Abstract
Purpose:
Methylphenidate (MPH) derivative drugs are used because of psychostimulants effects on attention-deficit hyperactivity disorder in children and adults. As far as we know, toxic or anti-proliferative effects of MPH against cartilage tissue were not studied in the literature. The present study was carried out to investigate the possible effects of MPH on the proliferation, viability and differentiation of primary human chondrocytes, in vitro.
Methods:
Monolayer primary chondrocyte cultures were prepared using osteochondral tissue obtained from patients who underwent a total knee prosthesis operation. Stock solution of MPH was prepared and aliquots having 1–1000 µM concentrations of the drug was composed. These solutions were applied to the wells containing cultured chondrocyte samples within the well plates. Control groups were composed of pure chondrocyte culture and no solution was added into them. All groups were evaluated at 24, 48 and 72 h in order to determine the possible negative effects of the drug on the chondrocytes. The data were evaluated by Tukey’s honestly significantly different test following analysis of variance.
Results:
In the group where MPH was applied, it was found that viability, proliferation and stage-specific embryonic antigen-1 protein expression were decreased in comparison to the control group.
Conclusions:
It was emphasized that clinicians should not disregard the fact that this drug might suppress chondrocyte cell proliferation and chondrogenic differentiation.
Introduction
For the treatment of attention-deficit/hyperactivity disorder (ADHD) in children and adults, many agents including dextroamphetamine, dextroamphetamine plus amphetamine derivatives such as levoamphetamine, dextromethylphenidate and methylphenidate (MPH) are used as psychostimulants.
As far as the pharmacokinetics mechanisms of MPH are concerned, no difference between children and adults exists. Although the mechanism of action of MPH has not been fully explained, it was proposed to act as a stimulant in reticular formation and cortical regions. 1 –3
These pharmaceuticals were found to cause side effects in immune, gastrointestinal, lymphatic, musculoskeletal and central and peripheral neural systems, as well as a number of organs including eye, heart and connective tissue. 4
In clinical studies, it has been found that when high-dose MPH is used in the long-term, it suppresses the rate of increase in height, which is an important indicator of child development and determinant of the general state of health. 5 –15 It is known that growth is affected in almost any period of childhood due to bone development defects, especially those are associated with chondrodystrophy, chronic systemic diseases, psychosocial factors, drugs or traumas. 15 Chondrocytes play a key role in forming growth plates, which provide height growth from the prenatal to the adult stage. 16 –25
As it is well known, cartilage tissue is devoid of neural and vascular structures and nourished by the synovial fluid. Many drugs taken via oral route is known to accumulate in the synovial fluid. 26
Not only orthopaedic surgeons but also other researchers were reported to spent considerable efforts in repairing the injured tissues using molecular methods. Many researchers are focusing on the repair of various injured tissues, primarily the joint cartilage. As a consequence, researches investigating the possible chondrotoxicity of the frequently prescribed drugs are gaining popularity. For maintaining the equilibrium between the anabolic and catabolic pathways within the cartilage tissue, the pH level of the medium that surrounds the cartilage tissue should be kept constant. In case pH decreases to acidic levels, chondrotoxicity and cartilage degeneration were reported to be inevitable. 27
The majority of drugs have an organic acid structure and are greatly bound to plasma proteins. When the drug accumulates in the synovial fluid, the medium becomes more acidic. This, in turn, would increase the amount of the fat soluble and non-ionized portion of the drug. Hence, the interaction between the lipid structures of the cell membranes and the drug would increase. The alkaline cartilage tissue, therefore, would be easily destructed by the rather acidic drug. 27
Despite these considerations, no experimental or clinical study investigating the possible adverse effects of MPH on chondrocytes and/or extracellular matrix formation was conducted so far. In addition, the effects of MPH on chondrocytes with a large dose–response curve at the molecular level were not investigated. Thus, this is a pioneering study that aims to address whether MPH has any toxic effects on chondrocytes.
Materials and methods
The present scientific research study was carried out with the approval of the ethical committee and consent forms were obtained from the volunteers. Primer chondrocyte cultures in which MPH was not applied were used as the control group. Researchers did not know which groups received drugs and were blind to the content of the drug applied in the culture. All experiments were repeated at least three times.
Materials
Collagenase type II (Clostridium histolycum; 1 mg/mL; Invitrogen Corporation, United States) was used for chondrocyte isolation. Hank’s balanced salt solution (HBSS-1X, 14025; Gibco, ThermoFisher Scientific, United States), penicillin–streptomycin (PS), fetal bovine serum, Dulbecco’s modified Eagle’s medium (DMEM; 1000 mg glucose/L) and
Methods
Selection criteria of the volunteers
The study was carried out with cartilage tissues of 20 patients with gonarthrosis. However, 1 case with kidney function defect and 12 cases who had used methotrexate, fludarabine, cyclophosphamide, high doses of steroid and/or pressor substances, and monoamine oxidase, coumarin group anticoagulants, antidepressants such as phenytoin, primidone, phenylbutazone and tricyclic in the past year were excluded from the study. 26,27 One case was found to have a protein allergy and was excluded from the study. Remaining cases (n = 6) who did not respond to medical and conservative treatments and had large osteophytes, graded using the Kellgren–Lawrence radiological grading scale, were selected. The average age of the patients was 65.83 ± 4.12 (mean ± standard deviation; Figure 1).
