Abstract
Activation of peroxisome proliferator–activated receptor α (PPARα) by di-(2-ethylhexyl) phthalate (DEHP) has an anti-inflammatory effect. This study investigated the potential combined influence of PPARα, tumor necrosis factor α-induced protein 3 (TNFAIP3/A20), and tumor necrosis factor receptor–associated factor 6 (TRAF6) on interleukin (IL)-12p40 production by macrophages exposed to DEHP and stimulated with lipopolysaccharide (LPS). LPS upregulated IL-12p40 expression by granulocyte-macrophage colony-stimulating factor–dependent macrophages (on day 9 of culture), whereas adding DEHP to cultures significantly attenuated the response of IL-12p40 to LPS stimulation. PPARα protein was also reduced by DEHP. Interestingly, transfection of macrophages with small interfering RNA (siRNA) duplexes for PPARα, TNFAIP3/A20, or dual oxidase 2 restored the response of IL-12p40 protein to LPS stimulation in the presence of DEHP. siRNAs for various protein kinase Cs (PKCs) (α, β, γ, or δ) also restored IL-12p40 production by macrophages exposed to LPS and DEHP. While LPS upregulated both IL-12p40 and TNFAIP3/A20 production, adding DEHP to cultures dramatically reduced IL-12p40 and TNFAIP3/A20 levels. Silencing of PKCα reduced TNFAIP3/A20 production, whereas PKCγ siRNA (but not PKCβ or δ siRNA) significantly increased TNFAIP3/A20. TRAF6 was also attenuated by macrophages with DEHP. The PPARα/TNFAIP3/TRAF6 axis may have an important role in the mechanism through which DEHP reduces IL-12p40 production by LPS-stimulated macrophages.
Keywords
Introduction
Interleukin-12 (IL-12) is a heterodimeric pro-inflammatory cytokine that induces the production of interferon-γ (IFN-γ), promotes differentiation of T helper type 1 (Th1) cells, and provides a link between innate and adaptive immunity. 1
IL-12p70 is a heterodimer composed of p35 and p40 subunits and is a major Th1 cytokine that promotes cell-mediated immunity. The p35 molecule is unique to IL-12, while p40 is shared by both IL-12 and IL-23. IL-12 promotes Th1 T cell responses, while IL-23 promotes Th17 T cell responses. 2 Differential regulation of IL-12p35 and p40 gene expression has been reported. Indeed, the p35 gene is “constitutively” and “ubiquitously” expressed and therefore has not been studied as much as the more inducible p40 gene. 3 IL-12 is primarily produced by macrophages and dendritic cells as part of both innate and adaptive immune responses. There is evidence that lipopolysaccharide (LPS) induces IL-12p40 production. 4
In various industrial processes, plasticizers are added to polyvinyl chloride (PVC) to increase its flexibility and di-(2-ethylhexyl) phthalate (DEHP) is the most commonly used plasticizer. Indeed, DEHP is found in many consumer products, including plastics, vinyl flooring, and personal care products. DEHP also continually enters the body from various foods, water, and the atmosphere. Moreover, substantial exposure to DEHP occurs via PVC-containing medical devices that are used for intravenous therapy, enteral and parenteral nutrition, blood transfusion, hemodialysis, cardiopulmonary bypass, and extracorporeal membrane oxygenation. Phthalates may have an influence on immunity to infection, 5 and macrophages are thought to be a sensitive target of DEHP. 6 The peroxisome proliferator–activated receptors (PPARs) are transcription factors belonging to the nuclear hormone receptor superfamily that consist of three subunits: PPARα, PPARβ/δ, and PPARγ. 7 Binding of DEHP to PPARα induces the proliferation of peroxisomes and regulates the expression of specific target genes, 8 and PPARα has been suggested to be involved in DEHP toxicity. 9 Species differences in the toxicity of PPARα agonists have been reported, and the major reasons for this are considered to be differences in the level of PPARα expression. 10 The PPAR family has recently been the focus of much interest with regard to a possible role in the regulation of inflammation and immune responses. Indeed, PPARα has been implicated in regulation of macrophage and endothelial cell inflammatory responses. PPAR activation generally seems to have an anti-inflammatory effect, although opposite effects have also been reported. 11 For example, activation of PPARα can lead to inhibition of nuclear factor kappa-B (NF-κB) activation and inflammatory gene expression. 12 This raises the question of whether DEHP suppresses NF-κB activation in response to stimulation by LPS. NF-κB is the central transcription factor for inflammatory signaling and activation of the innate and adaptive immune responses, so the NF-κB pathway is tightly controlled by several negative feedback mechanisms. DEHP enhances the production of tumor necrosis factor (TNF) α by macrophages, 13 and it has been suggested that TNFα-induced protein 3 (TNFAIP3) may be a key player in negative feedback regulation of NF-κB signaling responses to various stimuli. 14 A20 (also known as TNFAIP3) is a cytokine-inducible protein. It was first identified as a TNFα-induced primary response gene in human umbilical vein endothelial cells that encoded a 790-amino acid protein containing seven Cys2/Cys2-zinc finger motifs. 15
It has been reported that TNFAIP3/A20 is a potent anti-inflammatory signaling molecule that suppresses multiple intracellular signaling cascades. 16 Induction of A20 expression by TNFα is mediated via glycogen synthase kinase 3 (GSK3), and TNFα also induces endothelial activation that is modulated by PKCα-dependent inhibition of GSK3. DEHP has been reported to increase the production of reactive oxygen species via nicotinamide adenine dinucleotide phosphate (NADPH) oxidase in a PPARα-independent manner. 17 The NADPH oxidase (Nox) family has seven isoforms, including Noxes 1–5 and dual oxidase (DUOX) 1 and DUOX2. ROS promotes sustained activation of PKC. 18 Phosphorylation of DUOX1 is mediated via protein kinase A, whereas DUOX2 is phosphorylated via PKC, with DUOX2 being stimulated by both the phospholipase C (PLC) cascade and PKC-dependent phosphorylation. 19 It was also reported that TNFα activates NADPH oxidase via p22phox. 20 Therefore, we investigated the influence of PKC, DUOX2, and p22 phox on IL-12p40 expression by macrophages treated with DEHP.
