Abstract
Arsenic (As) is commonly associated with natural and human processes such as volcanic emissions, mining and herbicides production, being an important pollutant. Several studies have associated As intake with male fertility reduction, thus the aim of the present study was to evaluate whether vitamin C and/or zinc would counteract As side effects within the testicles. Adult male Wistar rats were divided into six experimental groups: control, sodium arsenite (5 mg/kg/day), vitamin C (100 mg/kg/day), zinc chloride (ZnCl2; 20 mg/kg/day), sodium arsenite + vitamin C and sodium arsenite + ZnCl2. Testicles and epididymis were harvested and either frozen or routinely processed to be embedded in glycol methacrylate resin. As reduced the seminiferous epithelium and tubules diameter due to germ cell loss. In addition, both the round spermatids population and the daily sperm production were reduced. However, ZnCl2 and vitamin C showed to be effective against such side effects, mainly regarding to sperm morphology. Long-term As intake increased the proportions of abnormal sperm, whereas the concomitant intake of As with zinc or vitamin C enhanced the proportions of normal sperm, showing that such compounds could be used to protect this cell type against morphological defects.
Keywords
Introduction
Arsenic (As) is a natural element, widely distributed over the soil, air and water. The free As is commonly associated with a variety of natural processes, as weather, volcanic emissions as well as a result of human activities such as mining, metal casting and herbicides and fossil fuel production. Such human processes deteriorate soil and water contaminations, affecting millions of people worldwide. 1,2
As is found in different oxidation states, being the most reactive, the pentavalent As (V) or arsenate, and the trivalent As (III) or arsenite. 3 The arsenate shows a relevant toxicity capacity due to its physical–chemical similarity to the phosphate, replacing it in cell respiratory mechanisms. On the other hand, the arsenite is considered the most toxic form due to its higher reactivity with sulphur-based compounds, as thiol, sulphydryl and cysteine radicals. 4 In the intracellular space, the arsenite becomes a complex with the glutathione (GSH), leading to a dramatic decrease in the GSH content. In addition, the redox potential increases at the same magnitude, leading to a mass formation of reactive oxygen species (ROS). 5 The oxidative stress takes place when there is an imbalance between ROS production and removal, which may lead to the oxidation of organic substrates. 6
The male reproductive system requires the right amounts of ROS to provide the sperm with the capacity of fertilizing the oocyte; therefore, the excess of ROS induces male infertility. 7 During spermatogenesis, severe Deoxyribonucleic acid (DNA) damage in germ cells is associated with high ROS levels, leading to higher apoptotic rates, whereas lower oxidative stress levels can induce compensatory mechanisms to promote their survival. 8
An interesting strategy can enhance the cell antioxidant capacity against some heavy metals, such as As: the supplementation with exogenous antioxidant substances. 9 Despite several studies confirmed that there is increased oxidative stress due to As consumption, the use of antioxidants both isolated or in association with a given chelation therapy is still poorly investigated. 10 Among the antioxidants are the enzymatic superoxide dismutase, catalase and GSH reductase, and the non-enzymatic zinc, selenium, vitamins C and E, lycopene and bioflavonoids. 11
Zinc, in normal biological concentrations, is essential to several biological processes, including genetic expression, DNA synthesis, enzymatic catalysis, neurotransmission, apoptosis 12 and germ cell development. 13 Zinc is a dismutase superoxide cofactor, showing the chelating ability, thus stabilizing cell membranes, inhibiting lipid peroxidation and inducing metallothionein synthesis. 14
Other antioxidant, vitamin C, plays an important role in metabolic and physiological processes as the vitamin B activation, the conversion of cholesterol in bile acids and the conversion of tryptophan into the neurotransmitter serotonin. 15 It is considered to be the most important hydro soluble antioxidant, since it is capable of neutralize ROS in the aqueous phase prior to the lipid peroxidation. 16 In the seminal plasma, vitamin C protects the sperm against lipid peroxidation, thus preventing DNA damage. 17
Therefore, due to the current environmental pollution by heavy metals and considering the toxicity of As and its role in oxidative stress, this study investigated the antioxidant capacity of vitamin C and zinc in the tubular morphology and in seminal parameters of adult rats exposed to chronic sodium arsenite.
