Abstract
Introduction:
This study provides an epidemiological description of non-health care facility medication errors involving anticonvulsant drugs.
Methods:
A retrospective analysis of National Poison Data System data was conducted on non-health care facility medication errors involving anticonvulsant drugs reported to US Poison Control Centers from 2000 through 2012.
Results:
During the study period, 108,446 non-health care facility medication errors involving anticonvulsant pharmaceuticals were reported to US Poison Control Centers, averaging 8342 exposures annually. The annual frequency and rate of errors increased significantly over the study period, by 96.6 and 76.7%, respectively. The rate of exposures resulting in health care facility use increased by 83.3% and the rate of exposures resulting in serious medical outcomes increased by 62.3%. In 2012, newer anticonvulsants, including felbamate, gabapentin, lamotrigine, levetiracetam, other anticonvulsants (excluding barbiturates), other types of gamma aminobutyric acid, oxcarbazepine, topiramate, and zonisamide, accounted for 67.1% of all exposures.
Conclusions:
The rate of non-health care facility anticonvulsant medication errors reported to Poison Control Centers increased during 2000–2012, resulting in more frequent health care facility use and serious medical outcomes. Newer anticonvulsants, although often considered safer and more easily tolerated, were responsible for much of this trend and should still be administered with caution.
Introduction
Medication errors are a leading cause of mortality in the United States, resulting in more than 7000 deaths per year. 1 Non-health care facility (HCF) medication errors pose a serious yet preventable threat to individuals of all ages and result in more than 200,000 calls to US Poison Control Centers (PCCs) annually. 2 –4 One class of pharmaceuticals that presents a particular risk of harm due to its prevalence and potentially life-threatening effects is anticonvulsant drugs.
Anticonvulsant drugs are primarily prescribed to treat seizures; however, they are also commonly prescribed to treat other conditions such as neuropathic pain, migraines, tremors, and psychiatric disorders. 5 Anticonvulsants cause varying degrees of adverse effects to the central nervous system, gastrointestinal tract, blood, liver, and skin. 6 In the 1990s, pharmaceutical manufacturers began marketing “new-generation” anticonvulsants in the United States, including felbamate, gabapentin, lamotrigine, levetiracetam, oxcarbazepine, topiramate, and zonisamide. These newer drugs are generally considered to be safer and easier to tolerate; however, research to date has yielded inconsistent findings. 7 Anticonvulsant drugs are becoming more widely used. From 1999 to 2009, the number of individuals in the United States who had obtained at least one prescription for anticonvulsant medications increased from 8.6 million to 15.2 million people. 8 Due to the growing number of people being prescribed anticonvulsants and the variety of conditions and age groups being treated, it is important that these medications be administered correctly. One study found that anticonvulsants were among the 10 classes of drugs most frequently associated with out-of-hospital medication errors. 2
Much of the prior research on anticonvulsant medication errors has been limited to cases treated in emergency departments or admitted to hospitals, exposures reported to a single PCC, or exposures to a specific anticonvulsant medication. 9 –16 This study provides a comprehensive epidemiological description of unintentional non-HCF anticonvulsant medication errors reported to US PCCs.
Methods
Study design
This retrospective study analyzed data from the National Poison Data System (NPDS) over a 13-year period to evaluate characteristics and trends of non-HCF unintentional medication errors involving anticonvulsant medications. The NPDS is maintained by the American Association of Poison Control Centers (AAPCC) and includes poison exposure data collected during telephone calls received by regional PCCs in the United States and its territories. PCC specialists provide medical advice to callers on exposure management and document each exposure in the NPDS. Follow-up is provided for each exposure as needed and to the extent possible. The NPDS contains more than 50 million poison exposure records and more than 2 million new exposures are added to the database annually. 17
Case selection criteria
NPDS data were obtained for all non-HCF unintentional therapeutic errors involving anticonvulsant pharmaceutical substances as determined by the AAPCC generic substance coding system (Appendix 1) from January 1, 2000 through December 31, 2012. The NPDS defines an unintentional therapeutic error as “an unintentional deviation from a proper therapeutic regimen that results in the wrong dose, incorrect route of administration, administration to the wrong person, or administration of the wrong substance.” 17 Excluded from this study were exposures involving intentional drug misuse or abuse, suicidal drug ingestions, and pediatric drug exposures resulting from exploratory behavior. Exposures were also excluded if (1) the exposure site was an HCF, nursing home, or prison; (2) a PCC professional determined that the event was a non-exposure or the clinical effects were not attributed to an anticonvulsant drug; or (3) the call originated from outside the United States. Many calls to PCCs involve a single individual with more than one substance exposure. In such cases, the PCC specialist ranks each substance according to its likely relative contribution to the observed clinical effects. This study includes only exposures in which an anticonvulsant drug was ranked first.