TNF receptor–associated factor 6 (TRAF6), an E3 ubiquitin ligase involved in innate immunity, is an adaptor protein with an important role in intracellular signaling, and TRAF6-dependent phosphorylation of p38 regulates the production of IL-12p40. 21 TNFAIP3/A20 can bind to TRAF6, suggesting that A20 prevents IL-1-induced activation of NF-κB by direct interaction with TRAF6. 22 Indeed, it was reported that A20 ubiquitinates TRAF6, which binds to the IkappaB kinase (IKK) complex (composed of IKKγ, IKKα, and IKKβ), after which the IKK complex is phosphorylated and promotes proteasomal degradation of Ikβα. Furthermore, it has been reported that TNFAIP3/A20 protects against lethal hepatic ischemia–reperfusion injury in mice by increasing PPARα expression. Thus, PPARα seems to be a target of TNFAIP3/A20. 23 This raises the possibility that the mechanism of DEHP toxicity involving PPARα may also be associated with TNFAIP3/A20. We hypothesized that DEHP activates PPARα to influence TNFAIP3/A20 expression, and we investigated whether PPARα/TNFAIP3/TRAF6 regulated IL-12p40 production by human granulocyte-macrophage colony-stimulating factor (GM-CSF)-dependent macrophages in response to LPS stimulation with or without co-exposure to DEHP.
Materials and methods
Ethics statement
Human peripheral blood samples were obtained from healthy, nonsmoking Japanese volunteers aged 18 to 22 years (30 men and 30 women). The volunteers were all shown to be healthy according to the results of an annual medical examination performed at Kumamoto Health Science University. This study was approved by the Institutional Review Board of Kumamoto Health Science University. Written informed consent was obtained from all of the volunteers.
Chemicals and reagents
Human recombinant GM-CSF was obtained from Tocris Bioscience, Bristol, UK.
DEHP was purchased from Sigma Chemicals Industries, Perth, WA, Australia.
Isolation of adherent monocytes from peripheral blood mononuclear cells
Lymphocyte medium for thawing (BBLYMPH1) was obtained from Zen-Bio, Inc. (Research Triangle Park, North Carlonina, USA). Peripheral blood mononuclear cells (PBMCs) were isolated as described previously. 24 Briefly, heparinized blood samples were obtained from nonsmoking healthy volunteers and were diluted 1:1 with pyrogen-free saline.
Further, PBMCs were isolated immediately after collection using Lymphoprep gradients (Axis-Shield PoC As, Norway). Then, cells were suspended with BBLYMPH1 and incubated for 3 h. For monocyte isolation by plastic adherence, 1 × 10
6
cells per well were distributed into 12-well plates (Corning Inc. Costar, New York, USA) and allowed to adhere in a 5% CO2 incubator at 37°C for 2 h and washed 3 times with warm phosphate-buffered saline to remove nonadherent cells. Then monocytes were cultured in complete medium consisting of RPMI 1640 supplemented with 10% heat-inactivated fetal calf serum (FCS) and 10 × 103 μg/L gentamicin at 37°C in 5% CO2 humidified air. Adherent monocytes were recovered with a cell scraper. To quantify the number of cells with membrane expression of CD14, fluorescein isothiocyanate (FITC)-conjugated anti-CD14 antibody (clone My4, no. 6603511, isotype: mouse IgG2b FITC) was employed. This antibody and the isotype control were purchased from Beckman Coulter (Krefeld, Germany). PBMCs were labeled with the FITC-conjugated anti-CD14 antibody (1:2000) for 3 min according to the manufacturer’s instructions and fluorescence-activated cell sorting analysis was performed. The recovery of monocytes was also evaluated by trypan blue staining and counted using a Zeiss microscope (Jena, Germany). CD14+ monocytes had a purity of 85.99
Induction of monocyte/macrophage differentiation
Monocytes were seeded at 1 × 106 cells/mL into 12-well tissue culture plates containing RPMI-1640 medium with 10% FCS and 4 mM
Enzyme-linked immunosorbent assay for PPARα, IL-12p40, TNFAIP3/A20, or TRAF6
GM-CSF-dependent macrophages (on day 9 of culture) were incubated with DEHP (0, 10, 20, 50, 100, or 200 nM) for 24 h and the PPARα protein level in whole-cell lysates was determined by enzyme-linked immunosorbent assay (ELISA; Abcam, Cambridge, UK) with an anti-PPARα antibody. A specific double-stranded DNA sequence containing the peroxisome proliferator response element was immobilized in the wells of a 96-well plate and the absorbance was determined at 450 nm.