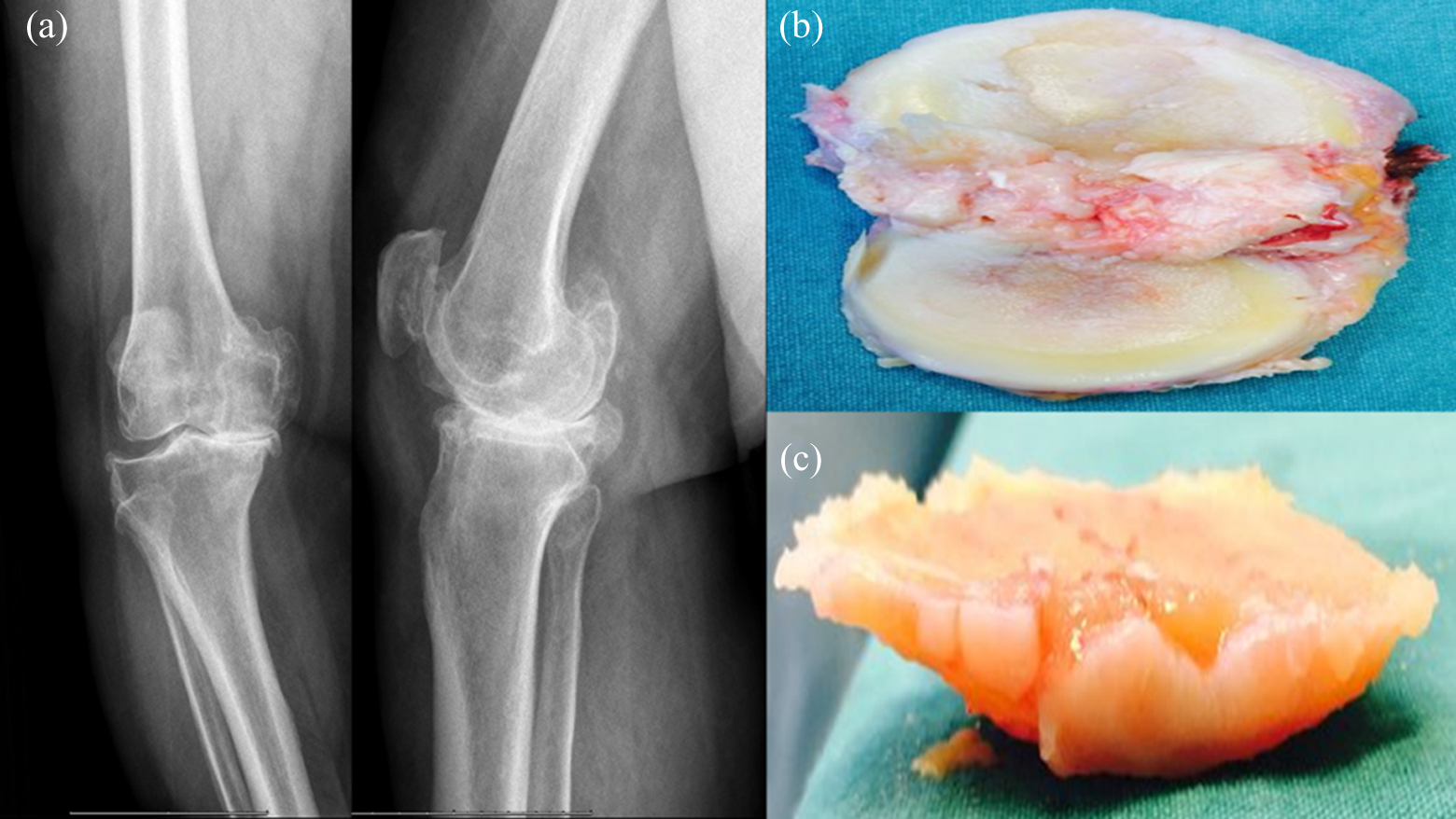
Resection of the osteochondral tissues that the chondrocyte cultures were derived. (a) Radiological image of right knee (anteroposterior [AP] and lateral; (b) osteochondral tissue; (c) chondral tissue.
Preparation of chondrocyte primer cultures
Tissues were transferred to the laboratory in DMEM medium containing 5% PS under aseptic conditions. Chondral tissues were washed with HBSS to remove red blood cells. Cartilage was separated from the bone tissue and cut into pieces. Tissues that were washed using HBSS solution in 50 mL 0.1% (w/v) were transferred to falcon tubes and letter coded.
Enzymatic digestion of cartilage tissue was performed with collagenase type II enzyme (0.375 g/mL; HBSS was used as solvent). Tissue samples were incubated in collagenase solution at 5% CO2 and 37.4°C for 24 h. Following incubation, the samples were centrifuged at 1200 r/min at 4°C for 10 min. Cartilage pellets were suspended using fresh culture medium prepared with DMEM. Cells were transferred into flasks and incubated for 72 h. Standard primary culture protocols were followed to obtain a monolayer human primary chondrocyte culture. 28 –31
Chondrocytes were detached from the culture vessel surfaces using trypsin–ethylenediaminetetraacetic acid and then stained with Trypan blue and counted with a Neubauer Chamber. Approximately 1.6 × 104 cells/well were placed to each well in a 96-well plate for MTT analysis and SSEA-1 ELISA, while 3.7 × 104 cells/well in 24-well plates and 4.6 × 106 cells in 35 mm petri dishes for further evaluations. All cultures were incubated for 24 h in order for cells to attach and proliferate. Cell culture media was supplemented with ITS media supplement and standard ingredients were refreshed every other day.
