Abstract
Postmenopausal patients with breast cancer have two options for adjuvant endocrine therapy, tamoxifen and aromatase inhibitors (AIs) as well as radiotherapy (RT) and chemotherapy. However, there is limited data regarding the optimal sequencing of RT and tamoxifen/AIs. Thus, we aimed to evaluate the effects of tamoxifen and AIs on radiation-induced cardiotoxicity. Eighty ovariectomized rats were divided into eight groups (G). G1 was defined as a control group; G2, G3, G4, and G5 were RT, tamoxifen, anastrozle, and letrozole groups, respectively; G6, G7, and G8 were RT plus tamoxifen, anastrozle, and letrozole groups, respectively. Drugs were started 1 week before RT and continued until the animals were killed 16 weeks after RT. The heart tissues were then dissected and examined with light microscopy to determine endocardial thickness and cardiac fibrosis. The endocardial thickness scores of both RT alone and the tamoxifen groups as well as the cardiac fibrosis score of RT alone were higher than that the control group (p < 0.05 for all). There was no difference in the endocardial thickness and cardiac fibrosis scores of the RT-only group and the RT plus hormonotherapy groups (p > 0.05 for all). Concurrent administration of RT and hormonal therapy with either tamoxifen or AIs did not further amplify radiation-induced cardiac toxicity. This issue warrants further study.
Introduction
Adjuvant therapies for the treatment of breast cancer, including radiotherapy (RT), chemotherapy (CT), targeted therapy, and hormonal treatment, significantly decrease the risk of recurrence and increase patient survival. However, cardiovascular problems such as the treatment complications caused by many of these treatment modalities are responsible for most common causes of noncancer mortality in breast cancer survivors. 1 Until now, few studies have examined treatment-related cardiovascular disease (CVD) in breast cancer patients, 2 –5 and the results that do exist are inconsistent. How the changes that occur in breast cancer might affect the presence of CVD in this population is not fully understood. Because these studies examined cardiovascular mortality rather than morbidity 2,3 or included few patients, 4 it remains unclear whether patients treated with RT for breast cancer have an increased risk of CVD. Cardiovascular injury is thought to be due to the generation of reactive oxygen species, 6 but there is no definitive information indicating why individuals with breast cancer are susceptible to CVD.
Postmenopausal patients with breast cancer have two options for adjuvant endocrine therapy, tamoxifen and aromatase inhibitors (AIs) as well as RT and systemic CT, which are usually given sequentially. Several agents used in breast cancer treatment are radiosensitizers, and the concurrent administration of drugs such as doxorubicin and methotrexate is associated with an increased risk of radiation complications. 7 On the other hand, there is limited evidence of the radiosensitizing properties of hormonal therapies, either with tamoxifen or AIs. To the best of our knowledge, tamoxifen has no significant radiosensitizing properties; therefore, it is often given concurrently with radiation. However, tamoxifen is a cytostatic drug that arrests cells in G1, a relatively radioresistant cell cycle phase. 8 The radiosensitizing property of letrozole was also observed in a study using a human breast cancer cell line that was stably transfected with the human aromatase gene. 9 In that study, it was found that letrozole inhibits cell proliferation by invoking a transition delay or by blocking the G1 phase of the cell cycle. In addition, the combination of radiation and letrozole produced a significant decrease in radiation-induced G2 phase arrest and a decrease of cells in the S phase, with cell redistribution in the G1 phase, which explains the radiosensitization effect of letrozole. 9 To the best of our knowledge, there is no study evaluating the radiosensitizing profile of anastrozole.
The potentially increased risk of adverse effects in patients receiving concurrent hormonal therapy and RT is a concern. Both tamoxifen and AIs have been shown to increase the risk of venous thrombosis, 10,11 but it remains controversial whether tamoxifen 12 –15 and AIs 16 –19 have positive, 12 negative, 15 –18 or no effects on cardiovascular events. 19 Given the widespread use of adjuvant hormonal therapy, it is important to assess the safety and efficacy of RT and these systemic agents relative to their sequence of administration. However, there is limited data regarding the optimal sequencing of RT and tamoxifen/AIs. Therefore, we conducted this experimental study to determine the effects of tamoxifen and AIs on radiation-induced cardiac toxicity.