Methods
Animals
Adult Wistar rats (80 days old) were purchased from the Centro de Ciências da Saúde/Universidade Federal do Espírito Santo Biotery and treated daily for 60 days. Commercial chow and water were provided ad libitum and the animals kept under standard conditions: 25 ± 1°C and 12 h light:12 h dark hours. The experimental design was approved by the University Ethics Committee (protocol no. 041/2014) and was in agreement with the National Institutes of Health guide for the care and use of laboratory animals.
The animals were randomly divided into six experimental groups (n = 6, each) as follows: control (distilled water), sodium arsenite (5 mg/kg/day), vitamin C (100 mg/kg/day), zinc chloride (ZnCl2; 20 mg/kg/day), sodium arsenite + vitamin C and sodium arsenite + ZnCl2. Each animal received 0.5 mL of the treatment daily by gavage.
The sodium arsenite dosage (Vetec, lot: 1000282) is lower than the Lethal Dose 50 (LD50), which is 20 mg/kg for rats. 18 The ZnCl2 and vitamin C were purchased and kept refrigerated in the dark (4°C). Their dosages were applied the following previous protocols. 19 –22
Biological samples
Twenty-four hours after the last gavage, the animals were weighed and anesthetized with xylazin and ketamine (5 and 80 mg/kg, respectively, intramuscular). Thus, the testes and epididymis were harvested and weighed after euthanasia.
The right testes were immersed in Karnovsky fixative 23 for 24 h (4°C). Testicular fragments were dehydrated in ethanol and routinely embedded in glycol methacrylate resin (Leica Historesin, Leica Microsystems Nussloch, Germany). The histological sections were made in a rotary microtome (Leica; 3-µm thick) and stained with haematoxylin and eosin. The left testes and epididymis were frozen (−20°C) for the calculation of daily sperm production (DSP) and the number of sperm within the epididymis and sperm transit, respectively.
Body weight and gonadosomatic index
The body weight gain was recorded, as well as the testicular relative weight, or gonadosomatic index (GSI). The GSI (%) was calculated dividing the organ weight by the body weight and multiplying the result by 100.
Morphometry and stereology
The morphometrical and stereological analyses were made using digital images of the testicular parenchyma taken with a digital camera coupled to the light microscope (Leica Microsystem DM500). The proportions between parenchyma elements (seminiferous tubules and interstitium) were calculated by counting the intersections of a square grid (121 intersections) placed on the digital image. The volumetric proportions (%) between seminiferous tubules (tunica propria, seminiferous epithelium and lumen) and interstitium were estimated after counting 2040 intersections (per animal) from random fields (400× magnification). The volume of each component (mL) was estimated from the previous knowledge of the proportions occupied by each of them within the testicle. The testicular weight was considered equal to its volume, discounting the albuginea weight. 24 –27
For the morphometric analyses, the software Image Pro Plus (MediaCybernetics) was used the following previous protocols. 28,29 The seminiferous tubules diameter and the epithelium height from each animal were calculated after measuring 30 tubules cross sections. The total length of the seminiferous tubules was estimated from the volume they occupy within the parenchyma and the tubular diameter, as described by Attal and Courot 30 and Dorst and Sajonski. 31
Cell counts and indexes
Eight seminiferous tubules cross sections in stage I of the seminiferous epithelium cycle, 32 per animal, were used in order to provide the number of germ and Sertoli cells. All type-A spermatogonia (A), pre-leptotene/leptotene (PL-L) and pachytene (P) spermatocytes and round spermatids (RS). The results were corrected regarding the nuclear/nucleolar diameter and the section thickness as described by Amann and Almquist. 33
The following indexes were calculated from the germ cell counting: the coefficient of spermatogonial mitosis efficiency (PL/A) and the meiotic index (RS/P) that quantifies the efficiency of meiotic divisions and the overall spermatogenesis yield (RS/A), thus demonstrating the efficiency of this process as a whole.