Variables
Categories of anticonvulsant pharmaceuticals analyzed in this study included the following: carbamazepine, phenytoin, primidone, succinimides, valproic acid, long-acting barbiturates, unknown types of anticonvulsants (excluding barbiturates), and other types of anticonvulsants (excluding barbiturates). The category other types of anticonvulsants (excluding barbiturates) comprises mostly newer anticonvulsants, which were first introduced in the 1990s. 18 The newer anticonvulsants in the “other” category began receiving individual generic codes in 2012. For sub-analyses of 2012 data, the medications were classified as “newer anticonvulsants” (felbamate, gabapentin, lamotrigine, levetiracetam, other anticonvulsants (excluding barbiturates), other types of gamma aminobutyric acid, oxcarbazepine, topiramate, and zonisamide); “older anticonvulsants” (carbamazepine, fosphenytoin, phenytoin, primidone, succinimides, valproic acid, and long-acting barbiturates); and “unknown anticonvulsants.”
The variable HCF level of care received was used to describe whether individuals were treated at an HCF (treated/evaluated and released; admitted to critical care unit; admitted to noncritical care unit; or admitted to a psychiatric care facility) or not (no healthcare received; patient lost to follow-up or left against medical advice; or patient refused referral/did not arrive at HCF). NPDS defines medical outcomes as minor effect (minimal symptoms that resolve rapidly), moderate effect (pronounced and prolonged symptoms that usually require treatment), or major effect (life-threatening symptoms that may result in significant disability or disfigurement). In this study “serious medical outcome” includes exposures resulting in death, major effects, or moderate effects. Chronicity of the exposure was categorized as acute (a single, repeated, or continuous exposure occurring over a period of ≤8 h), acute-on-chronic (a single exposure preceded by a continuous, repeated, or intermittent exposure occurring over a period of >8 h), or chronic (a continuous, repeated, or intermittent exposure occurring over a period of >8 h). Subjects were categorized by age as follows: younger than 6 years, 6–12 years, 13–19 years, by decade of life (20–79 years), and 80 years or older. Other variables included were gender, route of exposure, exposure site, management site, scenario, clinical effects, and therapeutic interventions.
Data analysis and ethical statement
Data analysis was performed using SPSS 21.0 statistical software (IBM Corp., Armonk, New York, USA). National exposure rates were calculated using intercensal and postcensal estimates for US residents from 2000 through 2012. Linear regression was used to analyze exposure trends. The estimated slope from the regression model (m) is reported with the associated p value. Statistical significance was determined using α = 0.05. The Institutional Review Board at our institution judged this study exempt from review.
Results
General characteristics
From 2000 through 2012, there were 108,446 calls made to PCCs in the United States reporting unintentional non-HCF medication errors involving anticonvulsant drugs. The annual frequency of unintentional anticonvulsant medication errors increased by 96.6% during the study from 5253 to 10,327 exposures (m = 407.38, p < 0.001). Likewise, the rate of exposures per 100,000 population increased 76.7%, from 1.86 to 3.29 (m = 0.11, p < 0.001; Figure 1). Females accounted for a greater proportion of exposures (55.9%; Table 1). Nearly all exposures (99.7%) resulted from ingestion of the medication. Most exposures occurred at the individual’s own residence (91.9%). Three-quarters (74.7%) of exposures were associated with one pharmaceutical substance; 12.9% involved two, and 12.4% involved three or more. Among all unintentional anticonvulsant medication exposures included in this study, 81.4% were managed onsite and did not receive treatment at an HCF. More than half of the exposures were acute (55.7%); 40.0% were acute-on-chronic, and 4.2% were chronic. Over the study period, the age groups with the highest average rates of anticonvulsant medication errors per 100,000 population were children 6–12 years (3.71), children <6 years (3.47), and children 13–19 years (2.82), compared with an overall average rate of 2.79.
