Abstract
Recently, palladium nanoparticles (PdNPs) have been increasingly used in many industrial sectors, and this has led to a significant release of nano-sized palladium particles into the environment. However, despite the increase in occupational and general population exposure, information on the potential adverse effects of these PdNPs is still limited and their impact on the immune system constitutes a major health concern. Therefore, the aim of this study was to investigate the potential adverse effects induced by subchronic intravenous administration of PdNPs on the immune system of female Wistar rats by evaluating alterations in Interleukin (IL)-1α, IL-2, IL-4, IL-6, IL-10, IL-12, granulocyte-macrophage colony-stimulating factor (GM-CSF), Interferon (INF)-γ, and Tumor Necrosis Factor (TNF)-α serum levels. Exposed and control animals were randomly divided into five groups (0, 0.012, 0.12, 1.2, and 12 μg PdNPs per kg body weight) which were treated with repeated intravenous injections of vehicle or PdNPs (on day 1, 30, and 60). Subchronic exposure to PdNPs induced a decreasing trend in serum levels in most of the cytokines investigated, with the highest concentration (12 μg/kg) determining significant inhibitory effects. Overall, these results showed that PdNPs are able to alter cytokine serum levels in subchronically treated Wistar rats, suggesting a possible impact of these xenobiotics on the immune system after long-term exposures.
Introduction
The advent and rapid development of nanotechnology have led to a significant increase in the number of manufactured products containing innovative engineered nanomaterials (NMs) and nanoparticles (NPs). In fact, due to their unique electrical, optical, and mechanical properties, nano-sized materials have been developed and employed in a wide range of industrial fields, including electronics, medicine, construction, food, clothing, and cosmetics. 1 Moreover, several palladium (Pd)-based nanostructures with different morphologies have been increasingly synthesized and used in catalytic and electrocatalytic chemical reactions, wastewater treatment, jewelry, electronics, and in biomedical fields. 2 –4 Furthermore, modern three-way automobile catalytic converters (containing Pd) 5,6 can cause the release and the accumulation of Pd ultrafine particles (PdUFPs) in several environmental matrices. 7 –9 Therefore, considering that both Pd nanoparticles (PdNPs) and PdUFPs are characterized by particular physicochemical features that may make their properties more harmful than those of larger particles of the same material, 10 it seems vital to understand the risks that the probable increase in exposure to nano-sized Pd particles may pose to human safety and health.
In recent years, scientific interest in the possible alterations induced by PdNPs on the immune system has been growing both on account of the role such a system plays in protecting the body’s homeostasis from external insults and the previous immunological results obtained with Pd salts. Indeed, it is widely acknowledged that one of the most important adverse health effects induced by Pd exposure is the sensitization to the metal 11,12 since Pd ions are known to be potent reacting sensitizers. 13 Several studies have observed cases of Pd sensitization, and almost all of these are related to exposure to dental restorations or jewelry containing Pd. Strongly positive reactions to Pd dichloride (PdCl2), indicating a hypersensitivity to the metal, have been observed by performing patch tests in patients (with metal bridges, crowns, or dental alloys containing Pd) who have experienced swelling and pain of the oral mucosa, itching, dizziness, xerostomia, and urticaria. 14 –19 Similarly, Pd hypersensitivity reactions caused by jewelry were observed when granulomatous contact dermatitis was associated with this metal in ear piercing in female patients, that, after patch testing, showed a distinctly positive reaction to PdCl2. 20,21 With regard to occupational exposures, Daenen et al. 22 reported a case of asthma in a worker exposed to the fumes of an electrolysis bath containing Pd chloride, whereas other studies that investigated hypersensitivity to Pd in a number of different workplaces revealed the presence of positive skin prick and patch test reactions in refinery and catalyst industry workers. 23 –25 Moreover, researchers have demonstrated the ability of Pd salts to affect the immunological functions of different cell types by altering the production of several cytokines 26 –28 and also to induce important immunomodulating modifications in Wistar rats (Figure 1(a) and (c)). 11,12

Effects of Pd and PdNP exposure on cytokine production and release observed in in vitro and in vivo studies. (a) In vitro studies that investigated the alterations in serum cytokines following exposure to Pd salts; (b) in vitro studies that investigated the alterations in serum cytokines following exposure to PdNPs; (c) in vivo studies that investigated the alterations in serum cytokines following exposure to Pd salts; (d) in vivo studies that investigated the alterations in serum cytokines following exposure to PdNPs. A549: human alveolar carcinoma cells; GM-CSF: granulocyte-macrophage colony-stimulating factor; IL: Interleukin; INF: Interferon; PBEC, human primary bronchial epithelial cells; PBMC: peripheral blood mononuclear cells; Pd: palladium; PdNP: Pd nanoparticle.
