Abstract
Introduction:
Despite an increasing awareness that the activity of excitable membranes is determined by the underlying ionic gradients across them, and their importance in drug dependency, we were not able to identify any reports of comparing the electrolyte composition of opioid-dependent and non-addicted controls.
Methods:
Linear regression was used to compare clinical pathology blood results taken from 2699 opioid-dependent patients (ODP) and 5307 medical control (MC) patients on a total of 21,734 occasions for the period 1995–2015. The presence of a hepatitis C antibody test was used to separate OPD and MC patients.
Results:
The mean age among ODP and MC was 33.51 ± 0.16 and 37.99 ± 0.23 years, respectively (p < 0.0001). The groups were 71.5% and 54.2% male (p < 0.0001). Drug use in this cohort has been reported previously. Analysis of sodium, haemoglobin and albumin were used to exclude marked effects of haemodilution/haemoconcentration. Repeated measures linear regression against age and time showed depressed levels of bicarbonate (p < 0.0001) and potassium (p < 0.05) and elevated levels of chloride (p < 0.025) and anions (p < 0.01) in ODP in both sexes. Multiple regression in mixed-effects models showed that these effects were all worse in females (p = 0.0001).
Conclusion:
This data shows that opioid dependence is associated with significant changes in chloride, potassium, bicarbonate and anions in both sexes, and worse in females. This likely has implications for the electrophysiological properties of excitable membranes. It is consistent with the reported impairment of potassium-chloride exchangers in opioid dependence. Explication of the mechanisms responsible must await further studies.
Introduction
The multiple roles of the ion transporters which establish the transmembrane ionic gradients upon which membrane excitation depends have become more evident in recent times. These transmembrane ionic gradients are determined by the activity of the membrane ion pumps, and also the associated passive ion exchangers, antiporters and symporters which set the basal ionic gradients. 1 –4
Numerous changes have been described in opioid dependence. An expansion of the tissue extracellular fluid has long been known, 5 which has been attributed to an opioid-dependent increase in antidiuretic hormone. 6 –8 One of its features on clinical pathology testing is haemodilution. 5 It is likely that the elevated immune environment of opioid dependence, including higher levels of circulating cytokines, 9 has a major effect upon myelopoiesis. 10 Similarly, dehydration can occur during opioid withdrawal, 7 which would be marked by a relative increase in sodium, albumin and haemoglobin. Changes in the circulating levels of calcium and phosphate have been noted previously by our group. 11
Our literature search has not identified prior reports of changes in the serum electrolytes in opioid dependence. It should be noted that the blood–brain barrier in both the brain and the choriod plexus is a significant barrier to the direct impact of changes in the serum on central nervous system extracellular milieu. 12
Changes in the activity of some of the key membrane ion transporters in the brain and the ventral tegmental area in opioid dependence have recently been elucidated. 12 –14 We were, therefore, surprised when comparison of the serum ion levels obtained in longitudinal studies from our patient cohort displayed differences between opioid-dependent and non-dependent groups. This report then is an observational study describing these changes in clinical opioid dependence in the hope that further work might elucidate the mechanisms responsible, and also the possible significance of these findings.
Patient selection
Patients were not selected. The data presented represents the totality of our clinical pathology data set which has been analysed by Queensland Medical Laboratory (QML). Patients were assigned to either the medical control (MC) group or the drug-dependent group based upon whether or not they had had serology performed for Hepatitis C antibody. In our clinic, this test is only performed on drug-dependent patients, so it is a useful marker for the drug-dependent state in our cohort. Blood was taken as clinically indicated for patient care in the course of their routine clinical care.
Pathology analysis
Pathology testing was all performed by QML. QML is accredited by the National Association of Testing Authorities Australia to the Australian Laboratory standard AS-15189. QML is also accredited to the international standard ISO 9001, the international laboratory clinical standard.
Statistics
The pathology results were downloaded as an Excel spreadsheet from QML for the period 1995–2015. Categorical data were compared in EpiInfo 7.1.4.0 from Centres for Disease Control in Atlanta, Georgia, USA. Bivariate statistics were compared by categories and groups in Statistica 7.1 from Statsoft, Oklahoma, USA. All t tests were two tailed. Continuous data was compared in ‘R’ version 3.2.3. Multiple regression was performed and graphs were drawn in R. R was downloaded from the Cloud source of Central R Archive Network mirror. R packages Base, nlme, Reshape and gglot2 were utilized. Linear regression was performed by the classical method with deletion of the least significant term until only significant terms remained. Variables were log transformed to improve the assumptions of the normal distribution as indicated by the Shapiro test. Linear regression compared the parameter of interest against the age and addictive status. Mixed-effects repeated measures statistical equations were used for time-dependent measures. Mixed-effects regression models compared the parameter of interest against age, time and addictive status. Random effects were unity and the unique patient identifier. A p value <0.05 was considered significant.