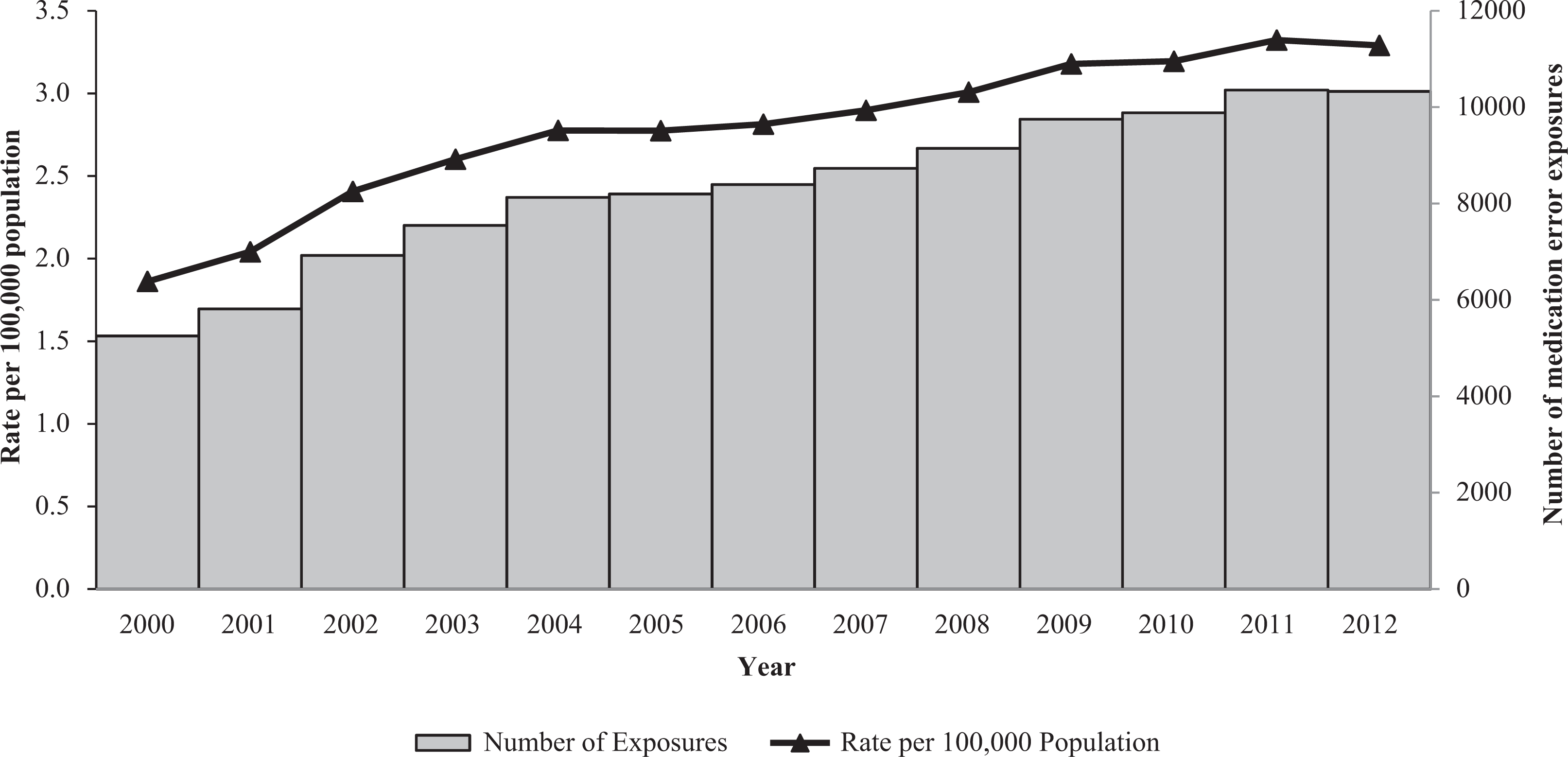
Annual number and rate of exposures associated with anticonvulsant medication errors, National Poison Data System 2000–2012.