On the other hand, recent evidence has shown that also PdNPs are able to influence the in vitro production of several cytokines (Figure 1(b)), 29 –31 although quite different results are observed compared to those produced by Pd salts. Furthermore, our previous investigation showed a generalized stimulatory action of PdNPs on the immune system of Wistar rats after a subacute treatment (Figure 1(d)). 32 However, current knowledge of the biological effects of PdNPs on the immune system is far from complete. For example, the immunological response induced by PdNPs after prolonged or repeated exposure, the underlying molecular mechanisms of toxic action, the possible consequences in terms of autoimmune/inflammatory disease manifestations, or conversely, compensatory anti-inflammatory responses, as well as the role of PdNPs in sensitization to the metal, all need to be further clarified. Therefore, the aim of the present study was to evaluate the effects induced on the immune system of female Wistar rats by repeated intravenous administrations of PdNPs in a subchronic time frame (90 days) by assessing possible quantitative changes in the serum concentrations of a number of cytokines (Interleukin (IL)-1α, IL-2, IL-4, IL-6, IL-10, IL-12, granulocyte-macrophage colony-stimulating factor (GM-CSF), Interferon (INF)-γ, and Tumor Necrosis Factor (TNF)-α).
Materials and methods
Preparation and characterization of uncoated PdNP hydrosol
PdNPs were prepared and characterized as previously described. 32,33 Briefly, for preparation of a NP stock solution, 500 μL of a commercially available Pd (II) standard solution (1000 mg/L, Pd(NO3)2 in 0.5 mol/L HNO3; Merck, Darmstadt, Germany) was added to 100 mL of a reducing solution and shaken thoroughly. The reducing solution was obtained by diluting 300 μL of a freshly prepared 0.029 molar sodium borohydride aqueous solution (NaBH4; pro analysis; Merck) in 100 mL of ultrapure water. Immediately after mixing the reducing solution with Pd(II), the formerly clear and transparent solutions change to dark gray, indicating the formation of PdNPs. The size distribution of NPs and the Pd concentration of the stock PdNP hydrosol were determined using transmission electron microscopy (TEM; Zeiss EM 10, Carl Zeiss Microscopy GmbH, Jena, Germany; operated at 80 kV) and high-resolution continuum source graphite furnace atomic absorption spectrometry (contrAA 600; Analytik Jena, Jena, Germany; spectral line at 244.791 nm), respectively. The Pd concentration of the stock hydrosol was found to be 4.71 ± 0.05 mg/L. The average size of the NPs of 10 ± 6 nm (see Figure 2) was evaluated by measuring the greatest particle length of approximately 500 individual particles as depicted by TEM images using ImageJ software (National Institutes of Health, Bethesda, Maryland, USA).

PdNP characterization. (a) Size distribution histogram of PdNPs. Mean size was 10 ± 6 nm; (b) TEM images of PdNPs. The mean size was obtained by evaluating and measuring maximum particle length of 500 individual nanoparticles depicted by TEM images. PdNP: palladium nanoparticles; TEM: transmission electron microscopy.
The hydrosol is stable for at least 2 weeks when stored at 4°C. Before use, the stored PdNP hydrosol was homogenized by shaking vigorously, then final concentrations and doses used in the experiments were obtained by diluting adequate aliquots of the PdNP hydrosol in ultrapure water.
