Abstract
Introduction:
Anthrax is a bacterial disease caused by the aerobic sporeforming bacterium Bacillus anthracis. It has been suggested that oxidative stress plays an important role in the pathogenesis of B. anthracis. The aim of this study was to investigate serum paraoxonase 1 (PON1) activity, catalase activity, malondialdehyde (MDA) levels, and superoxide dismutase (SOD) levels in patients with cutaneous anthrax.
Materials and methods:
Fifteen patients with cutaneous anthrax and 15 healthy controls were enrolled in this study. The serum MDA levels, SOD levels, paraoxonase, arylesterase, and catalase activities were measured using a spectrophotometer.
Results:
The serum SOD levels, paraoxonase, arylesterase, and catalase activities were significantly lower in patients with cutaneous anthrax than in controls (for all, p < 0.001), whereas MDA levels were significantly higher (p < 0.001). No significant correlation was found between serum paraoxonase activity, arylesterase activity, SOD levels, and MDA levels (all, p > 0.05) in patients with cutaneous anthrax.
Conclusions:
The current study was the first to show decreased antioxidant levels and increased oxidant levels in patients with cutaneous anthrax. Therefore, decreased PON1 activity may play a role in the pathogenesis of cutaneous anthrax.
Introduction
Anthrax is a rare, potentially fatal zoonotic disease caused by the bacterium Bacillus anthracis, which can infect both animals and humans. Anthrax is a bacterial disease caused by the aerobic spore forming bacterium B. anthracis, an important pathogen owing to its ability to be used as a bioterrorism agent. 1 Infection by inhalation of B. anthracis spores can result in a mortality rate of up to 96%. 2 The major routes of infection have been confirmed through inhalation of, skin contact with, or ingestion of B. anthracis spores. 3 Anthrax is an endemic zoonosis in Turkey, and anthrax remains a health problem in eastern Turkey. 4 –6 Cutaneous anthrax, the most common form of the disease, accounts for 95% of cases. 7 It is acquired when spores enter through a cut or abrasion in the skin. The most common areas of exposure are the hands, arms, face, and neck. 8 Diagnosis is based on clinical grounds in the majority of patients.
It has been demonstrated with many infectious diseases that various inflammatory cells are activated, leading to the production of reactive oxygen species (ROS), which kill intracellular and extracellular parasites. 9 ROS are crucial molecules that kill bacteria internalized by phagocytic cells, such as polymorphonuclear neutrophils and macrophages. 10 Evidence suggests that anthrax lethal toxin initiates ROS accumulation; in particular, the generation of superoxide and other ROS by macrophages and neutrophils. 11
Antioxidant enzymes, such as superoxide dismutases (SODs), catalases, and peroxidases, are the primary defense mechanisms utilized by bacteria for preventing oxidative damage, 12 and all are present in multiple copies in the B. anthracis genome. Catalase is an antioxidant enzyme converting hydrogen peroxide (H2O2) produced from highly reactive superoxide (O2 −) by SOD to water and oxygen molecules. Autophagy has been implicated in anthrax infection. 13 SODs are important antioxidant enzymes that are widely distributed in prokaryotic and eukaryotic cells. They catalyze the reduction of the superoxide radical to hydrogen peroxide and dioxygen in a critical reaction that protects aerobic organisms against oxidative damage. 14
Paraoxonase 1 (PON1) is a 354 amino acid, Ca2+-dependent serum esterase synthesized in the liver. 15 Human serum PON1 is an enzyme associated with high-density lipoprotein (HDL) that prevents the oxidative modification of low-density lipoprotein (LDL). 16 It has been suggested that decreased serum PON1 activity might be associated with increased oxidative stress in the serum and macrophages. 16 PON1 activity has been reported to decrease in patients with virtually all risk factors of atherosclerosis including hypercholesterolemia, obesity, and diabetes mellitus. 17 –19 Moreover, low serum PON1 activity is significantly associated with a risk of atherosclerotic disease. 20,21 It is well known that PON1 activity may decrease during the course of infection and inflammation. 22 –26
To the best of our knowledge, there are no studies addressing serum PON1 activity in patients with cutaneous anthrax in the literature. The aim of this study was to determine serum PON1 activity, catalase activity, oxidative stress, and SOD levels in patients with cutaneous anthrax.
