Abstract
It is reported that methanol is generally used as an industrial solvent, antifreeze, windshield washer fluid, cooking fuel and perfume. Methanol ingestion can lead to severe metabolic disturbances, blindness, or even death. So far, few studies about its negative effects on cardiovascular system have been reported. The purpose of this study was to determine the vasoactive effect of methanol and roles of ion channels and signal transduction pathways on isolated rat aorta. The results suggested that the mechanism of methanol-induced vasorelaxation at low concentrations (<500 mM) was mediated by ATP-sensitive K+ (KATP) and L-type Ca2+ channels, but the mechanism at high concentrations (>600 mM) was related to KATP, voltage-dependent K+, big-conductance Ca2+-activated K+, L-type Ca2+ channels as well as prostacyclin, protein kinase C, β-adrenoceptors pathways. In addition, methanol induced a dose-dependent inhibition of vasoconstrictions caused by calcium chloride, potassium chloride, or norepinephrine. Further work is needed to investigate the relative contribution of each channel and pathway in methanol-induced vasoactive effect.
Introduction
Methanol (CH4O, methyl alcohol) toxicity occurs via ingestion of antifreeze, windshield washer fluid, cooking fuel or perfume. Outbreaks of methanol toxicity are also associated with consumption of methanol-contaminated beverages. Methanol is always ingested as a substitute for ethanol, which is either too expensive or unavailable. 1,2 Low-level exposure to methanol occurs from ingestion of vegetables, fresh fruit, or artificial sweeteners when it is metabolized and through endogenous demethylation metabolic pathways. 3 It is reported that methanol is generally used as an industrial solvent like formaldehyde, acetic acid, glycol ethers or denaturants of ethanol. Human exposed to a large toxic quantity of methanol via the oral route rarely happens in modern society, basically from consumption of methanol in illicit whiskey. 4 Gil et al. reported a case of acute intoxication of methanol which occurred after the ingestion of pesticide containing methanol as an additive. 5 Hence, the toxicity caused by methanol has been a health concern and further evaluation of its toxicological properties is needed.
Toxicity of methanol, in humans and primates, is reported to be mediated via its metabolites. Methanol is first oxidized to methanoic acid, which acts as metabolic poison to induce histological abnormalities and vasorelaxation, and finally it is detoxified to carbon dioxide (CO2). 4,6 Compared to other animals, humans are especially sensitive to toxicity caused by methanol, because the lower mammals can oxidize the methanoic acid to CO2 immediately. 7 Anuria, shock, and vasodilatation are prognostic signs of methanol toxicity. The concentrations of methanol in blood and its metabolites, formate and methanoic acid are the determining factors of the toxicity in humans and primates. 4 Methanoic acid is considered to be the metabolite which is responsible for the methanol-induced toxicity in rats, 8 –10 nonhuman primates, 11 and humans. 5
Based on this background, we prompted to investigate the vasorelaxant effect of methanol on isolated rat artery and determine its underlying mechanism.
Materials and methods
Chemicals and solution preparation
N-nitro-
Krebs buffer contains 11.5 mM of glucose 11.5, 1.2 mM of sodium dihydrogen phosphate, 15.4 mM of sodium bicarbonate, 5.9 mM of potassium chloride (KCl), 1.2 mM of magnesium chloride, 120.6 of sodium chloride (NaCl), and 2.5 mM of calcium chloride (CaCl2). The components of Ca2+-free Krebs buffer were same as those of Krebs buffer but with ethylene glycol-bis(β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid at 0.2 mM instead of CaCl2. High K+, Ca2+-free Krebs buffer consisted of KCl 100 mM, NaCl 17 mM, and other components closely resembled to those of Ca2+-free Krebs buffer. 12
Preparation of isolated rat thoracic aorta rings
Healthy adult male Wistar rats, weighing 182–200 g, were purchased from Animal Center of Hebei Medical University (Shijiazhuang, China). Isolated thoracic aortic rings of rat were prepared following the procedure showed in our previous paper. 12 In brief, the rats were killed by intraperitoneal injection of pentobarbital sodium (90 mg/kg). Then, rat thoracic aorta was removed rapidly and cut into rings approximately 3 mm long. The rings were put in an organ bath containing Krebs buffer at 37°C (pH 7.4). The buffer was gassed continuously with 5% CO2 and 95% oxygen. The rings were equilibrated for 1 h at a resting optimal tension of 1.5 g and the Krebs buffer was changed every 15 min before experiment. Tensions were recorded with a MedLab Biological Signal Collection System (Medease Science and Technology, Nanjing, China) during the whole experiment. The viability of the rat aorta ring preparation was estimated by contracting vessels with 60 mM KCl before each experiment. The endothelium integrity and viability of rat artery were tested by the dilatory response of the rings to 10−6 M ACh. 12 –14
All animal care and experimental protocols complied with the Guide for the Care and Use of Laboratory Animals published by Ministry of Health, People’s Republic of China, and the Institutional Animal Care and Use Committee of Shanxi University.
