Abstract
Objective:
Cytokines, molecules within the immune system that affect either a pro- or anti-inflammatory response, have previously been shown to influence birth outcomes. The maternal cytokine gene–environment interactions are thought to alter their expression, potentially influencing susceptibility to adverse birth outcomes. The aim of this study was to determine the association between the maternal interleukin-1β (IL-1β) haplotype and expression variation with oxides of nitrogen (NOx) levels, and thereafter investigate the IL-1β haplotype-specific effects of NOx exposure levels, IL-1β mRNA expression and other variables on gestational age.
Material and methods:
Using the prospective Mother and Child in the Environment (MACE) birth cohort in Durban, South Africa, 335 participants were genotyped for the IL-1β haplotype. Previous studies showed that three single nucleotide polymorphisms (SNPs), IL-1β-1464G/C, -511C/T and -31C/T, constitute the IL-1β functional haplotype. These SNPs were genotyped using a restriction fragment length polymorphism assay, while IL-1β mRNA expression was measured using a quantitative real-time polymerase chain reaction assay. Individual estimates of NOx exposure were obtained by land use regression modelling. A multivariate linear regression analysis was employed to test for significant effects on gestational age.
Results:
IL-1β mRNA expression was found to possess a haplotype-dependent effect (p = 0.0001) and its expression levels positively correlated with NOx levels (r = 0.34; p = 0.006). In the high haplotype model, a unit increase in NOx exposure level was associated with a decrease in gestational age by 1 week (p = 0.02). Furthermore, gestational age decreased by 0.9 weeks for every unit increase of IL-1β mRNA expression level (p = 0.025). HIV-1 positivity was associated with a 0.2-week decrease in gestational age (p = 0.035) in the intermediate haplotype model and a 0.4-week decrease in the high haplotype model (p = 0.044).
Conclusion:
These data have implications for better understanding the effect of prenatal NOx exposure on gestational age and demonstrate the role of the IL-1β haplotype in modulating the effects of NOx exposure.
Keywords
Introduction
Ambient air pollution has been linked to adverse birth outcomes, including increased rates of mortality and various diseases in children, the elderly and pregnant women, who are particularly sensitive. 1,2 The risk for neonatal and infantile morbidity and mortality increases with decreasing gestational age 3 and is a predictor of mortality and chronic morbid conditions later in childhood and adulthood, namely developmental disabilities and cardiovascular and respiratory diseases. Such conditions are associated with impaired responses to environmental agents, further impacting morbidity.
Oxides of nitrogen (NOx), that is, nitrogen dioxide (NO2) and nitric oxide, are a component of industrial and vehicular exhaust emissions. Traffic is reportedly the principal source of NO2 and the residential distance from roadways impacts levels. 4,5 In Durban (South Africa), the primary source of NOx includes mobile sources, responsible for emitting 11,500 tons per annum and over 60% of the total emissions, and industry (largely concentrated in the Durban South Industrial Basin (DSIB)), responsible for emitting 5400 tons per annum (29% of the total emissions). 6 The DSIB comprises communities residing among heavy industry and significant roadways and is notorious for being among the most highly polluted regions in South Africa. 7 The region is associated with a greater prevalence of adult respiratory conditions (including higher adult asthma rates than those internationally), childhood asthma and marked airway hyperreactivity compared to the northern region in Durban. 6 –9
NOx, a strong oxidant, generates reactive oxygen species thereby inducing oxidative stress and the resultant pro-inflammatory response in vivo. 10,11 Pregnancy is an inflammatory state characterized by gestational stage-specific immune responses, thus pollutant-induced alterations in response may adversely affect birth outcomes. 12 Interleukin-1β (IL-1β), a mediator of the pro-inflammatory response to infection and oxidative stress, is implicated in the pathophysiology of various chronic diseases through dysregulated or increased synthesis. 13 Data on the maternal IL-1β response to NOx exposure, however, are scarce or non-existent. Most studies on air pollution and birth outcomes emanate from high-income countries, with none or few from low- or middle-income countries. In this study, we investigated the effect of prenatal NOx exposure on the maternal IL-1β response and explored the association between maternal IL-1β expression on gestational age in a predominantly African birth cohort in Durban, South Africa. Since single nucleotide polymorphisms (SNPs) affect transcription in a haplotype-dependent manner, 14 we characterized the maternal IL-1β haplotype distribution and associated expression profile using three major functional IL-1β promoter SNPs (IL-1β-1464, -511 and -31). 15 We also explored the role of such haplotypes in determining the effects of NOx exposure levels, IL-1β expression and other variables on gestational age, thereby identifying vulnerable subgroups to NOx-induced and IL-1β haplotype-dependent gestational age outcomes. We hypothesized that chronic elevated NOx exposure may play a significant role in altering maternal IL-1β expression and negatively influencing gestational age outcomes, while IL-1β haplotype-specific associations between maternal haemoglobin (Hb), body mass index (BMI; trimester 3), NOx levels, IL-1β mRNA levels, HIV-1, income and education may play a role in influencing gestational age.
