Abstract
Objective:
We investigate the effects of bone morphogenetic protein-7 (BMP-7) on models with silica-induced and macrophage-mediated fibrosis and its possible mechanisms in vitro.
Methods:
Rat alveolar II epithelial (RLE-6TN) cells were incubated with the supernatant of mouse macrophage-like cells (RAW264.7) and treated with 0, 25, 50, and 100 μg/mL silica. Using Western blotting, the epithelial markers (surfactant proteins-C and E-cadherin) and the mesenchymal markers (fibronectin (FN) and viminten (Vim)) were detected. After neutralizing the BMP-7, the progress of fibrosis was assessed by the content of hydroxyproline (Hyp) and collagen I, III protein levels as well as the Smad signaling pathway proteins, including phosphorylated Smad1/5(P-Smad1/5) and phosphorylated Smad2/3(P-Smad2/3). Collagen I was also identified by immunofluorescence and pretreated with SB-431542, LDN-193189, or anti-BMP-7-neutralizing antibody. In addition, the levels of matrix metalloproteinase-2 (MMP-2) and MMP-9 were detected using Western blotting.
Results:
The model of RLE-6TN cells was established successfully, the expressions of Vim, FN, MMP-2, and MMP-9 were upregulated, while the concentration of silica is increased. Neutralizing BMP-7 stimulated the decrease of P-Smad1/5 and the increase of P-Smad2/3, as well as the collagen I, collagen III, FN, and Hyp via Smad signaling pathway. Furthermore, pretreated with LDN-193189 or anti-BMP-7-neutralizing antibody, the expression of collagen I was increased, yet it was decreased with SB-431542 intervention.
Conclusion:
The activated BMP/Smad and suppressed transforming growth factor-β/Smad pathways could suppress silica-induced fibrosis via a MMP-dependent mechanism. BMP-7 is expected to be the optimized strategy of delaying the interstitial changes.
Introduction
Silicosis, caused by long-term inhalation of silica particles, is characterized by a persistent inflammatory response and progressive pulmonary fibrosis and has been the most common and severe forms of pneumoconiosis. 1,2 Nowadays, silicosis has the highest incidence among workers exposed to dust, but the pathogenic mechanisms are still unknown and an effective treatment is not yet available. 3
Macrophage activation and subsequent endogenous transforming growth factor-β (TGF-β) upregulation and secretion are essential steps in initiating a fibrotic response. 4 TGF-β is an important cytokine involved in the pathogenesis of fibrosis. Fibroblasts play a central role in the pathogenesis of fibrosis, and more evidences show these fibroblasts originate from alveolar epithelial cells and switch to myofibroblasts via local epithelial–mesenchymal transition (EMT). During an EMT, alveolar epithelial cells lose their polarity and certain proteins such as surfactant proteins-C (SP-C) and E-cadherin (E-cad) that are important for maintaining cellular adhesion of the basement membrane to other cells. In addition, the cells acquire mesenchymal characteristics such as viminten (Vim), fibronectin (FN), and migratory properties. 5 Smad-dependent pathway represented the dominant role on silica-induced EMT and fibrosis, and the signaling activity of bone morphogenic protein (BMP) seemed to oppose the actions of TGF-β’s profibrotic activity. 6 After an epithelial injury, EMT repairs damaged tissue by producing fibroblasts and extracellular matrix (ECM), and it is also associated with fibrosis, in many organs, 7 –11 including silicosis. 12,13
Matrix metalloproteinases (MMPs) are a family of zinc-dependent proteases that are responsible for ECM turnover as well as degradation of bioactive proteins and are involved in EMT. 14 –16 Furthermore, upregulation of MMPs is often detected in fibrosing lung diseases. 17,18 Bone morphogenic protein-7 (BMP-7) is the most prominent member of the BMP family, 19 which together with related BMP/Smad1/5/8 signaling pathway have been reported to be able to inhibit renal, intestinal, hepatic, and cardiac fibrosis where it appears to have a strong antifibrotic activity. 20 –24 However, MMPs are the major components of the matrix proteolytic network, and their roles in the pathogenesis of silica-induced fibrosis remain unknown. The relationship between BMP-7 and MMPs involved in silica-induced EMT and fibrosis was seldom investigated. It is increasingly being recognized that epithelial injury and EMT can lead to subsequent lung fibrosis. Therefore, preventing epithelial cell injury and inhibiting EMT have great potential to be an effective method of inhibiting pulmonary fibrosis. This study is to address the complicated mechanisms that how BMP-7 plays an antifibrotic role against epithelial injury and MMPs upregulation induced by silica and find a practicable strategy to inhibit or delay the fibrosis process induced by silica.