Materials and methods
All patients (4 males and 11 females) were diagnosed with cutaneous anthrax at outpatient clinics at the Department of Infectious Diseases and Emergency Medicine (Medical Faculty, Yuzuncu Yıl University, Van, Turkey) and 15 healthy controls (10 males and 5 females) were without a history of chronic or recurrent disease and had normal physical examination results.
All of the cutaneous anthrax cases were characterized by evolving skin lesions and had a history of animal contact. The diagnosis of cutaneous anthrax was based on dermatologic findings including the following: papules from the vesicular stage; pruritic ulcers covered by characteristic black eschar; and edema, erythema, or necrosis without ulceration. The cases were confirmed by a positive smear or by isolation of B. anthracis in clinical specimens. 27 B. anthracis isolates were identified on the basis of conventional methods such as gram-positive bacilli with spores present in the smear, the presence of a capsule, the lack of motility, or catalase positivity.
The control group consisted of 15 healthy subjects with stable weight who were asymptomatic had an unremarkable medical history and normal physical examination. None of the control subjects were receiving antioxidant vitamin supplementation such as vitamin E or C. In addition, the subjects were not taking any medication and were not smoking or consuming alcohol; the subjects had no known acute or chronic diseases.
The study protocol was conducted in accordance with the Helsinki Declaration, as revised in 1989, and was approved by the local ethics committee. All subjects were informed about the study and written consent was obtained from each subject.
Exclusion criteria
Exclusion criteria included the following: a history of alcohol abuse, habitual smoking, intravenous drug abuse, pregnancy, antioxidant supplement use, hypertension, diabetes mellitus, liver or renal disease, infectious diseases such as Salmonella and Brucella; autoimmune diseases such as systemic lupus erythematosus, inflammatory bowel disease, and rheumatoid arthritis; pulmonary diseases; and coronary artery disease.
Blood samples
Blood samples were collected at 8:00 and 11:00 a.m. after an overnight fasting period. The blood samples were collected into empty tubes and immediately stored at 4°C. The serum was then separated from the cells by centrifugation at 3000 r/min for 10 min, and the lipid parameters were measured immediately. The serum samples were stored in plastic tubes at 80oC and used to analyze the malondialdehyde (MDA) levels, PON1 activity, SOD, and catalase activities.
Measurement of paraoxonase and arylesterase activities
The paraoxonase and arylesterase activities were measured using commercially available kits (Relassay, Gaziantep, Turkey). The PON1 activity was assayed using two different substrates, 28 expressed as units per liter and defined as 1 mmol p-nitrophenol generated per minute under well-established conditions. The arylesterase activity was expressed as kilounits per liter and defined as 1 mmol of phenol generated per minute under the reaction conditions. 29
Measurement of the serum SOD activity
The serum SOD activity was determined according to a method previously described by Sun et al. 30 The results are expressed in units per milliliter.
Measurement of serum catalase activity
The catalase activity was measured using H2O2 as a substrate. 31 The disappearance of H2O2 was monitored at 240 nm, and the enzyme activity was expressed in units per liter of serum (U/L) at 25°C.
Measurement of the serum lipid hydroperoxide levels
The MDA level of the serum was measured spectrophotometrically as previously described by Tomotsu et al. 32 A 0.5 mL plasma sample was shaken with 2.5 mL of 20% trichloroacetic acid in a 10 mL centrifuge tube. This mixture was combined with 1 mL 0.6% 2-thiobarbituric acid, shaken, and warmed for 30 min in a boiling water bath followed by rapid cooling. The solution was then shaken into a 4 mL n-butylalcohol layer in a separation tube. The results were expressed as nanomole/milliliter serum.