Escherichia coli 0111:B4 LPS was purchased from Sigma-Aldrich (St. Louis, Missouri, USA). After GM-CSF-dependent macrophages (on day 9 of culture) were stimulated with LPS (0, 10, 20, or 50 ng) for 6 h, the IL-12p40 protein level in whole-cell lysates was measured by ELISA with an anti-IL-12p40 monoclonal antibody (Abcam). In addition, GM-CSF-dependent macrophages (day 9) were incubated with DEHP (0 or 200 nM) for 24 h and stimulated with LPS (10 ng) for 6 h, after which IL-12p40 protein was measured by ELISA (Abcam).
Furthermore, GM-CSF-dependent macrophages (day 9) were pretreated with DEHP (0 or 200 nM) for 24 h and stimulated with or without LPS (10 ng) for 6 h, after which TNFAIP3/A20 was measured by ELISA with an anti-TNFAIP3/A20 monoclonal antibody (MyBioSource, San Diego, California, USA).
The protein level of TRAF6 in whole-cell lysates derived from macrophages stimulated with DEHP (0 or 200 nM) for 24 h was measured by ELISA with an anti-TRAF6 monoclonal antibody (MyBioSource).
Effect of silencing various molecules on IL-12p40 production by macrophages exposed to DEHP after stimulation with LPS
Small interfering RNA (siRNA) duplexes for PPARα, PKCα, PKCβ, PKCγ, PKCδ, TNFAIP3/A20, epidermal growth factor receptor, TRAF6, TNFα, toll-like receptor (TLR) 4, p22phox, or DUOX2 were purchased from Santa Cruz Biotechnology (Dallas, Texas, USA). Transfection of GM-CSF-dependent macrophages with siRNA for PPARα (50 nM), PKCα (50 nM), PKCβ (50 nM), PKCγ (50 nM), PKCδ (50 nM), TNFAIP3 (50 nM), EGFR (50 nM), TRAF6 (50 nM), TNFα (50 nM), TLR4 (50 nM), p22phox (50 nM), or DUOX2 (50 nM) was performed on days 7 to 8 of culture according to the manufacturer’s protocol for Lipofectamine™ RNAiMAX (Life Technologies, Carlsbad, California, USA). Control siRNA-A (Santa Cruz Biotechnology) was utilized as the negative control for each experiments.
Transfected macrophages (day 9, 1 × 106 cells) were incubated with DEHP (200 nM) for 24 h and then stimulated with LPS (10 ng) for 6 h, after which the IL-12p40 protein level in whole-cell lysates was determined by ELISA using an anti-IL-12p40 monoclonal antibody (Abcam).
Effect of apigenin or GW7,647 on IL-12p40 production by macrophages exposed to DEHP after LPS stimulation
Apigenin (a selective inhibitor of protein kinase casein kinase 2: CK2) was purchased from Sigma-Aldrich (St. Louis, Missouri, USA), while GW7,647 (a specific PPARα ligand) was obtained from Cayman Chemical (Ann Arbor, Michigan, USA). GM-CSF-dependent macrophages (on day 9 of culture) were pretreated with Apigenin (50 μM), incubated with DEHP (200 nM) for 24 h, and stimulated with LPS (10 ng) for 6 h. Then the IL-12p40 protein level in whole-cell lysates was measured by ELISA using an anti-IL-12p40 monoclonal antibody (Abcam).
GM-CSF-dependent macrophages (day 9) were also pretreated with GW7,647 (6 nM) for 24 h and then stimulated with LPS (10 ng) for 6 h, after which the IL-12p40 protein level in whole-cell lysates was measured by ELISA.
Statistical analysis
Results are expressed as the mean ± SE. Analysis of variance and the t-test for independent means were used to assess differences between multiple groups and differences between two groups, respectively. When the F ratio was found to be significant, mean values were compared using a post hoc Bonferroni test. A probability (p) <0.05 was considered to indicate significance in all analyses.