Animal husbandry
Twenty-five 3-month-old female, pathogen-free Wistar rats were supplied by the Experimental Animal Production Plant of the Catholic University of the Sacred Heart (Rome, Italy) and allowed to acclimatize for 2 weeks before starting the experiment. Wistar rats are an outbred strain of albino rats employed in all fields of medical and biological research as a general multipurpose model. 34 In fact, the use of rats offers a series of advantages such as a metabolic pathway similar to that of humans, similar anatomical and physiological characteristics, and a large database for comparative purposes. 35 For this reason, the rat is the species selected for nonclinical immunotoxicity studies and outbred Wistar rats in particular are often used on account of their acceptable inter-animal variability. 36,37
The animals were maintained during the entire experiment in Makrolon cages (model 1291, with overall dimensions of 425 × 266 × 185 mm and floor area of 800 cm2; Tecniplast S.p.A., Buguggiate, Italy) containing a wooden dust-free bedding (model Scobis Uno, Mucedola s.r.l., Settimo Milanese, Italy), at a room temperature of 23.1°C, a relative humidity of 55%, and a 12-h light/dark cycle. The animals had a mean weight of 265 ± 13 g and were fed with the solid “R” maintenance diet for rats (Altromin Rieper A. S.p.A., Vandoies, Italy). Diet and purified water were administered to the animals ad libitum. No significant changes in body weight were observed during or at the end of the experiments.
Ethics statement
The animal study was approved by the Ethical Committee “Commissione per la Valutazione Etica di Sperimentazioni Animali e di Correttezza della Gestione dell’Animal Care” of the Catholic University of the Sacred Heart of Rome, Italy, under permit number 20H and was authorized by the Italian Ministry of Health, in compliance with Legislative Decree 116/92, which implemented the European Directive 86/609/EEC on laboratory animal protection in Italy. Animals used in this study were housed and treated in compliance with Legislative Decree 116/92 guidelines, and all efforts were made to minimize animal suffering.
Animal administration and sampling of biological material
The 25 female Wistar rats were randomly divided into four exposure groups and one control group, with five rats per group. Rats were given with repeated (on day 1, 30, and 60) intravenous injections of vehicle (control group) and varying concentrations of PdNPs (0.012, 0.12, 1.2, and 12 μg PdNPs per kg body weight) via the tail vein. The experimental design involved doses resembling possible occupational and/or environmental exposure scenarios. On the basis of Pd airborne levels (highest mean level of 7.70 ± 4.15 µg/m3) reported in the literature for an occupational setting 38 and a human breathing rate of around 20 m3/day (for a man with a mean weight of 70 kg), a potential occupational exposure to Pd via inhalation corresponds to a dose of 2.20 µg/kg, which was in the range of doses used in our experiments. Therefore, the higher exposure treatments (1.2 and 12 μg PdNPs per kg body weight) were chosen to simulate possible occupational exposures both under normal job activities and accidental conditions during which repeated chemical contacts may occur. The lower doses (0.012 and 0.12 μg PdNPs per kg body weight) were used to investigate potential adverse effects at exposure levels closely resembling those of the general population. Moreover, the intravenous route of treatment and the wide range of applied doses provided a suitable experimental framework for extrapolating a preliminary dose–response curve to define the immunotoxicological behavior of PdNPs in a subchronic time period. On 90th day of exposure, rats were anesthetized with 0.5 mg of medetomidine and 75 mg of ketamine per kg body weight. Subsequently, blood from each animal was drawn by cardiac puncture and collected in a 1.5 ml vial (Eppendorf srl, Milan, Italy). Serum samples were obtained from blood by centrifugation (3500 r/min per 15 min) and stored at −28°C until analysis. After the blood sampling, rats were euthanized via exsanguination by cutting both the abdominal aorta and vena cava. The intravenous mode of administration was chosen to reach a 100% bioavailability of PdNPs in order to prevent possible confounding issues affecting the xenobiotic internal doses due to the variable NP adsorption and distribution rates that occur with other routes of entry. The doses used to treat animals were the same as those employed in a previous study of our group that evaluated the same immunological endpoints during a shorter period of treatment. 32 This may enable us to have a clear “follow-up” of our previous subacute data and to verify possible differences in responses caused by longer periods of exposure.
Analysis of serum cytokines
The assessment of serum cytokines was performed according to the experimental protocol previously described in the study by Iavicoli et al. 32 Briefly, the simultaneous detection and quantitation of the different cytokines were carried out using a multiplex biometric enzyme-linked immunosorbent assay-based immunoassay (Bioplex Rat Cytokine 9-Plex A panel; BioRad Inc., Hercules, California, USA), whereas their serum levels were obtained using a Bio-Plex array reader (Bioplex, Biorad).