Methods and materials
Study design
The study included 80 virgin female Wistar-Albino rats (250–300 g), the use of which was approved by the Necmettin Erbakan University Animal Care and Use Committee. The animals were randomly stratified into 8 groups with 10 rats in each group. The animals were maintained under a controlled temperature (25 ± 2°C) with a 12-h light/12-h dark cycle. They were fed standard laboratory diet and water ad libitum.
The rats were anesthetized with 90 mg/kg ketamine hydrochloride and 10 mg/kg xylazine administered intraperitoneally and ovariectomized using a bilateral dorsal approach. No procedure-related deaths were reported in the groups. There were no significant differences in the rats’ body weight between groups before the operation or at the end of the study. The rats were divided into 8 equal groups (G) composed of 10 animals. G1 was defined as the control group, and the rats in this group were sham irradiated. G2 was an RT-only group. Groups 3, 4, and 5 were tamoxifen, anastrozole, and letrozole groups, without RT, respectively. Groups 6, 7, and 8 were RT plus tamoxifen, anastrozole, and letrozole groups, respectively (Table 1). Tamoxifen, anastrozole, and letrozole were started 1 week before the RT and continued until the animals were killed 16 weeks after the RT. As an end point, the extent of cardiac fibrosis and endocardial thickness for each rat was quantified with image analysis of histological sections of the heart.
Abbreviations used for the study groups.

RT: radiotherapy.
Irradiation and drug protocols
RT was applied under general anesthesia with 90 mg/kg ketamine hydrochloride and 10 mg/kg xylazine administered intraperitoneally. A single dose of 12 Gy, which was shown to be a sufficient dose to determine cardiac fibrosis in Wistar-Albino rats in our previous study, 20 with 6 MV photon beams was applied via a single anterior field to a depth of 2 cm using the source–axis distance technique. A 1 cm elasto-gel bolus was used to build up the radiation dose and to provide contour regularity. The field size was 4 cm × 4 cm and included the mediastinum.
Tamoxifen, anastrozole, and letrozole equivalent to 20 mg, 1 mg, and 2.5 mg adult doses, respectively, were calculated for each rat and administered orally with a feeding tube once daily, including weekends, until the animals were killed at the end of 16th week after the RT. These doses were shown to be effective in previous experimental studies of Wistar-Albino rats. 21,22 In order to achieve adequate plasma concentration of the drugs, they were started 1 week prior to the RT.
Morphologic study and light microscopy
The animals were anesthetized with 90 mg/kg ketamine hydrochloride and 10 mg/kg xylazine administered intraperitoneally and killed by cervical dislocation 16 weeks after the start of irradiation. The hearts were excised and fixed in 10% formaldehyde solution and embedded in paraffin for light microscopic examination. The slices obtained were stained with hematoxylen and eosin (H&E) to evaluate inflammation, and then with immunohistochemical triple staining to identify cardiac fibrosis. As a quantitative end point, the extent of radiation-induced fibrosis was graded on a scale of 0–3 (0: normal heart; 1: minimal fibrosis; 2: moderate fibrosis; and 3: severe fibrosis). Using the digital images obtained from the selected areas by light microscopy (Nikon, Labophot-2, Japan) at 20× magnification, the thickness of the endocardium was measured using the BAB system (BAB Bs200 Pro Image Analysis Software, BAB Yazılım Donanım Mühendislik Medikal San. ve Tic. Ltd. Sti. Ankara, Turkey). The pathologist was not aware of the treatment groups at the time of the histological examination of the specimens. After examining the whole sections of each rat, the average value was taken as the fibrosis score and mean values of the groups were calculated. Additionally, the endocardial thicknesses and cardiac fibrosis scores of all groups were compared.
Statistical method
The SPSS version 16.0 was used for statistical analyses. As the pathological scores were ordinal in nature, the differences in pathological findings between the study groups were analyzed using the Kruskal–Wallis test. When an overall statistically significant difference was observed, pairwise comparisons were performed using the Mann–Whitney U test. Bonferroni correction was used for multiple comparisons. A 5% type-I error level was used for the statistical significance cutoff for overall comparisons.