Materials and methods
Silica particles
The crystalline silica particles (Sigma-Aldrich Co. LLC, Dorset, UK) with a nominal size of 0.5–1.0 μm were used. Stock solution was prepared in phosphate-buffered saline (PBS) with a final concentration of 2 mg/mL after sonication.
Reagents
The BMP inhibitor LDN-193189 (4-(6-(4-(piperazin-1-yl)phenyl) pyrazolo[1,5-a] pyrimidin-3-yl) quinoline), purchased from Sigma-Aldrich, was synthesized as a hydrochloride salt. The TGF-β inhibitor SB-431542 ([4-(5-benzo [1, 3] dioxol-5-yl-4-pyridin-2-yl-1H-imidazol-2-yl)-benzamide]) was obtained from Sigma-Aldrich. Stock solution of the LDN-193189 was prepared in distilled water (ddH2O) and that of the SB-431542 was prepared in dimethyl sulfoxide and subsequently dissolved in culture medium for treatment. 25 Anti-BMP-7-neutralizing antibody was purchased from Sangon Biotech (Shanghai, China). Chemicals used were of the highest grade available commercially.
Cell culture and treatment
RLE-6TN cells (rat alveolar II epithelial cells) and RAW264.7 cells (mouse macrophage-like cells) were purchased from XiangYa Central Experiment Laboratory (Changsha, Hunan province, China). RLE-6TN cells were cultivated in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 14% fetal bovine serum (FBS) and 1% (w/v) penicillin/streptomycin, and RAW264.7 cells (RAW) were cultivated in the same condition with 10% FBS at 37°C in an incubator with 5% CO2 and 95% humidified air. Silica was added to the RAW cells culture at 0, 25, 50, and 100 μg/mL, respectively, and after 24 h of incubation, the cell-free supernatant was harvested for use in the next experiment. 26
The RLE-6TN cells were cultivated in six-well tissue-culturing dishes at 1 × 105 cells per well with a full-growth medium. The cells were cultured to an approximately 80% of confluence and then cultured in serum-free DMEM overnight. To determine the optimal silica concentration through the dose gradient experiment, the cells were incubated for 48 h in DMEM, each containing one of the following conditions: (1) DMEM only, as a negative control (control group); (2) RAW supernatant (50 μg/mL) only, as a positive control (silica group); and (3) incubated together with additional RAW supernatant (50 μg/mL) and anti-BMP-7-neutralizing antibody (10 μg/mL) (anti-BMP-7 group). In the neutralization experiment, neutralizing antibody was added in the cell cultures 1 h before adding RAW supernatant (50 μg/mL).
Assay of cell hydroxyproline
Reagent kit was obtained from Nanjing Jiancheng Biotechnology Research Institute (Nanjing, China). Culture mediums were kept for measurement of hydroxyproline (Hyp) content, and cells were harvested for protein extraction. All procedures were done according to the manufacturer’s instruction, and the absorbance was determined at 550 nm wavelength. The Hyp content of the RLE-6TN cells was expressed in micrograms per milliliter. All Hyp assays were performed three times.
Western blotting
The RLE-6TN cells were washed three times with ice-cold PBS and disrupted in lysis buffer. The concentration of protein was determined using the Bicinchoninic Acid (BCA) protein assay (Pierce, Rockford, IL, USA). Total protein was extracted and an equal amount of protein (20 μg) was size-fractionated in 8% sodium dodecyl sulfate polyacrylamide gel and then transferred to Polyvinylidene Fluoride (PVDF) membranes. The membranes were blocked in 3% BSA solution and then incubated overnight at 4°C, respectively, with the primary antibodies against collagen I (1:5000; Abcam, Burlingame, California, USA), collagen III (1:5000; Abcam), FN (1:500; Santa Cruz Biotechnology, Inc., Santa Cruz, California, USA), E-Cad (1:500; Santa Cruz Biotechnology, Inc.), Vim (1:5000; Abcam), MMP-2 (1:500; Santa Cruz Biotechnology, Inc.), MMP-9 (1:500; Santa Cruz Biotechnology, Inc.), SP-C (1:500; Santa Cruz Biotechnology, Inc.), P-Smad1/5 (1:1000; Cell Signaling Technology, Boston, Massachusetts, USA), and P-Smad2/3 (1:1000; Cell Signaling Technology). Proteins of interest were detected using the Esports Champion League (ECL)-enhanced chemiluminescence method, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (1:1000; Cell Signaling Technology) was used as loading controls to normalize the objective bands. Western blots were scanned and quantified using Tanon-5200 (Beijing Yuan Ping Hao Biotech Co., Ltd., Beijing, China).
