Abstract
The cancer threshold of toxicological concern (TTC) as determined by Kroes et al. in 2004 (0.15 µg/day and 1.5 µg/day at risk levels of 1 in 106 or 105, respectively) has been uncritically employed as a key metric in multiple regulatory guidance documents. There are numerous concerns regarding transparency and the highly conservative methodology in relation to its derivation; moreover, no formal confirmation has been undertaken by any regulatory body prior to its adoption. A recent joint report from the European Food Safety Authority and World Health Organization follows this trend, largely replicating previous conclusions and downplaying the need for a re-assessment. This view is challenged by Boobis et al. who confirm concerns regarding lack of transparency and stress that several of the assumptions and approaches used previously have been superseded by advances in knowledge; they recommend as a first step construction of a new dataset derived from the Carcinogenic Potency Database focusing on mutagenic DNA-reactive rodent carcinogens and a critical assessment of the relevance and reliability of carcinogenicity data. This type of approach is supported with two key exceptions: inclusion of data from the ToxTracker assay which provides a direct readout of DNA reactivity, and use of appropriate epidemiological data on acrylamide (AA) to determine a benchmark for human exposure to a typical DNA-reactive rodent carcinogen. It is concluded that a robust re-evaluation using an appropriate dataset and methodology is urgently needed to ensure the integrity of the cancer TTC before it is employed in its present form even more widely.
Keywords
Introduction
The cancer threshold of toxicological concern (TTC) is a critical element of guidance documents impacting particularly on foodstuffs and pharmaceutical impurities. In relation to the former, the latest iteration by the European Food Safety Authority (EFSA), jointly with the World Health Organization (WHO), of its position on the non-cancer and cancer TTC was published on 16th March 2016. 1 (Since EFSA was the lead contributor, citation of this report will refer only to this organization.) Consideration of the cancer TTC formed a minor yet important part of the report and EFSA’s approach showed few differences from that described by Dewhurst and Renwick in 2013. 2 Although various criticisms made by independent commentators are mentioned, these are ignored rather than rebutted. And the misalignment of the EFSA approach with that adopted on pharmaceutical impurities in ICH M7 3 is considerably underplayed.
This article is intended to critique the derivation of the cancer TTC. Although the principal point of reference is the latest EFSA report mentioned above, this is only because no other regulatory body has provided a similar detailed justification for the derivation and adoption of the cancer TTC. An earlier article 4 on this topic focused on EFSA’s initial position 2,5 ; since many of the arguments used previously still apply these have been restated (and augmented) in order to ensure completeness. In addition, there are important differences between the approaches used by EFSA and those for pharmaceuticals in relation to the application of the TTC concept. Such differences refer mainly to the categorization of compounds of concern as ‘genotoxic’ (foodstuffs) or ‘mutagenic’ (pharmaceuticals); use of structural alerts; numerical value of the TTC and lifetime versus less-than-lifetime (LTL) exposure.
Use of the cancer TTC in regulatory toxicology
EFSA has confirmed its adoption of the cancer TTC (0.15 µg/day or 0.0025 µg/kg/day – using 60 kg as an adult body weight – in relation to a compound containing a structural alert for genotoxicity) as derived by Kroes et al. 6 (who refer to structural alerts in relation to carcinogenicity). In coming to this position, EFSA 5 supported the methodology employed by Kroes et al. which involved linear extrapolation of cancer potency data for several hundred ‘carcinogens’ to a risk level of 1 in 106. EFSA deemed the current cancer TTC value to be ‘sufficiently protective’ and considered that a re-assessment was not a priority. 1
In 2006, an industry group 7 suggested by reference to the Kroes et al. publication (and based on a risk level of 1 in 105) a cancer TTC of 1.5 µg/day as the lifetime limit for a genotoxic impurity in a pharmaceutical product or drug substance, excepting compounds in the CoC (cohort of concern) containing high-potency substances such as N-nitroso, (alkyl)azoxy and aflatoxin-like compounds. At the time, industry scientists were concerned about the potential requirement for carcinogenicity data on such impurities and perceived the TTC as a viable alternative. However, the numerical value of the TTC was accepted without any independent re-evaluation. 8 The same value was subsequently adopted by the European Medicines Agency (EMA), Food and Drug Administration (FDA) and International Conference on Harmonization (ICH) in their guidance documents on genotoxic/mutagenic impurities in pharmaceuticals. The Kroes et al. publication was cited in the EMA and FDA guidances, but in ICH M7 3 (Assessment and control of DNA-reactive (mutagenic) impurities in pharmaceuticals to limit potential carcinogenic risk), where the methodology used to derive the cancer TTC is described as ‘very conservative’ (without any further explanation), no supporting references are provided. (Note that the cancer TTC in ICH M7 is expressed only in µg/day since it is used to determine allowable concentrations of (potentially) mutagenic impurities; for example, in the case of a mutagenic impurity in a drug used at a maximum daily dose of 150 mg for the treatment of a chronic condition, application of the default lifetime TTC produces an impurity limit of 10 ppm.)