Results
DEHP significantly reduced PPARα protein production by GM-CSF-dependent macrophages, as determined by Western blotting or ELISA (Figure 1). Stimulation of GM-CSF-dependent macrophages with LPS increased IL-12p40 production, whereas DEHP significantly attenuated this response of IL-12p40. However, incubation with DEHP alone did not affect IL-12p40 production by macrophages (Figure 2). Interestingly, transfection of macrophages with PKC siRNA (for PKC α, β, γ, or δ) restored IL-12p40 expression by these cells after DEHP exposure and LPS stimulation (Figure 3). In addition, siRNA for PPARα significantly restored IL-12p40 production by DEHP-treated macrophages after LPS stimulation. GW7,647 (a specific PPARα ligand) attenuated the response of IL-12p40 production to LPS stimulation. IL-12p40 protein production by DEHP-treated macrophages after LPS stimulation was also reduced by TLR4 siRNA (Figure 4). DEHP treatment reduced TRAF6 expression, as determined by ELISA (Figure 5). Stimulation of macrophages with LPS upregulated both IL-12p40 and TNFAIP3/A20 expression. Surprisingly, the TNFAIP3/A20 level remained high after LPS stimulation, while IL-12p40 and TNFAIP3/A20 were dramatically reduced by exposure of macrophages to DEHP (Figure 6). Next, we studied the effects of PKC isoforms on TNFAIP3/A20 production by DEHP-treated macrophages after LPS stimulation. We found that PKCα siRNA further reduced TNFAIP3/A20, whereas transfection with PKCγ siRNA (but not PKCβ or δ siRNA) significantly upregulated TNFAIP3/A20 expression (Figure 7). Moreover, we investigated the influence of NADPH oxidase p22phox or DUOX2 on IL-12p40 production by macrophages after incubation with DEHP and LPS stimulation. Silencing of DUOX2 led to dramatic restoration of IL-12p40, but p22 siRNA only had a partial effect (Figure 8). Interestingly, silencing of TNFAIP3/A20 dramatically restored IL-12p40 production by DEHP-treated macrophages after LPS stimulation and TNFα siRNA partially restored IL-12p40 production, but EGFR siRNA did not affect it. Apinenin (a specific inhibitor of protein kinase CK2) only had a marginal effect on IL-12p40 production (Figure 9).

PPARα protein levels in DEHP-treated macrophages. PPARα protein levels were determined after incubation of GM-CSF-dependent macrophages (day 9 of culture) with DEHP (0, 10, 20, 50, 100, or 200 nM) for 24 h. Data were obtained from the cells of three donors and are shown as the mean ± SE. The level of significance after Bonferroni correction was set at *p < 0.05, **p < 0.01, N.S., not significant. PPAR: peroxisome proliferator–activated receptor; DEHP: di-(2-ethylhexyl) phthalate; GM-CSF: granulocyte-macrophage colony-stimulating factor; SE: standard error.

Effect of DEHP or LPS on IL-12p40 production by GM-CSF-dependent macrophages. IL-12p40 protein levels were determined after GM-CSF-dependent macrophages (day 9 of culture) were incubated with DEHP (0, 10, 20, 50, 100, or 200 nM) for 24 h or stimulated with LPS (10 ng) for 6 h. Data were obtained from the cells of three donors and are shown as the mean ± SE. The level of significance after Bonferroni correction was set at **p <0.01; N.S., not significant. DEHP: di-(2-ethylhexyl) phthalate; LPS: lipopolysaccharide; IL: interleukin; GM-CSF: granulocyte-macrophage colony-stimulating factor; SE: standard error.
