Abstract
The study reports antidotal efficacy of three HNK [bis quaternary 2-(hydroxyimino)-N-(pyridin-3yl) acetamide derivatives] and pralidoxime (2-PAM), against soman and tabun poisoning in Swiss albino mice. Protection index (PI) was determined (treatment doses: HNK oximes, ×0.20 of their median lethal dose (LD50) and 2-PAM, 30 mg/kg, intramuscularly (im)) together with atropine (10 mg/kg, intraperitoneally). Probit log doses with difference of 0.301 log of LD50 of the nerve agents administered and inhibition of acetylcholinesterase (AChE) activity by 50% (IC50) was calculated at optimized time in brain and serum. Using various doses of tabun and soman (subcutaneously (sc)), in multiples of their IC50, AChE reactivation ability of the oximes was studied. Besides, acute toxicity (0.8× LD50, im, 24 h postexposure) of HNK-102 and 2-PAM was also compared by determining biochemical, hematological variables and making histopathological observations. Protection offered by HNK-102 against tabun poisoning was found to be four times higher compared to 2-PAM. However, nearly equal protection was noted with all the four oximes against soman poisoning. HNK-102 reactivated brain AChE activity by 1.5 times more than 2-PAM at IC50 dose of soman and tabun. Acute toxicity studies of HNK-102 and 2-PAM showed sporadic changes in urea, uric acid, aspartate aminotransferase, and so on compared to control group, however, not supported by histopathological investigations. The present investigation showed superiority of newly synthesized HNK-102 oxime over standard 2-PAM, as a better antidote, against acute poisoning of tabun (4.00 times) and soman (1.04 times), in Swiss albino mice.
Keywords
Introduction
Organophosphorus compounds (OPCs) have been largely used as pesticides and insecticides in agriculture and public health usages. One of the most important aspects of OPC toxicity is their use as nerve agents in military and terroristic acts as weapons of mass destruction. In 1980s, the chemical attack during Iran–Iraq war (1980–1988), use of nerve agent sarin by terrorist during 1994–1995 in Matsumoto area and Tokyo subway system, Japan, and again sarin attack in Damascus, Syria (2013) often made headlines worldwide. Date back to 1980s, an infamous chemical attack in Iran–Iraq war, poisoned on more than 400 people, has strongly alleged the use of tabun (GA) by the investigators. 1 Such kinds of threat demands broad spectrum and more effective medical countermeasure(s) against the nerve agents.
To treat nerve agents or other toxic OPC poisoning, the available standard treatment includes use of anticholinergic drug (atropine) with an oxime (preferably pralidoxime (2-PAM)). 2 The function of anticholinergic drug is to competitively block the effects of accumulated acetylcholine (ACh) at muscauranic sites while oximes counteract the exacerbate effect of OPC on (AChE) by restoring its (AChE) activity by the process of dephosphorylation. The commonly used AChE reactivators are monopyrdinium (2-PAM) and some bis-pyridinium (obidoxime, TMB-4, methoxime, HI-6, K-series) oximes. 3 However, the only pitfall in their potency is to eliminate acute lethal effects of tabun, soman, or cyclosarin. The deleterious effect of tabun poisoning is due to the lone pair present on amidic group of this compound which is difficult to counteract by oxime, while soman proved to be difficult to treat due to early aging effect (<2 h) on AChE enzyme. Previous studies showed HI-6 as the most efficacious oxime against the lethal effects of soman 4 but its poor reactivation efficacy against tabun inhibited AChE enzyme, urges the need for developing more efficacious antidote. 5
Since 2013, attempts are being made to evaluate antidotal efficacy of new bis-quaternary acetamide derivatives (HNK oximes) against organophosphorus toxicants. 6,7 Having shown better results in terms of therapeutic efficacy (protection index (PI)) and AChE reactivation potency, HNK series oximes found to be a better antidote against dichlorvos (unpublished data), diisopropylphosphorofluoridate (DFP), 6 and sarin 7 in comparison to 2-PAM, in vivo. In view of evaluating intrinsic toxicity of these new oximes, in vitro and in vivo acute toxicity of HNK oximes was also determined, with comparison to 2-PAM. 8
Succeeding the current research, the present study aimed to evaluate the efficacy of HNK oximes against acute poisoning of tabun and soman in Swiss albino mice. In view of studying acute toxicity profile of oxime, 9,10 biochemical, hematological, and histopathological variables were also studied at sublethal dose (0.8× LD50) of HNK-102 and 2-PAM in the mice. Overall, the study compares efficacy of HNK oximes and 2-PAM, in terms of protection against multiples of lethal dose, their AChE reactivation ability against tabun and soman, and acute toxicity.
Materials and methods
Compounds
Tabun (O-ethyl-N,N-dimethyl phosphoramidocyanidate; GA); soman (O-pinacolyl methyl phosphonofluoridate; GD); and oximes: 1,10-(ethane-1,2-diyl) bis(3-(2-(hydroxyl-imino) acetamido) pyridinium) dibromide (HNK-102), 1,10-(hexane-1,6-diyl) bis (3-(2-(hydroxyl-imino) acetamido) pyridinium) dibromide (HNK-106), and 1,10-(1,4-henylene) bis(methylene) bis(3-(2-(hydroxyimino) acetamido) pyridinium) dibromide (HNK-111; Figure 1) were synthesized with more than 98% purity (gas chromatography and nuclear magnetic resonance spectroscopy). 11 For AChE estimation, acetylthiocholiniodide (ASChI), 5,5-dithio-bis(2-nitrobenzoic acid) (DTNB), sodium chloride, atropine, and propylene glycol were procured from Sigma Chemicals Co. (St Louis, Missouri, USA). 2-PAM or pralidoxime chloride (ip) was obtained from Kwality Pharma (Amritsar, Punjab, India). All the oximes and nerve agents (tabun and soman) were injected using freshly prepared mixture solution of normal saline and propylene glycol in a ratio of 9:1 (v/v), respectively.
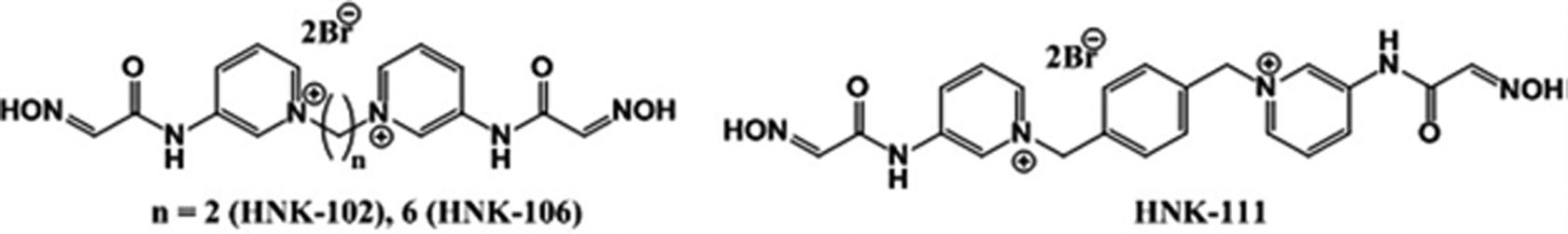
bis-Quaternary 2-(hydroxyimino)-N-(pyridin-3-yl) acetamide derivatives.
Animals
Randomly out bred Swiss albino male mice weighing 25–30 g were obtained from Animal Facility Division, DRDE, Gwalior, Madhya Pradesh, India. After acclimatization and randomization, the mice were housed in polypropylene cages having dust-free steam-autoclaved paddy husk as bedding material. The animals were maintained in environmentally controlled room (25 ± 2°C, relative humidity 40–60%) with 12-h dark/12-h light cycle. All the animals were offered free access to standard pellet feed (Ashirwad Industries, Chandigarh, India) and water ad libitum. Maximum four mice per dose were used for all the experiments. Prior approval for this study was obtained by institutional animal ethics committee, a statutory committee constituted by Committee for Purpose of Control and Supervision of Experiments on Animals, Animal Welfare Cell, Ministry of Environment, Forests and Climate Change, Government of India, Delhi.
Determination of LD50 and PI
LD50 of tabun and soman was determined by injecting subcutaneously (sc) and following method of “moving average.” 12 For LD50 determination, various doses were chosen on logarithm scale, increase or decrease by difference of 0.301 log, with four animals per group. The values are expressed as micrograms per kilogram of body weight.
In order to determine PI, tabun and soman were injected (sc) to the mice at 4, 6, 8, and 10.0× LD50 and 2.0 and 4.0× LD50, respectively. Atropine (10 mg/kg, intraperitoneally (ip)) was given ip as combination therapy with all the four oximes. The treatment doses of HNK-102, HNK-106, and HNK-111 were equal to 0.05, 0.10, and 0.20 of their LD50 (LD50; HNK-102 = 282.8 mg/kg, HNK-106 = 35.35 mg/kg, and HNK-111 = 35.35 mg/kg) and 2-PAM at 30 mg/kg (as standard oxime) 13 were administered intramuscularly (im) within 20–30 s post nerve agent exposure. Volume of all the injections was kept between 0.10 and 0.20 mL. PI of each oxime was determined against tabun and soman poisoning using the following formula:
The animals were observed for body weight and mortality up to 10 days. The higher treatment dose which offered best protection (equal to 0.20 LD50 of the oxime) was selected for in vivo determination of AChE enzyme inhibition/reactivation studies.
Sample preparation for AChE estimation
Animals were anesthetized by Anesthetic ether I.P. (Narsans Pharma, Chittoor, Andhra Pradesh, India) and blood samples were collected in sterilized tubes by puncturing the orbital plexus gently. The collected blood samples were allowed to clot at 37°C. Thereafter, blood samples were further centrifuged for 10 min at 513g for better serum separation. The collected serum was stored at −80°C until use. Parallel to it, the whole brain of anesthetized animals was dissected out quickly and stored at −80°C until use. Serum and processed tissue samples were analyzed for AChE activity using modified Ellman’s method. 14 At the time of assay, the whole brain tissue was thawed, diluted 1:10 (w/v) in 0.25 M sucrose solution, and homogenized using vertical homogenizer (REMI Motors, Mumbai, Maharashtra, India). The homogenates were twice centrifuged (Model 3-18K; Sigma Laborzentrifugen, Germany) at 8500 × g at 4°C for 10 min. The supernatant was decanted and the pellet was diluted in 0.35 M sucrose solution for assay. For AChE estimation, 180 µL phosphate buffer (pH 8.0), 80 µL of DTNB (prepared in phosphate buffer pH 7.0), and 10 µL of test sample (either homogenized brain or serum) were added together in a 96-well plate (Nunc Immuno, Thermo Fisher, Roskilde, Denmark). The reaction mixture was incubated for 3 min at 37°C and then 10 µL of ASChI (prepared in phosphate buffer pH 7.0) was added to this mixture. The blank was taken by adding the phosphate buffer in the place of substrate. The enzyme activity was read in kinetic mode (µQuant™ Microplate spectrophotometer, BioTeK Instruments Inc, Vermont, USA) at 412 nm up to 4 min. AChE activity was expressed as micromoles of ASChI hydrolyzed per minute per gram of brain tissue and micromoles of ASChI hydrolyzed per minute per 10 µL of serum.
Tabun and soman exposed time-dependent peak (maximum) AChE inhibition, in vivo study
Tabun and soman were injected sc at 0.10, 0.20, 0.40, 0.80, and 1.60× LD50 (LD50 = tabun, 282.8 µg/kg, and soman 133.2 µg/kg; sc) followed by atropine (10 mg/kg, ip) administration, to avoid the chances of mortality. Serum and whole brain were collected at 15, 30, 60, 120, 240, and 960 min intervals post nerve agents exposure. AChE estimation was performed by procedure as mentioned earlier. The maximum AChE inhibition in brain by soman was observed at 60 min while for tabun, 30 min obtained as optimized time for peak AChE inhibition. The determined time periods of soman and tabun, 60 min and 30 min, respectively, were used in further AChE enzyme inhibition/reactivation studies.
Tabun and soman IC50 value and IC50 shift for AChE activity (in vivo)
Inhibition concentration 50% (IC50) value was determined using doses 0.10, 0.20, 0.40, 0.80, and 1.60× LD50 of tabun and soman by linear regression analysis for brain and serum AChE at optimized time (30 and 60 min, respectively) in the mice. Using log values (difference of 0.301 log) of IC50, that is, 0.35, 0.70, and 1.40 for tabun and 0.50 and 1.0 for soman, AChE enzyme reactivation was studied at 0.20 LD50 as treatment dose of HNK oximes and 30 mg/kg dose of 2-PAM (im) in mice. The animals of positive control group were injected with tabun or soman and atropine, in the same volume of diluent as used for oxime-treated groups.
Acute toxicity studies of oximes
Toxicity profile of oximes was studied using a sublethal dose of 0.8× LD50 of HNK-102 and 2-PAM im at 24 h postexposure, in the mice. The following variables were determined: 1. Organ body weight index (OBI)
Lung, liver, kidney, brain, spleen, and heart were excised quickly, rinsed in normal saline (ip), gently dried on blotting paper, and weighed to measure OBI.
2. Hematological variables
The blood parameters, RBC, WBC, platelet count, hemoglobin (Hb) and % hematocrit were determined within 2 h of blood collection by automated blood analyzer (Mindray BC3000, Shenzen Mindray Bio-medical Co Ltd, Shenzen, China)
3. Biochemical assay
Biochemical parameters including urea, uric acid, creatinine, total protein, albumin, alkaline phosphatase, bilirubin, glucose, serum glutamic oxaloacetic transaminase (SGOT or aspartate amino transferase (AST), and serum glutamic pyruvic transaminase (SGPT) or alanine amino transferase (ALT) were determined by commercially available diagnostic kits (ERBA Diagnostics Mannhein Gmbh, Mannhein, Germany).
Serum cholesterol was estimated following Zak’s method using spectrophotometer. FeCl3-CH3COOH reagent (working reagent) was prepared at a concentration of 0.05%. Stock solution of standard cholesterol was prepared at a concentration of 0.04 mg/mL in FeCl3-CH3COOH reagent. Test serum samples (30 µL) were added to 3 mL working reagent and centrifuged at the rate of 1500 × g for 10 min. To the collected supernatant, H2SO4 was added in ratio 1:66. The mixed solution was then kept for incubation at 37°C for 20–30 min. The intensity of developed red color was measured at 560 nm.
4. Lipid hydroperoxidase (LPO) assay
As a biomarker of lipid peroxidation, concentration of malondialdehyde (MDA) was measured in liver, brain, and kidney. MDA was estimated by measuring the formation of thiobarbituric acid reactive substances according to Ohkawa et al. 15 Briefly, 100 mg of tissue (liver, brain, and kidney) was homogenized in 1.15% potassium chloride; 100 µL of the tissue homogenate was added to 8.01% sodium dodecyl sulfate (SDS), 20% CH3COOH, and 0.8% thio Barbituric Acid (TBA) (pH 7.0) and was made up to 700 µL by adding ultrapure water. Subsequently, the reaction mixture was then heated at 95°C up to 60 min and centrifuged at 3000 × g for 15 min. The intensity of the developed color was measured at 532 nm and the concentration of MDA was calculated as: MDA (nmol/g of tissue) = OD × 400.
5. Histopathology
The tissues were dissected immediately after killing, freed from adhered tissues, washed with normal saline, and fixed in 10% formalin. After 16–24 h, the tissues were continuously washed overnight under slow running tap water to remove excessive fixative, subjected for automated processing (Tissue Processor, Leica TP 1020, Germany) for dehydration in graded alcohol and xylene treatment, followed by embedding in paraffin blocks. Multiple tissue sections of 4–5 μ thickness were cut with the help of microtome (Microm, 9HM 360, Germany) which were then stained with hematoxylin and eosin, observed and analyzed under light microscope (Leica, DMLB, Microsystems, Wetzlar GmbH, Wetzlar, Hesse, Germany) for changes, if any.
Statistical analysis
Results are expressed as mean ± standard error of the mean. Data were analyzed by one-way analysis of variance followed by Dunnett test and Student’s t-test. Probability of 95% or more (p < 0.05) was considered as a level of significance.
Results
Sign of toxicity and LD50
The animals when treated with 1.0 LD50 dose of soman and tabun showed signs of convulsions, tremors, seizures, and muscles fasciculation. The dose of 1.0 LD50 or higher of soman and tabun caused death of animals within 5 min postexposure. LD50 of soman and tabun was found to be 133.4 ± 9.84 and 282.8 ± 0.00 µg/kg (sc), respectively (Tables 1 and 2). Determination of LD50 of soman repeated five times due to variation in mortality pattern, however, twice with tabun.
PI of oximes (2-PAM, HNK-102, HNK-106, and HNK-111) in combination with atropine against soman poisoning in Swiss albino mice.