Criticisms of the TTC derivation
A number of criticisms of the Kroes et al. methodology and the validity of the conclusions have been made by Delaney,
9
Snodin and McCrossen
10
and Snodin.
4
In summary, these are as follows: Skewed dataset(s) derived from the Carcinogenic Potency Database (CPDB) based on a preponderance of compounds expected to be carcinogenic/mutagenic; Use of the lowest statistically significant TD50 (a cancer-potency metric) instead of the default harmonic-mean TD50 leads to the phenomenon of false carcinogens and often overestimates carcinogenic potency. (Alternatively the most reliable/appropriate TD50s could be used as described in ICH M7 R1
11
) Linear extrapolation of cancer potency values for non-mutagenic carcinogens, which are generally considered to be thresholded, is inappropriate.
A further issue in relation to point 3 is the growing concern about the use of the linear no-threshold (LNT) concept for assessment of carcinogenic risk (initially for radiation and subsequently adopted for chemical carcinogens) in that there appears to be clear evidence of misrepresentation and lack of transparency dating back to its development and promulgation in the 1950s and 1960s. 12,13 Discontinuities between the effects of chemicals at high and low doses often make simple linear extrapolation unrealistic. Since multiple mechanisms operate in mammalian systems, the assumption of a simple dose–response relationship and a single outcome over the low- to high-dose range is likely to be highly misleading.
Skewed dataset for determination of cancer TTC
The dataset used by Kroes et al., 6 stated to contain 730 compounds, for the determination of the cancer TTC was non-transparent, which still remains the case. Various unsuccessful attempts have been made to retrieve the dataset, described as ‘no longer available’. 14 Several of the co-authors of the 2004 article appear to remain professionally active and it seems strange that none has come forward to clarify the position and/or release a copy of the dataset. However, it can be reasonably assumed 10 that the vast majority of the compounds (claimed to be 709; 706 listed) were those employed to support an earlier evaluation by Cheeseman et al. 15 using closely similar inclusion and cancer-potency criteria.
Cheeseman et al. commented on the skewed composition of their dataset as follows: Another conservativism is the nature of the CPDB itself as a basis for establishment of a regulatory threshold. The overall percentage of positive carcinogens in the CPDB is approximately 54%, which is comparable to the level of 52% reported for the smaller NCI/NTP (National Cancer Institute/National Toxicology Program) subset of carcinogenicity studies. Huff et al. (1995) evaluated several aspects of the NCI/NTP database, including the basis for nomination of the substances for study. In general, substances were nominated either because they were suspected of carcinogenic activity based on structural concerns or they represented substances of potentially high exposure. Substances nominated as suspected carcinogens accounted for 86% of those substances positive in at least one species in the NCI/NTP database. These compounds represent 90% of compounds positive in more than one species. This high rate of positivity is indicative of a database that is (justifiably) skewed towards expected carcinogens. In contrast, Huff et al. (1995) postulated that an expectation of a reasonable range of human carcinogenicity is likely to be no higher than 5-10% of chemicals in production. From this, we can expect that a regulatory threshold based on the CPDB or the NCI/NTP subset would be anywhere from fivefold to 10-fold more conservative than a threshold derived from a subset more representative of the larger population of commercially produced substances.
EFSA asserts that the dataset is broadly representative of the ‘world of chemicals’ and comments as follows: The term “world of chemicals” is a poorly defined term and can be interpreted in various ways. In the present context the term is meant to address the question whether the structure(s) of a chemical under consideration is represented by the chemicals in the database used to derive the respective TTC value.