Methods
Subjects
Study subjects included a subset of 338 participants from the Mother and Child in the Environment (MACE) prospective birth study that commenced in 2013 in Durban, KwaZulu-Natal, South Africa. MACE participants were recruited from public sector antenatal clinics, during the first trimester. The cohort comprised of individuals from African, Caucasian, Indian and Coloured ethnicity. Informed and signed consent was obtained from each participant. Study exclusion criteria were twin births, pregnancy complications and smoking. The study was approved by the Biomedical Research Ethics Committee of the University of KwaZulu-Natal (reference number: BF263/12).
Genotyping
Maternal DNA was extracted from third trimester whole blood using the FlexiGene DNA Kit (QIAGEN GmbH, Hilden, Germany) as per manufacturer’s protocol. DNA concentration was quantified spectrophotometrically (NanoDrop Spectrophotometer, Biolab, Scoresby, VIC, Australia) and standardized to 10 ng/µl. The IL-1β gene SNPs at the following positions, IL-1β-511 C>T, IL-1β-31 T>C and IL-1β-1464 G>C comprising the IL-1β haplotype, were genotyped by the polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) method. The PCR primers and cycling conditions used for IL-1β amplification are shown in Table 1. The PCR mixture included 30 ng DNA, 5 μl buffer (5×), 3 μl magnesium chloride (25 mM), 2 μl deoxynucleotide (10 mM), 0.125 µl Taq polymerase (5U/µl; GoTaq®G2 Flexi DNA Polymerase (Promega, Madison, Wisconsin, USA)), 0.125 μl of each primer (20 pmol/μl) and nuclease-free water (25 µl total volume). To confirm specificity, PCR products were visualized on a 1% agarose gel with a 50-bp molecular marker. PCR products were digested for 16 h at 37°C, IL-1β-511 C>T (10 U AvaI), IL-1β-31 T>C (10U AluI) and IL-1β-1464 G>C (10U StyI). Digested PCR products were visualized on a 2% agarose gel with a 50-bp molecular marker.
Primers used in study. Primers and cycling conditions used are listed in the table below.

IL-1β: interleukin-1β; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
RNA isolation and analysis
RNA was isolated from a subset (n = 66) of subjects based on haplotype, HIV status and NOx exposure. NOx exposure was categorized into high NOx (HN) and low NOx (LN) groups using the median, that is, 36 μg/m3 as the split point. RNA was extracted from 34 HIV-1-positive and 32 HIV-1-negative subjects. RNA was extracted immediately after thawing from 500 μl of whole blood using the TRIzol method (Invitrogen, California, USA) as per manufacturer’s instructions. The total RNA concentration was quantified using a spectrophotometer and all RNA samples were DNase treated. One microgram of total RNA from each sample was reversed transcribed using the iScript cDNA synthesis kit (Bio-Rad, California, USA).
Real-time PCR RNA quantitation
Quantitative real-time PCR was performed for the IL-1β gene on RNA isolated from participants within high and low NOx exposure groups with comparable haplotypes and HIV-1 infection status. The IL-1β gene was amplified using primers obtained from a public database, PrimerBank, to uniquely amplify the IL-1β gene (NM_000576.2 GenBank accession no.). The primers used were 5′-ATGATGGCTTATTACAGTGGCAA-3′ (forward) and 5′-GTCGGAGATTCGTAGCTGGA-3′ (reverse). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was validated as the most suitable reference gene, based on its PCR efficiency. The GAPDH (NM_002046) primers used were 5′ -AAGGTCGGAGTCAACGGATT-3′ (forward) and 5′-CTCCTGGAAGATGGTGATGG-3′ (reverse).