Statistical analyses
Statistical analysis was carried out using SPSS statistics software (Statistical Package for Social Sciences, version 17.0, Chicago, Illinois, USA). We assessed the response to four different levels (0.012, 0.12, 1.2, and 12 μg PdNPs per kg body weight) of PdNPs and compared the level of different cytokines (IL-1α, IL-2, IL-4, IL-6, IL-10, IL-12, GM-CSF, INF-γ, and TNF-α) with the control animals. One-way analysis of variance was performed to test the significance of differences between the parameter means in the exposed and control rat groups. The Dunnett post hoc multiple comparison test was used to test the significance (p-value Dunnett t test <0.05) of differences between the values for each parameter at different exposure levels against the control group. Box plot or linear graphs were obtained for all analyzed parameters at different exposure levels.
Results
The serum levels of cytokines (IL-1α, IL-2, IL-4, IL-6, IL-10, IL-12, GM-CSF, INF-γ, and TNF-α) detected in female Wistar rats following the intravenous treatment with 0.012, 0.12, 1.2, and 12 μg PdNPs per kg body weight are reported in Table 1 and Figures 3 and 4.
Mean serum levels (standard error) and statistical significance of IL-1α, IL-2, IL-4, IL-6, IL-10, IL-12, GM-CSF, INF-γ, and TNF-α in control and PdNP-exposed groups (0.012, 0.12, 1.2, and 12 μg PdNPs per kg body weight).a
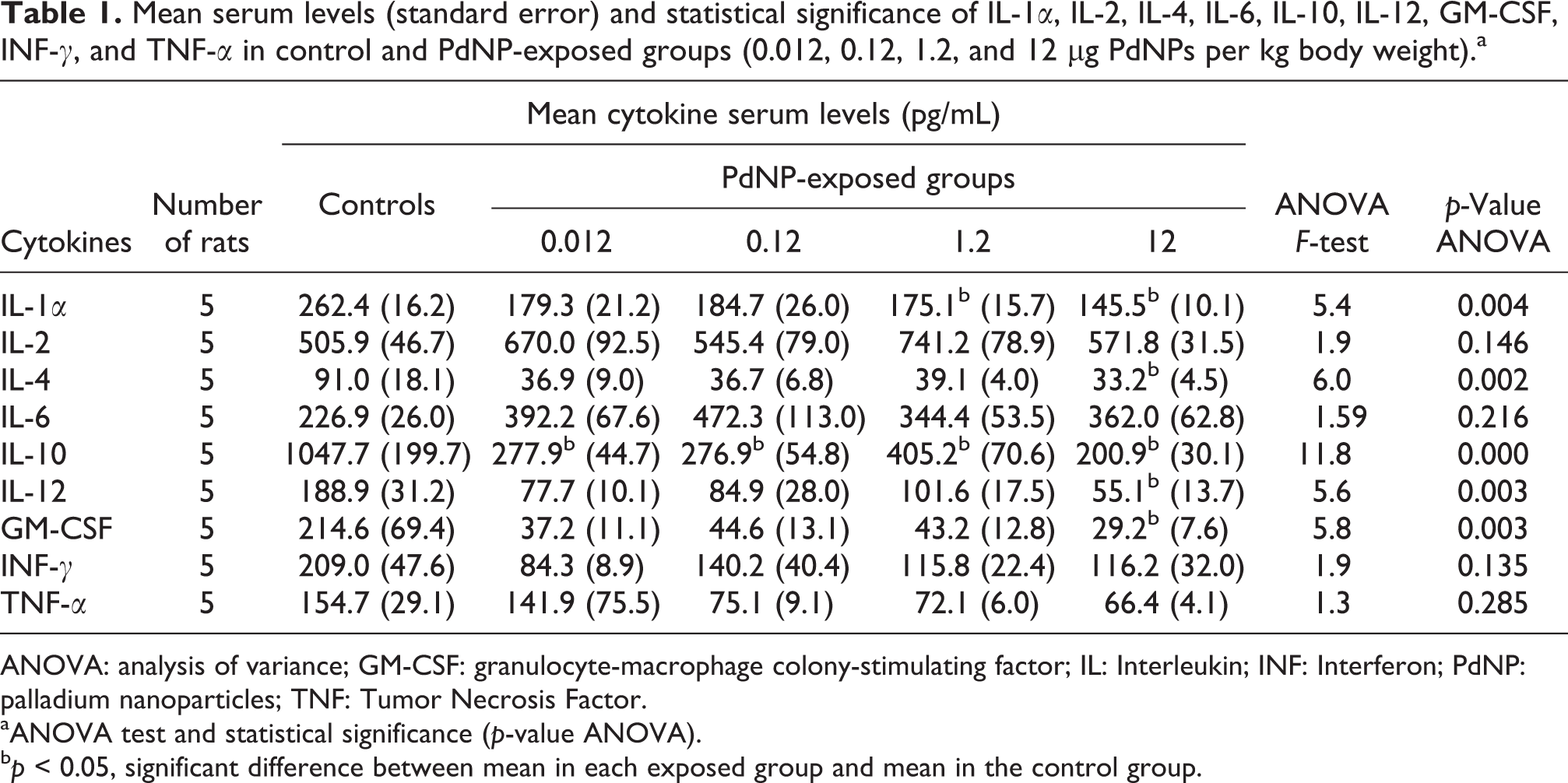
ANOVA: analysis of variance; GM-CSF: granulocyte-macrophage colony-stimulating factor; IL: Interleukin; INF: Interferon; PdNP: palladium nanoparticles; TNF: Tumor Necrosis Factor.