Results
Endocardial layer thickness
There was a significant difference among the study groups with respect to the endocardial thickness scores (p < 0.001; Table 2). Histological examinations of the heart sections revealed that endocardial thickness was significantly increased in all the RT groups compared to the control group (p values were <0.001). Severe fibrinous exudate was also prominent in RT groups. Adding hormonal treatments to RT (G6, G7, and G8) did result in a further increase in endocardial thickness compared to the RT-only group (G2).
Endothelial thickness values of each study group.

In terms of hormonal treatments, tamoxifen, even used alone, caused deterioration in inflammatory cells and endocardial thickness compared to the control group (p < 0.001). H&E staining showed prominent hemosiderin-laden macrophages in the tamoxifen-only group. In addition, tamoxifen itself led to fibrinous exudates. There were some spaces between atrophic myocytes in the RT groups as well as in the tamoxifen-only group. The administration of anastrozole and letrozole alone did not cause a significant change in endocardial thickness (p values were 0.14 and 0.10, respectively; Table 3 and Figure 1).
Comparison of the control group and combination groups with only RT or only HT groups according to endocardial thickness

a p < 0.05.

H&E staining: section from the hearts of the rats from (a) control group, (b) radiotherapy-only group, (c) tamoxifen-only group, (d) radiotherapy + tamoxifen group. Normal cardiac structure (×20, H&E). There is moderate fibrosis under the endocardium. Endocardium is thickened by connective tissue. Severe fibrinous exudate is prominent. There are some spaces in between atrophic myocytes (×20, H&E). There are prominent hemosiderin-laden macrophages. The number of inflammatory cells is increased. Endocardium is thickened. There is moderate fibrinous exudate. There are some spaces in between atrophic myocytes (×40, H&E). There is severe fibrosis under the endocardium. Endocardium is thickened by fibrotic connective tissue. There are many spaces in between atrophic myocytes (×20, H&E). H&E: hematoxylen and eosin.
Fibrosis scores
We found a statistically significant difference between the study groups in terms of cardiac fibrosis (p < 0.001; Table 4). Pairwise comparisons revealed that there was a significant difference in the cardiac fibrosis scores between the control and RT-only groups (p value for G1–G2 was <0.001) as well as the hormonal treatment-only groups (G3, G4, and G5) and RT plus hormonotherapy groups (G6, G7, and G8; p values were <0.001 for all). Again, concomitant RT and hormonotherapy did not result in significant changes in fibrosis caused by the RT-only group (Table 5). There were fibrotic bands extending through the myocytes in all the RT groups. The most prominent fibrosis was observed in the tamoxifen plus RT group (G6; Figure 2).