The problem of selection bias is confirmed by Boobis et al.
18
: these databases are impacted by a large selection bias because those substances tested were predominantly already suspected of being carcinogens, based on structural features or toxicological effects. Indeed, Fung et al. (1995) reported in their analysis that whilst of 400 substances tested in cancer bioassays by NCI/NTP, 210 were positive, of these, 181 had been selected for the testing program based on suspected carcinogenicity, while only 29 positive substances (7% of 400) had been chosen due to high exposure potential.
Regarding the Cheeseman et al. dataset, if various compound types are eliminated from further consideration – CoC; CoC substances, endocrine disruptors, organophosphorus compounds, heavy-metal compounds and low/moderate-potency carcinogens (TD50 > 50 mg/kg/day) – only around 150 critical compounds remain of which up to 20% appear to be non-mutagens (based on data presented by Snodin and McCrossen 10 ).
Lowest statistically significant TD50 versus default harmonic-mean TD50
EFSA released a scientific opinion (dated 1 March 2016) on approximately 140 substances from the CPDB that are of particular concern. 19 A high proportion of the compounds involved can be classified as belonging to the CoC; for example, around 50% are N-nitroso compounds. It is noteworthy that harmonic-mean TD50s were employed throughout, and in general, the lowest statistically significant TD50s cited in Cheeseman et al. are distinctly lower. The latter values are 50–90% of the default value for several non-mutagenic pharmaceuticals/pesticides such as dieldrin, enovid, kepone, methimazole, phenesterin and heptachlor. On the one hand, EFSA is content to employ harmonic-mean TD50s for this exercise, whereas the (presumed) use of the lowest statistically significant TD50 by Kroes et al. 6 is supported by EFSA in relation to derivation of the cancer TTC. Concerning false carcinogens, an important example is carbamate esters for which Cheeseman et al. list around 18 compounds, all claimed to be carcinogens, whereas the CPDB indicates that all but five show no positive test. In addition, the majority are non-mutagenic. (Some carbamates that are reported to be mutagenic may reflect the presence of mutagenic impurities in technical-grade test materials.) A further example is acrolein which is listed in the CPDB as showing no positive test in either rats or mice 20 ; on the other hand, a TD50 of 14.4 mg/kg/day is cited by Cheeseman et al. 15 Methyl methanesulfonate and captan are two examples where the use of the lowest statistically significant TD50 (as listed by Cheeseman et al.) grossly overestimates carcinogenic potency (based on the geometric-mean value).
Evaluation of non-mutagenic carcinogens
Gold et al. 21 determined that for compounds in the CPDB at the time a high proportion of both mutagens and non-mutagens were carcinogenic, but the proportion was higher for mutagens (75%) than non-mutagens (45%).
A statement confirming the generally thresholded nature of non-mutagenic carcinogens is provided by the EFSA in their 2016 report,
1
as follows: Carcinogens which are not directly DNA-reactive often have a threshold mode of action and, in general, NOAELs for these are in the same range or higher than NOAELs for other types of toxicity. Thus, EFSA (2012) concluded that TTC values that are higher than the value of 0.15 μg/person/day are appropriate for any chemical where the weight of evidence for DNA reactivity is negative. The expert group concurred with this statement. Although further expansion of the TTC cancer dataset and an updated carcinogenicity potency database is desirable, it is not expected that the overall threshold for carcinogenicity based on linear extrapolation for genotoxic carcinogens would significantly change.