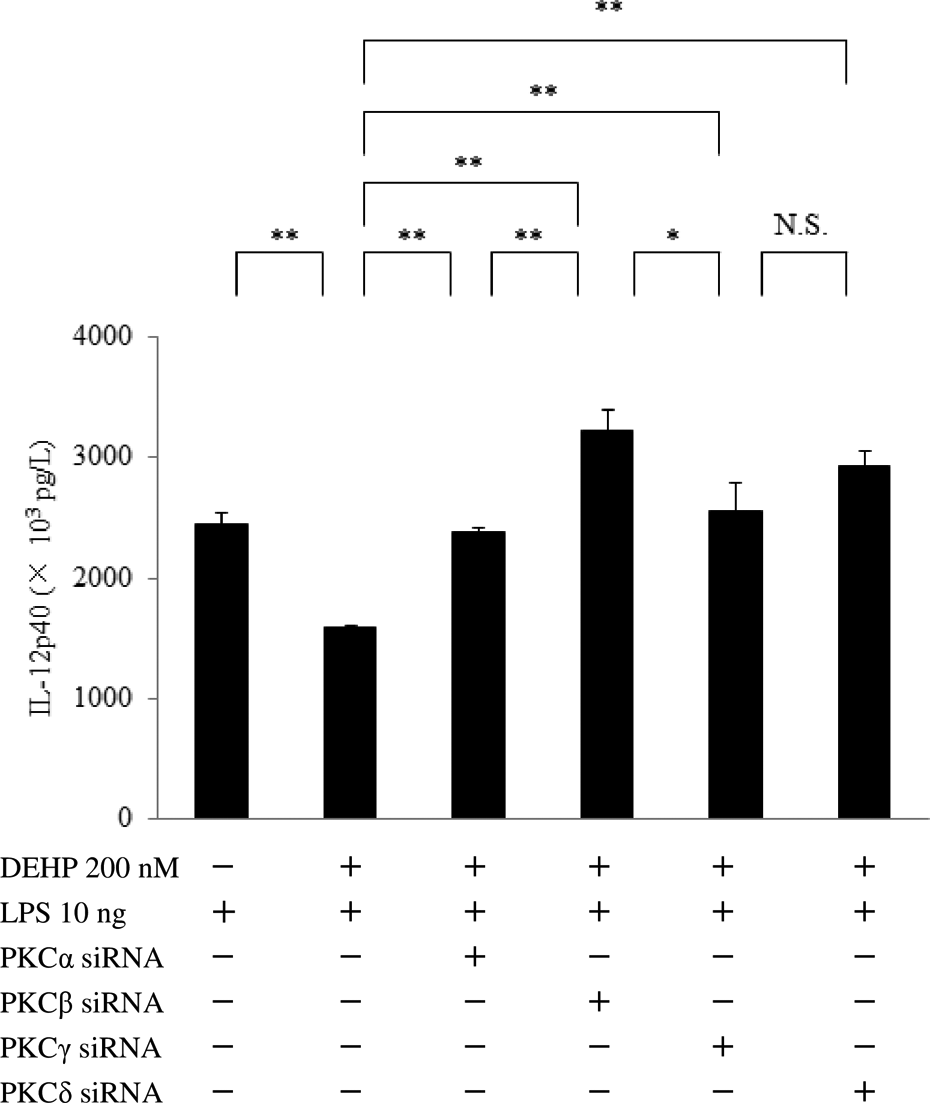
Effect of siRNA for PKCα, β, γ, or δ on IL-12p40 production by DEHP-treated macrophages after LPS stimulation. IL-12p40 protein levels were determined after GM-CSF-dependent macrophages (day 9 of culture) were transfected with PKCα, β, γ, or δ siRNA, exposed to DEHP (200 nM) for 24 h, and stimulated with LPS (10 ng) for 6 h. Data were obtained from the cells of three donors and are shown as the mean ± SE. The level of significance after Bonferroni correction was set at *p < 0.05, **p < 0.01, N.S., not significant. siRNA: small interfering RNA; PKC: protein kinase C; IL: interleukin; DEHP: di-(2-ethylhexyl) phthalate; GM-CSF: granulocyte-macrophage colony-stimulating factor; SE: standard error.

Effect of PPARα siRNA, TLR4 siRNA, or GW7,647 on IL-12p40 production by LPS stimulated macrophages with or without DEHP treatment. IL-12p40 protein levels were determined after GM-CSF-dependent macrophages (day 9 of culture) were transfected with PPARα or TLR4 siRNA, incubated with DEHP (0 or 200 nM) or GW7,647 (6 nM) for 24 h, and stimulated with LPS (10 ng) for 6 h. Data were obtained from the cells of three donors and are shown as the mean ± SE. The level of significance after Bonferroni correction was set at **p < 0.01. PPAR: peroxisome proliferator–activated receptor; siRNA: small interfering RNA; TLR: toll-like receptor; IL: interleukin; LPS: lipopolysaccharide; DEHP: di-(2-ethylhexyl) phthalate; GM-CSF: granulocyte-macrophage colony-stimulating factor; SE: standard error.

Effect of DEHP on TRAF6 expression by macrophages. GM-CSF-dependent macrophages (day 9 of culture) were incubated with DEHP (0 or 200 nM) for 24 h and whole-cell lysates and the protein level of TRAF6 in whole-cell lysates were also measured by ELISA. Data were obtained from the cells of three donors and are shown as the mean ± SE. The level of significance after Bonferroni correction was set at **p < 0.01. DEHP: di-(2-ethylhexyl) phthalate; TRAF6: tumor necrosis factor receptor–associated factor 6; GM-CSF: granulocyte-macrophage colony-stimulating factor; ELISA: enzyme-linked immunosorbent assay; SE: standard error.

IL-12p40 and TNFAIP3/A20 production by LPS-stimulated macrophages with or without DEHP pretreatment. Changes of IL-12p40 or TNFAIP3/A20 were determined after GM-CSF-dependent macrophages (day 9 of culture) were incubated with DEHP (0 or 200 nM) for 24 h and stimulated with LPS (0 or 10 ng) for 6 h. Data were obtained from the cells of three donors and are shown as the mean ± SE. The level of significance after Bonferroni correction was set at **p < 0.01, N.S., not significant. IL: interleukin; TNFAIP3: tumor necrosis factor α–induced protein 3; LPS: lipopolysaccharide; DEHP: di-(2-ethylhexyl) phthalate; GM-CSF: granulocyte-macrophage colony-stimulating factor; SE: standard error.