Each PCR reaction comprised of either 0.5 pmol/μl for IL-1β or 0.25 pmol/μl for GAPDH of each primer, 5 μl SYBR Green I Master Mix (2X) (Bio-Rad), 1 μg cDNA and water to 10 μl. Reactions were run in duplicate on a CFX real-time machine (Bio-Rad) with cycling condition as follows: 1 cycle at 95°C for 10 min, followed by 40 cycles at 95°C for 30 s, at 60°C for 30 s and at 72°C for 30 s. Relative mRNA levels were calculated by generating standard curves from serial dilutions of a known concentration of cDNA sample for IL-1β and GAPDH. Individual samples were then analysed against the standard curve and represented as levels of mRNA per microgram of cDNA. Results are depicted as a ratio of IL-1β to GAPDH per microgram of cDNA. To confirm amplification specificity, the PCR products were subjected to a melting curve analysis and agarose gel electrophoresis.
NOx exposure data
An estimate of individual exposure levels for NOx, a widely-utilized surrogate for other traffic-related pollutants including particles, was available. A NOx passive sampling programme was used to characterize community exposure levels. NOx monitoring locations were randomly selected for each of the study areas (Durban North and South). Monitoring efforts focused on characterizing the spatial variation in air pollution exposure given the surrounding environmental factors such as land use and topography. Data generated from this monitoring were used in developing land use regression models, which permitted the prediction of exposure at each of the participant’s homes, through geographical positioning data.
For the model, NOx was measured at selected locations and regressed against site specific a priori defined (i.e. direction of effect) geographic predictors such as road length, land use types, topography and population and housing density in a multivariate regression model. The NOx measurements were conducted using Ogawa samplers (Ogawa & Co., Inc., V3.98, USA), which were deployed at 40 randomly selected sites (Durban North and South) over a once repeated 2-week periods during midwinter and midsummer, 16,17 thus accounting for seasonal variation. The results from the two sampling periods were adjusted using data from an existing air quality monitoring station consistent with the sampling periods as per the European Study of Cohorts for Air Pollution Effects approach. 18 The annual adjusted averages of NOx were obtained by adjusting the arithmetic mean of the available measurements (i.e. both sampling periods) per site using the difference between the sampling period and the annual average at the air quality monitoring station. The adjusted NOx measurements were then used in model development of which the regression coefficients were applied to participants.
Statistical analysis
All statistical analyses were performed using InStat GraphPad Prism version 5 and SPSS Statistics version 25 (SPSS Inc., Chicago, Illinois, USA). The IL-1β haplotypes were constructed using Haploview version 4.2 from IL-1β-1464, -511 and -31 SNPs. The high haplotype (GTC/CTT/CCT), the intermediate haplotype (CTC) and low haplotype (GCT/CCC) were defined as previously published. 15 IL-1β mRNA expression values are expressed as medians. Differences between the medians of comparable haplotype LN and HN groups were determined using the Student’s t-test. High and intermediate haplotypes were combined and compared to the low haplotype for both HIV-1 positive and negative groups. Differences in expression between haplotypes were determined using an analysis of variance. Pearson’s correlation, χ 2 or Fisher’s exact test was used to analyse genetic and clinical data. Scatter plots and bar graphs were generated using InStat GraphPad Prism version 5. p Values of <0.05 were considered as statistically significant. SPSS Statistics version 25 was used to conduct a multivariate linear regression analysis for gestational age on all participants for which NOx data were available (n = 272) with adjusting the following variables chosen a priori: maternal Hb, BMI, NOx exposure levels, IL-1β/GAPDH, HIV-1 status, education and income.
Results
Cohort characteristics
The multi-ethnic cohort was predominantly African (76%) and had a 34% HIV-1 prevalence (Table 2). The preterm birth (PTB) and low birth weight prevalence were 10% and 15%, respectively. The NOx exposure range for the cohort was 12.8–56.9 µg/m3 with a median of 36.23 µg/m3. There was no significant difference in the IL-1β SNPs (-1464, -511 and -31) and haplotype distribution between the low and high NOx exposure groups (p = 0.8, 0.3, 0.5, 0.1, respectively; Table 3). The predominant cohort haplotype is the high producer and the distribution for the African group was 40%, 35% and 25% for the high, intermediate and low haplotypes, respectively.