Inverted light microscopy
Microimages of cell organization in the cartilaginous tissue were recorded confocally at magnifications of 4×, 10×, 20× and 40× under phase-contrast microscopy before and after the application of MPH. The images were analysed using the CX41 Olympus microscope and Cell Imaging software.
Preparing pharmacological agents
MPH with 5 mg of main stock was dissolved in DMSO, at a concentration of 3.7 µM/mL under sterile conditions. The final concentrations were diluted from 1000, 500, 250, 100, 10 and 1 µM/mL, respectively. The first group comprised the control group with no medication (Table 1).
Agent: commercial stock solution concentrations and final concentrations.
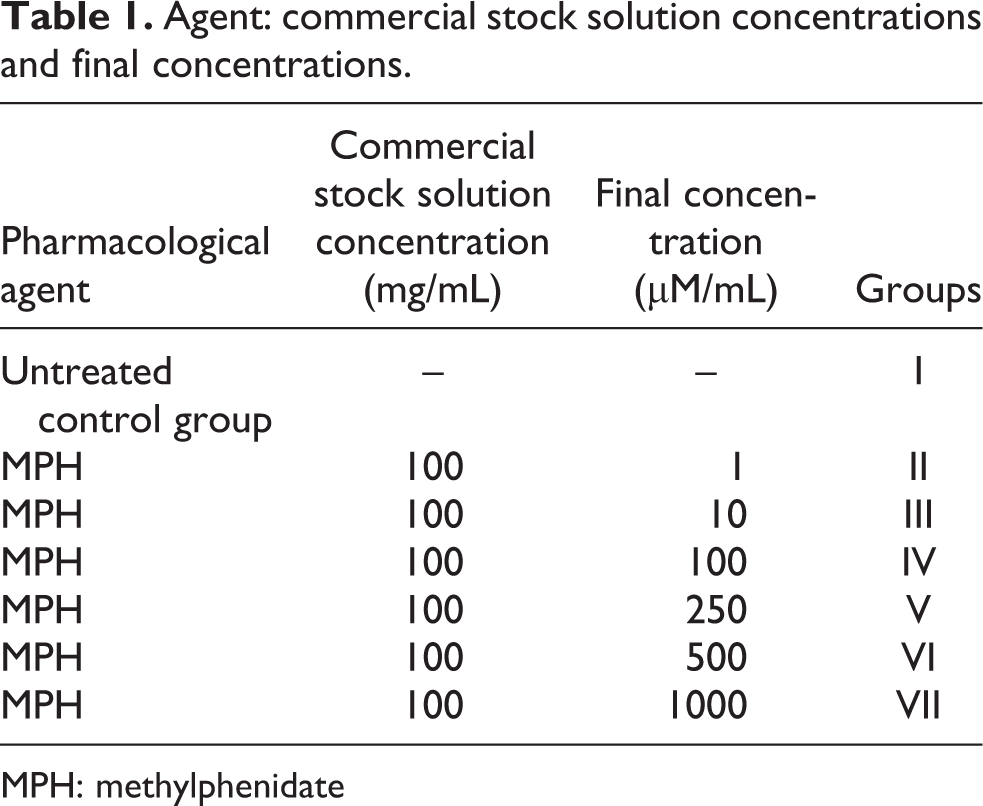
MPH: methylphenidate
In the seven main groups, drugs were applied for 24, 48 and 72 h. Following this application, the MTT test was carried out to determine toxicity and proliferation, while SSEA-1 analyses were performed to determine differentiation. MTT and SSEA-1 analyses were carried out at least three times for all drug concentrations. 27
Solutions in light-proof bottles were delivered to the researchers to carry out the analyses following letter coding.
ESEM analysis
ESEM analysis was carried to assess the surface topography and composition of the cells. The culture medium was removed from the culture vessels using a gun pipettor. A 2.5% glutaraldehyde solution, composed of 97.5 mL of cacodylate buffer and 2.5 mL of glutaraldehyde, was added to the vessel covering the cells. The glutaraldehyde solution was then removed and the cells were maintained at room temperature for 2 h, prior to washing three times using cacodylate buffer. After the last wash, the cells were analysed with ESEM device. 27 –32
A device with a lifting system and the ability to transfer the electron beam in a high vacuum was used. In this manner, we have obtained images of the extracellular matrix as well as images of characteristic cellular structures. Images were recorded at a pressure of 219–231 Pa in ESEM vacuum mode, under magnifications of 5000×, at resolution depths (horizontal field width) of 41.4 and 82.9 μm, at an operating voltage of 5.00 kV, and at a Working distance (WD) of 9.8–10.7 mm.
MTT–ELISA viability, toxicity and proliferation analyses
Viability tests were carried out using a commercial MTT kit. Viable cells digest tetrazolium ring forming formazan crystals with the activity of mitochondrial dehydrogenase enzymes. Density of the resulting formazan crystals could be detected with spectrophotometer. Unlike viable cells, dead cells do not form these formazan crystals. In this way, MTT analysis viability and proliferation of chondrocytes were evaluated and toxicity of MPH was detected. MTT analysis was performed to a series of MPH dosages at 24, 48 and 72 h.