Immunofluorescence
RLE-6TN cells were pretreated with SB-431542 (20 μM), LDN-193189 (10 nM), or anti-BMP-7-neutralizing antibody (10 μg/mL) for 1 h, which are grown on chamber slides for 48 h, then rinsed twice in PBS and fixed with 4% paraformaldehyde for 20 min, and followed by 0.3% Triton X-100 for 10 min at room temperature (RT) RLE–6TN cells were pretreated. 27 After rinsed in PBS and blocked with 5% BSA at RT for 10 min, slides were incubated with primary antibodies against collagen I (1:500; Abcam) overnight at 4°C. After washed, they were lucifuge incubated with the secondary antibodies conjugated to fluorescein isothiocyanate for 1 h at RT and rinsed again. Cells were counterstained with 4′,6-diamidino-2-phenylindole (DAPI). Immunolabeling was examined by Eclipse 80i microscope (Nikon, Tokyo, Japan).
Statistical methods
Quantitative data are reported as mean ± standard deviation, and the significant difference was determined by one-way analysis of variance followed by the Dunnett’s test. A p value of 0.05 was considered to be statistically significant. All statistical analyses were done with the SPSS 17.0 software.
Results
Silica-induced epithelial injury and MMPs upregulation on RLE-6TN cells in dose-related manner
When RLE-6TN cells were exposed to different doses of RAW supernatant induced by silica, we observed the upregulation of MMPs and mesenchymal markers during the downregulation of epithelial markers. Western blotting analysis showed that RAW supernatant significantly increased the expression of the Vim and FN by approximately 1.5-fold and 1.69-fold (p < 0.05; Figure 1(c), (d), and (g)), accompanied with decreased E-cad by 40% (p < 0.05; Figure 1(b) and (g)). As a marker of alveolar type II epithelial cells, the ratio of SP-C/GAPDH in silica group was lower than that in the control group (p < 0.05; Figure 1(a) and (g)). Similarly, the expressions of MMP-2 and MMP-9 were also presenting a certain dose-related trend, and they increased 1.49-fold and 1.48-fold in silica group compared with the control group (p < 0.05; Figure 1(e) to (g)). All these changes had dose-related trends, which reached the peak or nadir at 50 μg/mL. Thus, these results indicated that silica could induce epithelial injury and upregulate MMPs in RLE-6TN cells.