Characterization of the methanol-induced vasorelaxation
Isolated thoracic aorta rings of rat were precontracted by 10−6 M NE, methanol at 0–2000 mM was added, when the vasoconstriction curves of rings reached the plateau phase of the maximum tension, the tensions were recorded, and then, the percentage of dilation was calculated. In our study, saline was used as a control group. NE at 10−6 M was used, and vasodilator effect of methanol was expressed as a percentage of relaxation to 10−6 M NE-induced maximum constriction.
Endothelium was removed gently by a cotton ball in order to examine the role of endothelium in the relaxation response. Confirmation of endothelium removal was affirmed by the failure of rat aorta rings to relax >20% in response to 10−6 M ACh. 13,14
Involvement of ion channels in the methanol-induced vasorelaxation
To elucidate the contribution of K+ channels in the vascular effects of methanol, aortic rings were preincubated with iberiotoxin (100 nM), TEA (10 mM), glibenclamide (10 μM), and 4-AP (2.5 mM) for 10 min, respectively, before the contraction caused by NE. 14,15 Then, the methanol buffers of various concentrations were added while the tensions were being recorded.
To determine whether the L-type Ca2+ channels were involved in the vasoactive effect caused by methanol, rat aorta rings were preincubated with an L-type Ca2+ channel blocker, nifedipine (1 μM), for 10 min before contraction with 1 μM NE. Then, methanol buffers of various concentrations were added with the tensions being recorded.
The signal transduction pathways of vasorelaxation caused by methanol
To examine the signal transduction pathways involved in the methanol-induced vasoactive effect, the rat thoracic aorta rings were preincubated with multifarious inhibitors: indomethacin (10 μM), staurosporine (30 nM), propranolol (10 μM),
Effects of methanol on vasoconstriction
Effects of methanol on vasoconstriction evoked by CaCl2
To test the involvement of Ca2+ channels in the vascular effects of methanol, the isolated rat aortic rings were balanced in Ca2+-free Krebs buffer (37°C), washed three times for 20 min, and the rings were equilibrated in Ca2+-free, high K+ Krebs buffer for 15 min to depolarize the membrane. Then, CaCl2 was added accumulatively from 0.01 mM to 4.44 mM to observe the normal dose–effect curve. Finally, after methanol at 300, 600, or 900 mM was added and bathed for 10 min, the dose–effect curves of CaCl2 from 0.01 mM to 4.44 mM were derived in the presence of methanol at various concentrations. 12
Effects of methanol on vasoconstriction evoked by KCl or NE
After the aortic rings of rat equilibrated in Krebs buffer (37°C), KCl from 10 to 100 mM or NE from 10−8 to 10−4 M was accumulatively added to the solution to obtain the normal dose–effect curves of KCl and NE. Then, the rings were washed to normal tension and bathed in Krebs buffer at 37°C for 30 min. 12,17 Finally, methanol at 300, 600, or 900 mM was added. Ten minutes later, dose–effect curves of KCl and NE were derived from methanol at various concentrations, respectively.
Statistical analysis
All values were expressed as mean ± standard deviation. The data was analyzed by one-way analysis of variance for significant differences between the methanol groups and the control group, followed by a post hoc test. A value of p < 0.05 was accepted as statistically significant.
Results
The vasodilator effects of methanol
Figure 1 shows that methanol caused relaxation of rat aorta in a dose-dependent manner for both endothelium-intact (half maximal effective concentration (EC50), 840.32 ± 2.12 mM) and endothelium-denuded aortic rings (EC50, 1255.35 ± 3.43 mM). These vasorelaxant effects of methanol at all tested concentrations could be reversible to the levels after methanol was washed out with Krebs buffer or before the administration of methanol.

Relaxation effects of methanol on the endothelium-intact or endothelium-denuded thoracic aorta rings of rat precontracted by 10−6 M NE. The vasorelaxan effect was presented as a percentage of decrement to the maximum tension induced by 10−6 M NE. n = 6 (six aorta rings from six rats); *p < 0.05: compared with the methanol group of endothelium-intact rings. NE: norepinephrine.