The culture media containing MPH discarded from wells 100-μL MTT (12 mM/5-mg MTT prepared by adding 1 mL sterile PBS) were added. Cultures were incubated at 37°C for 2 h protected from the light. DMSO was added to these samples to stop reaction and were incubated at 37°C for an additional 10 min, prior to photometric analysis of a 540-nm wavelength absorbance. For assessment of proliferation, 500 μL of SDS–HCl solution was added to the remaining cells and incubated at 37°C for 18 h. The reaction solution was subjected to photometric analysis at a 570-nm wavelength. The vitality of the control group was assumed to be 100%. 27 –35
SSEA-1 human pre-chondrocytic activity assay through protein components
During the differentiation of human mesenchymal stem cells containing embryonic stem cells, the SSEA-1 protein is upregulated; however, in undifferentiated cells, the SSEA-1 protein is downregulated. We used a pre-chondrocytic human characterization ELISA kit to analyse whether the cells in chondrocyte cultures underwent differentiation, undifferentiation, stimulation or inhibition by determining whether SSEA-1 increased or decreased in the cultured cells in the drug experiments. 27,36
Analyses of the samples were carried out at 24, 48 and 72 h, synchronously with MTT analysis, according to the manufacturer’s instructions.
Statistical analyses
Descriptive statistics were shown as mean ± standard deviation. In the analyses of the obtained data, results were evaluated by cell number, proliferation and SSEA-1 protein component expression. The Minitab R16 program was used for the statistical evaluation. The evaluations were made at a 95% confidence interval. Furthermore, the results were evaluated using analysis of variance to determine whether there were significant differences across the groups. When there were differences across the groups, Tukey’s honestly significantly different, one of the post hoc multiple pairwise comparison tests, was used to determine the difference and investigate false positive; thus, the different averages across the experimental groups were evaluated. A value of p < 0.01 was considered to be highly significant.
Results
Inverted light microscopy and ESEM evaluation
The viability and morphology of cells were examined microscopically using inverted light microscopy. MPH at different doses in the range of 1–1000 µM was applied to cell cultures for 24, 48 and 72 h, and changes were recorded (Figure 2).
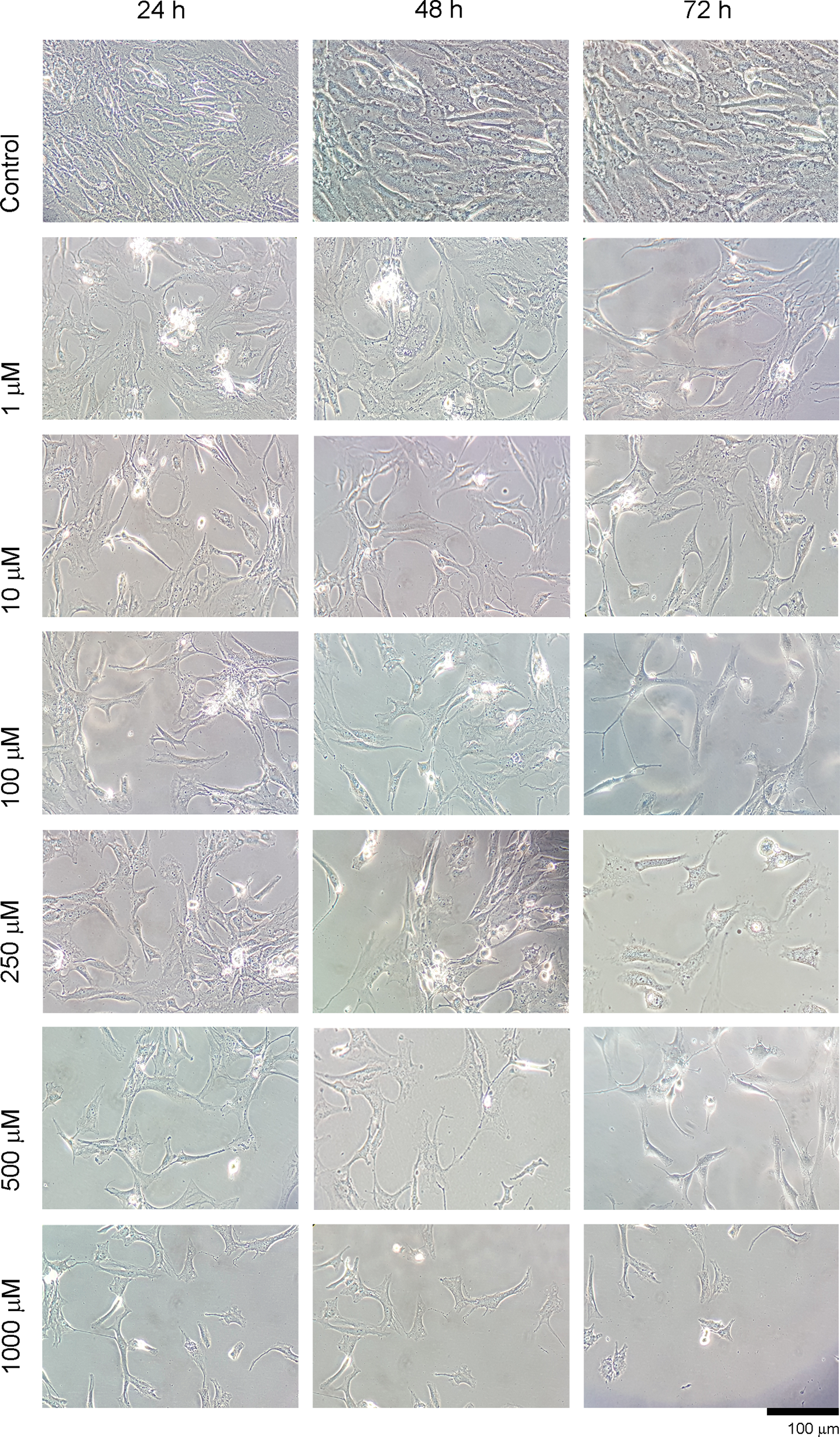
Evaluation of round shape that represents cytotoxicity through inverted light microscopy (×100 magnification). Cytotoxicity was not only represented by round shape but a decrease in the number of living cells and disruption in the integrity of extracellular matrix structure were also observed.