Statistical analysis
The results are presented as the mean ± standard deviation. The parametric variables were compared using Student’s t-test. Qualitative variables were assessed using a χ2 test. Correlation analyses were performed using Pearson’s correlation test. The results were considered to be statistically significant when the p-value was less than 0.05. The data were analyzed using the SPSS® for Windows computing program (Version 21.0).
Results
The demographic characteristics and laboratory findings of the patients with cutaneous anthrax and the controls are presented in Table 1. There were no significant differences between the groups with respect to age or gender (p > 0.05; Table 1).
Demographic characteristics and laboratory findings of the two groups.a
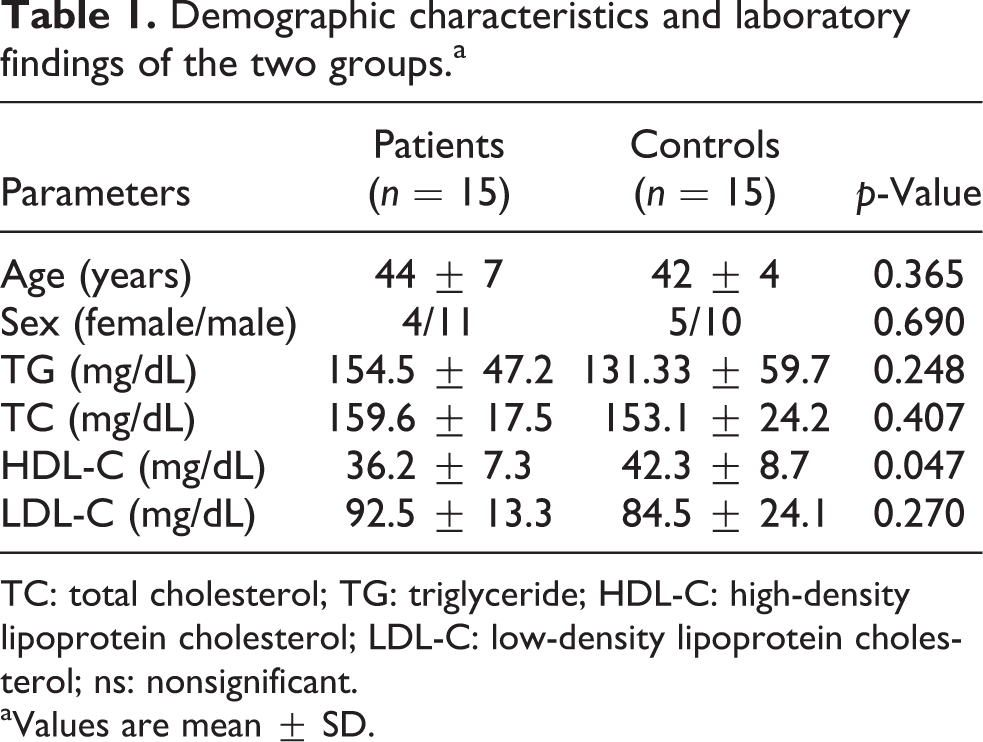
TC: total cholesterol; TG: triglyceride; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; ns: nonsignificant.
aValues are mean ± SD.
Serum triglyceride (TG), total cholesterol (TC), and LDL cholesterol (LDL-C) levels were significantly higher in the patients with cutaneous anthrax compared to the controls but the difference was not statistically significant (for all, p > 0.05). Serum HDL cholesterol (HDL-C) levels were significantly lower in the patients with cutaneous anthrax compared to the controls (p = 0.047; Table 1).
Serum SOD levels, paraoxonase, arylesterase, and catalase activities were significantly lower in patients with cutaneous anthrax than in controls (for all, p < 0.001), whereas MDA levels were significantly higher (p < 0.001; Table 2).