Involvement of K+ and Ca2+channels in the methanol-induced vasorelaxation
Figure 2 indicates that the vasorelaxant effects of 300 mM methanol were partially inhibited by TEA, glibenclamide, and nifedipine. However, the vasorelaxant effect of 600 or 900 mM methanol on the endothelium-intact aortic rings was blocked by TEA, 4-AP, glibenclamide, iberiotoxin, or nifedipine.

Inhibitory effect of 4-AP, iberiotoxin, TEA, nifedipine, and glibenclamide on the relaxation of aorta rings of rat caused by methanol. The isolated rings were preincubated with 4-AP (1 μM), TEA (10 mM), glibenclamide (10 μM), iberiotoxin (100 nM), and nifedipine (1 μM) for 10 min, respectively, before the rings precontracted by 10−6 M NE. *p < 0.05: compared with control group. 4-AP: 4-aminopyridine; TEA: tetraethylammonium; NE: norepinephrine.
The signal transduction pathways of methanol-induced vasorelaxation
From Figure 3, we can see that the vasorelaxant effects of high concentration (600 and 900 mM) methanol on the rat rings were partially inhibited by propranolol, indomethacin, or staurosporine. However, the vasorelaxation induced by methanol at low concentrations (300 mM) was not altered by those blockers.
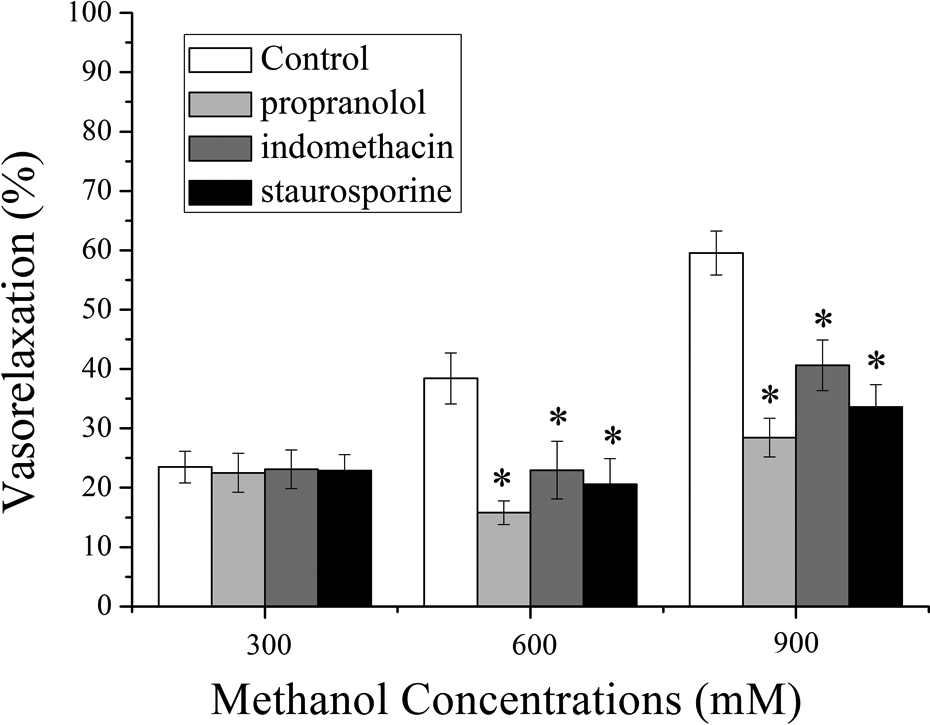
Inhibitory effect of propranolol, indomethacin, and staurosporine on the methanol-induced relaxation of aortic rings. The rat rings were preincubated with propranolol (10 μM), indomethacin (10 μM), and staurosporine (30 nM) for 10 min, respectively, before the rings precontracted by 10−6 M NE. *p < 0.05: compared with the control group. NE: norepinephrine.
Different relaxation potencies of methanol on rat aorta rings precontracted by low or high dose of KCl
Figure 4 shows that the vasorelaxant effect of methanol on the aorta rings precontracted by 20 mM KCl was more potent than that on the rings precontracted by 100 mM KCl. It indicated that the K+ channels were related to methanol-induced vasorelaxation.

The relaxant effect of methanol on the aortic rings of rat contracted with 20 or 100 mM KCl. Aortic rings were contracted with either 20 or 100 mM KCl and the vasorelaxant effect of methanol was then examined. *p < 0.05: comparison with the 20 mM KCl group. KCl: potassium chloride.
Effects of methanol on vasoconstriction
Effects of methanol on vasoconstriction evoked by CaCl2
From Figure 5, we can see that pretreatments with 600 and 900 mM methanol induced a dose-dependent inhibition of vasoconstrictions that was caused by CaCl2. Methanol shifted the dose–effect curves to the right, and the CaCl2-induced vasoconstrictions were significantly depressed.