It was observed that cell morphology changed along with the ascending doses and cells detach from culture vessel, lose their specific morphology and becomes spherical, which is a typical reaction of cells to a cytotoxic agent. It was found that the applied dose was more effective than the duration of the application. Morphological changes observed with microscope were verified with ESEM and MTT analyses. In healthy chondrocytes showing chondroblastic activity, all natural surface characteristics were visible in the control group images. Viable cells were observed at 24, 48 and 72 h after the application of MPH, and it was found that the viable cell concentration decreased with ascending doses. With ESEM analysis, deterioration of cell surface morphologies and loss of extracellular matrix were evident in chondrocyte cultures after a 1000 µM application of MPH (Figure 3).
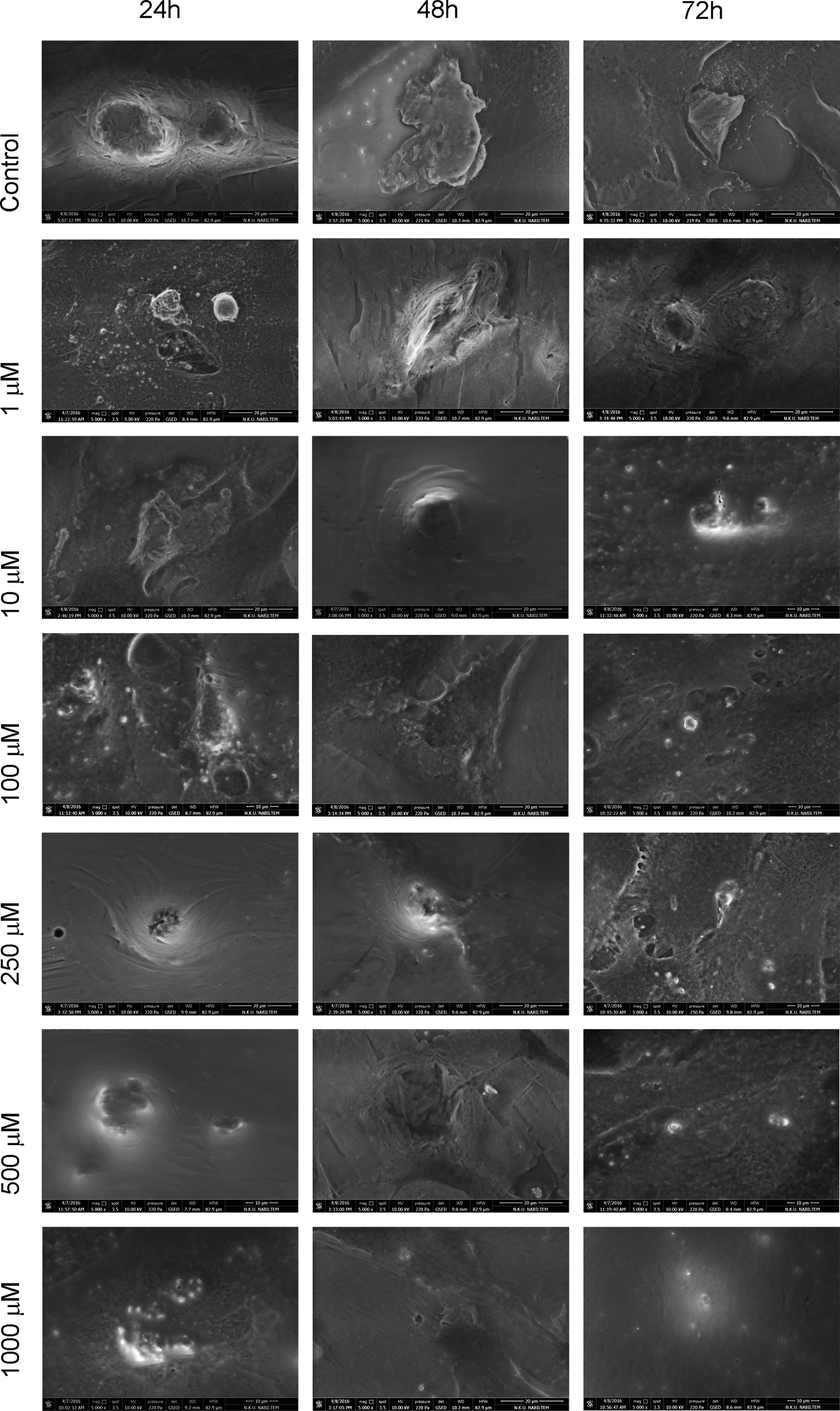
Evaluation of the surface morphology of the chondrocytes by ESEM. The images were recorded under magnifications of ×5000, distortion of cell surface morphology and loss of extracellular matrix were noted. ESEM: environmental scanning electron microscope.