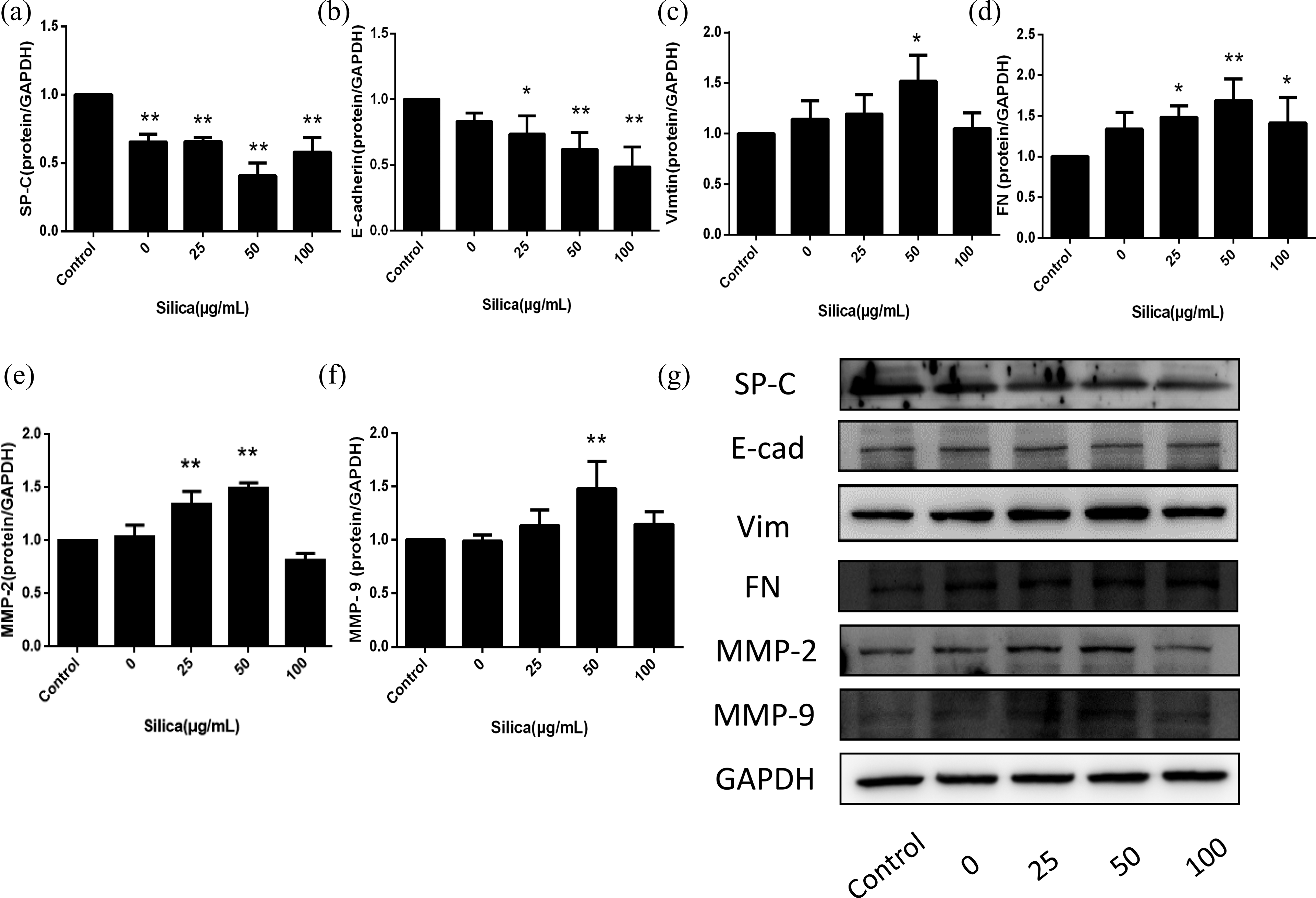
Silica promoted changes in the expression of SP-C, E-cad, Vim, FN and MMPs in a dose-related manner. RLE-6TN cells were incubated with DMEM (control) or 0, 25, 50, and 100 μg/ml RAW supernatant for 48 h. Western blot analyses revealed that the expression of SP-C (a) and E-cad (b) was decreased while Vim (c), FN (d), MMP-2 (e), and MMP-9 (f) were increased in a dose-related manner. When the concentration of RAW supernatant induced by silica reached up to 50 μg/mL, all the markers showed significant changes. The data were presented as the means ± SD (n = 3), the significance of differences was determined by ANOVA followed by the Dunnett’s test. *p < 0.05 versus control group and **p < 0.01 versus control group. SP-C: surfactant proteins-C; E-cad: E-cadherin; Vim: Viminten; FN: fibronectin; MMPs: matrix metalloproteinases; ANOVA: analysis of variance; SD: standard deviation.
Neutralizing BMP-7 promoted silica-induced fibrosis by suppressing BMP/Smad and activating TGF-β/Smad signaling pathway in RLE-6TN cells
The profibrotic effect of anti-BMP-7-neutralizing antibody on fibrosis resulted from blocking the expression of P-Smad1/5 and increasing the expression of P-Smad2/3 in RLE-6TN cells. The expression of P-Smad2/3 was increased after treated with silica plus anti-BMP-7-neutralizing antibody compared with silica alone (p < 0.05; Figure 2(b) and (c)). The expression of P-Smad1/5 in silica group decreased significantly compared with that in the control group and was further decreased when anti-BMP-7-neutralizing antibody was added (p < 0.05; Figure 2(a) and (c)). The profibrotic effect of neutralizing BMP-7 was characterized by higher levels of FN (1.12-fold), collagen I (1.24-fold), and collagen III (1.23-fold) in comparison to the silica group (p < 0.05; Figure 3(b) to (d) and (f)).