Effect of methanol on vasoconstrictions induced by CaCl2 on aorta rings of rat incubated in Ca2+-free solution with 100 mM. The rings were preincubated with 300, 600, or 900 mM methanol for 10 min, respectively, and then subsequent cumulative administration of CaCl2 was carried out. Vasoconstrictions were presented as a percentage of decrement to maximum constriction caused by CaCl2 in control group, which was taken as 100%. *p < 0.05: comparison with the control group. CaCl2: calcium chloride.
Effects of methanol on vasoconstriction evoked by NE or KCl
For the aortic rings, pretreatments with 600 and 900 mM methanol caused a dose-dependent inhibition of vasoconstrictions which were induced by KCl or NE. Methanol shifted the dose–effect curves to the right, and the vasoconstrictions caused by KCl or NE were depressed (Figure 6).

Effect of methanol on vasocontraction caused by KCl (a) and NE (b) on the rings of rat. The aortic rings were preincubated with 300, 600, or 900 mM methanol for 10 min, respectively, and then the subsequent cumulative administration of KCl and NE was carried out. The vasoconstriction effect was presented as a percentage of decrement to the maximum constriction caused by KCl or NE in control group, which was taken as 100%. *p < 0.05: comparison with the control group. KCl: potassium chloride; NE: norepinephrine.
Discussion
Methanol ingestion is a form of poisoning that can lead to severe metabolic disturbances, blindness, permanent neurologic dysfunction, or even death.
18,19
It is reported that methanol is frequently used as a component of washing fluids, paint removers, and photocopying fluids. Because being cheap and easy to be obtained, methanol is used in the production of illegal alcoholic beverages in many areas.
18
Methanol is a globally distributed and widely used product with numerous industrial applications. It is considered to be an ideal alternative fuel due to the fast dismissing of gas resources and current depletion of fossil fuel resources.
20
Methanol is absorbed by dermal exposure, inhalation, or ingestion, and then it is rapidly distributed to tissues with the circulation of body water.
11
In humans and nonhuman primates, it has been reported that the methanol-induced acute toxicity is associated with the accumulation of formate or methanoic acid, either of which is toxic metabolites of methanol. Then, methanoic acid was converted to water and CO2 rapidly.
4
Treatments of rats with methanol extremely increased lipid peroxidation level and modulated antioxidant enzymes activities.
21
In the present study, the vasoactive effects of methanol were explained. Methanol relaxed the aorta ring of rat precontracted by NE in a dose-dependent manner for both endothelium-intact (EC50, 840.32 ± 2.12 mM) and endothelium-denuded aortic rings (EC50, 1255.35 ± 3.43 mM, Figure 1). However, the mechanism of vasodilator effect caused by methanol is not clear yet. Thus, the inhibitors such as glibenclamide, TEA, iberiotoxin, apamin, 4-AP,
Opening of K+ channels, which leads to the membrane potential hyperpolarization in the vascular smooth muscle cells (VSMCs), provides a vital mechanism in vasorelaxant effect. 12,23,29 Our study showed that the vasodilation of methanol on the aorta rings of rat contracted by 20 mM KCl was more potent than that by 100 mM KCl (Figure 4). In order to elucidate the potential involvement of K+ channels, the rat aorta rings were pretreated with 100 mM iberiotoxin, 10 mM TEA, 50 mM apamin, 10 μM glibenclamide, and 1 μM 4-AP for 20 min, respectively, prior to the application of methanol. 14,26 The results indicated that (1) the methanol-induced vasorelaxation was inhibited by TEA that blocked the K+ channels in VSMCs, including voltage-dependent K+ (Kv), big-conductance Ca2+-activated K+ (BKCa), and ATP-sensitive K+ (KATP) channels. (2) 4-AP, a blocker of Kv channel, was failed to affect the vasoactive effects of methanol at 300 mM but partially inhibited the 600 and 900 mM methanol-induced vasorelaxation. This result showed that Kv channels may be responsible for the high concentrations methanol-induced vasodilator. (3) Glibenclamide inhibited the 300, 600, and 900 mM methanol-induced vasodilator for the aorta rings, indicating that KATP channels may be involved in vasorelaxation caused by methanol. (4) Iberiotoxin inhibited the 600 and 900 mM methanol-induced vasorelaxation for the rings, suggesting that BKCa channels may be responsible for the high concentrations of methanol-induced vasodilator effect (Figure 2). The methanol-induced vasodilator effect was not affected by apamin (data not shown), showing that the vasorelaxation caused by methanol was not related to the small-conductance KCa channels. These results illustrated that opening of K+ channels (BKCa, KATP, and KV) may contribute to the vasodilator effect of methanol.