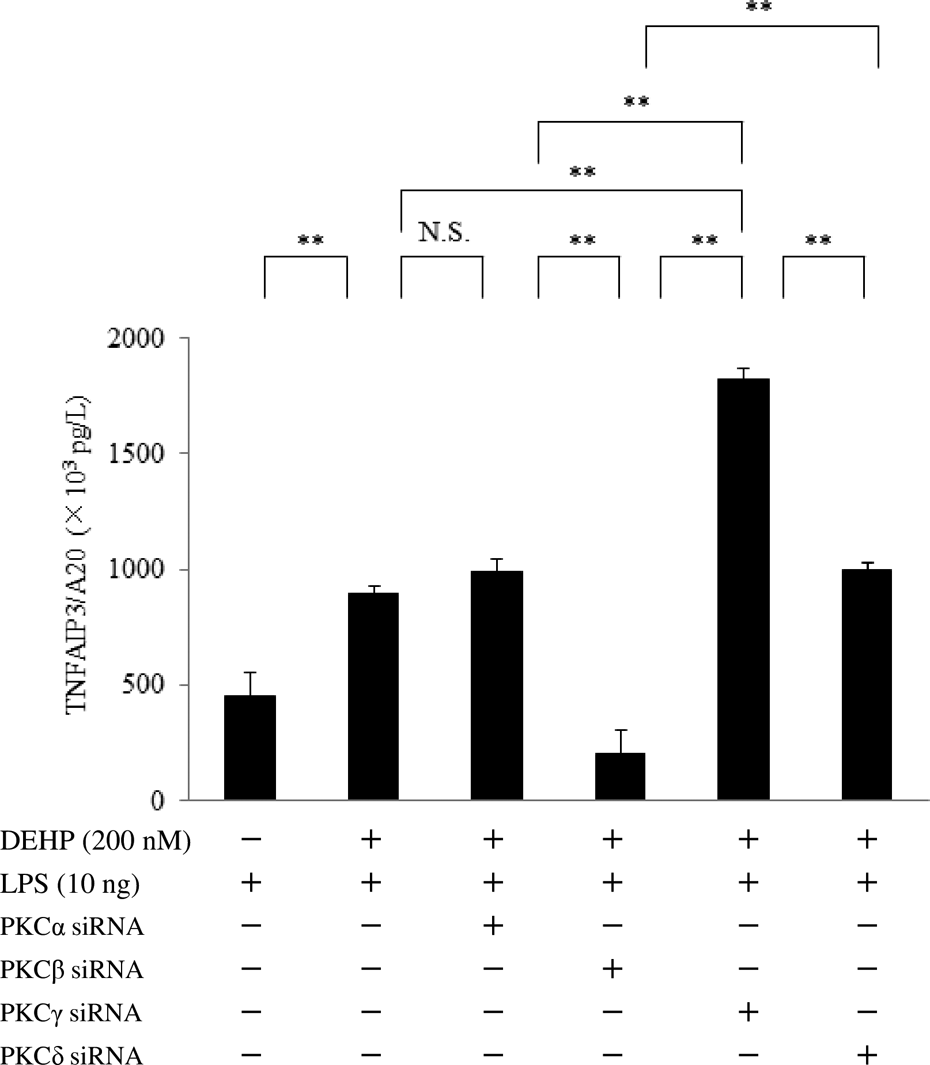
Effect of PKCα, β, γ, or δ siRNA TNFAIP3/A20 expression by DEHP-treated macrophages after stimulation with LPS. Changes of TNFAIP3/A20 protein were determined after GM-CSF-dependent macrophages (day 9 of culture) were transfected with PKCα, β, γ, or δ siRNA, exposed to DEHP (0 or 200 nM) for 24 h, and stimulated with LPS (10 ng) for 6 h. Data were obtained from the cells of three donors and are shown as the mean ± SE. The level of significance after Bonferroni correction was set at **p < 0.01, N.S., not significant. PKC: protein kinase C; siRNA: small interfering RNA; TNFAIP3: tumor necrosis factor α–induced protein 3; DEHP: di-(2-ethylhexyl) phthalate; LPS: lipopolysaccharide; GM-CSF: granulocyte-macrophage colony-stimulating factor; SE: standard error.

Effect of DUOX2 siRNA or p22phox siRNA on IL-12p40 production by macrophages after DEHP and LPS treatment. IL-12p40 protein levels were determined after GM-CSF-dependent macrophages (day 9 of culture) were transfected with DUOX2 siRNA or p22phox siRNA, exposed to DEHP (200 nM) for 24 h, and stimulated with LPS (10 ng) for 6 h. Data were obtained from the cells of three donors and are shown as the mean ± SE. The level of significance after Bonferroni correction was set at *p < 0.01, **p < 0.05, N.S., not significant. DUOX: dual oxidase; siRNA: small interfering RNA; IL: interleukin; DEHP: di-(2-ethylhexyl) phthalate; LPS: lipopolysaccharide; GM-CSF: granulocyte-macrophage colony-stimulating factor; SE: standard error.

Effect of TNFAIP3/A20 siRNA, TNFα siRNA, EGFR siRNA, or apigenin on IL-12p40 expression by macrophages after DEHP and LPS stimulation. Changes of IL-12p40 protein levels were determined after GM-CSF-dependent macrophages (day 9 of culture) were transfected with TNFAIP3/A20 siRNA or EGFR siRNA, or pretreated with apigenin (50 μM), followed by stimulation with DEHP and LPS. Data were obtained from the cells of three donors and are shown as the mean ± SE. The level of significance after Bonferroni correction was set at *p < 0.05, **p < 0.01, N.S., not significant. TNFAIP3: tumor necrosis factor α-induced protein 3; siRNA: small interfering RNA; TNF: tumor necrosis factor; EGFR: epidermal growth factor receptor; IL: interleukin; GM-CSF: granulocyte-macrophage colony-stimulating factor; SE: standard error.