Maternal clinical, demographic and birth outcome data.a

NOx: oxides of nitrogen; BMI: body mass index; Hb: haemoglobin.
aMaternal clinical, demographic and birth outcome data are shown with a comparison by NOx exposure categories, high (>=36 μg/m2) and low (<36 μg/m2). A p value of <0.05 for the Student’s t-test or χ 2 was considered as statistically significant.
IL-1β genotypic and haplotypic distribution in the cohort.a
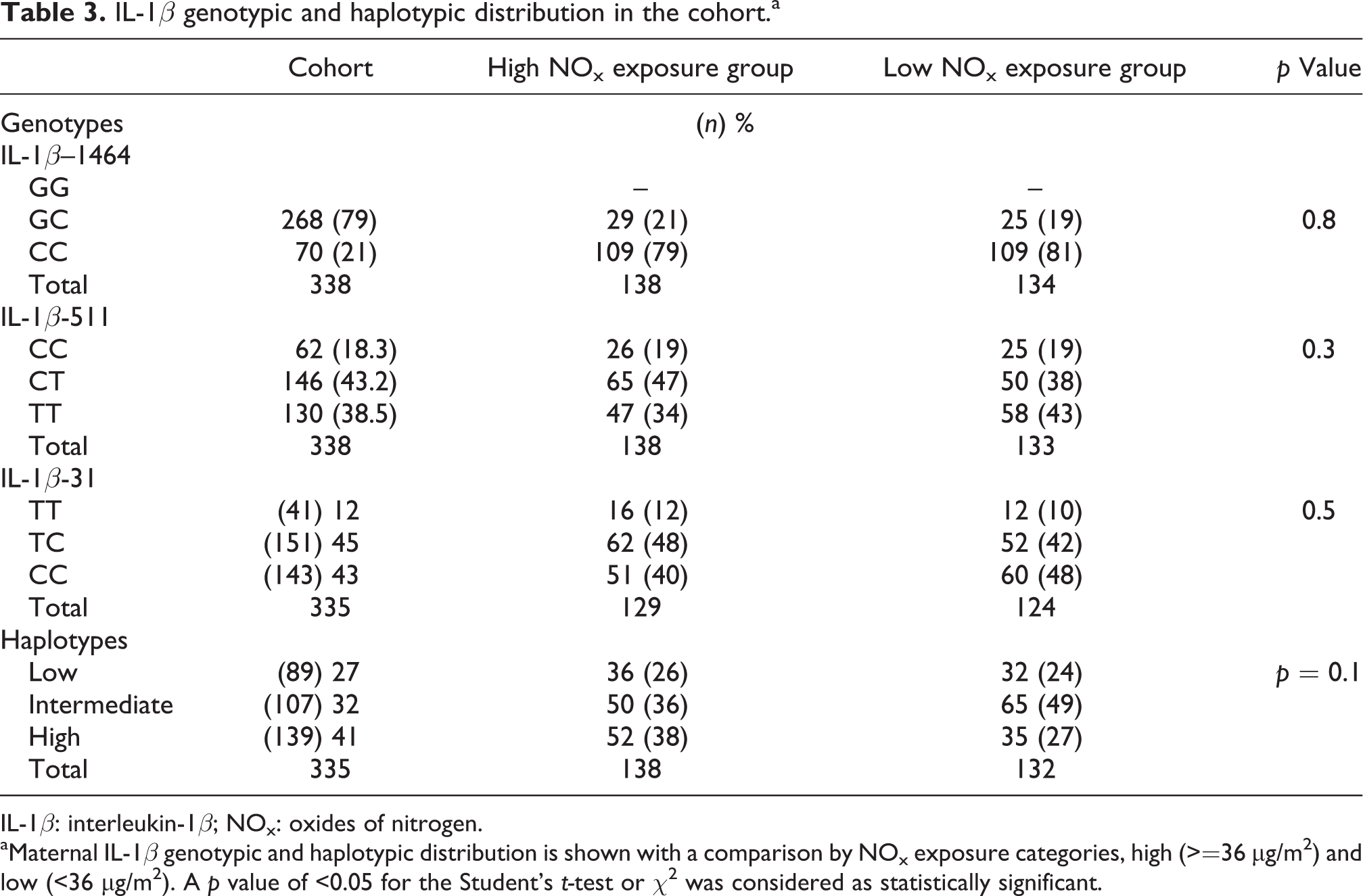
IL-1β: interleukin-1β; NOx: oxides of nitrogen.