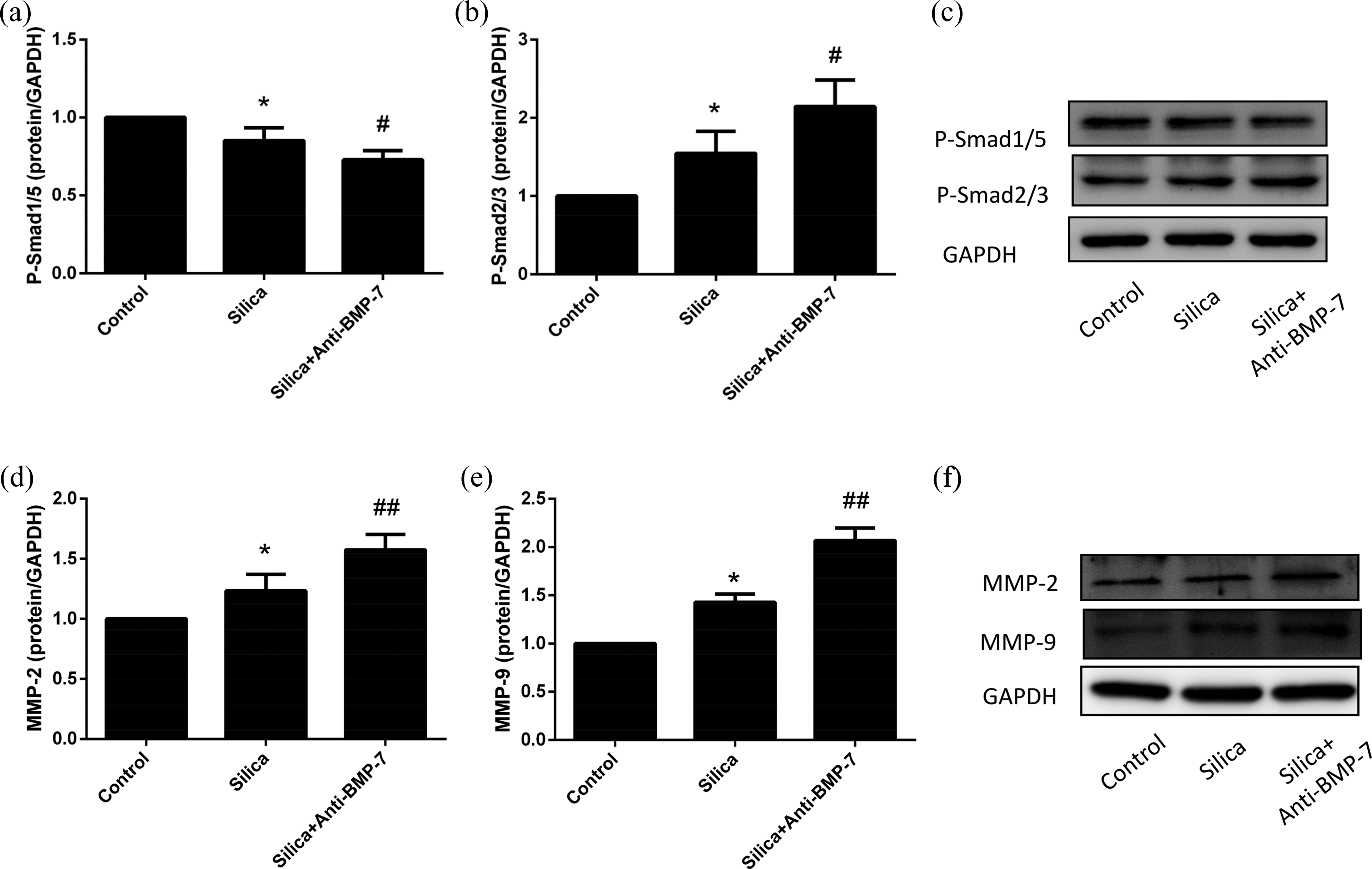
Effect of neutralizing BMP-7 on the Smad signaling pathway and MMPs induced by silica. Western blot showed that loss of BMP-7 further suppressed the expression of P-Smad1/5 (a) induced by silica and increased the expression of P-Smad2/3 (b), MMP-2 (c), and MMP-9 (d) in comparison with silica group. The data were presented as the means ± SD (n = 3), the significance of differences was determined by ANOVA followed by the Dunnett’s test. *p < 0.05 versus control group; #p < 0.05 versus silica group; and ##p < 0.01 versus silica group. BMP-7: bone morphogenetic protein-7; MMPs: matrix metalloproteinases; ANOVA: analysis of variance; SD: standard deviation.
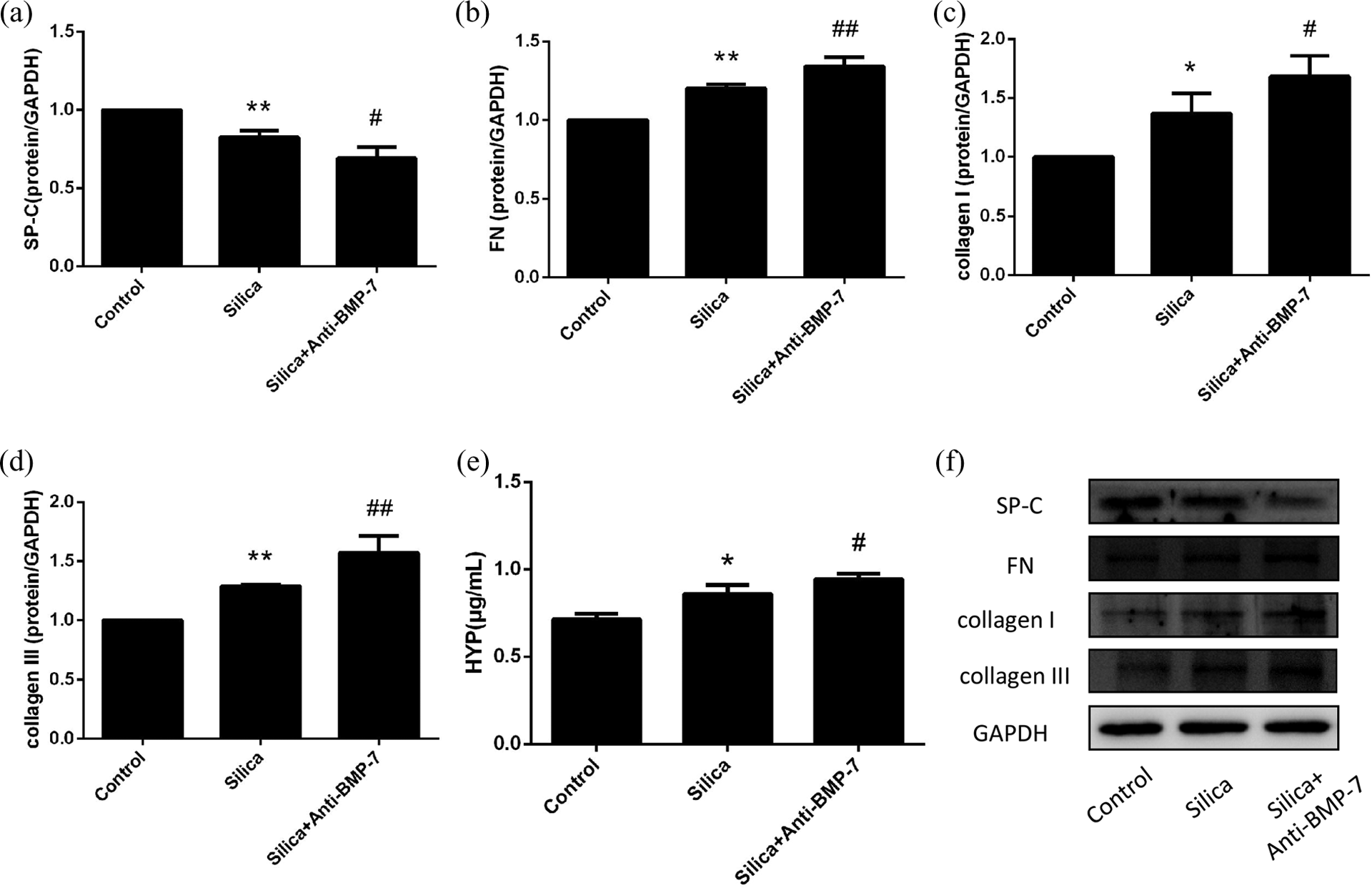
Effect of neutralizing BMP-7 on the silica-induced RLE-6TN cells. Western blot showed that the expression of FN (b), collagen I (c), and collagen III (d) were relatively higher in silica + anti-BMP-7 group than that in silica group. Western blot also showed that loss of BMP-7 further suppressed the expression of SP-C (a) induced by silica. In silica + anti-BMP-7 group, Hyp secretion into the culture medium was higher than that in silica group (e). The data were presented as the means ± SD (n = 3), the significance of differences was determined by ANOVA followed by