysphonia after exposure of his hands with 10% aqueous DQ dibromide. The patient also developed acute and persistent Parkinsonism, although it was not considered to be the direct cause of dysphonia.
As kidney is the primary excretory pathway for absorbed DQ, it specially causes kidney damage (oliguria, anuria, proteinuria, hematuria, pyuria, azotemia, acute renal failure, and acute tubular necrosis). Interestingly, DQ is structurally related to orellanine, a highly potent nephrotoxin (Figure 1). 201,202 Toxicity for the liver [elevations of serum alkaline phosphatase (ALP), aspartate aminotransferase (AST), alanine transaminase (ALT), and lactate dehydrogenase (LDH) as consequence of hepatic necrosis], heart (cardiovascular collapse, dysrhythmias, and hypertension), GIT (severe esophagitis, ulcers, intestinal paralysis, fluid loss, abdominal distension, nausea, vomiting, and diarrhea), skin (skin burn, contact dermatitis, pruritus, and edema), reproductive system, and central nervous system (hypotensive shock, seizures, coma, intracerebral and brainstem hemorrhagic lesions) have also been reported. DQ can also damage the lungs, leading to dyspnea, upper respiratory pain, pulmonary edema, and respiratory depression. However, unlike PQ, pulmonary fibrosis has not been reported. 22,40,49,50,113,114
According to the ingested dose, different clinical manifestations can be observed, and all implicate an urgent medical assessment
50
: Mild poisoning (< 1 g DQ2+, 20%): GIT symptoms with putative development of renal injury. Full recovery is possible. Moderate-to-severe poisoning (1–12 g DQ2+, 20%): multiple-organ dysfunction and acute renal failure. Most patients can still recover. Fulminant poisoning (> 12 g DQ2+, 20%): multiple organs severely affected, leading to failure and eventually death in all patients within 1–2 days.
Sometimes an asymptomatic period can occur, extending from 24 h to 48 h after DQ exposure.
Other laboratorial analysis
Some laboratory tests are performed, including specific serum levels of electrolytes, creatinine, glucose, liver transaminases, as well as urinalysis, chest X-ray, and arterial blood gases or oximetry. As the mechanism of toxicity of DQ involves formation of ROS and consequent damage to biomolecules, Zwart et al. 203 proposed that the urinary levels of aldehydes (including acetaldehyde, propanal, butanal, pentanal, hexanal, and malondialdehyde), acetone, coproporphyrin III, and 8-hydroxy-2′-deoxyguanosine are good effect biomarkers of DQ-induced free radical injury. It is important to highlight that DQ interfere with creatinine assays using the Jaffe method causing a false elevation, although this largely occurs at concentrations exceeding 100 mg L−1, which is rarely observed and likely to be associated with severe clinical toxicity. 33
Treatment
Currently, there is no antidote or effective treatment for DQ intoxications, the treatment being devoted to reduce absorption and/or enhance elimination. 48,50 Because DQ does not accumulate in the lungs, intensive care treatment is likely to lead to a better prognosis than PQ.