Cardiac fibrosis score values of each study group.

Comparison of the control group and combination groups with RT- or HT-only groups according to cardiac fibrosis.

RT: radiotherapy; HT: hormonotherapy.
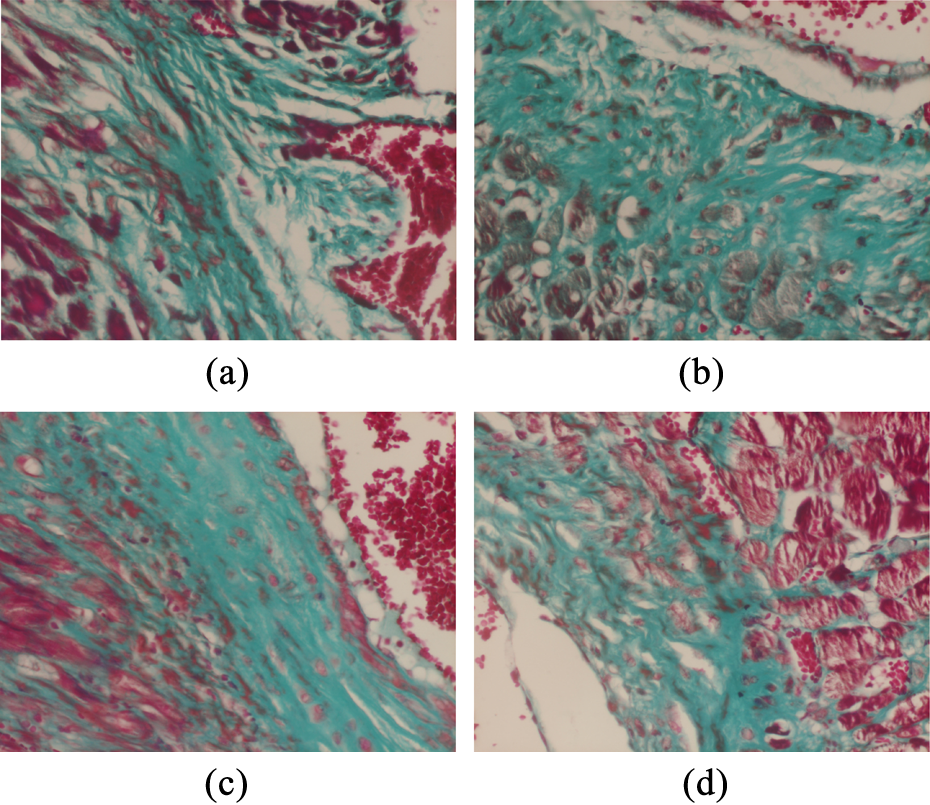
Immunohistochemical triple staining (×20): section from the hearts of the rats from (a) RT-only group, (b) RT + tamoxifen group, (c) RT + anastrozole group, and (d) RT + letrozole group. (a) There is moderate thickening in the endocardium. There is fibrosis in between the myocytes. Myocytes are degenerated. (b) There is extensive fibrosis and fibrotic band are extending through the myocytes. (c) There is moderate thickening in the endocardium. There is fibrosis in between the myocytes. Myocytes are degenerated just like the RT-only group. (d) There is moderate thickening in the endocardium. There is moderate fibrosis and the fibrotic bands are extending through the myocytes. Myocytes are degenerated just like the RT-only group. RT: radiotherapy.
Discussion
In the current study, we demonstrated that the concomitant use of RT and hormonal therapy either with tamoxifen or AIs (anastrozole and letrozole) did not increase cardiac fibrosis and endocardial thickness scores. Both the cardiac fibrosis and endocardial thickness scores were higher in the tamoxifen plus RT group when compared to the AIs plus RT groups; however, these differences were not statistically significant. Tamoxifen, even used alone, led to an increase in inflammatory cells and endocardial thickness compared to the control group. The use of AIs did not deteriorate the fibrosis or endocardial thickness in the rats’ cardiac tissue.
Microcirculatory damage appears to be a common pathophysiologic pathway of damage to the heart. Steward and Fajardo demonstrated that damage to the myocardium develops through three phases of injury. 23 The acute inflammation phase occurs about 6 h after RT, and a neutrophilic infiltrate develops involving all layers of heart. The second phase, also known as the latent phase, is characterized by a slight progressive fibrosis that begins about 2 days after exposure. However, electron microscopy of the myocardial capillary endothelial cells reveals progressive damage leading to obstruction of the lumen and thrombi of fibrin and platelets. Although healthy endothelial cell replication occurs in the vicinity, it is generally inadequate, and ischemia inevitably leads to progressive fibrosis. The animals begin to die on approximately the 70th day due to extensive fibrosis. Indeed, the hallmark of this late stage is extensive fibrosis. 23,24 Cardiac fibrosis, which is the focus of the current study, is a normal healing response to the scarring process in cardiac tissue.