the Dunnett’s test. *p < 0.05 versus control group; **p < 0.01 versus control group; #p < 0.05 versus silica group; ##p < 0.01 versus silica group. SP-C: surfactant proteins-C; E-cad: E-cadherin; Vim: Viminten; FN: fibronectin; BMP-7: bone morphogenetic protein-7; Hyp: hydroxyproline; ANOVA: analysis of variance; SD: standard deviation.
The Hyp level was increased in the silica group by approximately 1.24-fold (0.83 ± 0.08 μg/mL) in comparison to the control group (0.67 ± 0.06 μg/mL; p < 0.05). After anti-BMP-7-neutralizing antibody intervened, the Hyp level had significantly increased to 0.95 ± 0.03 μg/mL (p < 0.05; Figure 3(e)).
Neutralizing BMP-7 promoted silica-induced epithelial injury and MMPs upregulation in RLE-6TN cells
In order to examine whether BMP-7 has the ability to inhibit epithelial injury and MMPs expression, anti-BMP-7-neutralizing antibody was applied to RLE-6TN cells by 1 h before switching the cells to a 48-h treatment of RAW supernatant conditions. Then, the changes in the SP-C, MMP-2, and MMP-9 protein levels were assayed.
The expression of SP-C in silica group decreased significantly compared with that in the control group. In addition, it was efficiently suppressed by anti-BMP7-neutralizing antibody in comparison with the silica group (p < 0.05; Figure 3(a) and (f)).