PI: protection index; SEM: standard error of the mean; 2-PAM: pralidoxime; sc: subcutaneously; LD50: median lethal dose; ip: intraperitoneally; im: intramuscularly.
aLD50 is determined following the method of “moving average.” 12 The treatment doses of HNK-102, HNK-106, and HNK-111 correspond to their 0.20 LD50 (LD50: HNK-102 = 282.8 mg/kg, HNK-106 = 35.35 mg/kg, HNK-111 = 35.35 mg/kg, im). PI = LD50 of soman with treatment/LD50 of soman. Total number of animals used 108. All the injectable(s) prepared in freshly made diluent (9:1 ratio of normal saline Indian Pharmacopoeia (IP) and propylene glycol, v/v).
Protection offered by oximes (2-PAM, HNK-102, HNK-106, and HNK-111) in combination with atropine against tabun poisoning in Swiss albino mice.
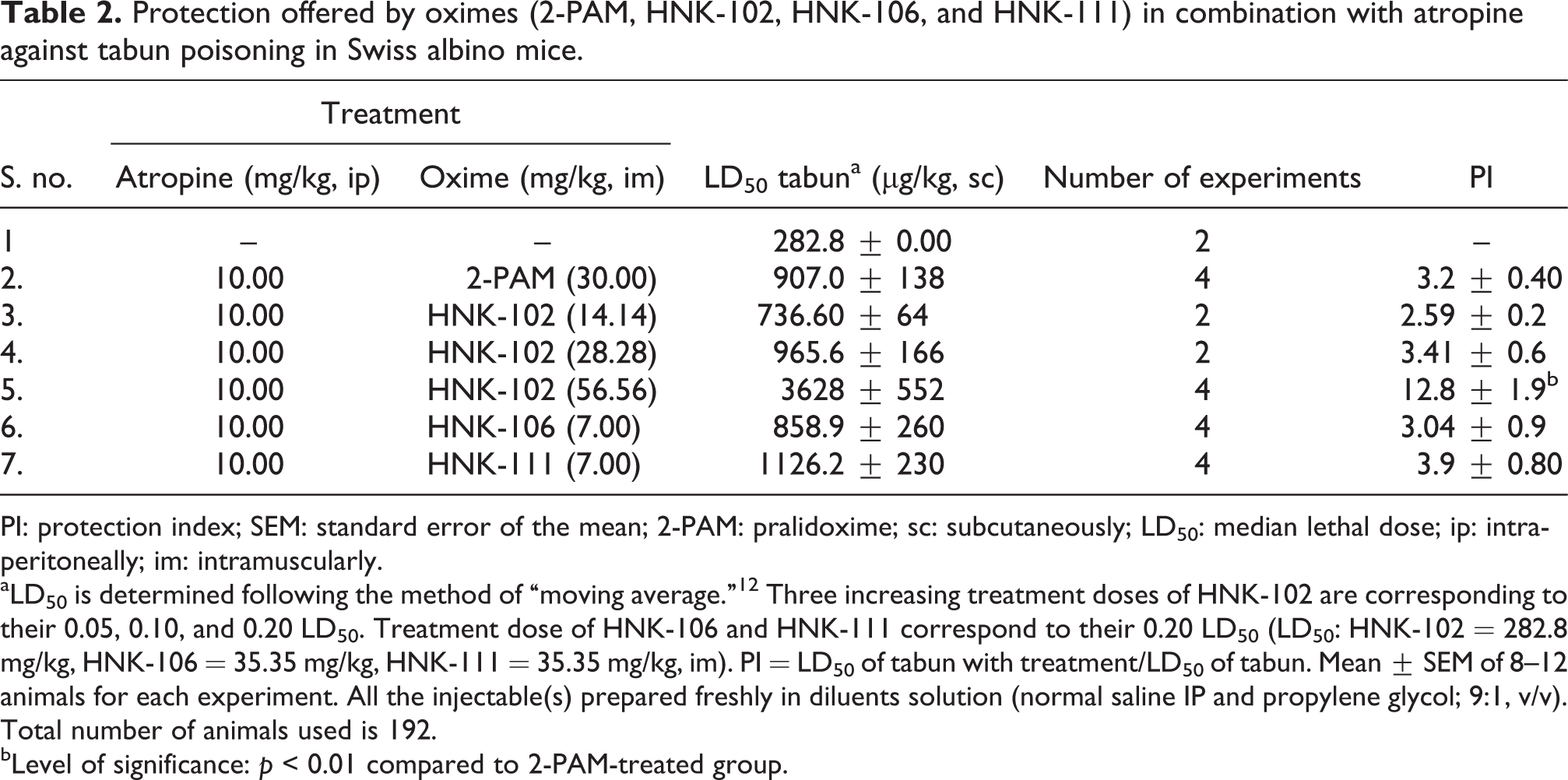
PI: protection index; SEM: standard error of the mean; 2-PAM: pralidoxime; sc: subcutaneously; LD50: median lethal dose; ip: intraperitoneally; im: intramuscularly.
aLD50 is determined following the method of “moving average.” 12 Three increasing treatment doses of HNK-102 are corresponding to their 0.05, 0.10, and 0.20 LD50. Treatment dose of HNK-106 and HNK-111 correspond to their 0.20 LD50 (LD50: HNK-102 = 282.8 mg/kg, HNK-106 = 35.35 mg/kg, HNK-111 = 35.35 mg/kg, im). PI = LD50 of tabun with treatment/LD50 of tabun. Mean ± SEM of 8–12 animals for each experiment. All the injectable(s) prepared freshly in diluents solution (normal saline IP and propylene glycol; 9:1, v/v). Total number of animals used is 192.
bLevel of significance: p < 0.01 compared to 2-PAM-treated group.
Protection index
For protection index (PI) determination of oximes, animals were treated with multiples of lethal doses of soman or tabun followed by administration of atropine and oximes within 10–15 s. Treatment with 2-PAM, HNK-102, HNK-106, and HNK-111 oximes showed PI of 1.8, 1.8, 1.8, 2.1, and 1.6, respectively against soman poisoning (Table 1 and Figure 2). And against tabun poisoning, PI of 3.2, 12.82, 3.04, and 3.98 were noted (Table 2 and Figure 2). HNK-102 also showed dose-related enhancement of protection, up to fourfold more compared to 2-PAM, against tabun poisoning (Table 2 and Figure 2). Animals exposed to multiples of lethal doses of tabun and saved by the treatments with oximes, showed a decrease in the body weight on day 1 postexposure. Second day onward, increase in body weight was noted in all the groups those poisoned and treated with 2-PAM and HNK oximes. Notably, marginal to significant increase in body weight was more pronounced with HNK-102, even at the massive dose (3200 µg/kg) of tabun (Table 3).