Paraoxonase activity and oxidative status levels of the two groups.a
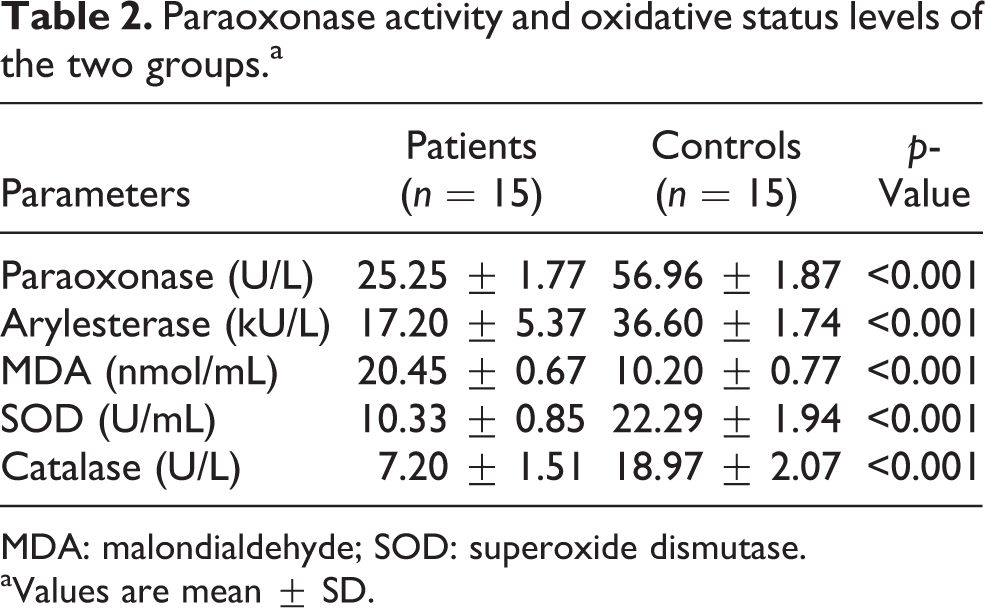
MDA: malondialdehyde; SOD: superoxide dismutase.
aValues are mean ± SD.
No significant correlation was found between serum paraoxonase activity, arylesterase activity, SOD levels, and MDA levels (all, p > 0.05) in patients with cutaneous anthrax. In addition, no significant correlations were found between lipid parameters and oxidant and antioxidant levels (all, p > 0.05).
Discussion
In the present study, we measured the enzymatic activities of PON1 in patients with cutaneous anthrax. We observed that cutaneous anthrax patients had decreased PON1, arylesterase, catalase activities, and SOD levels and increased MDA levels. To our knowledge, this study is the first to investigate serum PON1 activity in patients with cutaneous anthrax. We believe that the decreased serum PON1 activity could be attributed to the increased oxidative stress levels.
Anthrax is a bacterial disease caused by the aerobic spore forming bacterium B. anthracis, which is an important pathogen in the Bacillus cereus group. B. anthracis can infect multiple hosts, including humans, and anthrax can be contracted via intradermal inoculation or the ingestion or inhalation of spores.
Pathogens are endowed with protective mechanisms against ROS generated within the host, such as ones generated by professional phagocytic cells. 33 B. anthracis spores can escape phagocytosis and initiate the germination process even in antimicrobial conditions, such as oxidative stress. 34 B. anthracis spores can survive and germinate in oxidative environments. 12 As an aerobe, B. anthracis cells cannot avoid the oxidative stress created by endogenous respiration. In B. anthracis, ROS are generated as by-products of endogenous respiration, and phagocytes subject the bacteria to an oxidative environment during infection. 35 During the establishment and progression of anthrax, reactive oxygen metabolites may be encountered by B. anthracis exogenously, such as from the oxidative burst of phagocytic cells of the innate immune system 36,37 and in the extracellular milieu established in certain tissues of a host. 38,39
MDA is a potential biomarker for oxidative stress. 40 MDA levels have been investigated to examine oxidative stress and have been considered an indicator of oxidative imbalance during the onset of many diseases. 41 In the present study, we found that serum MDA levels were significantly higher in patients with cutaneous anthrax compared with controls.