DSP, sperm reserve and sperm transit time
Elongated spermatids, stage 19, 34 within the testis and sperm cells from the epididymis were counted using Neubauer chamber following the protocol described by Robb et al. 35 Thus, the DSP was calculated dividing the sperm number by 6.1, which is the number of days in which the mature spermatids are still attached to the epithelium. Finally, to evaluate the sperm transit time within the head/body and cauda epididymis, the sperm numbers in each fragment were divided by the DSP. 36
Sperm morphology
One centimetre of the initial part of the deferent duct (portion attached to the cauda epididymis) was removed, twisted with the tweezers and the contents immersed in phosphate buffer (0.1 mol/L and pH is 7.2) for 15 min. The free sperm within the buffer were placed in Neubauer chamber for morphological analyses. 37 The sperm (200 per animal) were observed under light microscopy (400× magnification) and classified in two categories: normal and abnormal. 37 –39 The abnormal sperm were divided into categories as follows: normal head apart from the tail, hook in different angles, coiled tail and abnormal head and normal tail. 40
Statistical analysis
The Shapiro–Wilk (BioEstat 5.0) test was used to evaluate normality. Data normally distributed were tested with ANOVA followed by Tukey’s test (Past 3.08). The Kruskal–Wallis test was applied to the data that did not follow normal distribution. The significance level was 5%. Results are shown as mean : standard deviation of the mean (SD).
Results
Body weight gain and GSI
There were no differences in the body weight gain or in the GSI after the treatments (Figure 1).
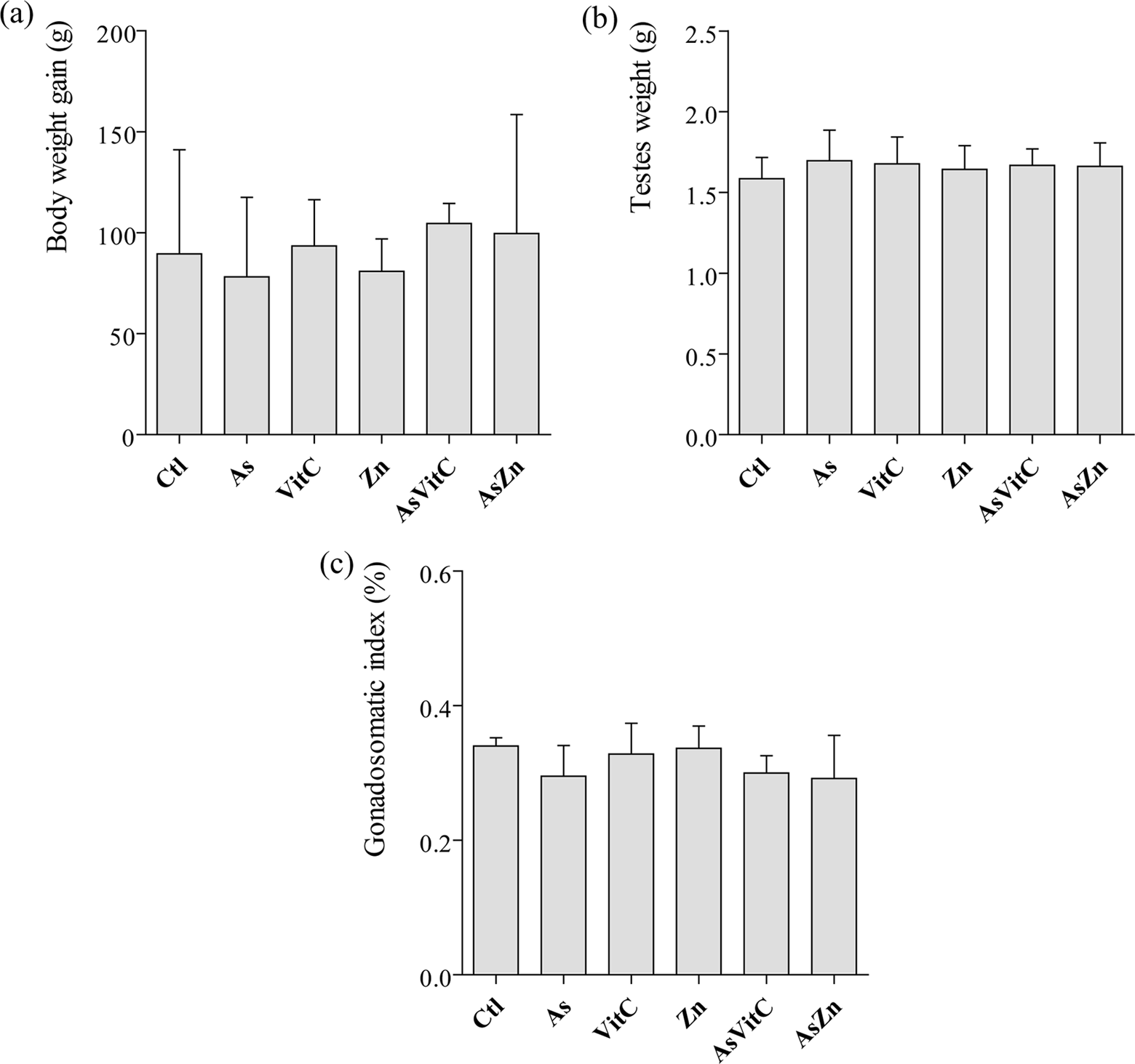
(a) Body weight gain (g). (b) Testes weight (g). (c) Gonadosomatic index (%). As: sodium arsenite (5 mg/kg/day); VitC: vitamin C (100 mg/kg/day); Zn: zinc chloride (20 mg/kg/day); AsVitC: arsenic and vitamin C; AsZn: arsenic and zinc chloride. Data were tested using ANOVA followed by Tukey test. Values are mean ± SD.