Protection offered in terms of PI by HNK-102, HNK-106, and HNK-111 at 0.05, 0.10, and 0.20 of their respective LD50 as treatments. PI = LD50 of soman or tabun with treatment ÷ LD50 of soman or tabun. LD50: HNK-102 = 282.8 mg/kg, HNK-106 = 35.35 mg/kg, and HNK-111 = 35.00 mg/kg. Values are mean ± SEM (n = 4). *Level of significance p < 0.01 compared to 2-PAM. PI: protection index; SEM: standard error of the mean; 2-PAM: pralidoxime; sc: subcutaneously; LD50: median lethal dose.
Percent body weight of control and all the groups of Swiss albino mice those poisoned and treated with atropine (10 mg/kg, ip) and 2-PAM or HNK series oximes.

PI: protection index; SEM: standard error of the mean; 2-PAM: pralidoxime; sc: subcutaneously; LD50: median lethal dose; ip: intraperitoneally; im: intramuscularly.
aLD50 of tabun is 282.8 µg/kg, sc. Initial body weight is 24.7 ± 1.23 (mean ± SEM of 66 animals). n = number of animals survived post tabun poisoning followed by the treatment.
b0.2× LD50 of HNK series oximes used 6 as treatment dose against tabun poisoning.
c p < 0.05 compared to control group.
Soman- and tabun-induced AChE inhibition in brain and serum, in vivo
At various doses of soman and tabun, that is, 0.1, 0.2, 0.4, 0.8, and 1.6 LD50, almost a linear trend in brain AChE inhibition was observed (Figures 3 and 4). At 60 min post soman exposure, 72% AChE inhibition was observed and at 30 min ca. 48% brain AChE inhibition was observed against tabun exposure (Figures 5 and 6). The maximum inhibition of brain AChE for soman at 60 min and for tabun at 30 min was noted, considered as optimized time for IC50 determination. Likewise, soman- and tabun-induced inhibition of serum AChE using different doses and time points were determined. In dose–time response study, nonlinear inhibition pattern of serum AChE was observed post soman and tabun exposure (Figures 5 and 6).