Ethics
The study was given ethical approval by the Human Research Ethics Committee of the South City Medical Centre which is registered with the National Health and Medical Research Centre. The study was conducted in accordance with the Declaration of Helsinki.
Results
Of the 8006 patients, 2699 were opioid dependent and 5307 were not. These groups were categorized as opioid dependent and MCs, respectively. These patients were studied biochemically on 4779 and 16,955 occasions.
The age of the two groups was 33.51 ± 0.16 and 37.99 ± 0.23 years, respectively (Student’s t = 15.77, degree of freedom (df) = 8001.6, p < 0.0001). Among the males the mean ages of the opioid dependent and MCs was 34.14 ± 0.09 and 38.95 ± 0.31, respectively (t = 13.17, df = 4557.7, p < 0.0001) and among females the mean ages were 31.94 ± 0.29 and 36.87 ± 0.36, respectively (t = 10.8, df = 2762.9, p < 0.0001). There were 1929 (71.47%) and 2875 (54.17%) males in the opioid-dependent and MC groups, respectively (χ2 = 221.99, p < 0.0001). These major differences are shown in Table 1.
Bivariate comparisons.

SEM: standard error of the mean.
Drug use and other demographic data are not available for this population as it is derived directly from our clinical pathology database. Drug use has been reported previously from this cohort in other reports, 15 –18 and is broadly comparable with other opioid-dependent cohorts reported nationally and internationally.
Two major possibilities potentially exist for explaining any ionic disequilibrium between opioid-dependent and non-dependent populations. They are withdrawal-associated dehydration, and the syndrome of inappropriate antidiuretic hormone secretion (SIADH) which is well described in opioid dependence. 6,7 For this reason, the bivariate comparison between the two groups indicated in Table 1 is presented in two sections. The first section compares three cardinal biochemical and haematological parameters of dehydration in each sex, namely sodium, haemoglobin and albumin. The lower half of the table shows bivariate differences in ionic composition.
These data indicate that the opioid-dependent and control groups were not comparable by age or sex. The age problem was addressed by multiple regression, and the sex differences were addressed by the analysis of each sex separately.
Figures 1 and 2 present the three indices of haemoconcentration by sex as a function of chronological age and of time, respectively. No clear pattern indicative of either haemoconcentration or SIADH emerges from these graphical analyses.

Haemoconcentration by age.

Haemoconcentration by time.
Figures 3 and 4 present the data for selected key serum electrolytes by chronological age and by time. Significant changes of bicarbonate with age, with the opioid-dependent group being more acidotic, and of potassium, chloride and bicarbonate with time are seen in both sexes.

Selected serum ions by age, dependency status and sex.
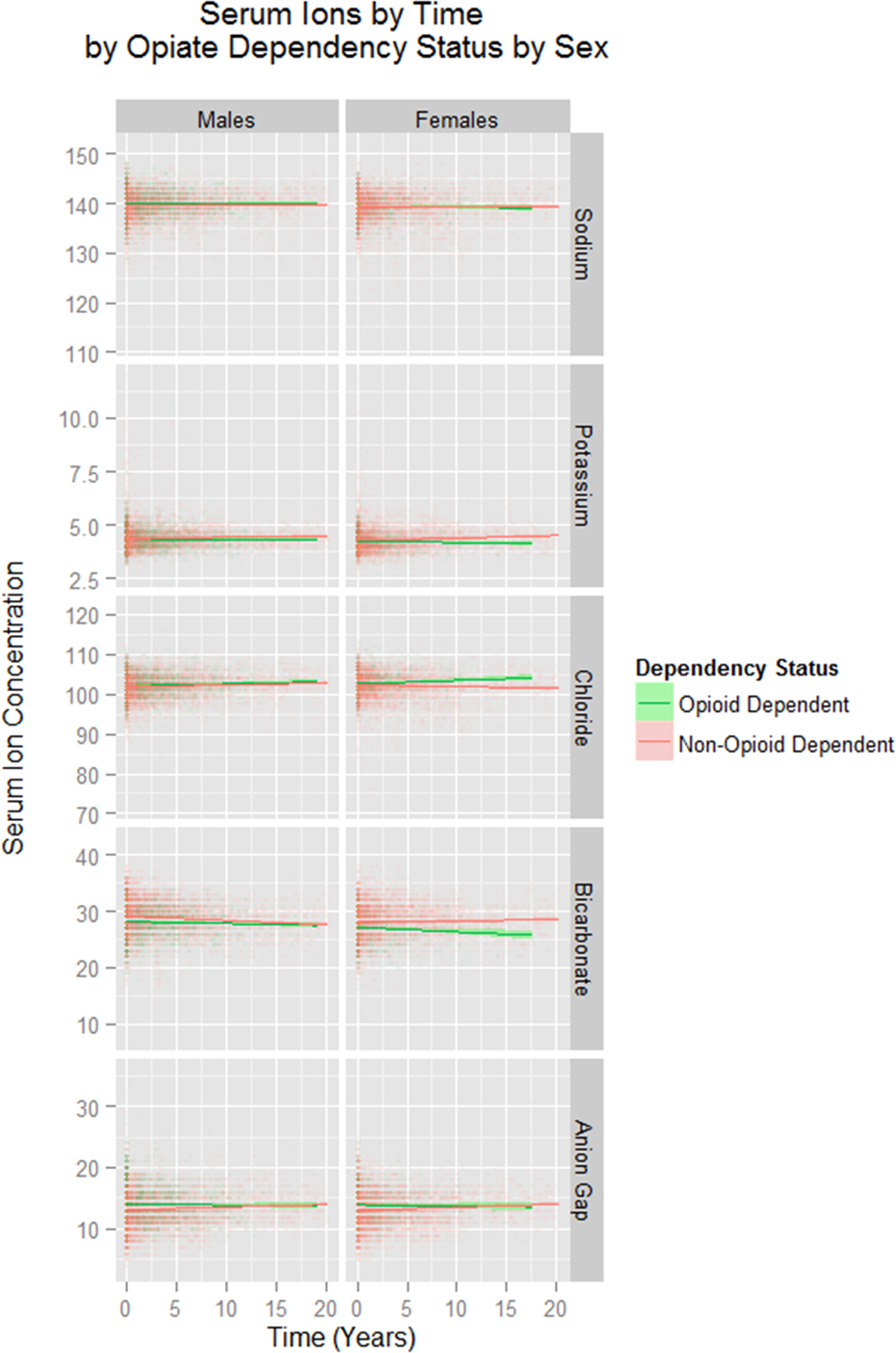
Selected serum ions by time, dependency status and sex.