Approaches for pharmaceutical impurities and LTL exposure
The LTL concept is described by the EFSA as follows: The current uses of TTC, with the exception of impurities in pharmaceuticals and possibly consumer products, are primarily for lifetime human exposure assessments. A number of proposals have been put forward for adjusting the TTC value for chemicals with a structural alert for genotoxicity for shorter than chronic durations of exposure (Müller et al, 2006, Humfrey 2007, Felter et al, 2009, Galloway et al. 2013), mainly in the context of impurities in pharmaceuticals and consumer products. The proposals made are mainly based on Haber’s law for cancer risk assessment, i.e. that higher exposures for shorter periods of time are essentially equivalent to lower exposures for longer periods of time. The EFSA (2012a) was not confident about the general applicability of these proposals. It therefore recommended that the issue of less than chronic exposure should be addressed on a case-by-case basis. This could be done for example by considering the margin between the appropriate TTC value (without any adjustment for duration of exposure) and the estimated dietary exposure. There is no mention of the ICH M7 consensus guideline on mutagenic impurities
3
; The EFSA statement mentions genotoxicity, whereas in ICH M7, structural alerts are interpreted in the context of mutagenicity; The determination of bacterial mutagenic potential in ICH M7 is a sophisticated and multi-layered procedure involving inter alia; In silico evaluation using two complementary systems (rule- and statistical-based); Follow-up using expert assessment; Use of read-across from appropriate reference compounds (e.g. evaluating the drug substance and impurities for the presence of similar structural moieties); Performing an Ames’ assay, as necessary; For human pharmaceuticals data from mammalian-cell assays are discounted if the impurity is predicted with confidence to be non-mutagenic and/or is an experimental non-mutagen – not the case veterinary; If an impurity with a conventional structural alert for bacterial mutagenicity is shown by either in silico prediction or experimental means to be a non-mutagen, it is treated as a normal (non-mutagenic) impurity with no structural alert; Compound-specific limits can be determined for mutagenic impurities mainly based on the determination of an in vivo threshold for mutagenic effects
22
establishing that the mechanism of carcinogenicity is unrelated to genotoxicity, or linear extrapolation of carcinogenic potency data.
11
Some examples are shown in Table 1.
Examples of compound-specific limits for mutagenic impurities in pharmaceuticals.
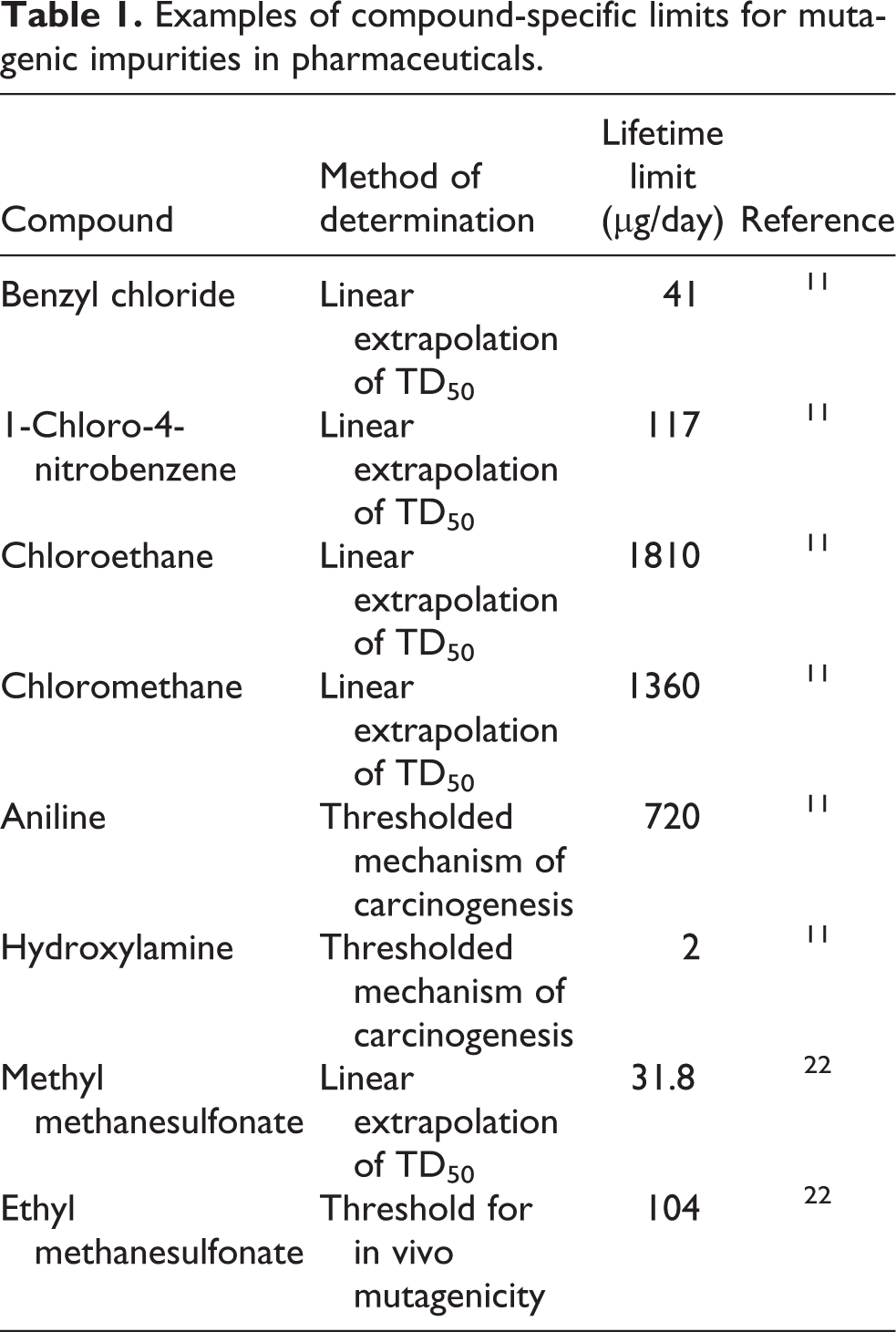
Thus, EFSA’s characterization of the ICH M7 approach as applying broadly to ‘chemicals with a structural alert for genotoxicity’ is extremely misleading. ICH M7 indicates: ‘The existence of impurity structural alerts alone is considered insufficient to trigger follow-up measures, unless it is a structure in the cohort of concern’. Whereas, EFSA relies on conventional structural alerts for genotoxicity, 23 which are often inappropriate and significantly over-predictive, the precise targeting of the potential for bacterial mutagenicity using in silico and other techniques enables a number of conventional