Morphometry and stereology
The volumetric proportion of the interstitium increased in AsZn animals, whereas the epithelium proportion decreased, the same observed for the arsenic and vitamin C (AsVitC) animals, when compared to control (Table 1). The proportion of the tubular lumen in As treated animals was the lowest in all groups (Table 1).
Volume (mL) and volumetric proportions (%) of the components of the testicular parenchyma.

As: sodium arsenite (5 mg/kg/day); VitC: vitamin C (100 mg/kg/day); Zn: zinc chloride (20 mg/kg/day); AsVitC: arsenic and vitamin C; AsZn: arsenic and zinc chloride. Values are mean ± SD. Different superscript alphabets show significant differences (p < 0.05) by ANOVA followed by Tukey’s test.
The tubular diameter and the epithelium height significantly decreased in all groups that consumed As, even combined with vitamin C and ZnCl2. The seminiferous tubules length per gram of testis increased in As and AsVitC animals (Figure 2).

(a) Seminiferous tubule diameter (μm). (b) Epithelial height (μm). (c) Length of the seminiferous tubules per gram of testes (m/g). As: sodium arsenite (5 mg/kg/day); VitC: vitamin C (100 mg/kg/day); Zn: zinc chloride (20 mg/kg/day); AsVitC: arsenic and vitamin C; AsZn: arsenic and zinc chloride. *p < 0.05 and ***p < 0.001. Data were tested using ANOVA followed by Tukey test. Values are mean ± SD.
Cell counts and indexes
The number of PL-L decreased in AsVitC animals comparing to its control. Also, the number of RS was reduced in all groups treated with As, even after association with zinc and vitamin C (Table 2). On the other hand, only the groups, As and AsVitC, showed an overall reduction in the total number of germ cells. The mitotic index was significantly reduced in the AsVitC group (Table 3).
Germ and Sertoli cells numbers per cross section of seminiferous tubule in stage 1 of the seminiferous epithelium cycle.