Table 2 lists the significant terms which emerge from linear regression of the various parameters against age, where the addictive status is significant and quantifies these changes between groups. Among males, the haemoglobin is slightly raised and the albumin is slightly lowered and sodium is unchanged. Among females while the sodium is unchanged, the haemoglobin and albumin are both significantly higher. Changes in the serum ions are also shown.
Linear regressions: cross-sectional studies.

Table 3 lists similar results for both haemoconcentration and ionic composition. This table reports the output of repeated measures mixed-effects models including time, chronological age and addictive status. It thus summarizes all the information which has been presented. Among males, haemoglobin changes are significant but not sodium or albumin. Comparison of Figures 1 and 2 shows that the relationship among males of the haemoglobin opioid regression line with the control regression line is different with age and time. Similar comments also apply in females. Changes in albumin among females are quantitatively small.
Repeated measures random effects: longitudinal studies.

Among the ionic changes in males, bicarbonate and potassium are lower and chloride and anions are higher both over time and by age. Among females, potassium and bicarbonate were lower and chloride and anions higher as a function of both age and time.
It is also clear from Figures 3 and 4 that the changes reported, although statistically significant, are generally not of large size. However, this generalization is not true for the changes seen in bicarbonate in females over time. This data was interrogated in a mixed-effects model against time (Akaike Information Criteria (AIC) = 38,844.69, Bayesian Information Criteria (BIC) = 38,886.46, log-likelihood = −19,416.35). Consideration of this model shows that after 20 years the modelled mean bicarbonate concentration among MC was 27.72 mmol/L and among opioid-dependent patients (ODP) was 25.63 mmol/L which represents a 2.09 mmol/L difference or 7.53% reduction in the opioid-dependent group.
An analysis has also been performed of the relatively abnormal results for the three ions of principal interest, potassium, chloride and bicarbonate. The detailed data are presented in Supplementary Tables 1 and 2 for males and females, respectively. Data are presented based on Figure 4 which suggests that potassium and bicarbonate tend to be elevated and chloride depressed by opioid dependence. Therefore, these tables list the abnormal results in the direction of the indicated abnormality. The tables do not list results which are abnormal in the direction opposite to the apparent main trend. The tables, therefore, do not sum to the total sample sizes. As indicated in the tables, in each case the opioid-dependent group has fewer abnormal than the MC group. The fourth column of these tables presents the applicable χ 2 test for the categorical assessment of each ion. For males, the overall corrected Mantel–Haenszel (MH) χ 2 in all groups was 88.77, p < 0.0001, adjusted MH odds ratio (OR) 0.54 (95% confidence intervals [CI] 0.47–0.61). For females, the comparable summary statistics were corrected χ 2 = 65.78, p < 0.0001, adjusted MH OR 0.39 (95% CI 0.31–0.39). If all abnormal results are considered, this result is unchanged (data not presented).