In RLE-6TN cells, the addition of RAW supernatant induced an increase of MMP-2 and MMP-9 expressions in protein level, by approximately 1.23-fold and 1.29-fold, respectively (p < 0.05; Figure 2(d) to (f)). We also found that the loss of BMP-7 further upregulated both of them at protein levels in the presence of silica. The effects were characterized by higher levels of MMP-2 (1.28-fold) and MMP-9 (1.6-fold) in anti-BMP-7 groups compared with the silica groups (p < 0.01; Figures 2(d) to (f)).
Regulation of BMP-7-related pathways affects the expression of collagen I in RLE-6TN cells
Immunofluorescence staining revealed that collagen I is mainly expressed in the cytoplasm. This protein had low background value, and the expression significantly increased with induction of silica but was suppressed when SB-431542 was added. On the other hand, LDN-193189 and anti-BMP-7-neutralizing antibody could promote the expression of collagen I (Figure 4).
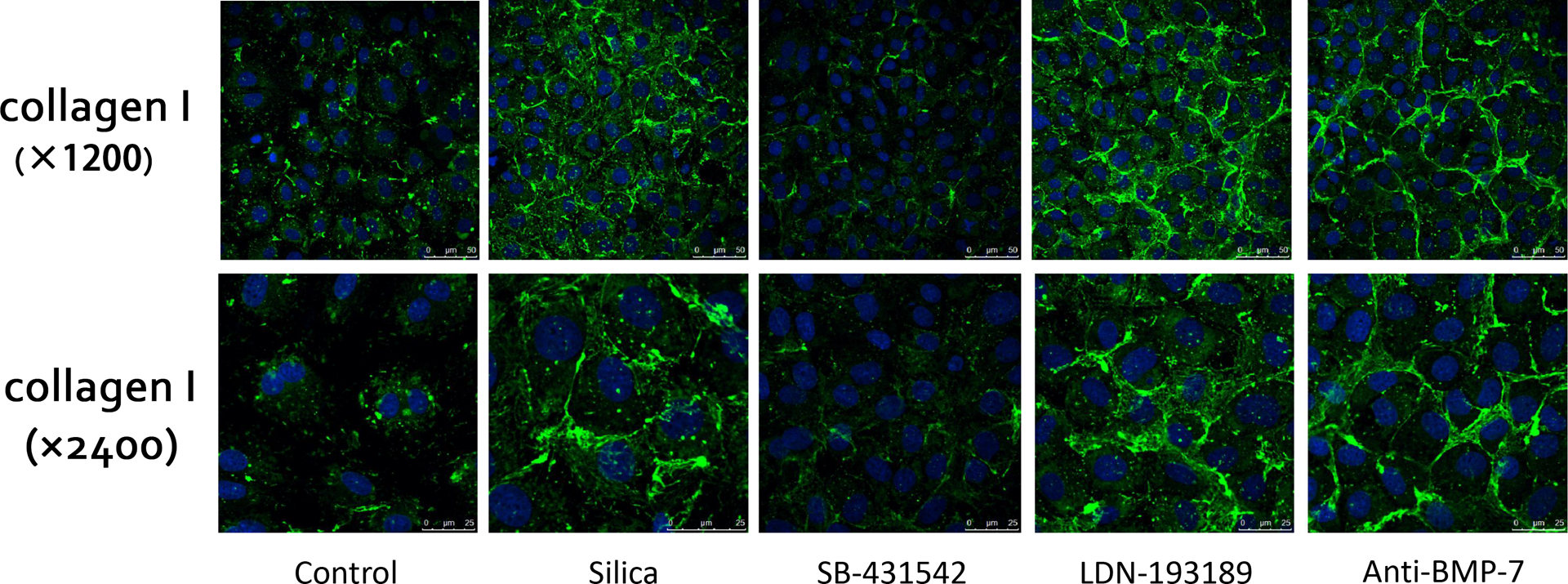
Effect of BMP-7 and related pathways on silica induced the expression of collagen I in RLE-6TN cells. Immunofluorescence staining revealed that collagen I was mainly expressed in cytoplasm. It was significantly increased following induction by LDN-193189 or anti-BMP7 neutralizing antibody yet decreased with SB-431542 intervention. BMP-7: bone morphogenetic protein-7.
Discussion
In our study, we developed a model of silica-induced EMT and fibrosis using culture supernatants of silica-treated RLE-6TN cells, a cell line stemmed from the rat normal alveolar epithelium, which simulated the interaction between macrophages and epithelium in vivo during silica-induced fibrosis. A critical step in the pathogenesis of silica-induced fibrosis was EMT, whereby cells phenotypically and functionally changed into myofibroblasts. The macrophages, activated by the ingestion of silica particles, released fibrogenic and inflammatory cytokines including TGF-β, 28 which stimulated pulmonary epithelium to produce collagen and Hyp via EMT. TGF-β has also been reported to induce EMT in cultured alveolar epithelial cells through Smad-dependent signaling, 29 which ultimately leads to the development of fibrosis.