alerts to be discounted. One such alert is the α, β-unsaturated ketone moiety, 10 and a key example is mesityl oxide (4-methylpent-3-en-2-one, CAS no 141-79-7; a potential minor contaminant in acetone) which was used frequently as a source of regulatory-agency deficiency questions if acetone was used as a solvent in the synthesis of a drug substance. In fact, mesityl oxide tests negative for mutagenicity in five strains of Salmonella typhimurium and in the in vitro micronucleus assay using human blood peripheral lymphocytes. 24
Structural alerts and in silico prediction of mutagenic potential are also discussed by Boobis et al. 18 who conclude that ‘It is thus critical in the application of the TTC approach that those compounds likely to be mutagenic via a DNA-reactive mode action can be reliably identified on the basis of structure alone’. As noted above, experience with pharmaceutical impurities indicates that employing structural alerts alone for predicting mutagenic potential can be erroneous in many cases. On the other hand, use of historical data and (commercial) in silico systems supported by large datasets provides the most reliable predictions, particularly when combined with an expert assessment (e.g. evaluating mode of action (MOA), structural analogues, data reliability and overall weight of evidence). 25 However, both structural alerts and in silico QSAR systems are reported to be over-predictive for compounds containing bulky or electron-withdrawing substituents because an alert is triggered when a particular toxicophore is present irrespective of the structural environment surrounding the toxicophore. 26
As regards the LTL concept, this is based on Haber’s rule, a fundamental concept in toxicology, where concentration (C) × time (T) = a constant (k), the carcinogenic effect being related to both dose and duration of exposure. The LTL levels proposed in ICH M7 are significantly lower than values calculated using Haber’s rule and are also in compliance with a 10−6 cancer risk level if treatment durations are no longer than 6 months. 3 LTL multiples of the generic lifetime limit (1.5 µg/day) or compound-specific lifetime limit are 80, 13.3 and 6.7 for exposure durations of 1 month, 1–12 months and 1–10 years, respectively. Thus, the limit for short-term (≤ 1 month) exposure to hydroxylamine (Table 1) would be 160 µg/day. Haber’s rule has been used extensively in the context of Registration, Evaluation, Authorisation and Restriction of Chemicals 27 and in regulations on the control of substances hazardous to health, in which both frequency and duration of exposure are considered to be critical. 28
The concept of a threshold for mutagenic carcinogens is a closely related issue for which there are a number of examples such as ethyl methanesulfonate
29
and AA (acrylamide, whose metabolite glycidamide is a direct-active bacterial mutagen
30
). In its most recent review
31
of AA, EFSA reported (inter alia) that: Mean and 95th percentile dietary AA exposures across surveys and age groups were estimated at 0.4–1.9 μg/kg body weight (b.w.) per day and 0.6–3.4 μg/kg b.w. per day, respectively. Although the epidemiological associations have not demonstrated AA to be a human carcinogen, the margins of exposure indicate a concern for neoplastic effects based on animal evidence.