Consideration of Figures 3 and 4 suggests that the changes were more significant in females. This was confirmed on multivariate mixed-effects regression when the interaction between potassium, chloride, bicarbonate and anions was regressed against age, time, addictive status and sex, and gender emerged as a highly significant variable (p = 0.0001, estimate = −8321.1, SE = 2133.63, df = 12,053, t = −3.9286; model parameters, AIC = 419,951, BIC = 420,031, and log-likelihood index = 209,962).
Discussion
These data from a large and 20-year longitudinal data set show a slight reduction of serum bicarbonate and potassium and elevation of chloride and anions in opioid dependence as a function of age and of time, which is worse in females. These changes imply a metabolic acidosis with reciprocal changes in other major anions and cations. There was for the most part no change in circulating sodium or albumin levels.
These changes are not accounted for by either the SIADH, which is known to occur in opioid dependence, or the dehydration which accompanies opioid withdrawal. While there was some evidence for haemoconcentration in both cross-sectional and longitudinal studies, the direction of these effects was not uniform between the sexes or between the time and age analyses, so that this can be largely discounted as an explanation for the observed effects. Interestingly, it has been previously shown that arginine vasopressin acting through luminal V1 receptors in the endothelium of the blood–brain barrier in a calcium-dependent manner can alter sodium–potassium–chloride co-transport acutely with an onset within minutes and lasting for several hours. 19
As this is an observational rather than a mechanistic study, it is not possible to state what the likely mechanism for these described effects. It is interesting to note that addiction has increasingly been noted to be accompanied by a relative impairment of the potassium–chloride exchanger of the brain KCC2, which underlies impeded activity of both inhibitory signalling by γ-aminobutyric acid (GABA) at the GABA(A)R 1 –4 and downstream altered processing of hedonic reward by the limbic system with rostral signalling from the ventral tegmental nucleus into the nucleus accumbens rather than the usual caudal signalling into the tegmentopontine nucleus. 13,14,20,21 KCC2 controls the transmembrane concentrations of potassium and chloride and effects on bicarbonate and protons are secondary to these actions. Hence, the findings in the present study would be consistent with what has recently been proposed as an important unifying theory of the electrophysiology of addiction.
Having said that KCC2 is only expressed in the brain. If the described changes are due to dysfunction of the ion exchangers, or any other membrane pumps, they must be acting organism wide including in the kidney, to be demonstrable at the organismal level as has been demonstrated in this study.
Normal membrane excitability is dependent upon tight control of transmembrane ionic gradients. The demonstrated changes in potassium, chloride and bicarbonate shown here imply an alteration of membrane excitability which may potentially have numerous downstream implications. In this regard, it is important to note the interposition of the blood–brain barrier in both the brain and the choroid plexus as a major regulator of the ionic milieu of the extracellular space of the central nervous system. 8
These changes are reminiscent and build upon those described previously in opioid dependence by our group for calcium and phosphate. 11
Most of the changes described are not large relative to the values seen among control patients. However, the reduction in the modelled level of bicarbonate among females after 20 years was 2.09 mmol/L, representing a 7.53% reduction from the control value of 27.72 mmol/L.
It was of interest that significantly less ODP had results outside the normal quoted laboratory reference range. The explanation for this must await further studies.
The observation that these changes were largely worse in females is of interest. Work from our own group 18 and from others 22,23 has similarly found that females seem to be more severely affected by opioid dependence. The biological basis for this is not understood. It is possible that it may have a hormonal, an immune, a genetic or epigenetic basis. Further explication would require additional mechanistic studies. This also has implications for future research in that many studies of opioid users, using opportunistic samples, have a larger proportion of male participants. Sampling methodologies should ensure that females are adequately represented so that studies are sufficiently powered to identify possible gender effects. The present study did not have data on blood pH.
In summary, we have observed significant changes in serum chloride, bicarbonate, anions and potassium levels in opioid dependence, particularly in females, which are not accounted for by known syndromes seen in drug dependency. While their mechanism is not known, they would be consistent with described impairment of ion exchangers in opioid dependence, albeit they imply an organism-wide effect not limited to the central nervous system.
Footnotes
Authors Contributions
ASR designed the study, treated the patients, conducted the PWA studies, analysed the data and wrote the initial draft of the article. GKH gave advice on study design, and data analysis, wrote the article and assisted with literature review. AN assisted with literature review and wrote the article.
Data Access
Access to data will be considered upon request to the authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