Severe injury and retarded repair of the alveolar epithelium were sufficient to promote the fibrotic process. 30,31 Loss of key epithelial features might disturb the integrity of the alveolar epithelium, and thereby influenced their functionality. 5 They comprised aggregates of mesenchymal cells that underlay sites of unresolved epithelial injury and were associated with the progression of fibrosis. In addition to classic mesenchymal markers, the newly formed mesenchymal cells expressed the fibrillar collagen I and III, that were likely to contribute to excessive accumulation of ECM. SP-C was synthesized exclusively by alveolar type II epithelial cells and was also an important marker for lung epithelial cells. 5 Lack of SP-C or pro-SP-C directly links to the pathogenesis of lung diseases. 32 As expected, injured RLE-6TN cells with the associated loss of SP-C production were observed in the silica group.
Traditionally, MMPs are thought to be antifibrogenic due to their ability to degrade ECM proteins, 33 but some MMPs, particularly MMP-2 and MMP-9, have been recognized as promoters of epithelium EMT via basement membrane disruption. 34 MMPs degrade components of the ECM and have been observed to be increased in patients with fibrosing lung diseases. The upregulation of MMPs is often detected in fibrosing lung diseases during EMT processes. 17,18,35 MMP-2 and MMP-9 were upregulated by TGF-β in parallel with changes in EMT markers in both our and other researches. 36,37 In conclusion, silica promoted EMT and fibrosis in a MMP-dependent manner, which resulted in consistently decreased expression of SP-C.
Moreover, in this study, we investigated the potential effects of BMP-7 on a model involving silica-induced and macrophage-mediated fibrosis. As the most prominent member of the BMP family, BMP-7 has been reported to inhibit intestinal, renal, and cardiac fibrosis. 10,21,38 Loss of BMP-7 protein may play multiple roles in the pathogenic development of silica-induced fibrosis by driving EMT via Smad-mediated signaling or by suppressing epithelial cell proliferation. Our results suggested that the majority of cells performed a fibroblastic appearance, along with the increase of Hyp, FN, collagen I, and collagen III when pretreated with anti-BMP-7-neutralizing antibody. At the same time, the upregulated P-Smad2/3 indicated the activation of the TGF-β/Smad pathway, whereas the downregulated P-Smad1/5 indicated the inhibition of BMP/Smad pathway. Myllarniemi observed that BMP-7 exerted antifibrotic effects on a mouse model with asbestosis. 39 Our previous research has shown that, in an animal model with silicosis and silica-induced fibrosis in A549 and RLE-6TN cells, the addition of exogenous BMP-7 prevented EMT and subsequent fibrosis, suggesting that BMP-7 might be important in the fibrosis process from positive and negative aspects. 1,12,13,40 In addition, BMP/Smad signaling pathway has been found to be essential in animal lung injuries and human lung diseases, including silica-induced fibrosis, and it is important for the antifibrosis function of BMP-7.
Furthermore, dysregulation of MMPs may contribute to the pathogenesis of silica-induced fibrosis. The works of Perez-Ramos et al. showed an upregulation of MMPs in silica-exposed rats. 41 As initiators of this process, MMPs were upregulated during early stages of silica exposure. 42 In our and other researches, MMP-2 and MMP-9 are upregulated by TGF-β through signal mediators such as Smads and integrin-linked kinase. 43 As a key enzyme in lung interstitial fibrotic processes, the elevation of MMP-9 has a higher specificity. 2 Monitoring MMPs seems a legitimate target with the aim of inhibition of fibrogenesis. 44 Inhibition of BMP-7 often corresponds with elevated expression and activation of MMPs. BMP-7 may prevent the silica-induced fibrosis by inhibiting collagen synthesis and MMPs activation.
Conclusion
Alveolar epithelial injury appears to be a key pathological feature of silica-induced fibrosis, which results in the alterations in cell morphology, the loss of epithelial cell properties and function, and the acquisition of mesenchymal characteristics. Furthermore, MMPs suggest an EMT process in cells induced by silica. BMP-7 may be helpful in the restoration or maintenance of an epithelial phenotype, suppression of EMT findings, and inhibition of silica-induced fibrosis via BMP/Smad signaling pathway in MMP-dependent manner.
This study finds out the relationship between BMP-7 and silica-induced fibrosis and finds a practicable strategy to inhibit or delay the interstitial process induced by silica. It will provide the theoretical basis to the application of BMP-7 on silicosis.
Footnotes
Author Contribution
Di Liang and Guoliang An have contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants of National Natural Science Foundation of China (no. 81273047) and Key Projects of Science and Technology Program by Beijing Municipal Education Commission (no KZ20160025020).