Statistical evaluation of MTT viability, toxicity, proliferation and SSEA-1 protein components
In the non-drug control group, it was reported that cell viability, proliferation and SSEA-1 protein expression was enhanced with increased duration. In the groups where MPH was applied, it was found for all doses that viability, proliferation and SSEA-1 protein expressions decreased. However, it was statistically confirmed that the duration of exposition to drugs determined the toxic effect (p < 0.01). It was found that while the lowest rates of viability, proliferation and SSEA-1 protein expressions were exhibited in the group where a dose of 1000 µM was applied for 72 h, the lowest rate of toxicity was evident in the group where 1 µM MPH was applied for 24 h. The results were statistically significant (p < 0.0.01; Figure 4, Table 2).
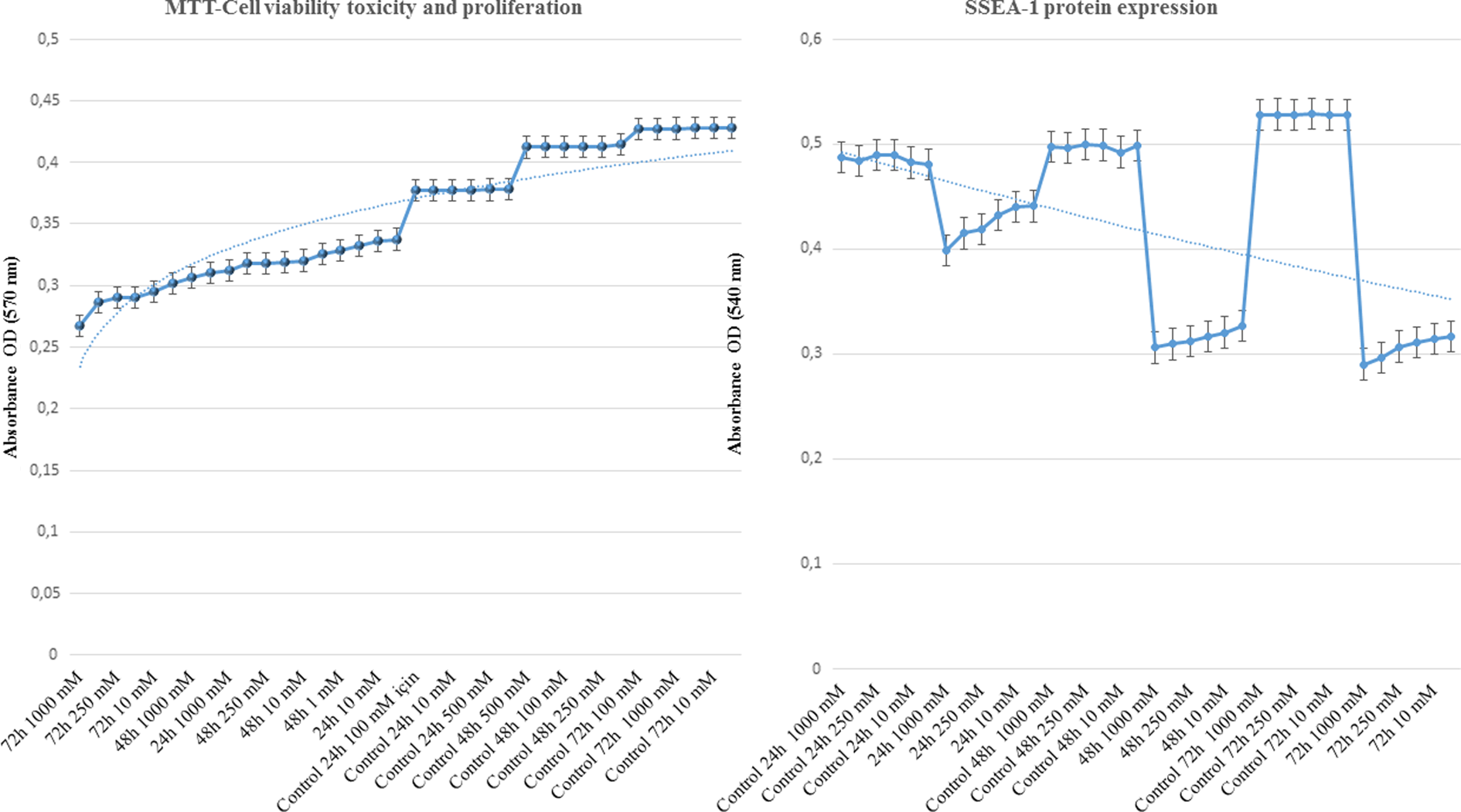
Change of expression pertaining to SSEA-1 protein components and comparison of the indicators of MTT cell viability, toxicity and proliferation. Following analysis of the interactions among the independent variables and the impact of these interactions on the dependent variables using analysis of variance, since the sample numbers in the groups were equal, we used Tukey’s HSD test, which would yield the strongest analysis report. The analysis of HSD test showed that, after 72 h following MPH application into the wells containing the culture medium, the MTT absorbance values, which represented the level of viability, toxicity and proliferation, were found to be significantly decreased. Meanwhile, SSEA-1 protein expression increase was suppressed, which represented a decrease in differentiation. These findings could be the evidence for the possible toxicity of the drug for the chondrocytes and the surrounding structures. SSEA-1: stage-specific embryonic antigen-1; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; HSD: honestly significantly different.
Statistical analyses: pairwise comparisons for SSEA-1 protein and MTT-cell proliferation (grouping information using the Tukey’s method and 95% confidence interval).