aANOVA test and statistical significance (p-value ANOVA).
b p < 0.05, significant difference between mean in each exposed group and mean in the control group.

Mean serum levels of different cytokines in female Wistar rats exposed to 0.012, 0.12, 1.2, and 12 μg PdNPs per kg body weight compared to control group. Outliers (°) measures between 1.5- and 3-fold that of the interquartile range (75° and 25° percentile); extreme values (*) more than 3-fold the interquartile range. GM-CSF: Granulocyte-Macrophage Colony-Stimulating Factor; IL: Interleukin; INF: Interferon; PdNP: palladium nanoparticle; TNF: Tumor Necrosis Factor.

Serum levels of IL-1α, IL-2, IL-4, IL-6, IL-10, IL-12, GM-CSF, INF-γ, and TNF-α in control and exposed (0.012, 0.12, 1.2, 12 μg PdNPs per kg body weight) Wistar rats. Outliers (°) measures between 1.5- and 3-fold that of the interquartile range (75° and 25° percentile); extreme values (*) more than 3-fold the interquartile range. GM-CSF: granulocyte-macrophage colony-stimulating factor; IL: Interleukin; INF: Interferon; PdNP: palladium nanoparticle; TNF: Tumor Necrosis Factor.
These findings show that the repeated subchronic administration of PdNPs affected the immune response in laboratory animals by inducing in the exposed groups a decreasing trend in the serum levels of most of the cytokines investigated (IL-1α, IL-4, IL-10, IL-12, GM-CSF, INF-γ, and TNF-α). In detail, statistical analysis revealed significant reductions in the mean serum concentrations of IL-1α, IL-4, IL-10, IL-12, and GM-CSF compared to controls, whereas the values of other cytokines such as INF-γ and TNF-α, although lower than in untreated rats, failed to reveal significant differences. Administration of the highest concentration of PdNPs (12 μg PdNPs per kg body weight) determined the most evident inhibitory effect on the cytokine serum levels, whereas in the exposed groups, the mean serum levels of IL-2 and IL-6 showed no significant differences compared to controls. Therefore, taken together these results suggest that repeated subchronic exposure to PdNPs is able to exert a significant effect on the immune system of female Wistar rats.
Discussion
The current increase in the production of PdNPs, the use of Pd nanostructures, and the environmental release of PdUFPs have aroused scientific concern regarding the potential adverse impact of rising levels of exposure in occupational and general living environments. In this scenario, in vivo experiments seem to be necessary in order to validate and improve in vitro results because no simple direct prediction can be made using the in vitro effects as cellular models are not able to exactly reflect the complexity of an in vivo response. 39 Furthermore, no clear definition of the chronic effects of NMs in animals and humans has yet been obtained. 40 This appears to be an even more urgent research issue since, realistically, the greatest impacts from NMs will probably be the result of long-term and low-dose exposures and little is currently known regarding these potential effects. 41
The results of our study showed that almost all of the mean cytokine serum concentrations investigated had a decreasing trend compared to the mean control values, throughout the whole exposure range, even if statistically significant differences (p < 0.05) were detected only for IL-1α, IL-4, IL-10, IL-12, and GM-CSF, particularly at the highest exposure dose (12 μg PdNPs per kg body weight; Table 1 and Figure 3). These findings may suggest that nano-sized Pd may act as a chemical insult able to induce an impact on several mediators of the immune response. However, further research is needed to determine whether such preliminary alterations are effective indicators of specific PdNP immunotoxic potential or the expression of a more complex interaction of these xenobiotics with biological environments.