aMaternal IL-1β genotypic and haplotypic distribution is shown with a comparison by NOx exposure categories, high (>=36 μg/m2) and low (<36 μg/m2). A p value of <0.05 for the Student’s t-test or χ 2 was considered as statistically significant.
IL-1β genotypes associate with mRNA expression levels
IL-1β gene expression was associated with the three major functional IL-1β promoter SNPs (IL-1β-1464, -511 and -31) and the IL-1β haplotypes. High levels of IL-1β expression were significantly associated with IL-1-31 T/T, IL-1-511 C/C and IL-1-1464 G/C (p = 0.0381, 0.038 and 0.046, respectively; Figure 1(a) to (c)). High levels of IL-1β mRNA were associated with GTC when compared to other haplotypes (p = 0.023; Figure 1(d)). Haplotypes CCC, CCT and CTT (Figure 1(e)) were grouped according to expression. Haplotype CCC showed similar expression patterns as the low haplotype, while CCT and CTT had similar expression patterns as the high haplotype. Expression in the high haplotype (GTC/CTT/CCT) was significantly higher than other haplotypes, that is, intermediate (CTC) (p = 0.0017) and low (GCT/CCC; p = 0.0001), and the intermediate haplotype expression was significantly higher than that of the low haplotype (p = 0.0025).

IL-1β genotypes associate with expression levels. Gene expression analysis was performed on 66 participants for IL-1β-1464, -511 and -31 genotypes and IL-1β haplotypes. Data are depicted as a normalized ratio of IL-1β versus GAPDH (Figure 1(a) to (f)). The horizontal line represents the median. Differences between groups were determined using an unpaired Student’s t-test. A p value of <0.05 was considered as statistically significant. IL-1β: interleukin-1β; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Effect of NOx exposure level on IL-1β expression in the cohort
IL-1β mRNA levels were positively correlated with NOx levels in the cohort (r = 0.34; p = 0.006; Figure 2). A significant positive correlation was shown independently in both HIV-1 positive and negative groups (p < 0.05; data not shown).

NOx exposure level versus IL-1β expression. We assessed whether there was an association between pro-inflammatory cytokine IL-1β mRNA expression and NOx exposure level in the cohort using the Pearson’s correlation. A p value of <0.05 was considered as significant. IL-1β: interleukin-1β; NOx: oxides of nitrogen.
Both high and intermediate haplotypes were significantly higher in expression than the low haplotype (Figure 3). In both the HIV-1 positive and negative groups, IL-1β expression in participants with the low haplotype was significantly elevated in the HN exposure group compared to the LN exposure group (p < 0.05; Figure 3). In the HIV-1 negative group, expression in HN was elevated in the high haplotype compared to the low haplotype (p = 0.03; Figure 3).

IL-1β expression in different NOx exposure groups. Gene expression analysis was performed on 66 participants by IL-1β haplotype: (high (CCT/CTT/CTC) versus low (CCC/GCT)), HIV-1 status and NOx exposure category: high (>=36 μg/m2) and low (<36 μg/m2). For this analysis, the IL-1β high and intermediate haplotypes were combined to increase the numbers. Data are depicted as a normalized ratio of IL-1β versus GAPDH (Figure 1(a) to (f)). The horizontal line represents the median. The differences between groups were determined using an unpaired Student’s t-test. A p value of <0.05 was considered as statistically significant. IL-1β: interleukin-1β; NOx: oxides of nitrogen; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Multivariate linear regression model for gestational age
A multivariate linear regression analysis for gestational age was conducted on 272 samples adjusting for maternal Hb (trimester 1), BMI (trimester 3), NOx exposure levels, IL-1β mRNA expression levels, HIV status, income level and education, with separate models for each haplotype: low (0), intermediate (1) and high (2) (Table 4). In the first model, that is, without stratifying by haplotype and the low haplotype model (haplotype 0), no significant associations were observed. In the high haplotype (haplotype 2), NOx exposure level, IL-1β mRNA expression level and HIV-1 positivity were negatively associated with gestational age (p < 0.05 for all). In this model (haplotype 2), for each unit increase in NOx level, a 1-week decrease in gestational age was observed (p = 0.02), while for every unit increase in IL-1β mRNA expression level, a 0.9-week decrease in gestational age was observed (p = 0.025); HIV-1 positivity was associated with a 0.4-week decrease in gestational age (p = 0.044). In the intermediate haplotype model (haplotype 1), HIV-1 positivity was associated with a 0.2-week decrease in gestational age (p = 0.035).