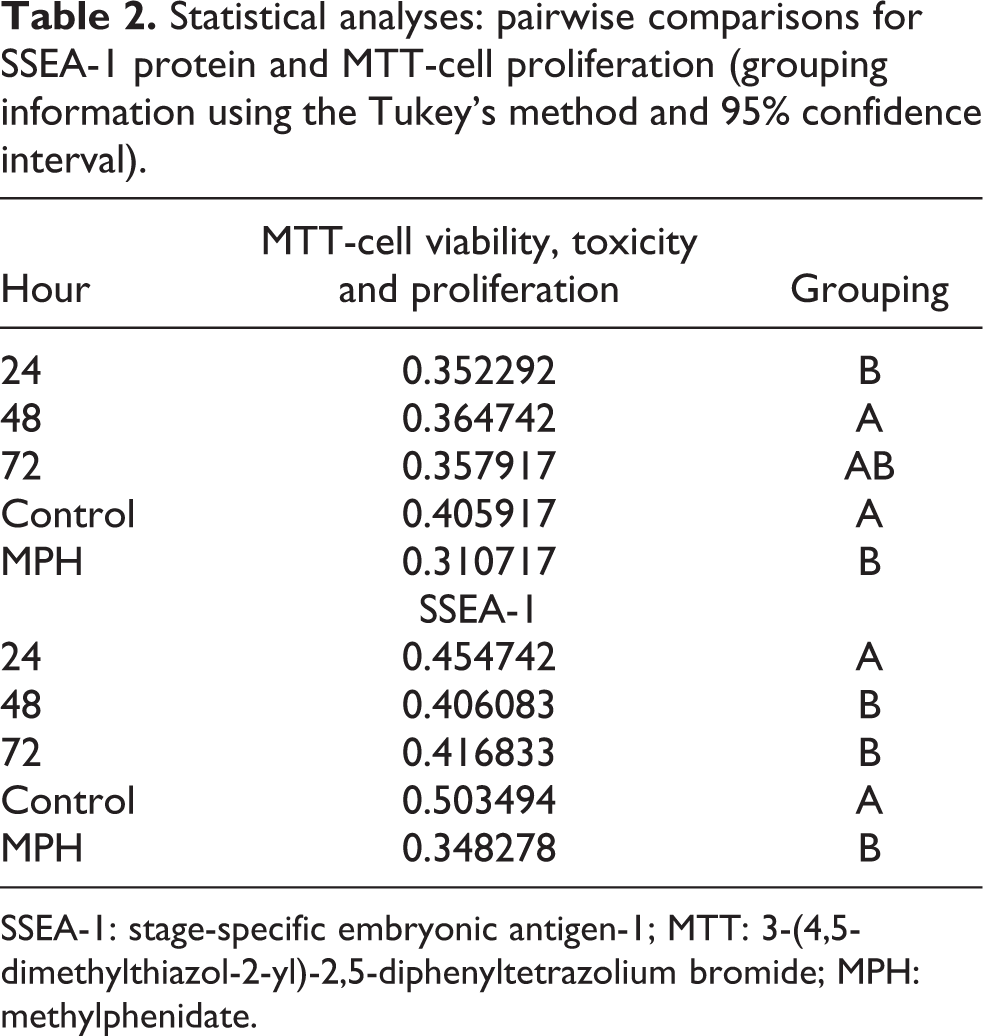
SSEA-1: stage-specific embryonic antigen-1; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; MPH: methylphenidate.
Discussion
Most of the drugs administered either systemic or oral routes accumulate in synovial fluid. 27,37 –39 Veins, lymphs and nerves are not present in cartilage tissue; therefore, cartilage cells are in contact with only a perichondrium layer and/or synovial fluid in the articular surface of the bone. Nourishment of chondrocytes is provided by double diffusion from this articular surface. The outer layer of synovial fluid is thicker, so drugs and/or nutrients diffuse primarily in synovial fluid before synovial tissue. Following this, a second diffusion occurs by passing through pores in synovial fluid, which are 6–8 nm in length and in contact with the chondrocytes. In addition, active transport from synovial fluid is also essential in the nutrition of cartilage tissue. 27,40,41
As a result of developments in pharmaceutic technology and generative medicine, the toxicity of drugs on a molecular level and repair of damaged tissues has been extensively researched. Orthopedic surgery, like all branches of medicine, utilizes tissue engineering and seeks to protect bone and cartilage tissues or repair damaged tissues. However, some studies were found that drugs may have toxic effects on healthy cartilage and bone tissues. 27,29 –35 For this reason, we studied whether MPH, which is frequently prescribed for children or adults with ADHD, has a toxic effect on human articular primary cartilage cells.
In a literature review, we found only one study in which MPH toxicity was investigated in the in vitro experimental setting. 42 Moreover, Ludolph et al. clarified that there have been almost no evidential preclinical or clinical studies concerning the pharmacological or toxicological effect mechanism of MPH. In their study, they applied MPH (up to 1 µM) to mesencephalic cell cultures composed of human embryonic kidney (HEK-293) and cultured rat embryonic (E14.5) commercial cell lines, and they analysed the proliferation and dopaminergic levels of cells at the end of 72 h. They concluded that no toxic effect of MPH was found in HEK-293 cells, whereas in HEK-human dopamine transporter cells and dopaminergic neurons, significant toxic effects were observed. 42
As a rule, in the present study and this type of cytotoxicity research, experimental settings are generated using animal tissues. However, it has been emphasized in the literature that analyses carried out on animal tissues might offer different results from those using human tissues, as the sensitivity may be different. Apart from this, in the previously performed in vitro studies, animal tissues were not used and commercial cell lines were applied. 27,42 However, since it is known that commercial cell lines contain monotypes, lack a micro-environment like the extracellular matrix, and exhibit changes in their phenotypic and genotypic features, the results may be unreliable. 27 –35
In the present study, we did not use animal tissue or commercial cell lines. Rather we used cartilage tissues belonging to patients with knee prostheses in the course of routine clinical practice. During the application of knee prostheses, primary chondrocyte cultures were prepared from undamaged chondral tissues of the resected articular surface.