Different types of NMs have proven their capacity to exert immunosuppressive or antiinflammatory effects per se. These include the inhibition of T-cell-mediated immunity, interference with functions or myelosuppression and toxicity in the cells of the immune system, inhibition of pro-inflammatory signaling pathways, antioxidant functions, and anticytokine activity. 42 In a few examples, citrate-stabilized gold NPs inhibited, in a size-dependent manner, the in vitro and in vivo production of TNF-α. 43,44 Other NP types exerted immunomodulatory effects alternatively causing an imbalance in the production of specific Th cytokine subsets. In fact, a single intravenous injection of iron oxide NPs (0.2–10 mg/kg) shifted the cytokine balance from Th1 to Th2 (decreased INF-γ and increased IL-4 release) in BALB/c mice, 45 and similar results (decreased production of Th1 cytokines such as INF-γ and TNF-α and enhanced release of Th2 cytokine IL-10) were obtained in an in vitro study that exposed dendritic cells to the same NPs. 46 An immunomodulatory effect was also observed by Yamashita et al. 47 who, following the intravenous treatment of female C57BL/6 mice with 5.5 μg/mL of fullerene C60, reported a suppression of IL-4, IL-6, and IL-17 with a concomitant induction of TNF-α.
However, while the aforementioned studies detected an immunomodulatory NP function characterized by a certain imbalance between the Th1 and Th2 subsets of cytokines, prevalently responsible for cell- and humoral-mediated immune responses, respectively, our findings failed to show such imbalance in the ratio of Th1 and Th2 key effector-cytokines, since both the serum levels of INF-γ (the signature cytokine for Th1 cell subsets) and IL-4 (the signature cytokine for Th2 cell subsets) showed a decreasing trend induced by PdNP treatment. IL-4 and other Th2 cytokines can promote the development of allergic and autoimmune diseases by inducing B-cell proliferation and isotype switching from IgM to IgE. 48 Consequently, the decreasing trend in IL-4 and in most of the cytokines assessed seems an even more surprising finding considering that Pd ions are potent and frequent reacting sensitizers 13 and that bulk Pd has frequently been the subject of conjecture in the etiopathogenesis of allergic disorders, 14 –22,49 that are traditionally associated with a typical imbalance in the Th1/Th2 cytokines with a predominant Th2 pathway. In this preliminary phase of research, and given the limited information available with regard to the immunopotential of PdNPs, we cannot exclude that the unique set of PdNP physicochemical characteristics may affect their toxicokinetic and toxicodynamic behaviors, thus influencing the interactions with the immune system, in a potentially different manner compared to that of their bulk counterparts.
Importantly, this subchronic experiment yielded quite different results from those of our previous experiment demonstrating that subacute intravenous exposure to PdNPs in rats can exert a stimulatory action on the immune system, with an overall upward trend for all cytokine serum levels, and statistically significant differences obtained in the highest treatment group (Figure 5). 32 These findings may suggest that these xenobiotics cause a more complex and generalized inhibitory action on the immune system, possibly associated with a compensatory response induced by prolonged/repeated PdNP chemical insults after an initial inflammatory activation. Although these preliminary results are interesting in so far as they underline the complexity of PdNP interaction with the immune system, they do not provide us with a full understanding of the different responses induced by PdNPs in subacute and subchronic conditions of exposure and their peculiar modes of action. A possible hypothesis is that the initial inflammatory host reaction induced by an acute–subacute PdNP exposure may induce a subsequent state of immune deactivation as an adaptive/compensatory antiinflammatory response aimed at restoring tissue homeostasis.

Serum levels of the cytokines in control and exposed (0.012, 0.12, 1.2, 12 μg PdNPs per kg body weight) Wistar rats after subacute and subchronic administration. GM-CSF: Granulocyte-Macrophage Colony-Stimulating Factor; IL: Interleukin; INF: Interferon; PdNP: palladium nanoparticle; TNF: Tumor Necrosis Factor.