Linear regression model for gestational age.
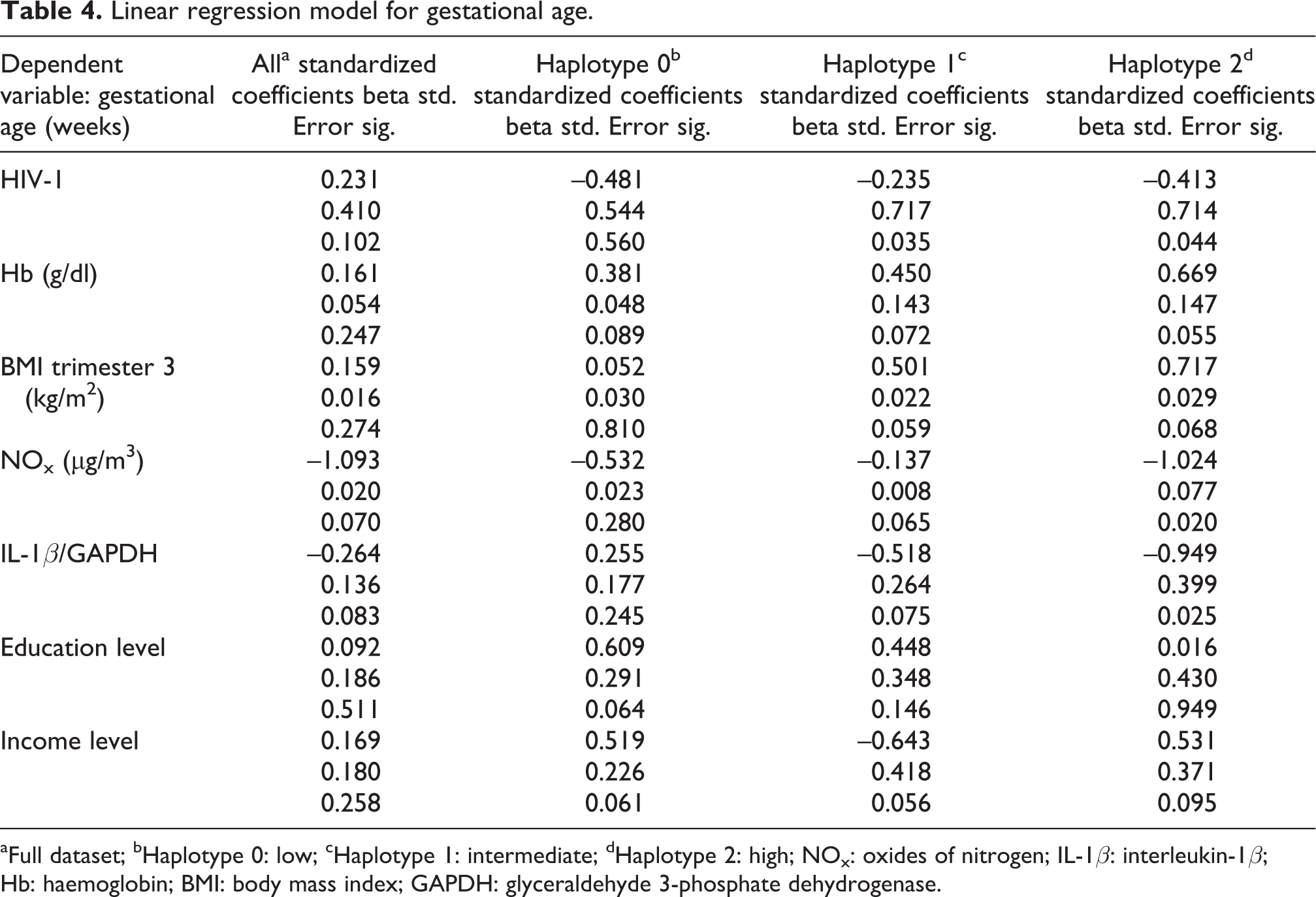
aFull dataset; bHaplotype 0: low; cHaplotype 1: intermediate; dHaplotype 2: high; NOx: oxides of nitrogen; IL-1β: interleukin-1β; Hb: haemoglobin; BMI: body mass index; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Discussion
In this study of a South African birth cohort, a significant association between NOx and gestational age was observed, with a strong suggestion of effect modification by the IL-1β haplotype. These findings are evident when adjusting for maternal IL-1β mRNA expression, HIV status, BMI, Hb level, income and education.