Numerous epidemiological studies have been undertaken on AA and in a 2012 review Lipworth et al.
32
state: we found no consistent or credible evidence that dietary acrylamide increases the risk of any type of cancer in humans, either overall or among non-smokers. In particular, the collective evidence suggests that a high level of dietary acrylamide intake is not a risk factor for breast, endometrial, or ovarian cancers, which have generated particular interest because of a conjectured hormonal mechanism of acrylamide.
Proposals for re-assessment of the cancer TTC by Boobis et al. 18
Following a detailed commentary on the current status of the cancer TTC, Boobis et al. conclude: it is apparent that the current TTC values for substances assumed to be DNA-reactive mutagens and carcinogens could be questioned, for a number of reasons. These values are likely to be conservative.…From a scientific perspective, there is a clear case to be made for the re-assessment of these values. While existing values appear to be adequately protective of human health, a number of assumptions and approaches used in their original derivation have been superseded by advances in knowledge. Hence, to ensure a robust, transparent basis for all aspects of the TTC approach, re-assessment of these values is timely. In addition, restrictions on the use of chemicals should be commensurate with their risk, which may not be the case with the application of the current values.
Principal requirements for constructing a suitable dataset for re-evaluation of the cancer TTC 18 .

Discussion and Conclusions
The three key criticisms of the derivation of the cancer TTC are considered to remain unanswered.
Concerning the issue of the skewed dataset, Cheeseman et al., 15 whose analysis employed a closely similar dataset to that used by Kroes et al. 6 , clearly state that their dataset is (justifiably) skewed to such an extent that any regulatory threshold would be 5- to 10-fold more conservative than that derived from a subset more representative of the larger population of commercially produced substances. The latter figure appears to be based on comparison of the proportion of carcinogens in the CPDB (50%) with that estimated in chemicals of commerce (5–10%). Thus, a random selection of 1500 substances from those used in commerce would be expected, on average, to contain 75–150 carcinogens most probably with potencies significantly lower than CPDB carcinogens which were for the most part selected on the basis of being likely (potent) carcinogens. EFSA has never mentioned this kind of evaluation and instead attempts to construct a contradictory argument based on a ‘world of chemicals’ chemoinformatics concept.
Regarding the use of the lowest statistically significant TD50s in the derivation of the TTC by Kroes et al., which is defended by EFSA, this stance appears to have been reversed by evidence from a recent EFSA report that uses harmonic-mean TD50s throughout. 19 Although EFSA has confirmed its position that linear extrapolation of carcinogenic potency data is inappropriate for non-mutagenic carcinogens, so far it has ignored the presence/impact of such compounds in the CPDB dataset. Over the last decade or so, there have been multiple missed opportunities to perform an ab initio evaluation of a suitable dataset using appropriate inclusion and evaluation criteria, and the outcome of the non-transparent and arguably flawed evaluation by Kroes et al. has been accepted by EFSA and other regulatory bodies. The possibility cannot be discounted that there has been a general policy of avoiding any re-evaluation for fear of a less conservative outcome. This is in marked contrast to the use of transgenic mouse models in carcinogenicity bioassays which, before their widespread acceptance, were subjected to a detailed review by a working party of the EMA. 38 A joint report by three EU expert committees (SCHER, SCENIHR, SCCP) was also somewhat critical of the cancer TTC in relation to being based on worst-case assumptions, assuming no threshold for carcinogenicity and databases requiring further development and validation. 39
A more general point on linear extrapolation down to extremely low risk levels is made by Cheeseman et al. 15 : ‘the procedure of linear extrapolation to low dose is highly conservative and may exaggerate risk by as much as two orders of magnitude. This may be particularly true for non-genotoxic carcinogens’. These remarks about significant overconservatism are consistent with recent challenges to the LNT concept and its adoption by regulatory agencies. 12,13 Three of the four criticisms mentioned in previous sections (skewed dataset, lumping together of genotoxic and non-genotoxic carcinogens and overconservatism of associated with the LNT concept) were openly acknowledged by Cheeseman et al. And so it’s alarming that these issues have not been evaluated by regulatory agencies.