Discussion
The present study demonstrated that LPS upregulated IL-12p40 expression by GM-CSF-dependent macrophages in a concentration-dependent manner, whereas incubation with DEHP significantly reduced IL-12p40 production after LPS stimulation. In patients with sepsis, the median endotoxin level was reported to be 300 pg/mL. 27 It has been reported that a DEHP concentration >1000 nM significantly decreased IL-8 release from human umbilical vein endothelial cells. 28 The DEHP level in blood bags ranges from 1.8 to 83.2 μg/mL. 29 Therefore, we utilized 200 nM DEHP in this study and found that DEHP promoted the ubiquitin-editing enzyme TNFAIP3/A20, which suppressed IL-12p40 production by macrophages after LPS (10 ng) stimulation.
DEHP attenuated PPARα protein production in a concentration-dependent manner, Interestingly, siRNA for PPARα restored IL-12p40 in DEHP-treated macrophages after LPS stimulation, while GW7,647 (a specific PPARα ligand) attenuated IL-12p40 production after LPS stimulation. These results suggest that inhibition of IL-12p40 production by macrophages in response to DEHP may be mediated through a PPARα-dependent mechanism.
PPARα governs inflammatory processes, mainly by downregulating gene expression via a mechanism generally referred to as transrepression. The anti-inflammatory effects of PPARα are probably explained by its interference with the activity of many pro-inflammatory transcription factors, including signal transducer and activator of transcription (STAT), activator protein-1 (AP-1), and NF-κB. 30
DEHP has been reported to increase the production of ROS in a PPARα-independent manner via NADPH oxidase. 17 The NADPH oxidase (Nox) family has seven isoforms, including Noxes 1 through 5 and DUOX1 and DUOX2. ROS promote the sustained activation of PKC. 17 DUOX1 phosphorylation is mediated via protein kinase A, whereas DUOX2 is phosphorylated via PKC, with DUOX2 being stimulated by the PLC cascade and PKC-dependent phosphorylation. 19 It was also reported that TNFα activates a p22phox-based NADPH oxidase. 20 Next, we investigated the role of DUOX2 and p22phox in the reduced response of IL-12p40 to LPS stimulation after DEHP treatment of macrophages. The IL-12p40 protein level was markedly restored by silencing DUOX2 but was only partially restored by p22phox siRNA. PPARα is known to suppress oxidative stress, and it has been reported that PPARα ligands/activators decrease p22phox expression. 31
DEHP induces oxidative stress and increases the generation of ROS, 32 while oxidative stress activates PKC. We found that transfection of macrophages with siRNA for PKCα, β, γ, or δ restored IL-12p40 production by macrophages after DEHP treatment and LPS stimulation. Inhibition of PKC impairs ligand-activated PPARα transcriptional activity, 33 and it has been reported that PKCs (α, β, δ, and ζ) affect PPARα activity. 34
Phosphorylation of specific serine residues of PPARα nearly doubles its transactivation activity. In human liver cells, PKCα and β were reported to phosphorylate PPARα at serines 179 and 230, thus increasing ligand-induced PPARα transcriptional activity. 33 Next, we investigated the effect of PKC isoforms on IL-12p40 production by macrophages. We found that silencing of PKCα, β, γ, or δ restored IL-12p40 production by DEHP-treated macrophages after LPS stimulation. In addition, siRNA targeting PPARα also restored the IL-12p40 protein level after DEHP treatment and LPS stimulation. These results suggest that PKC may play an important role in activation of PPARα.
Activation of the NF-κB pathway is tightly controlled by several negative feedback mechanisms, including A20 (also known as TNFAIP3), which is a ubiquitin-modifying enzyme encoded by the tnfaip3 gene. 35 Also, the intracellular ubiquitin-editing protein TNFAIP3/A20 is a key player in negative feedback regulation of NF-κB signaling in response to multiple stimuli. 14 Interestingly, we found that transfection of TNFAIP3/A20 siRNA dramatically restored IL-12p40 expression by DEHP-treated macrophages after LPS stimulation.
Expression of TNFAIP3/A20 is induced by TNFα, and it has also been reported that DEHP promotes TNFα secretion/production by monocytes or macrophages. 13 Furthermore, TLR activation induces the production of TNFα. We found upregulation of TNFAIP3/A20 expression by GM-CSF-dependent macrophages after LPS stimulation, whereas silencing of TNFα significantly reduced A20 expression. TNFAIP3/A20 is a key molecule in an inhibitory feedback loop downstream of the TNFα 36 or TLR 37 signaling cascades. We demonstrated that LPS stimulation upregulated both IL-12p40 and A20 expression by macrophages, while DEHP dramatically reduced both IL-12p40 and A20 protein levels.
Next, we investigated the effects of PKC isoforms on TNFAIP3/A20 expression by macrophages after DEHP treatment and LPS stimulation. Unexpectedly, we found that TNFAIP3/A20 was either induced or attenuated by differentially acting PKC isoforms. PKCα siRNA further reduced A20 protein levels in DEHP-treated macrophages after LPS stimulation, whereas PKCγ siRNA dramatically elevated A20, but siRNA for PKCβ or PKCδ did not. DEHP enhances the production of oxidants through activation of NADPH oxidase, 17 and it was reported that oxidative activation of PKCγ is mediated through the C1 domain. 38 Surprisingly, PKCγ siRNA was found to be sufficient for elevation of TNFAIP3/A20 production. Conversely, PKCγ may negatively modulate TNFAIP3/A20 expression by DEHP-treated macrophages after LPS stimulation.