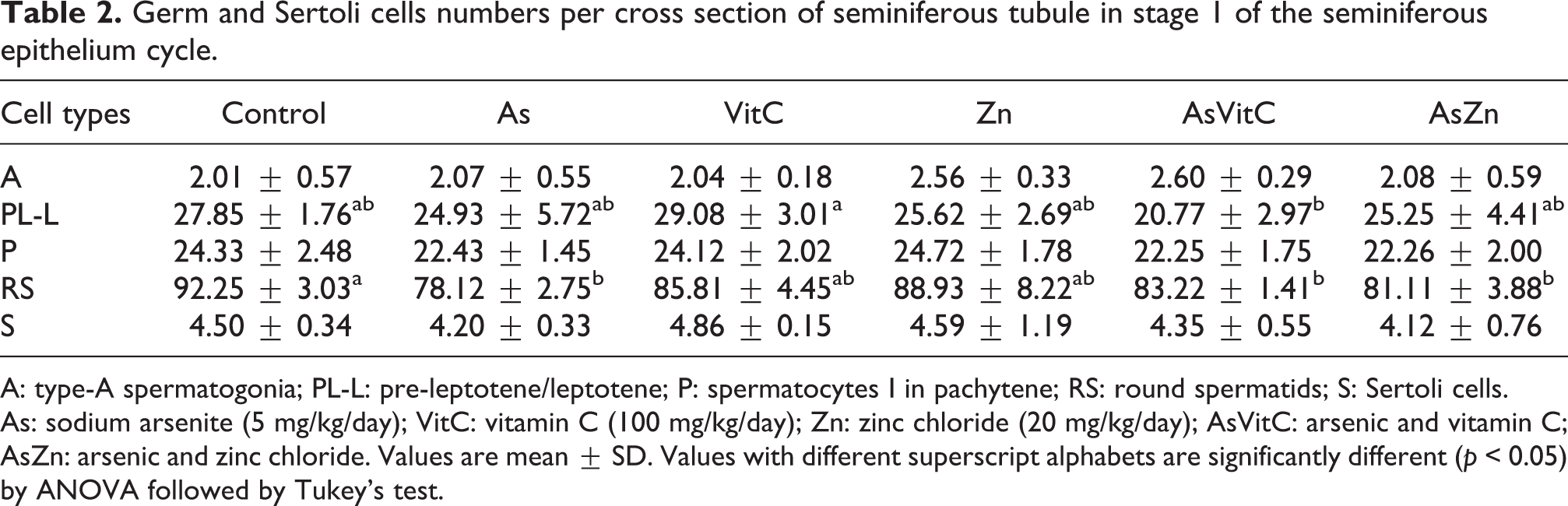
A: type-A spermatogonia; PL-L: pre-leptotene/leptotene; P: spermatocytes I in pachytene; RS: round spermatids; S: Sertoli cells. As: sodium arsenite (5 mg/kg/day); VitC: vitamin C (100 mg/kg/day); Zn: zinc chloride (20 mg/kg/day); AsVitC: arsenic and vitamin C; AsZn: arsenic and zinc chloride. Values are mean ± SD. Values with different superscript alphabets are significantly different (p < 0.05) by ANOVA followed by Tukey’s test.
Total germ and Sertoli cell’s number and spermatogenic indexes.

As: sodium arsenite (5 mg/kg/day); VitC: vitamin C (100 mg/kg/day); Zn: zinc chloride (20 mg/kg/day); AsVitC: arsenic and vitamin C; AsZn: arsenic and zinc chloride. Values are mean ± SD. Values with different superscript alphabets are significantly different (p < 0.05) by ANOVA followed by Tukey’s test.
DSP, sperm reserve and sperm transit time
The number of step 19 spermatids (sperm production rate) and the DSP were reduced in all animals after As and As + vitamin C intake (Table 4). The number of sperm in the cauda epididymis of AsVitC animals was lower than the control. The sperm transit time was the same in all groups (Table 4).
Spermatids counts in the testis and in the epididymis (head/body and cauda epididymis), daily sperm production and sperm transit time.

DSP: daily spermatic production; As: sodium arsenite (5 mg/kg/day); VitC: vitamin C (100 mg/kg/day); Zn: zinc chloride (20 mg/kg/day); AsVitC: arsenic and vitamin C; AsZn: arsenic and zinc chloride. Values are mean ± SD. Values with different superscript alphabets are significantly different (p < 0.05) by ANOVA followed by Tukey’s test.
Sperm morphology
The As group showed the higher proportions of abnormal sperm (p < 0.05). On the other hand, the concomitant intake of As with zinc or vitamin C enhanced the proportions of normal sperm (Figure 3). The main morphological alterations are shown in Figure 4.

Proportion of normal and abnormal sperm (%) presents in the vas deferens. As: sodium arsenite (5 mg/kg/day); VitC: vitamin C (100 mg/kg/day); Zn: zinc chloride (20 mg/kg/day); AsVitC: arsenic and vitamin C; AsZn: arsenic and zinc chloride. Values are mean ± SD and *p < 0.05 according to ANOVA followed by Tukey test.

Sperm abnormalities (vas deferens). (a) Normal. (b) Normal head separated from the tail. (c) Wrong-angled hook. (d) Folded sperm. (e) Amorphous head and normal tail. Bar: 50 μm.