Characteristics of exposures associated with anticonvulsant medication errors by age group, National Poison Data System 2000–2012.

AMA: against medical advice; HCF: health care facility.
aExcluding barbiturates. Percentages may not sum to 100.0 due to rounding error.
Substances
Other types of anticonvulsants (excluding barbiturates) accounted for 52.1% of all exposures and were the most frequently identified exposure source for each age group (Table 1). This medication category, which comprises primarily newer anticonvulsants, also had the highest rate of exposures overall (Figure 2). The rate of non-HCF medication errors associated with other types of anticonvulsants (excluding barbiturates) increased significantly by 437.2% from 2000 through 2012 (m = 1.49, p < 0.001; Figure 2). The second most common source of anticonvulsant exposures was valproic acid (17.8%), followed by carbamazepine (11.8%), phenytoin (8.4%), and long-acting barbiturates (8.0%).

Annual rate of exposures associated with anticonvulsant medication errors by type of anticonvulsant, National Poison Data System 2000–2012.
Scenario
The most common scenario for medication error among all age groups was “inadvertently took/given medication twice” (42.4%), followed by “inadvertently took/given someone else’s medication” (17.5%), “other incorrect dose” (12.4%), “wrong medication taken/given” (9.6%), and “medication doses given/taken too close together” (7.2%).
HCF utilization
From 2000 through 2012, the overall rate of anticonvulsant medication error exposures resulting in HCF utilization increased by 83.5% (m = 0.19, p < 0.001) from 2.84 to 5.21 exposures per 1,000,000 population. There was a 558.8% increase (m = 2.05, p < 0.001) in the rate of HCF utilization resulting from exposure to other types of anticonvulsants (excluding barbiturates; from 0.43 to 2.85 per 1,000,000 population), but no significant change in HCF utilization for exposures to older anticonvulsants (Figure 3). Among the 15.0% (n = 16,318) of exposures that resulted in HCF utilization, 69.5% (n = 11,346) were treated and released without hospital admission, 19.1% (n = 3115) were admitted to a noncritical care unit, 10.3% (n = 1673) were admitted to a critical care unit, and 1.1% (n = 184) were admitted to a psychiatric care facility. Compared with other age groups, a greater percentage of children younger than 6-years old were treated at an HCF (19.4%). The largest proportion of exposures resulting in admission to a critical care unit occurred among age groups ≥80 years (2.7%) and <6 years (2.1%; Table 1).

Annual rate of serious medical outcomes and health care facility utilization by type of anticonvulsant, National Poison Data System 2000–2012. Note: Health care facility utilization includes: treated and released, admitted to non-critical care, admitted to critical care, and admitted to psychiatric care facility. Note: Serious medical outcomes include: death, major health effect, and moderate health effect. *Other types of anticonvulsants (excluding barbiturates) are comprised of newer anticonvulsants.
Clinical effects, medical outcomes, and therapy
The overall rate of serious medical outcomes associated with anticonvulsant medication errors increased significantly by 62.3%, from 9.11 to 14.82 per 10,000,000 population (m = 0.46, p < 0.001) over the study period. The rate of serious medical outcomes resulting from exposure to other types of anticonvulsants (excluding barbiturates) increased 331.1% from 1.56 to 6.72 per 10,000,000 (m = 0.45, p < 0.001), while the rate associated with older anticonvulsants did not display a clear trend (Figure 3). At least one clinical effect was reported for 23.2% of exposures. The most commonly observed clinical effects were drowsiness (13.6%), dizziness (5.3%), ataxia (3.2%), nausea (2.4%), and vomiting (1.9%). Primidone accounted for 1.3% of all exposures, but represented the highest proportion of exposures resulting in drowsiness (27.2%), dizziness (14.2%), and nausea (7.6%). Phenytoin was associated with a substantial percentage of exposures resulting in ataxia (12.8%). Seizures occurred in 332 exposures, the majority of which were associated with phenytoin (n = 101), carbamazepine (n = 96) and “other types of anticonvulsants (excluding barbiturates)” (n = 91).