In order to understand toxicological differences induced by PdNPs in subacute and subchronic conditions of treatment, we cannot rule out the possible role of nano-biomolecular interactions in influencing the effects of NPs on the immune system. In fact, NPs are able to absorb different biomolecules upon contact with any kind of biological environment, thus forming the so-called “biomolecule” corona that can affect their toxicological profile. However, the composition of the protein corona can vary according to the NP physicochemical properties, as well as to the NP-protein binding affinity, that is, some proteins may stay on the surfaces as long as NPs circulate in the bloodstream, whereas others detach from the particle surface or get replaced by proteins with a higher binding affinity. 50 Moreover, the duration of exposure may affect the nature of corona by modifying the quantitative and qualitative total protein binding, the duration of NP in the bloodstream, the possible interactions with other macromolecules, and the NP clearance by cells of the immune system, in such a way that may determine a time-dependent variability in immunological results. 51 An additional aspect to take into consideration in order to achieve a correct interpretation of these data is the plasticity of different T-cell subsets and emerging evidence that subset-signature cytokine expression is not as stable as initially believed. 52 The cytokines produced together with metabolic regulation, diverse epigenetic modifications, microRNA expression, expression of transcription factors, and tissue specific environmental cues may all interact underlying T-cell plasticity, thereby possibly modulating immune responses following acute to subchronic exposures to PdNPs. These phenomena make it even more difficult to extrapolate definite conclusions from such a precise analysis and indicate the need for further research. Moreover, on the basis of these preliminary data, clearly we are not able to establish if the cytokine alterations observed are due to a direct toxic action of PdNPs on Th-lymphocytes or are the consequence of a more complex action involving their signaling pathways of differentiation and/or their mechanisms of activation. Future in vitro and in vivo investigation should focus on understanding PdNP interactions with both immune and nonimmune cells, that is, T and B lymphocytes, CD8+ cells, natural killer cells, activated dendritic cells, macrophages, and mast cells as well as fibroblasts, endothelial cells, glial cells, and keratinocytes able to secrete cytokines in response to a variety of external stimuli. 53 –55 In order to determine the biological effects of PdNPs on the immune system, a deeper knowledge of specific cellular interactions, in terms of internalization pathways and biomolecular effects, and of NP toxicokinetic and toxicodynamic behaviors could provide helpful data. Ad hoc long-term experiments should be planned to investigate how the immune system adapts to the insults of specific xenobiotics and activates possible antiinflammatory responses. Periods of investigation should be extended, since immunosuppression is not an acute toxicity response and thus its evaluation, which involves complex functional changes of the immune system, requires long-term experiments and multiparametric analyses. 42 From a public and occupational health perspective, the different responses induced by acute/subacute and subchronic/chronic exposures to PdNPs raise questions concerning the identification of possible, sensitive, and specific biomarkers that could be useful in detecting particular time-dependent biological alterations.
Since this study constitutes a first attempt to evaluate the subchronic effects of PdNPs on the immune system, the extrapolation of definite biological implications, in terms also of disease hypersusceptibility, induced by such cytokine alterations is challenging. Clearly, future in vitro and in vivo investigations are needed to confirm these preliminary results so that a deeper understanding can also be reached regarding the immunological effects of PdNPs particularly in conditions of long-term, low-dose exposure such as those experienced by occupational and general exposed populations.
Conclusions
Only recently research into the health effects of NMs focused on assessing the potential immunotoxic impact of both immunostimulatory and immunosuppressive effects. However, to investigate issues regarding the safety of NMs, it is essential to identify their unintended impact on the immune system and to comprehend the underlying NM mechanisms of action. Our current understanding of how NPs interact with the immune system is still very limited and too many questions remain unanswered, especially with regard to the possible effects induced by long-term exposure. In this study, we assessed alterations in serum cytokine levels to make a first in vivo attempt to evaluate the possible effects on the immune system of subchronic exposure to PdNPs. We demonstrated that PdNPs were able to induce immunological alterations in Wistar rats since almost all of the cytokine concentrations investigated showed a decreasing trend compared to the control values, with significant differences obtained at the highest exposure dose. However, since knowledge of the immunotoxic potential of PdNPs is limited, these preliminary findings should be considered with caution and should be carefully verified and confirmed by further studies. Further research is also needed to determine the mechanisms leading to the inhibition of the cytokine release and potential tolerance induction.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