Effect of soman (LD50 = 133.4 µg/kg, sc) on brain and serum acetylcholinesterase activity using 0.1, 0.2, 0.4, 0.8, and 1.6 LD50 (sc) at 15 min postexposure. Each bar represents mean ± SEM of four experiments. SEM: standard error of the mean; sc: subcutaneously; LD50: median lethal dose.

Effect of tabun (LD50 = 282.8 µg/kg, sc) on brain and serum acetylcholinesterase activity using 0.1, 0.2, 0.4, 0.8, and 1.6 LD50 (sc) doses at 15 min postexposure. Each bar represents mean ± SEM of four experiments. SEM: standard error of the mean; sc: subcutaneously; LD50: median lethal dose.

Effect of soman (LD50 = 133.4 µg/kg, sc) on brain and serum acetylcholinesterase activity at various time points using 0.71 LD50 dose. Each point represents mean ± SEM of four experiments. SEM: standard error of the mean; sc: subcutaneously; LD50: median lethal dose.

Effect of tabun (LD50 = 282.8 µg/kg, sc) on brain and serum acetylcholinesterase activity at various time points using 0.8 LD50 dose. Each point represents mean ± SEM of four experiments. SEM: standard error of the mean; sc: subcutaneously; LD50: median lethal dose.
IC50, in vivo
For determination of IC50, dose-dependent AChE inhibition at 60 and 30 min (optimized time) post soman and tabun exposure was performed (Figures 7 and 8). IC50 value for brain AChE was calculated as 112 µg/kg, corresponding to 0.84 LD50 of soman. Similarly, for serum AChE, IC50 calculated as 128 µg/kg, corresponding to 0.96 LD50 of soman. IC50 value for brain AChE calculated as 192.8 µg/kg, corresponding to 0.69 LD50 and for serum 129 µg/kg, corresponded to 0.46 LD50 of tabun.

Effect of soman (LD50 = 133.4 µg/kg, sc) on brain and serum acetylcholinesterase activity at various doses, 60 min postexposure. Each point represents mean ± SEM of four experiments. SEM: standard error of the mean; sc: subcutaneously; LD50: median lethal dose.

Effect of tabun (LD50 = 282.8 µg/kg, sc) on brain and serum acetylcholinesterase activity at various doses, 30 min postexposure. Each point represents mean ± SEM of four experiments. SEM: standard error of the mean; sc: subcutaneously; LD50: median lethal dose.
AChE reactivation, in vivo
Reactivation of phosphorylated brain AChE by 2-PAM and HNK oximes was estimated at the optimized time, 60- and 30-min post soman and tabun exposure, respectively, in vivo. At 0.50 IC50 dose of soman (56 µg/kg), significant reactivation of brain AChE enzyme, 74% and 80%, was observed with the treatment of HNK-102 and HNK-106 (p < 0.001), respectively. At LC50 dose of soman (112 µg/kg), the reactivation by all the oximes was not more than 20% (Figure 9). Approximately, 69% and 6% (p < 0.01) brain AChE reactivation was shown by HNK-102 at 197.9 µg/kg (IC50) and 395.8 µg/kg (2× IC50) doses of tabun, respectively (Figure 10).

Reactivation of brain and serum acetylcholinesterase activity by oximes at 60 min post soman exposure. Doses: soman, 56 µg/kg (0.50 IC50) and 112 µg/kg (equal to IC50), sc; Oximes: ×0.20 of HNK-102, HNK-106, and HNK-102 of their LD50 as treatment dose; 2-PAM 30 mg/kg, im. Each bar represents mean ± SEM of four experiments. * and #Level of significance p < 0.001 compared to soman. SEM: standard error of the mean; sc: subcutaneously; LD50: median lethal dose; IC50: inhibition concentration 50%; im: intramuscularly; 2-PAM: pralidoxime.