Overall, anticonvulsant exposures resulted in medical outcomes of minor effect in 13.9% of exposures, moderate effect in 3.9%, major effect in 0.3%, and death in 0.01% (13 exposures; Table 1). Deaths were associated with exposures to phenytoin (n = 7), other types of anticonvulsants (excluding barbiturates; n = 3), carbamazepine (n = 1), long-acting barbiturates (n = 1), and valproic acid (n = 1). Most fatal exposures were admitted to a critical care unit (n = 9), two were treated and released, one was admitted to a noncritical care unit, and one was not treated at an HCF. Three of the deaths were judged to be associated with health professional iatrogenic error. Among the 13 fatalities identified in this study, 10 were among individuals exposed to only 1 substance, 2 were exposed to 2 substances, and 1 individual was exposed to 7 substances. In all cases, an anticonvulsant medication was ranked first as the substance most likely to be the primary contributor to the observed clinical effects. A higher percentage of phenytoin exposures resulted in serious medical outcomes (13.2%) and hospital admissions (19.5%) than other medications. Most exposures (74.7%) did not require therapy.
Newer versus older anticonvulsant drugs
Older anticonvulsant exposures were compared with newer anticonvulsant exposures for the year 2012, the first year that newer anticonvulsants were coded with individual generic codes. In 2012, 32.8% of reported exposures involved older anticonvulsants and 67.1% were attributed to newer anticonvulsants. Among newer anticonvulsant medication error exposures in 2012 (n = 6930), 12.9% resulted in HCF utilization, compared with 21.8% of the 3384 exposures to older anticonvulsants. The percentage of exposures to older anticonvulsants resulting in serious medical outcomes in 2012 (7.5%) was more than double that for exposures to new anticonvulsant drugs (3.0%). Comparing 2012 data, older anticonvulsants were more likely to cause drowsiness, ataxia, nausea, vomiting, and confusion than newer anticonvulsants (Table 2).
Serious medical outcomes, health care facility treatment, and clinical effects by anticonvulsant type, National Poison Data System 2012.a
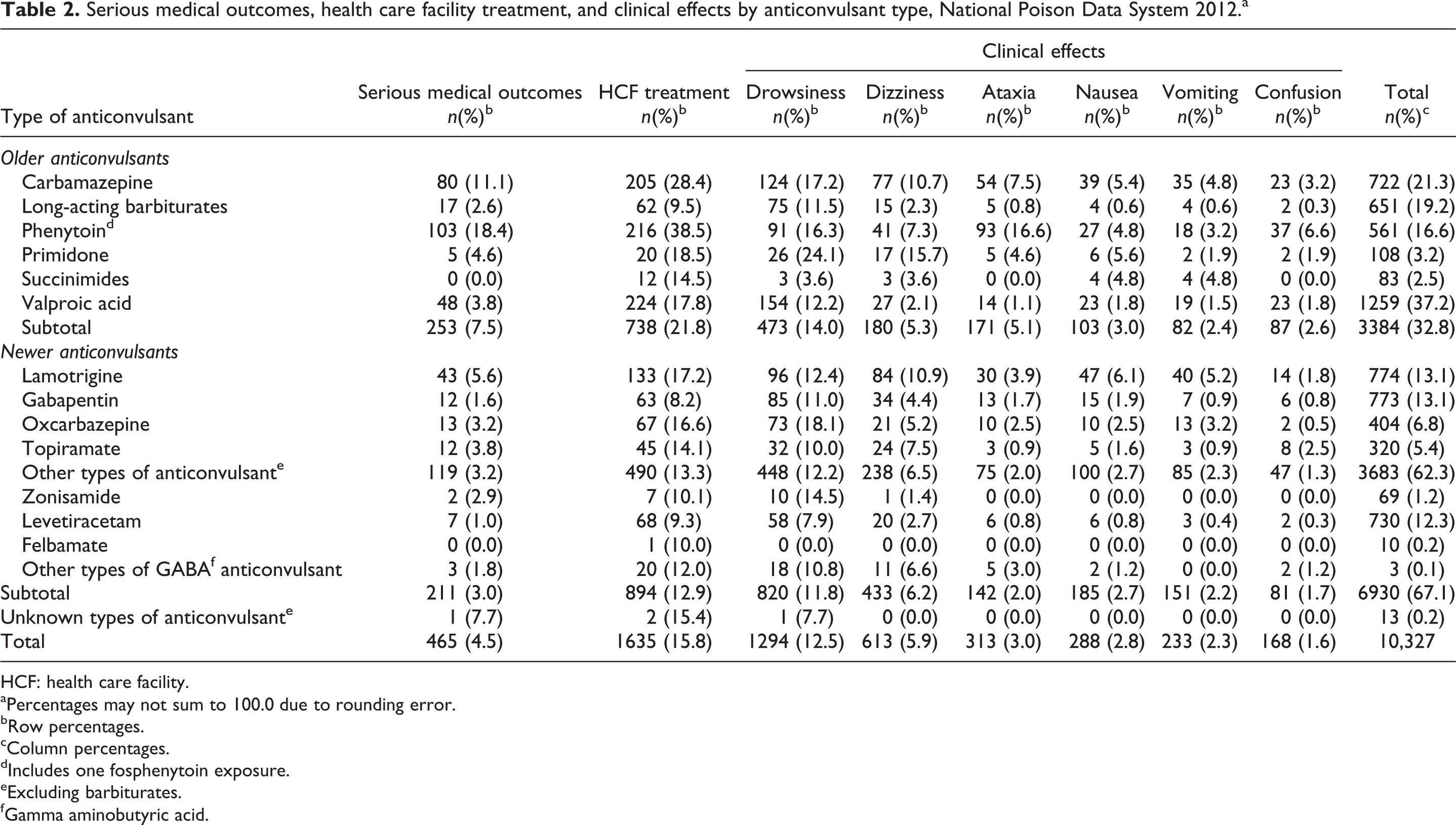
HCF: health care facility.
aPercentages may not sum to 100.0 due to rounding error.
bRow percentages.
cColumn percentages.
dIncludes one fosphenytoin exposure.