It is reported that Ca2+ influx plays a crucial physiological role in mediating the contraction of VSMCs. The opening of L-type Ca2+ channels permits Ca2+ influx and triggers intracellular Ca2+-induced Ca2+ release during sarcolemmal membrane depolarization, ultimately leading to the contraction of cells. 12,30 Changes of L-type Ca2+ channels activity in VSMCs related to diabetes and hypertension were reported. 30 Hence, we studied the role of L-type Ca2+ channels in vasodilator effect caused by methanol. The data indicated that the vasorelaxant effects of 300, 600, and 900 mM methanol (Figure 2) on the rat aortic rings were partially inhibited by nifedipine, an L-type Ca2+ channel blocker, which suggested the possible involvement of L-type Ca2+ channel during the vasorelaxant effects of methanol.
The β-adrenoceptors, in almost all VSMCs of mammals, could activate adenylate cyclase via G proteins to cause the relaxation of smooth muscles. In the present study, the vasorelaxant effect on rat aortic rings caused by methanol can be inhibited by the preincubation with propranolol, an inhibiter of β-adrenoceptor, at high concentration (600 and 900 mM; Figure 3). 27,28 It means that the methanol-induced vasorelaxation is related to the β-adrenoceptors.
Protein kinase C (PKC) plays a significant role in signal transduction for a lot of biologically active substances which activate cellular functions. It is no doubt that dysregulation of PKC leads to diseases such as cancer, diabetes, immune-mediated diseases, cardiovascular diseases, neurological conditions, and psychiatric diseases. 26,31 To examine if PKC pathways were involved in the vasodilator effects of methanol, the isolated aorta rings of rat were preincubated for 20 min with 30 nM staurosporine, a blocker of PKC. 22,26,28 Our results presented that staurosporine can inhibit the vasodilator effect of methanol at high concentration (600 and 900 mM; Figure 3). This result suggested that methanol-induced vasorelaxant effect was relevant to the PKC pathway.
Prostacyclin (PGI2), a member of the prostanoid family of lipid mediators, is expressed in VSMCs and endothelial cells. In the cardiovascular system, PGI2 is also an inhibitor of platelet aggregation and a potent vasodilator. 32 Moreover, PGI2 inhibits the proliferation of SMCs and reduces pulmonary blood pressure. 33 Indomethacin was a blocker of cycloxygenase, which is one of PGI2 synthetases. 25,26,28 Hence, the role of indomethacin in the vasodilator effect of methanol was further explored. The basal tension of the aortic rings was partly suppressed by indomethacin at high concentrations (600 and 900 mM; Figure 3), which suggested that the vasodilator effect of methanol was connected with the PGI2 pathway.
Cyclic guanosine monophosphate (cGMP), a second messenger of cellular signal transduction, plays a key role in the regulation of a range of physiologic functions including the relaxation of VSMCs.
28,34,35
Nitric oxide (NO) activates guanylate cyclase (GC) to synthesize cGMP, which will create a signal transduction pathway.
35,36
In this study, we observed the vasoactive effect of methanol in the presence of the NO synthase inhibitor
It is beyond any doubt that the constriction of VSMCs is caused by an increase of intracellular Ca2+ level. The vasoconstriction may be caused by two ways: one is the influx of extracellular Ca2+ via the receptor-operating Ca2+ channel (ROC) evoked by NE and the release of intracellular Ca2+, the other is extracellular Ca2+ influx via the voltage-dependent Ca2+ channel (PDC) evoked by depolarization with high K+ concentration. 12,39 Our results detected that pretreatments with methanol produced a dose-related inhibition of vasoconstrictions caused by NE, KCl, or CaCl2. However, whether methanol could block VDC and ROC is still unclear.
The results demonstrate that the vasorelaxation caused by methanol at low concentrations was probably related to KATP and L-type Ca2+ channels. While the methanol-induced vasorelaxation at high concentrations was partly mediated by KATP, Kv, BKCa, L-type Ca2+ channels as well as the β-adrenoceptor, PKC, and PGI2 pathways. Methanol exposure may be a potential inducer of cardiovascular diseases. Further work is surely needed to investigate the relative contribution of each channel and pathway in methanol-induced vasoactive effect.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grant 21107064 from the National Natural Science Foundation of China, by grants 2012021033-4 and 2016011082 from the Natural Science Foundation of Shanxi Province, and by Program for the Outstanding Innovative Teams of Higher Learning Institutions of Shanxi (OIT).