If the patient was exposed to spills and splashes of DQ-containing liquid formulations, one of the first preventive measures that has to be taken is to remove all contaminated clothing and to wash exposed skin and eyes with water. 17 When ingested, therapeutic approaches before absorption are of great importance, because DQ is not quickly absorbed by the GIT. Therefore, activated charcoal should be immediately administered; if not available, ingestion of any food may afford some protection. Gastric lavage may be helpful if executed within an hour of the ingestion. In either case, both procedures should be performed cautiously due to the risk of perforation, especially when treatment is delayed. 40,50 Furthermore, instillation of mineral adsorbents such as Fuller’s earth or bentonite with combined administration of a cathartic may be of great importance to reduce absorption of DQ, although they are possibly no more effective than charcoal. Diarrhea induction with purgative and high enemas have also been applied as an early treatment to reduce absorption from the GIT. 40
To eliminate DQ from circulation, measures as forced diuresis, hemoperfusion, and extracorporeal hemodialysis have been considered, although some reports do not support these techniques as sufficiently effective, as they do not remove clinically and toxicologically significant quantities of the herbicide, probably due to the rapid distribution of the absorbed DQ. 40,17 In cases with significant hypovolemia, immediate fluid and electrolyte replacement must be done. 50
Administration of antioxidant compounds could be useful for scavenging or preventing the ROS and RNS formation, which play a major role in DQ poisoning. Melatonin, for example, is a broad-spectrum antioxidant and a potent free radical scavenger responsible for scavenging O2 ●−, HO●, ●NO and singlet oxygen (1O2) as well as preventing LPO and reducing the serum levels of blood urea nitrogen. 119 Furthermore, it has low toxicity and its highly lipophilic and slight hydrophilic nature makes it capable to cross all morphophysiological barriers, accumulating in all tissues and subcellular compartments. Of note, the efficacy of melatonin depend on the administered dose, as it can protect against DQ-induced acute hepatic and renal toxicity and reduce mortality due to oxidative stress at low concentrations, whereas at high doses it can exert pro-oxidant effects. 119,126,137 Polymeric sulfates such as heparin, 51 the potent iron chelator desferrioxamine 204 and the climbing plant Forsythia suspense 120 were also demonstrated to inhibit significantly the DQ-induced redox cycling procedures.
Concluding remarks
Since the discovery of its herbicidal potential in the 1950s, DQ has been extensively used as a nonselective contact herbicide to control terrestrial and aquatic vegetation by commercial and domestic gardeners. It is rapidly absorbed by green plant tissue and inhibits the photosynthetic process to “starve” and expose the weeds to ROS over several days, leading to plant cells necrosis. It is inactivated on contact with soil and not taken up by plant roots. DQ has been commercially available both combined with PQ and, less commonly, alone, namely in countries where PQ is severely restricted. It is less toxic than PQ and therefore when formulated with PQ allows a lower concentration of PQ to be used.
Much more scientific human reports and animal experimental data exists for PQ than DQ. 106 –112,205 The majority of poisoning cases are related to intentional ingestion of concentrated liquid formulations, resulting in life-threatening renal, hepatic, cardiac, and GIT complications. Humans may also be exposed to DQ by pulmonary, ocular, or dermal routes. Its main mechanism of toxicity is related to redox cycling, leading to ROS and RNS generation, oxidative stress, and potentially cell death. DQ redox cycling continues as long as NADPH and oxygen are available. Depletion of NADPH prevents recycling of GSH and interferes with other intracellular processes, including energy production and active transporters, exacerbating toxicity. Intracellular protective mechanisms such as GSH, SOD, and CAT are overwhelmed or depleted following large exposures. Taken together, these cytotoxic reactions induce cellular necrosis, which is followed by an influx of neutrophils and macrophages.
Many therapeutic measures have been applied to minimize the effects of DQ poisoning and the delay between DQ ingestion and the onset of treatment is critical. Despite all therapies, for several times, it was not possible to prevent a fatal outcome of DQ poisoning. Therefore, an antidote or effective treatment that is specific for DQ is needed. Specifically, in vitro and/or in vivo studies should be performed, to evaluate a potential inducer of efflux transporters to release DQ residues from cells and being excreted through urine, minimizing the effects of poisoning. On the other hand, a compound that can form complexes with DQ inhibiting its entry to the cells could also be an interesting approach.
Footnotes
Acknowledgements
The authors are grateful for the extensive and knowledgeable feedback provided by the anonymous peer reviewers selected by the Editor of the journal. Ricardo Dinis-Oliveira acknowledges his Investigator Grant (IF/01147/2013) from the Fundação para a Ciência e a Tecnologica (FCT).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The employment affiliation of the authors is as shown on the first page of this article. The review was prepared during the normal course of the authors’ employment. The authors have sole responsibility for the writing and content of the article. Authors also declare that they have not been involved during the past 5 years in any legal or regulatory proceedings related to the contents of the article. The authors declare they have no conflicts of interest with regard to preparation of the article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NORTE-01-0145-FEDER-000024, supported by Norte Portugal Regional Operational Programme (NORTE 2020), under the PORTUGAL 2020 Partnership Agreement, through the European Regional Development Fund (ERDF) and CESPU projects TramTap-CESPU-2016 and ChronicTramTap_CESPU_2017.