Reactivation of brain and serum acetylcholinesterase by oximes at 30 min post tabun exposure. Doses: tabun, 98.9 µg/kg, 197.9 µg/kg, and 395.9 µg/kg (sc); Oximes: HNK-102, HNK-106, and HNK-111, ×0.20 of their LD50 as treatment; 2-PAM, 30 mg/kg (im). Each bar represents mean ± SEM of four experiments. *Level of significance p < 0.001 compared to tabun. SEM: standard error of the mean; sc: subcutaneously; LD50: median lethal dose; IC50: inhibition concentration 50%; im: intramuscularly; 2-PAM: pralidoxime.
Serum AChE reactivation, in vivo
Equivalently, serum AChE activity (%) was determined at log IC50 doses of soman and tabun with 2-PAM and HNK analogs. HNK analogs did not show AChE reactivation at all the used doses of soman and tabun. 2-PAM at 198 µg/kg dose of tabun showed significant (ca.70%; p < 0.001) reactivation of AChE in comparison to HNK-102, in vivo (Figures 9 and 10).
Acute toxicity of oximes, in vivo
For comparison and firsthand information, acute toxicity studies were carried out by administering 0.8× LD50 (im) of 2-PAM and HNK-102 in the mice. Biochemical, hematological, and histopathological variables and OBI were determined. No significant changes were observed in OBI, compared to control group (Table 4). Examination of the hematological parameters showed increase in total erythrocyte count (TEC), Hb, hematocrit, and decrease in platelet counts (Table 5).
Acute toxicity of 2-PAM and HNK-102 oximes (0.8 LD50) on OBI of Swiss albino mice, 24 h postexposure.a
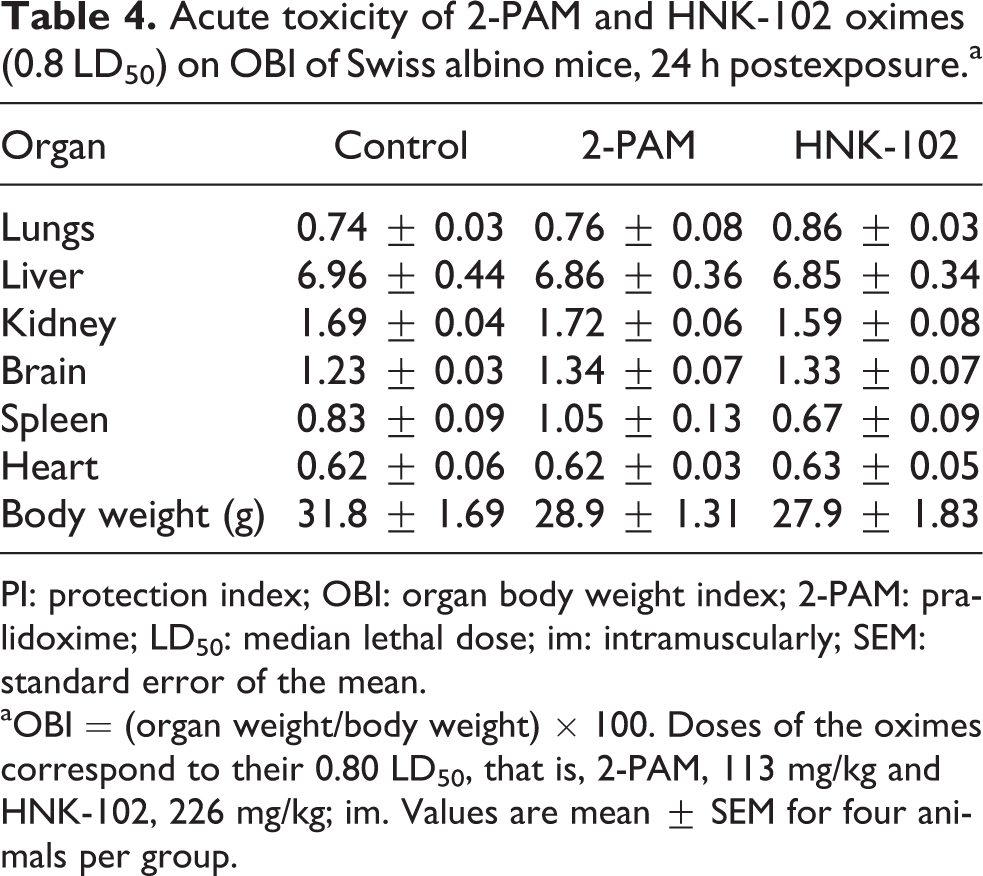
PI: protection index; OBI: organ body weight index; 2-PAM: pralidoxime; LD50: median lethal dose; im: intramuscularly; SEM: standard error of the mean.
aOBI = (organ weight/body weight) × 100. Doses of the oximes correspond to their 0.80 LD50, that is, 2-PAM, 113 mg/kg and HNK-102, 226 mg/kg; im. Values are mean ± SEM for four animals per group.
Acute toxicity of 2-PAM and HNK-102 oximes (0.8 LD50) on hematological profile of Swiss albino mice, 24 h postexposure.a

2-PAM: pralidoxime; LD50: median lethal dose; im: intramuscularly; Hb: hemoglobin; RBC: red blood cell; WBC: white blood cell; SEM: standard error of the mean.
aDoses of the oximes correspond to their 0.80 LD50, that is, 2-PAM, 113 mg/kg and HNK-102, 226 mg/kg; im. Values are mean ± SEM for four animals per group.
bLevel of significance: p < 0.05 compared to control.
cLevel of significance: p < 0.01 compared to 2-PAM.
Among various biochemical variables studies, HNK-102 group showed increase in serum urea and SGOT (p < 0.05 and p < 0.01) compared to 2-PAM and control groups. 2-PAM induced increase in total protein (p < 0.05) and decrease in SGOT (p < 0.05). Both the oximes induced decrease in uric acid and creatinine (p < 0.05) compared to control group (Table 6).
Acute toxicity of 2-PAM and HNK-102 oximes (0.8 LD50) on various biochemical variables in Swiss male mice, 24 h postexposure.a

SGOT: serum glutamic oxaloacetic transaminase; SGPT: serum glutamic pyruvic transaminase; ALP: alkaline phosphatase; 2-PAM: pralidoxime; LD50: median lethal dose; im: intramuscularly; SEM: standard error of the mean.
aDoses of the oximes correspond to their 0.80 LD50, that is, 2-PAM, 113 mg/kg and HNK-102, 226 mg/kg; im. Values are mean ± SEM for four animals per group.
bLevel of significance: p < 0.05 compared to control.
cLevel of significance: p < 0.01 compared to 2-PAM.
The effect of 2-PAM and HNK-102 oximes at sublethal dose (0.8× LD50) on LPO in liver, kidney, and brain are depicted in Table 6. HNK-102 decreased (p < 0.05) the values of LPO in all three organs (liver, brain, and kidney) in comparison to control. However, a significant decrease in LPO values of brain was observed in 2-PAM-treated mice, compared to control group (Table 7).
Acute toxicity of 2-PAM and HNK-102 oximes (0.8 LD50) on LPO,a in liver, brain, and kidney of Swiss albino mice, 24 h postexposure.b

LPO: lipid peroxidation; 2-PAM: pralidoxime; LD50: median lethal dose; im: intramuscularly; SEM: standard error of the mean.
aOhkawa et al. 15
bDoses of the oximes correspond to their 0.80 LD50, that is, 2-PAM, 113 mg/kg and HNK-102, 226 mg/kg; im. Values are in mean ± SEM for four animals per group.
cLevel of significance: p < 0.05 compared to control.
dLevel of significance: p < 0.01 compared to control and 2-PAM.
Histopathological examination of liver, brain, kidney, and heart of both the treated and control groups showed normal tissue architecture and vasculocellular appearance at sublethal dose post 24 h treatment of both oximes (Figure 11).