Antioxidant enzymes, such as catalases and SODs, play a pivotal role in facilitating the survival of pathogens within a host and, thus, indirectly contribute to the pathogenesis of bacteria. 42 Catalase, a ubiquitous metalloenzyme guards aerobic organism as an intracellular H2O2 scavenger. Catalases relieve almost all organisms from deleterious peroxide stress by catalyzing the reaction to free oxygen and water. One of the major protective mechanisms employed by the host against an intracellular pathogen is the generation of reactive oxygen intermediates such as H2O2. 43 SODs are enzymes that catalyze the dismutation of the ROS superoxide anion (O2−) to hydrogen peroxide and molecular oxygen. 44 In the current study, we found that serum SOD and catalase activities were significantly lower in patients with cutaneous anthrax compared with controls.
Infection and inflammation are associated with a decrease in HDL-C levels. Several studies have indicated that chronic infection is closely associated with alterations of lipid metabolism. 45 A decrease in the cholesterol levels in various infections is well known, but the underlying mechanisms remain unclear. Several studies showed a reduction in cholesterol levels in patients with various infections. 26,46 In the present study, we found that serum TG, TC, and LDL-C levels were significantly higher in patients with cutaneous anthrax compared with controls, but there was no statistically significant difference. Moreover, serum HDL-C levels were significantly lower in the patients with cutaneous anthrax compared with the controls.
PON1 activity may also be altered as a part of the inflammatory response. During infection and inflammation, serum PON1 activity decreases due to changes in the synthesis and/or secretion of HDL particles and Apo AI. 47 Van Lenten et al. 48 reported that HDL becomes proinflammatory during the acute phase response, possibly due to a loss of PON1 activity from HDL. Paraoxonases are a new addition to the antioxidant enzyme family. PON1 is one of the antioxidants that act as enzymatic defense against lipid hydroperoxides. 49 Additionally, oxidative stress has been reported to affect the expression and activity of PON1. 50,51 The increased inactivation of PON1 according to the increased generation of ROS in brucellosis can explain the decrease in serum PON1 activity. 51 Furthermore, PON1 is an HDL-associated enzyme, and it has been suggested that reduced serum PON1 activity might be associated with decreased HDL-C levels. 52
Several studies reported that PON1 activity may decrease during the course of infection and inflammation. 22 –26 Aslan et al. 26 reported reduced levels of PON1 activity in the serum of Helicobacter pylori infected patients. Moreover, Apostolou et al. 25 observed decreased PON1 activity in patients with acute brucellosis compared with controls. Karakucuk et al. 23 investigated serum paraoxonase activity in the active stage of Behcet’s disease. They found decreased serum paraoxonase activity in the active stage of Behcet’s disease compared with controls. Moreover, reduced PON1 activity has been reported in diabetes mellitus, atherosclerotic heart disease, and hypercholesterolemia. 17 –19 In the present study, we found that serum paraoxonase and arylesterase activities were significantly lower in patients with cutaneous anthrax compared with controls.
There were several limitations in the present study. First, this study was cross-sectional. Second, the number of patients with cutaneous anthrax who were enrolled in the study was relatively small. A larger sample would have increased the power to detect serum PON1 activity in patients with cutaneous anthrax. Third, the PON1 genotype was not determined in the study population. However, it has been reported that serum PON1 activity is a better predictor of the risk of cardiovascular diseases than the PON1 genotype. 53
Conclusion
This study is the first study to investigate serum PON1 enzyme activity in patients with cutaneous anthrax. In the current study, we concluded that oxidative stress was increased whereas serum PON1 activity was decreased in patients with cutaneous anthrax. These results indicate that lower PON1 activity is associated with an oxidant–antioxidant imbalance. It would be useful to recommend drugs with antioxidant effects along with medical treatments in the treatment of cutaneous anthrax. These results suggest that lower PON1 activity contributes to the inflammation observed in cutaneous anthrax and might be used in the clinical setting.
Footnotes
Acknowledgments
The authors thank the staffs of Yuzuncu Yıl University, Faculty of Science and Art, Department of Chemistry, for their generous and friendly assistance in every step of this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