Estrogen can impact cardiovascular health and disease directly through effects on vascular cells and cardiomyocytes or indirectly through systemic effects. Estrogen can modulate vascular function, inflammatory response, metabolism, insulin sensitivity, cardiac myocyte and stem cell survival, and the development of hypertrophy, 25 as well as improving vascular function, reducing atherosclerosis, 26 and inhibiting the development of cardiac fibrosis. 27,28
It is estimated that hormonal therapies with agents that cause a decrease in estrogen levels or those acting on estrogen receptors could have some effects on the cardiovascular system and CVD. Moreover, when hormonal therapies are used with concomitant RT, they may act as radiosensitizing agents, which may eventually potentiate both the effects and the side effects of RT. The radiosensitizing properties of tamoxifen 8 and letrozole 9 were studied; however, there are no data with respect to the radiosensitizing profile of anastrozole. Tamoxifen is a cytostatic drug that arrests cells in G1, a relatively radioresistant cell cycle phase. 8 On the other hand, it was shown that the combination of radiation and letrozole produced a significant decrease in radiation-induced G2 phase arrest and a decrease of cells in the S phase, with cell redistribution in the G1 phase, which explains the radiosensitization effect of letrozole. 9
Despite this evidence, the cardiovascular effects of tamoxifen and AIs remain unclear. Clinical studies concerning the cardiotoxicity profiles of both tamoxifen and AIs have conflicting results. In fact, these studies were not designed to evaluate cardiotoxicity as a primary end point. Additionally, most of these studies failed to address the cardiac complications of hormonal treatments other than CAD, such as cardiomyopathies, valvular heart disease, and arrhythmia. The studies comparing tamoxifen and AIs as adjuvant hormonal treatment for breast cancer patients have a problem with regard to interpreting their results on the cardiac side effects of AIs. In these trials, AIs were compared with tamoxifen, not with a placebo. Therefore, it is impossible to determine the pure cardiotoxic profile of AIs. Limited preclinical data on tamoxifen-related accelerated atherosclerosis and CAD are available. 29 In an experimental study, Cihan and colleagues investigated whether RT plus trastuzumab, a humanized anti-ErbB2 antibody that has proven to be an essential tool in the immunotherapy of breast cancer, followed by subsequent hormonotherapy either with tamoxifen or AIs (letrozole, anastrozole, and exemestane), increase the cumulative toxic effect on cardiac functions in rats. 22 The authors concluded that there was no additional cardiotoxicity of adjuvant hormonotherapy following concomitant RT and trastuzumab treatment compared to RT with respect to cardiac fibrosis. In the current study, we also demonstrated that concurrent administration of RT and hormonal therapy using either tamoxifen or AIs did not further amplify radiation-induced cardiac fibrosis. Therefore, our results were consistent with the findings of Cihan and colleagues. Additionally, we found that tamoxifen increased endocardial thickness, likely by infiltration of inflammatory cells, but it did not cause an increase in cardiac fibrosis scores. AIs did not cause such effects.
The concomitant hormone-radiotherapy (CO-HO-RT) study was a phase II randomized trial evaluating whether letrozole given concomitantly or sequentially after breast-conserving surgery and adjuvant RT altered radiation-induced side effects and cosmetic outcomes. 30 This trial was supported by the fact that letrozole showed radiosensitizing effects on breast cell lines in vitro. 9 Based on long-term follow-up, the authors demonstrated that letrozole could be safely delivered concomitantly with adjuvant breast RT. 31 Moreover, the authors reported no cardiac toxicity. In our study, we also demonstrated that letrozole did not deteriorate radiation-induced cardiac fibrosis.
In a recent study, Skyttä et al. evaluated whether the acute effects of RT on cardiac functions are augmented by the concurrent use of AIs. 32 The authors performed 2-D echocardiography, electrocardiogram, and cardiac biomarker measurements before and after adjuvant breast RT in 60 patients who had been diagnosed with breast cancer. They observed a significant decrease in right ventricular systolic function during RT in tricuspid annular plane systolic excursion. In addition, left ventricular diastolic function decreased among patients using AI. The authors concluded that the concurrent use of AI during RT for left-sided breast cancer led to a more pronounced change on right ventricular systolic function and left ventricular diastolic functions compared to RT alone. The result of this study is inconsistent with the current study; however, the authors observed acute cardiac toxicity with 2-D echocardiography. In the current study, we investigated cardiac fibrosis and endocardial thickness scores, which indicate chronic cardiac toxicity. Thus, using AIs with concomitant RT may lead to acute subclinical cardiac dysfunction. We believe that the authors should follow up these patients in order to observe chronic cardiac toxicity.
Our study has some limitations. We did not use electron microscopy. We might have seen further ultrastructural changes if we had evaluated the samples with electron microscopy in addition to light microscopy. Moreover, there is a paucity of both clinical and experimental data regarding the effects of hormonal treatments on the cardiovascular system. Our results suggest that the use of hormonal treatments did not cause cardiac fibrosis and did not potentiate radiation-induced cardiac fibrosis. However, our results should be clarified with further experimental and clinical studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Selcuk University. The study’s sponsor had no role in the study design, in the collection, analysis and interpretation of data, in the writing of the manuscript, or in the decision to submit the manuscript for publication.