Photomicrographs of control, 2-PAM-, and HNK-102-exposed (0.80 LD50, 24 h postexposure) mice brain, heart, liver, kidney, and lung (H&E). (a) Control mice brain: 2-PAM- and HNK-102-treated brain showing normal histoarchitecture of cerebrocortical cells. (b) Control mice heart: 2-PAM- and HNK-102-treated mice heart showing normal cardiac muscles and myocardium. (c) Control mice kidney: 2-PAM- and HNK-102-treated mice kidney showing normal glomerulus, Bowman’s space, and renal parenchyma. (d) Control mice liver: 2-PAM- and HNK-102-treated mice liver showing normal hepatic cord pattern of lobules, hepatocytes, central canal, and bile duct. (e) Control mice lung: 2-PAM- and HNK-102-treated mice lung showing homogeneous parenchyma with normal airways, air sacs, alveolar septa, and bronchiole. SEM: standard error of the mean; LD50: median lethal dose; 2-PAM: pralidoxime; H&E: hematoxylin and eosin.
Discussion
Since decades, many efforts are being made to develop broad-spectrum antidote against toxic OPC and nerve agent poisoning. Although currently used oximes (2-PAM, obidoxime, trimedoxime, HI-6) are potent antidotes against majority of structurally different OPC, however, unequal therapeutic efficacy, varying AChE reactivation ability and certain stability issues necessitates search of novel and more efficacious oxime.
The new bis-pyridinium acetamide derivatives (HNK series oximes) have been found to be more efficacious than presently used antidote 2-PAM against (DFP, 6 sarin 7 and dichlorvos (unpublished data) poisoning. In the present study, therapeutic efficacy as antidote, HNK oximes were examined and compared, against acute poisoning of soman and tabun in Swiss albino mice. Treatment with HNK-102 shifted LD50 of tabun by 12.80 times upward or in other words, PI was found to be 12.80. Parallel to it, 2-PAM could show PI of 3.20. Thus, HNK-102 was found to be four times better antidote against acute poisoning of tabun in the mice. Previous studies 16 have shown trimedoxime as a replacing antidote against tabun poisoning with PI of 1.71 in mice. The lower PI by the available oximes against tabun poisoning, explained by the fact that the existence of lone pair on dimethylamide group of tabun, makes impossible for oximes to undergo nucleophilic attack for desired dephosphorylation. 17 However, our findings are not in agreement with earlier proposed explanation as HNK-102 has shown significantly high PI against tabun poisoning in Swiss mice. This is attributed to fulfilling two of the structural requirements, oxime group on position 3 in the pyridinium group and connecting linker between the pyridinium rings, present in HNK-102 oxime.
Following the acute tabun poisoning and treatment with oximes (in combination with atropine), body weight changes was recorded up to 10 days. A dose-dependent decrease in the body weight was noted up to 48 h post tabun exposure, recovered with progression of days. This observation grossly indicated that all the four used oximes (2-PAM, HNK-102, HNK-106, and HNK-111) are equally safer when used against the nerve agents. Among all the oximes including 2-PAM, HNK-102 showed higher regain in body weight even at 12-fold higher lethal doses of tabun. This may be an indication of apparent lesser intrinsic toxicity or/and greater efficacy of HNK-102 oxime against tabun poisoning.
All the four oximes with atropine fairly protected intoxicated mice against acute soman poisoning up to PI of 1.80–2.18. HI-6, having the lowest toxicity, has been shown higher therapeutic potency with protection ratio 1.74 18 against soman poisoning, compared to available oximes, that is, 2-PAM, obidoxime, trimedoxime, and K series. But its poor solubility and instability in aqueous media limits its use as an emergency first aid antidote. In the present study, HNK-102 was found to be four times and marginally better compared to 2-PAM and HI-6 as antidote against acute poisoning of tabun and soman, respectively.
Selection of treatment dose of newly synthesized molecules of HNK oximes was based on their LD50, that is, the maximum treatment dose was always kept × 0.20 of their LD50, keeping in consideration that therapeutic index should not be less than 5. HNK-102 followed the dose–response relationship as antidote against acute poisoning of tabun, where, 0.05, 0.01, and 0.20× LD50 were used as treatment doses and PI were 2.59, 3.41, and 12.80, respectively.
Previously reported in vivo study of HNK series oximes demonstrated that HNK-102 significantly reactivates DFP and sarin intoxicated brain AChE activity at log doses of IC50. 6,7 Similarly, the reactivation study of HNK series oximes in comparison to existing antidote 2-PAM was performed against soman and tabun poisoning in vivo. Soman at LD50 caused 50% inhibition of AChE activity in brain and serum at optimized time, 60 min postexposure. Dissimilar to soman, tabun caused 50% inhibition of brain AChE activity at 0.68× LD50 dose and in serum at 0.45× LD50. These findings indicated that there is no dependable correlation between lethality and inhibition of AChE activity. 6,7 Reason could be related to the structural difference (responsible for difference in toxicity) in the two nerve agents. Soman consists of pinacolyl group whereas tabun consists of ethyl cyanophosphate group. The rate of spontaneous reactivation of AChE is also variable, which partly accounts for differences in acute toxicity between the nerve agents. 19,20
To evaluate the AChE reactivation efficacy of the oximes at optimized time for maximum AChE inhibition (60 min for soman and 30 min for tabun), percent AChE activity was estimated at log doses of IC50. With soman at 56 µg/kg (ca. 0.4× LD50), corresponding to 0.5 IC50 dose, reactivation obtained by HNK-102 and HNK-106 was 73.6% and 79.6% of AChE activity, 1.5-fold higher compared to 2-PAM. Similarly, at IC50 dose corresponding to 112 µg/kg (0.84× LD50) of soman, ca. 1.5 times higher AChE reactivation was shown by HNK-102 in comparison to 2-PAM. Contrary to these results, no significant reactivation of AChE activity was shown by any of the oximes (2-PAM, HNK-102, HNK016, and HNK-111) in serum against soman poisoning.