We observed a positive association between IL-1β expression and NOx exposure, and in a multivariate analysis observed further IL-1β haplotype-specific effects of NOx and IL-1β mRNA expression on gestational age, with an almost twofold decrease in gestational age with the high haplotype when compared to the other haplotypes.
Several epidemiological studies in predominantly Caucasian or Asian cohorts have linked air pollution with adverse birth and health outcomes 2,19,20 , including an association between maternal NOx and NO2 exposure with either shortened gestational age or PTB. 21 –24 Other studies have shown increased risk for PTB by proximity to traffic or major motorways. 25 –27 Our finding of a negative association between NOx exposure and gestational age is consistent with other studies. The pathogenetic mechanisms, however, are not completely understood.
Data on the maternal IL-1β response to NOx exposure are scarce or non-existent. In this study, the observed positive association between NOx exposure levels and maternal IL-1β expression may indicate an inductive effect of NOx on oxidative stress, 28 which leads to an elevated pro-inflammatory IL-1β response. DNA and cellular component damage 29 and tissue damage in target organs, including the placenta, may ensue. 30 An excessive pro-inflammatory response may induce further free radical activity, leading to further tissue destruction. In the multivariate models, we observed that NOx effects on gestational age are haplotype-specific for the high-producer IL-1β haplotype in which a unit increase in NOx exposure level is associated with a 1-week shortened gestational age. This may suggest that elevated maternal IL-1β plays a role in mechanisms by which NOx affects gestational age.
During labour and PTB, the level of IL-1β mRNA in peripheral blood was shown to be significantly elevated in comparison to pregnant women not in labour 31 and in spontaneous PTB. 32 Exacerbation of pro-inflammatory responses due to cytokine gene variants and gene-environment interactions at varying stages may threaten the Th2 dominant maternal tolerance of the semiallogeneic foetal-placental unit and increase the risk for decreased gestational age or PTB. 12 Elevated IL-1β contributes to induction of labour by stimulating the expression and production of uterine activation proteins and prostaglandin synthesis, and decreasing tissue progesterone responsiveness. 33 Significant down-regulation of 11β-HSD2 mRNA expression and activity by IL-1β increases the amount of cortisol entering the foetal circulation 34 and increases intrauterine cortisol concentrations which drive prostaglandin synthesis potentially contributing to PTB. We observed in the multivariate models that a unit increase in IL-1β mRNA expression level associates with a decrease in gestational age by almost 1 week in the high haplotype only (Table 4), highlighting a vulnerable group to shortened gestational age by elevated IL-1β gene expression.