As noted by Galloway 40 in the United States, an individual has a 40% chance of developing cancer over a lifetime. Exposure to a mutagen/potential carcinogen at a level estimated to increase risk by 1 in 106 would elevate the individual cancer risk from 0.400000 to 0.400001.
Overall, the lifetime cancer TTC is likely to be over-conservative by possibly 2 orders of magnitude. Moreover, deriving a TTC metric accurate to two significant figures (i.e. 0.15 or 1.5 µg/day) seems extremely implausible given that the procedure involved linear extrapolation of a highly variable dataset of carcinogenicity potency data over 5 or 6 orders of magnitude. Alternative cancer-potency metrics such as the T25 41 –43 (which is much simpler to determine than the TD50 and so would most likely increase the size of the dataset) could be employed as mentioned by EFSA. 1 (Although these two metrics are reasonably well correlated, 41 they appear to be sufficiently different to negate the determination of a TTC value to two significant figures.) Anyone familiar with pharmaceutical regulatory affairs would most likely be able to quote the value employed for lifetime exposure to a mutagenic impurity (1.5 µg/day), but based on the author’s experience relatively few have any clear notion of its derivation. For example, a 2011 article 44 asserted that the cancer TTC dataset was obtained from the Registry of Toxic Effects of Chemical Substances rather than the CPDB. It’s therefore unsurprising that a limited number of scientists have taken the time to delve into the somewhat opaque background to the cancer TTC.
EFSA’s concerns about use of the LTL principle are understandable to some extent given that the possibility of short-term exposure is much more limited for foodstuffs compared to pharmaceuticals. On the other hand, EFSA has not provided any rational argument and evidence to support its position and, moreover, the flow chart in its 2016 report
1
appears to indicate an increased limit of 1.5 µg/day for genotoxic compounds when the duration of exposure does not exceed 12 months. Two further considerations are: The highest LTL multiple mentioned in ICH M7 is 80 in relation to exposure duration of 1 month, which is lower than the estimated extent of overconservatism of the lifetime TTC. Research on smoking-related mortality shows a dramatic beneficial effect of early cessation. A report on one million women smokers indicated that stopping before age 40 years avoids more than 90% of the excess mortality caused by continued smoking; stopping before age 30 years avoids more than 97% of excess mortality.
45
Given the foregoing, the position of EFSA on the cancer TTC is considered to be non-robust when subjected to a critical analysis of relevant data and regulatory precedents. Furthermore, there appears to be no example of any regulatory body, including EFSA, adopting the cancer TTC only after performing an independent re-assessment in order to accommodate the various criticisms of the Kroes et al. derivation. This state of affairs could be attributed to a pattern of confirmation bias presumably because no agency would want to put itself in a position of challenging the current consensus, which ignores the general philosophy that science advances in an iterative fashion by deliberately challenging hypotheses and poorly established concepts.
The cancer TTC has gradually gathered more ‘superstructure’ as it has been applied or proposed for application in areas other than foodstuffs and pharmaceuticals, such as food contact materials, flavouring substances, herbal products, cosmetic ingredients and personal and household care products. 39 Recent examples are the EMA guidance on setting health-based exposure limits 46 and draft guidance on mutagenic impurities in veterinary medicines. 47 It would be a great pity and a major disservice to toxicological risk assessment if the cancer TTC as proposed by Kroes et al. were allowed to continue on its current trajectory. However, ILSI (International Life Sciences Institute) Europe is sponsoring an initiative on a re-evaluation as described by Boobis et al. 18 In order to initiate this process, the authors believe that the principal requirement is the construction of a fully curated, publicly available dataset on genotoxic and non-genotoxic compounds using predetermined inclusion and exclusion criteria. In addition, assessment of the relevance of tumours for human health, dose–response and MOA for tumours detected in rodent carcinogenicity bioassays are important issues. Distinguishing compounds that are carcinogenic by a DNA-reactive mutagenic MOA from those that act via a non-genotoxic MOA is a critical consideration. While such an exercise is to be greatly encouraged, there may be difficulties in harmonizing this kind of approach and that in ICH M7 which employs bacterial mutagenicity as a surrogate for DNA-reactivity and excludes use of data from a mammalian-cell assay such as the in vitro micronucleus test. 48 There are often potential issues on deciding whether a particular compound should be tagged as a ‘mutagenic carcinogen’ or ‘genotoxic carcinogen’, and Boobis et al. 18 focus on carcinogens that have a DNA-reactive mutagenic MOA (determined on the basis of a positive Ames’ test plus data from other genotoxicity assays because 20–30% of Ames’-positive compounds are reported to test negative for carcinogenicity). Unfortunately, there is no mention of the newly developed ToxTracker assay 49 which has the ability to provide a direct readout of DNA-reactivity and so should be superior to most other genotoxicity assays in providing MOA data. (Clearly weight-of-evidence evaluations will be critical across the board particularly in relation to mechanism and human relevance of rodent carcinogenesis 50 –52 ). An additional concern is that the initiative by Boobis et al. mentions no role for epidemiological evidence although it is acknowledged that the enzymes involved in producing DNA-reactive metabolites are frequently less active in humans than in rodents. As described previously, AA is an example of a potent genotoxic carcinogen in rodents for which extremely comprehensive epidemiological studies indicate no overall association with an increased cancer risk, and thus, the current human exposure to AA could provide a benchmark and ‘reality check’ impacting on determination of the cancer TTC.