HNK-102 showed 69.09% AChE activity which was 1.4 times higher than 2-PAM against tabun at IC50 dose (193 µg/kg equal to 0.7× LD50). Likewise, at 2× IC50 dose of tabun (1.4× LD50), ca. 2.2 times higher (6.28%) AChE reactivation was shown by HNK-102 as compared to 2-PAM. These findings attributed to the physicochemical properties of HNK-102. Earlier it was reported that HNK series oximes exhibit lower dissociation constant (K D value) and pK a value within range 7.95–8.20. 11 Additionally, the unique structure of HNK series oximes containing the oxime (=NOH) group in conjugation to amide may assist for dissociation of the oxime group into more nucleophilic oximate anion. These structural differences make HNK-102 to show higher reactivation in comparison to 2-PAM against OPC including nerve agent poisoning. Although the reactivation of brain AChE by HNK-102 at IC50 dose was equal or more than 1.5 times higher than 2-PAM against soman and tabun poisoning, total percentage of AChE reactivation was very low (ca. 20%) in soman in contrast to tabun (ca. 70%) poisoning. This is explainable in light of “aging” involving monodealkylation of the dialkylphosphonyl enzyme (intoxicated or inhibited AChE enzyme), which is quite important in soman poisoning. 21 As reported elsewhere, aging within soman exposure occurs within 2 min while at acute tabun poisoning, it takes longer than 40 h for AChE enzyme to completely get inhibited. It may be stated that differences in functional groups are responsible for differences in acute toxicity, also accounts for AChE enzyme inhibition and its reactivation by HNK oximes in soman and tabun nerve agents poisoning.
It was felt pertinent to undertake acute toxicity studies of newly synthesized HNK-102 and compare with established 2-PAM oxime. Sublethal doses (0.8× LD50; HNK-102, 262.24 mg/kg; and 2-PAM, 113.1 mg/kg) of both the oximes were injected im in Swiss mice and were killed at 24 h postexposure. No significant changes were observed in OBI. A marginal variation was observed in spleen to body weight ratio with both the oximes compared to control group. Single administration of the oximes caused alterations in hematological variables. A significant increase was observed in TEC, Hb, and hematocrit in HNK-102-exposed animals in comparison to control group. At sublethal dose, moderate side effects such as hyperventilation, muscular weakness, and tachycardia were also observed in 2-PAM- and HNK-102-treated groups. Increased RBC has been reported in partial asphyxia, after administration of certain drugs. 22 Using the high dose of oxime (0.8× LD50) might have caused increase in TEC, Hb, and hematocrit values. Obviously, increase in Hb level was due to increase in TEC. 2-PAM injected im at dose of 30 mg/kg, caused slight changes in blood pressure and ECG. However, no conclusion can be drawn from these observations as to whether the toxicity is due to the oxime or any other by-product formation in the treated subjects. 9
There were mild changes observed in liver- and kidney-related biochemical variables. SGOT (AST and SGPT (ALT are common liver enzymes for the evaluation of liver function. 23,24 SGOT was increased significantly in HNK-102-treated animals in comparison to control. SGPT (ALT) is considered as liver-specific enzyme and used as a marker of hepatocellular necrosis or increased cell membrane permeability. 25 AST (SGOT) is not a specific enzyme but occurs in most cells; however, it is useful in evaluating hepatocellular and muscular injury because of its high activity in these tissues. The level of SGPT was not found to be changed by both the oximes compared to control group. Alike control group, increased level of urea, decreased level of uric acid and creatinine was observed by HNK-102; however, no significant different was found between HNK-102- and 2-PAM-treated groups. As reported earlier, elevated levels of blood urea are associated with marked damage to functional nephrons. 26 Excelling over all the tested oximes, HNK-102 was found least toxic (i) in human cell lines (up to 1 mg/mL dose) (ii) via im, ip, and intravenous routes of administration in Swiss male mice compared to 2-PAM. 8 Histopathological findings did not show change(s) in histoarchitecture of liver, kidney, brain, and lung, ruled out hepatocellular toxicity, nephrotoxicity, cerebrocortical cells degeneration, or airways obstruction compared to control group. The findings related to kidney function need further investigation following long-term toxicity studies.
A hallmark of oxidative stress is LPO which disrupts the structural integrity of cell membrane and can lead to formation of aldehydes which in turn lead to lipid, protein, and DNA damage. 27 MDA is the byproduct of LPO of mainly phospholipid of cell membrane, where its level is likely to be increased in oxidative stress. MDA was measured as oxidative markers in vital organs such as liver, brain, and kidney in the present study, where HNK-102 decreased the LPO levels (p < 0.05) in all the three organs as compared to control, whereas 2-PAM could decrease LPO in brain only. This was quite interesting to note that HNK-102 also showed protective efficacy against LPO in the three vital organs. This effect of HNK-102, corroborated to some structural specificity for scavenging lipid peroxides, might have contributed to additional beneficial effect. An extensive study is further needed to establish its antioxidative efficacy, if any.
In conclusion, HNK-102 was found to be more efficacious as antidote against soman and tabun poisoning compared to presently used 2-PAM in vivo.
Conclusion
HNK series of oximes showed better potency in Swiss mice with (i) fourfold better PI (in terms of survival) shown by HNK-102, in comparison to 2-PAM against tabun poisoning and (ii) brain AChE reactivation by 1.5 times at IC50 dose of soman and tabun poisoning, compared to 2-PAM in Swiss male mice.
Footnotes
Acknowledgements
The authors thank Dr Lokendra Singh, the director, Defence Research and Development Establishment, Jhansi Road, Gwalior, India for his encouragement and providing necessary facilities.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