Thanks to primary cultures taken from chondral cut materials that were derived from the lateral compartment, analyses in an environment that included extensions, such as the extracellular matrix, were carried out in addition to analyses of cartilage cells.
The possibility that other drugs might have accumulated in synovial tissue and passed through the cartilage tissue was also taken into account. When MPH was added to the cell culture environment, there was a possibility that it might interact with other previously accumulated drugs to generate toxicity. Thus, the selection criteria of the cases included drug use history, in order to avoid drug–drug interactions. For the above-mentioned reasons, we are of the opinion that the results of the present study carried out in human primary articular chondrocyte cultures are substantial.
Growth is an essential indicator in determining the general health of children. At times, it may be difficult to determine the difference between normal and abnormal growth. Normal growth is influenced by genetic, hormonal and environmental factors. Although genetic factors are effective in all periods of growth, including intrauterine life, nutrition becomes important in the early periods of life and hormonal factors in pubescence. In contrast, environmental factors such as chronic systemic diseases, irradiation, trauma, drugs and psychosocial factors may give rise to bone growth defects associated with chondrodystrophies. 16
It has been reported that in cases where MPH was applied in clinics, short stature is seen. 12 –15,43 It is known that short stature may be related to injury to chondroblasts. 44
Drawing out in long bones occurs via the increase of chondrocytes on epiphyseal plates neighbouring the epiphysis. In addition, chondrocytes on the side of the plate facing the diaphysis grow better, while matrices calcify and cells die. The growth of cells in humans is the fastest at birth and slows down in childhood; however, it again gains momentum in puberty. Growth plates are composed of hyaline cartilage and appear as a line between the epiphysis and metaphysis in roentgenography. Bone develops in growth plates and grows mature in the metaphysis; these structures are the most important elements responsible for increased bone length. 17 –22
More importantly, in the fibrous part, called the node of Ranvier, which encircles the growth plate and is composed of fibrous, cartilaginous and bone parts, chondrocytes are required for growth in both length and size. 17 –24 In the cartilaginous portion, the chondrocytes should divide rapidly and pile up as pillars that are perpendicular to the long axis of the bone. In the cytoplasms of these hypertrophic regions, glycogen-accumulated chondrocytes have an important place. 17 –25 In the mature zone of bone, stimulation of hypertrophic chondrocytes is one of the important characteristics of growth plates. 17 –25,45,46
In our study, we analysed whether chondrocytes and chondroblasts, which have vital roles in growth plates, were affected by mitochondrial activity assay through MTT cell viability, toxicity and proliferation assays; however, we also analysed chondrogenic differentiation by the SSEA-1 protein component, which is a pre-chondrocytic cell precursor. In addition to molecular quantitative analysis, we demonstrated cytotoxicity through visual images obtained from invert light microscopy and ESEM scanning; this represents another outstanding point of our study.
One of the advantages of our study is that we used cartilage cultures containing a natural microenvironment that were obtained from cartilage tissues instead of standard commercial cartilage cultures. However, as it is not ethical to obtain such tissues from healthy children, cartilage tissues were obtained from cases with a diagnosis of gonarthrosis who underwent knee arthroplasty. One of the constraints of our study is that the results – which showed the effects of MPH on cartilage tissue in the elderly cases with a diagnosis of gonarthrosis was examined in vitro – cannot be generalized to in vivo effects of frequent application of MPH on young cartilage tissues in children and adolescents.
As a result, in our study where different doses ranging from 1 to 1000 µM were evaluated on a molecular level for different periods of time, it was recorded not only mathematically but also visually that MPH showed chondrotoxic effects by repressing proliferation and differentiation (p < 0.01). It was observed that cell morphology changed along with the ascending doses, and cells dematerialized and took a round form, which is a typical reaction to cytotoxic agents. It was found that the applied dose (p = 0.340) was more effective than the duration of the application (p = 0.000). However, cells exposed to a particular dose in in vitro conditions remained stable at the designated time. In contrast, when active substances accumulated in the synovial fluid during systemic drug intake are taken into consideration, incessant drug use would mean exposure to continuously increasing doses of drugs. For this very reason, clinicians should closely follow bone development of children in long-term treatments. It is obvious that the same experimental setting should be repeated by applying it to chondrocyte cultures obtained from children within an ethical framework.
Conclusion
Our study is valuable in that it compared the effects of MPH on cell viability, which is frequently prescribed for children or adults with ADHD, using samples obtained from human articular primer chondrocyte cartilage cultures. The data obtained from in vitro experiments cannot determine the effects of clinical application directly; still, it is a crucial point for a clinician that a drug used for another disease could repress chondrocyte cell proliferation and chondrogenic differentiation. The attention increases brought about by various preparations of MPH are known to be limited to their effect time, but clinicians do not yet know how long-term treatment may affect cartilage.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