The As group showed increased values for all categories compared to the controls (Table 5). In addition, zinc and vitamin C supplementation protected the sperm against abnormalities, since all values were lower compared to the As ones. Only AsZn animals did not show any two of the head phenotypes (Table 5).
Abnormalities found in the Wistar rat spermatozoa treated with sodium arsenite and supplemented with zinc and/or vitamin C.

As: sodium arsenite (5 mg/kg/day); VitC: vitamin C (100 mg/kg/day); Zn: zinc chloride (20 mg/kg/day); AsVitC: arsenic and vitamin C; AsZn: arsenic and zinc chloride. Values are mean ± SD. Different superscript alphabets show significantly different results (p < 0.05) by ANOVA followed by Tukey’s test.
Discussion
The present study described the adverse effects of the sodium arsenite long-term intake within the seminiferous parenchyma as well as the protective role of vitamin C and zinc supplementation after such contamination.
Quantitative evaluations regarding the seminiferous tubules, such as the tubule diameter, the epithelial height and the total tubular length, are good indicators of the spermatogenic activity as a whole, being widely used in toxicological approaches. 26,34,41,42 Such parameters could be useful to evaluate the DSP 43 and the spermatogenic dynamics, helping to describe possible alterations within the tubular compartment due to a given treatment. For instance, the increase of the tubular diameter could be an indication of fluid retention from the abnormal emptying through the vas deferens, while the decrease of the diameter normally indicates loss of germ cells. 44
The treatment with sodium arsenite reduced the tubule diameter and the epithelial height, however, the total tubular length increased as an attempt to keep the balance unaltered. Such alterations lead to the reduction of the epithelial volumetric proportion in all groups that received As, corroborating the results found by Sanghamitra et al. 45 and Ferreira et al. 46 In later studies, the epithelium was recovered after 70 days without As intake.
Since the epithelial height was affected, it was necessary to evaluate the spermatogenic process as a whole. Thus, the efficiency of spermatogonial divisions, the meiotic yield and the overall spermatogenic yield was evaluated, since they quantify the amount of cell loss and the efficiency of the meiotic divisions within the spermatogenic process. 34 Normal cell loss is expected due to hormonal and temperature variations, 47 being an efficient mechanism to regulate germ cell population. It is mainly noticed in the mitotic phase and less pronounced in the meiotic phase. 48 The meiotic and spermatogenic yields did not vary after As intake; however, the mitotic divisions were reduced after vitamin C supplementation (AsVitC group). Despite the aforementioned indexes did not vary in the As group, the germ cell number and the DSP were significantly reduced after As intake, except after ZnCl2 supplementation. Thus, even after supplementation, especially with vitamin C, the overall spermatogenic process was affected due to cell loss.
The oral administration of As, either alone or conjugated with ZnCl2 and vitamin C, significantly reduced the number of RS within the epithelium. Such result corroborates the reduction found in the stage 19 spermatids and the DSP, mainly in the animals from As and AsVitC groups, although the sperm transit time was not affected. In some situations, the association between antioxidants, such as vitamin C and ROS, induces the formation of pro-oxidant species, inducing cell death. 49 A reasonable explanation could rely on the amount of ROS generated after As intake, since the cell ability to overcome and neutralize ROS reaches a limit from which lipid and protein alterations could irreversibly compromise cellular viability. 50,51 Other important variable that was remarkably affected by As in all analysed categories was the sperm morphology. Previous studies have shown that similar treatments decreased sperm number and viability, as well as increased morphological alterations, affecting male fertility. 52 –54 On the other hand, our results indicated that vitamin C and zinc supplementation protected the sperm from the oxidative stress caused by As, as also suggested by Eskenazi et al. 55 while working with human cells.
Overall, the sodium arsenite long-term intake at 5 mg/kg showed itself harmful to the germ cell line, especially to the step 19 spermatids, thus reducing the DSP. On the other hand, the antioxidant supplementation with vitamin C and zinc proved to be efficient to prevent such damage. Further transmission electron analyses are still needed to describe the potential effects of As within the Sertoli cell barrier.
Footnotes
Acknowledgement
The authors wish to thank Dr. Gustavo Rocha Leite for the statistical analysis and Celina Almeida Lamas for the kind help with sperm analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LS Altoé’s Masters’ scholarship was provided by Fundação de Amparo à Pesquisa do Estado do Espírito Santo for 24 months.