eExcluding barbiturates.
fGamma aminobutyric acid.
Discussion
On average, there were more than 8300 exposures associated with anticonvulsant medication errors reported to PCCs in the United States each year from 2000 through 2012. The frequency of anticonvulsant medication errors nearly doubled over the study period. This is likely due to increases in anticonvulsant drug prescribing and usage. 8 As reports of anticonvulsant medication errors rose, the frequency and rate of HCF utilization and serious medical outcomes among exposed individuals also increased. This trend was observed among all age groups.
The observed upward trends in HCF utilization and serious medical outcomes are largely a result of medication errors associated with drugs in the other types of anticonvulsants (excluding barbiturates) category, which almost entirely comprises newer anticonvulsants. This is likely a result of increased prescribing of newer anticonvulsant medications. In contrast, rates of HCF utilization and serious medical outcomes associated with older anticonvulsants changed minimally between 2000 and 2012. Therefore, although newer anticonvulsant drugs are reportedly better tolerated and have fewer adverse side effects, 19 –22 they still pose a risk of adverse clinical effects when administered incorrectly.
For the year 2012, we were able to compare exposures to individual newer and older anticonvulsant drugs. For example, a smaller percentage of reported exposures to a newer sodium channel blocker, lamotrigine, resulted in serious medical outcomes or HCF treatment, compared with an older sodium channel blocker, carbamazepine. This finding supports previous studies demonstrating that lamotrigine is safer and causes fewer side effects than carbamazepine. 20 –23 Both topiramate (new) and valproic acid (old) use multiple mechanisms to inhibit seizures and are often used to treat generalized seizures. 18 When compared in this study, both were associated with similar percentages of serious medical outcomes and HCF usage. Based on 2012 data, medication errors involving older anticonvulsants had higher percentages of HCF usage or serious medical outcomes than those related to newer anticonvulsants. However, increased prescribing and use of these newer drugs likely is contributing substantially to the rising frequency of anticonvulsant medication error exposures.