PPARα has been reported to induce the inhibitory protein IκBα 39 and rapid IκBα turnover is implicated in high basal NF-κB activity. Protein kinase CK2 has traditionally been classified as a messenger-independent protein serine/threonine kinase, and CK2 regulates the NF-κB pathway as well as being responsible for phosphorylation of IκBα. 40 Apigenin, a selective inhibitor of protein kinase CK2, was reported to decrease IκBα turnover and reduce nuclear accumulation of NF-κB. 41 We found that apigenin had a marginal effect on the restoration of IL-12p40 production by macrophages after DEHP treatment and LPS stimulation, and this inhibitory effect of DEHP on IL-12p40 expression may be partly IκBα dependent.
Importantly, PPARα ligand has been shown to induce mitogen-activated protein kinase (MAPK) signaling by EGFR transactivation. 42 Peroxisome proliferators activate protein kinase cascades and promote the phosphorylation of kinases related to the extracellular signal-regulated kinase (ERK) pathway, including EGF. 43 Therefore, we investigated the effect of EGFR kinase on ERK signaling for suppression of IL-12p40 production by silencing of EGFR, but we found that EGFR siRNA did not affect IL-12p40 production. Thus, the reduction of IL-12p40 by DEHP is independent of ERK signaling via EGFR transactivation.
TRAF6 has a critical influence on TLRs. 44 Human TLRs employ myeloid differentiation primary response gene 88 (MyD88) as an adaptor protein and induce activation of NF-κB via IL-1 receptor-associated kinase (IRAK) and TRAF6 protein, similar to IL-1R-mediated NF-κB activation. 45 Thus, TRAF6 activates NF-κB by different mechanisms depending on the signals. 46 MyD88-IRAK-TRAF6-NF-κB signaling is the major signaling pathway for the response of IL-12p40 to LPS stimulation, and we have previously reported that silencing TRAF6 inhibits LPS-induced production of IL-12p40. 47 Interestingly, we also found that DEHP attenuated TRAF6 expression and we showed that TNFAIP3/A20 was upregulated in macrophages after LPS stimulation. Importantly, TNFAIP3/A20 has been reported to attenuate TRAF6 expression, 48 while silencing TNFAIP3/A20 restores TRAF6 expression, and it has been suggested that this TNFAIP3/TRAF6 axis is essential for preventing activation of NF-κB by LPS. 49
Humans absorb and metabolize DEHP differently to rats and mice. 50 Thus, environmental levels of DEHP are not expected to cause adverse effects on human health. However, aging alters the immune system, which may contribute to the increased susceptibility of older people to infection and cancer. Profound suppression of the protective immune response to infection occurs in the absence of the IL-12p40 subunit. Because DEHP decreases IL-12p40 production, it could potentially impair the immune response. Figure 10 displays the proposed protein–protein interactions and signaling pathways addressed in this study.

Proposed mechanisms through which DEHP may suppress IL-12p40 production by LPS-stimulated macrophages via the PPARα/TNFAIP3/TRAF6 axis. DEHP induces ROS via the PKC/DUOX2 signaling pathway. ROS or PPARα agonists promote production of the ubiquitin-editing enzyme TNFAIP3/A20, which deubiquitinates TRAF6 and IκBα, thus inhibiting NF-κB activation. DEHP: di-(2-ethylhexyl) phthalate; IL: interleukin; LPS: lipopolysaccharide; PPAR: peroxisome proliferator–activated receptor; TNFAIP3: tumor necrosis factor α–induced protein 3; TRAF6: tumor necrosis factor receptor–associated factor 6; ROS: reactive oxygen species; PKC: protein kinase C; DUOX: dual oxidase.
Conclusion
Stimulation of GM-CSF-dependent macrophages with LPS increased IL-12p40 production, whereas DEHP significantly attenuated the increase of IL-12p40. siRNA for PPARα significantly restored IL-12p40 production by DEHP-treated macrophages after LPS stimulation. Stimulation of these macrophages with LPS upregulated TNFAIP3/A20 expression, while both IL-12p40 and TNFAIP3/A20 were dramatically reduced by exposure of macrophages to DEHP.
Furthermore, silencing of TNFAIP3/A20 substantially restored IL-12p40 production by DEHP-treated macrophages with LPS stimulation. Moreover, DEHP treatment reduced TRAF6 expression by macrophages. These findings suggest that the PPARα/TNFAIP3/TRAF6 axis may play an important role in suppressing IL-12p40 production by DEHP-treated macrophages after LPS stimulation.
Footnotes
Author’s note
The funding source had no role in study design; the collection, analysis, and interpretation of data; in writing of the report; and in the decision to submit the article for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by (Kumamoto Health Science University special fellowship grant no. 27-A-01).