In their derivation of the cancer TTC, Kroes et al. show that 3.9% (17 out of 434; Table 3) of compounds (i.e. with exclusion of CoC (cohort of concern) compounds, heavy-metal-containing compounds, highly chlorinated compounds, organophosphorus compounds, steroids and tetrahalogenated dibenzodioxins and dibenzofurans) possess TD50 values < 1.25 mg/kg/day (corresponding to an exposure less than 1.5 µg/day using linear extrapolation of 1 in 105). Presently, this small group of potent carcinogens has been effectively ignored in relation to the derivation of the lifetime TTC of 1.5 µg/day for pharmaceuticals. Consequently, based on this kind of issue and regulatory pragmatism retention of the current value of the TTC has been advocated, while accepting that the Kroes et al. derivation has many deficiencies. 53 Since no information is available in the public domain on the precise makeup of the dataset employed by Kroes et al., it is not possible to identify with certainty the 17 high-potency compounds. However, using the current version of the CPDB 14 mutagenic (Ames’-positive) compounds have been identified with (oral) TD50s < 1.25 mg/kg/day (Table 4). Although clearly not a perfect match with the 17 high-potency compounds identified by Kroes et al., this group of compounds is considered sufficiently representative for current assessment purposes. Data from Table 4 indicate that the potency metrics for six compounds are based on studies with a single dose level and so would be excluded from any re-evaluation of the TTC using criteria set out by Boobis et al. 18 Thus, on the basis of this initial analysis only 8/434 high-potency compounds (1.8%) remain, and there could be further attrition if other criteria shown in Table 2 were applied. It is believed that these ‘outlier’ compounds are likely to be of negligible significance in any re-evaluation of the cancer TTC.
Numbers and fractions of compounds in different structural categories (excluding CoC compounds and other categoriesa) estimated to produce a risk ≥ 1 in 106 at different intake levelsb.

aHeavy-metal-containing compounds, highly chlorinated compounds, organophosphorus compounds, steroids and tetrahalogenated dibenzodioxins and dibenzofurans.
cCorresponding TD50 s for the two intake levels are: < 1.25 and 1.25–12.5 mg/kg/day.
Fraction = compounds in particular intake category divided by total number in structural category; numbers and fractions are shown cumulatively.
Data from CPDB on mutagenic compounds in different structural categories with high carcinogenic potencya.

aTD50 < 1.25 mg/kg/day; TO = (main) target organ(s).
bGeometric mean TD50 in rat or mouse, whichever is lower.
Several lines of evidence support the notion that the current values of the cancer TTC values for foodstuffs and pharmaceuticals are over-conservative and not commensurate with the actual risk to human health posed by DNA-reactive mutagenic carcinogens. It is concluded that there is a strong case for re-evaluation of the cancer TTC, which is supported by Boobis et al. 18 who have developed a detailed strategy for this purpose. While the Boobis et al. approach is endorsed in general, two modifications are suggested: use of the ToxTracker assay for determination of DNA-reactivity and incorporation of epidemiological data on compounds such as AA and aflatoxins.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