In a predominantly Black African birth cohort, we report the maternal IL-1β haplotype (-1464, -511, -31) distribution and associated expression, which has been described in African Americans, Caucasians and Koreans 14,15 but not in an African cohort. Similar to the reported distribution in African Americans, the highest transcriptional activity haplotype is most prevalent in Africans at a comparable 40% versus 46.1% in African Americans, while haplotype-associated expression matches previously published data (Figure 1). 14,15,35 The PTB and low birth weight prevalence matches the estimated global prevalence of 9.6% and 16%, respectively. 36
We report a positive correlation between NOx exposure level and IL-1β mRNA expression levels in the cohort. Further study is required to determine the association between elevated IL-1β in response to NOx and the occurrence and exacerbation of asthma and respiratory morbidity. In immunostimulated pregnant dams, a transient increase in IL-1 among other cytokines in serum and amniotic fluid was shown, and their offspring displayed accelerated development and heightened responsiveness of Th1, Th17 and cytotoxic effector T-cell subsets. 37 –39 Morelli et al. (2015) thus hypothesized that elevated maternal IL-1 among other pro-inflammatory cytokines may influence programming of the foetal immune system towards a pro-inflammatory phenotype with exacerbated responses to immune stimuli that may persist into adulthood. 40 Interestingly, in a Chinese cohort comprising 2598 children, maternal exposure to NO2 was significantly associated with increased risk for childhood asthma, allergic rhinitis and eczema. 41
Maternal HIV-1 infection is associated with increased risk for adverse birth outcomes, including lower gestational age, PTB and intrauterine growth retardation; however, the mechanisms underlying these associations are not completely understood. 42 –44 Indeed, in vitro studies show the inductive effect of IL-1β on HIV expression in the latently infected human promonocytic cell line U1. 45,46 In this study, we observe an IL-1β haplotype-specific negative association between HIV-1 positivity and gestational age for the intermediate and high-producer haplotypes with an almost twofold greater effect in the high haplotype, while no association is shown in the low-producer haplotype. We postulate that higher IL-1β expression associated with the high and intermediate haplotypes in comparison to the low haplotype may potentially play a role in mediating or synergizing the association between HIV-1 positivity and lower gestational age outcomes, via a pro-inflammatory state which alters maternal tolerance of the foetal-placental unit and potentially via facilitation of HIV-1 replication. We thus identify vulnerable subgroups of the HIV-1 population to lower gestational age by the IL-1β haplotype. We speculate that significantly elevated IL-1β in response to elevated NOx exposure may potentially further increase susceptibility of HIV-1-positive individuals to lower gestational age.
Inter-individual variability in HIV-1 infection outcome is an indicator of the role of host genetic and environmental factors in influencing disease progression. The reported positive association between IL-1β expression and elevated NOx exposure is shown independently in the HIV-1-positive group and (data not shown) is a cause for concern. We speculate that chronic NOx-induced increase in IL-1β expression may potentially influence HIV-1 infection outcome, disease progression, pathogenesis and transmissibility. It has been shown that increased plasma IL-1β is associated with disease progression in both children and adults, 47 while in vitro studies show the inductive effect of IL-1β on HIV expression in the latently infected human promonocytic cell line U1. 45,46 IL-1β also plays a key role in pyroptosis, a major mechanism of CD4 T-cell depletion 48 and IL-1β production is lower in T-cell cultures from women with controlled viral load, 19,49 essential to prevent mother-to-child transmission. However, further work is needed to address whether NOx exposure levels may impact HIV-1 progression and pathogenesis in a larger characterized cohort matched for IL-1β haplotype and stage of infection.
Conclusion
In conclusion, we report the maternal IL-1β (-1464, -511, -31) haplotype distribution in a predominantly African pregnant cohort and show maternal IL-1β haplotype-specific effects of NOx levels, IL-1β expression and HIV-1 positivity on gestational age in a birth cohort, which has not been previously reported. These data have implications for better understanding the effect of prenatal NOx exposure on gestational age outcomes and demonstrate the role of the IL-1β haplotype in modulating the effects of NOx exposure therein. The study identifies vulnerable subgroups to shortened gestational age, due to IL-1β haplotype and NOx interactions. The study identifies vulnerable subgroups among HIV-1 infected mothers to lower gestational age outcomes by IL-lβ haplotype and contributes to the understanding of the association between HIV-1 infection and gestational age, which is currently limited. The study provides a basis for further work to explore the association between maternal IL-1β haplotypes and morbid outcomes among children of the cohort.
Footnotes
Acknowledgement
The authors wish to thank the Mother and Child in the Environment cohort study participants and nursing staff at the participating clinics and hospitals.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Research Foundation of South Africa (grant number: 90550), the Medical Research Council of South Africa (SIR Grant) and the AstraZeneca Research Trust.