Newer anticonvulsants are often used as add-on therapy. When these medications were introduced to the market, the majority were not yet approved for monotherapy. 6 Previous studies have shown that the more medications a patient takes, the greater the risk of experiencing adverse drug reactions and medication errors. 24,25 One-quarter (25.3%) of individuals in this study were reportedly exposed to two or more pharmaceutical substances at the time of the call to the PCC. Use of multiple medications may be contributing to the observed increase in medication errors involving other types of anticonvulsants (excluding barbiturates).
The most common reasons for anticonvulsant medication errors were medication being unintentionally taken twice, inadvertently taking someone else’s medication, other errors related to dosing, and taking the wrong medication. Several prevention strategies exist to reduce the frequency of unintentional non-HCF medication errors. Health care providers should take the time to educate patients about their medications and proper dosing. 26,27 Use of child-resistant weekly pill boxes may be helpful for increasing medication adherence and preventing double dosing of medications, but could increase the risk of taking the wrong medication or other errors. 27 –32 The pharmaceutical industry should work to improve drug names and abbreviations to avoid similarities that may lead to medication errors. 26,33,34 It is also important that all prescription labels give clear dosing instructions. 26,34 Lastly, to better understand the etiology of medication errors and identify effective prevention strategies, additional funding should be allocated to study this issue. 26
Study limitations
There are limitations to this study. The NPDS gathers self-reported data from voluntary calls to PCCs, and because not all exposures are reported to PCCs, the number of exposures associated with anticonvulsant medication errors observed in this study is an underestimate. PCCs and the AAPCC cannot completely verify the self-reported data received. Reported exposures do not necessarily represent poisonings or overdoses, and the frequencies reported in this study include all exposures reported to PCCs, even if not resulting in adverse clinical effects. We excluded cases judged by the PCC to have clinical effects unrelated to the exposure. There is potential for error in that initial judgment, and to the extent the toxicology of newer drugs is less well known than the toxicology of older agents, a potential for bias as well. Also, there is the potential for error in multiple substance calls when PCCs must assign the drug most responsible for the observed clinical effects; however, the majority of calls in this study were single-substance exposures. In years 2000–2011, the newer anticonvulsant medications were not coded separately; therefore, analysis of exposures involving this category of drugs was possible only for 2012. Also, we did not have access to NPDS product-specific codes for the medications included in this study, limiting our ability to describe and compare medication errors related to individual products. Because data on the number of doses of each drug taken or prescribed are unavailable, we had limited ability to compare the relative toxicities of these drugs. However, use of population data is an acceptable method for calculating rates of exposure, and exposure frequencies provide a useful measure of the public health burden of these medication errors. Despite these limitations, the NPDS is a comprehensive national database that provides extensive information on non-HCF medication errors in the United States.
Conclusions
This study provides a comprehensive overview of non-HCF anticonvulsant medication errors reported to US PCCs. From 2000 through 2012, there was a significant increase in the rate of these exposures, with a corresponding rise in HCF use and serious medical outcomes. The drug category containing primarily newer anticonvulsants was responsible for the majority of the observed rate increase. Newer anticonvulsants, although often considered safer and more easily tolerated, still need to be used with caution.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Author Emily A DeDonato received a research scholarship stipend from the Medical Student Research Scholarship Program at The Ohio State University while she worked on this study.
Appendix 1: American association of poison control centers generic codes and descriptions of anticonvulsants included in this study

| Code | Description |
|---|---|
| 249000 | Carbamazepine |
| 201176 | Oxcarbazepine* |
| 201172 | Gabapentin* |
| 201178 | Other Types of Gamma AminobutyricAcid Anticonvulsant* |
| 201170 | Fosphenytoin* |
| 105000 | Phenytoin |
| 201171 | Felbamate* |
| 201173 | Lamotrigine* |
| 201175 | Levetiracetam* |
| 201073 | Primidone |
| 230000 | Succinimides |
| 201174 | Topiramate* |
| 260000 | Valproic Acid |
| 201177 | Zonisamide* |
| 77730 | Other Types of Anticonvulsant (Excluding Barbiturates) |
| 77735 | Unknown Types of Anticonvulsant (Excluding Barbiturates) |
| 5000 | Long Acting Barbiturates |
*Generic codes added in 2012.
