Abstract
Background:
Colorectal cancer is the fourth leading cause of death. Various natural compounds are known to have antitumor properties. Garcinol, a polyisoprenylated benzophenone, has antioxidant and anti-inflammatory properties. In the current study, we investigated the anticancer activity of garcinol on human colorectal adenocarcinoma cell line (HT-29) human colon cancer cells.
Methods:
HT-29 cells were treated with various concentrations of garcinol for 24 h. The effect of garcinol on HT-29 cells proliferation was assessed by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay; the mRNA expression of microsomal prostaglandin E synthase-1 (mPGES-1), hypoxia-inducible factor-1α (HIF-1α), vascular endothelial growth factor (VEGF), C-X-C chemokine receptor type 4 (CXCR4), matrix metalloproteinase-2 (MMP-2), and matrix metalloproteinase-9 (MMP-9) were examined by quantitative real-time polymerase chain reaction; apoptosis was detected by proportion of sub-G1 cell; caspase 3 activity and prostaglandin E2 (PGE2) level were determined by enzyme-linked immunosorbent assay and HT-29 cells migration was assessed using scratch test.
Results:
Garcinol preconditioning markedly decreased the expression of mPGES-1, HIF-1α, VEGF, CXCR4, MMP-2, and MMP-9. The proportion of cells in sub-G1 phase and caspase 3 activity were increased by garcinol treatment whereas the cell proliferation, PGE2 level, and cell migration were decreased in these cells, compared to the control group.
Conclusion:
Our findings suggest that garcinol plays a critical role in elevating apoptosis and inhibiting HT-29 cells proliferation, angiogenesis, and invasion by suppressing the mPGES-1/PGE2/HIF-1α signaling pathways.
Introduction
Colorectal cancer (CRC) is the third most common cancer and the fourth cause of cancer-related death throughout the world. 1,2 Lifestyle factors such as diet, obesity, and exercise have been linked to CRC risk. Approximately, 25% of CRC occurs in individuals with a family history of the disease. The risk is, also, higher in individuals with inflammatory bowel disease. 3 Despite current treatments, such as surgical intervention, chemotherapy, and radiotherapy, survival is still very poor especially in the advanced stage of disease. 4 Colonoscopy is a gold standard of the CRC prevention. This invasive procedure helps to identify precancerous lesions. Another complementary strategy of cancer prevention is chemopreventive agents. 5 Cancer chemoprevention is the use of natural or synthetic agents to suppress or delay cancer progression in normal or preneoplastic tissue. 6 It has been explored that CRC is associated with overexpression of cyclooxygenase-2 (COX-2) and subsequent overgeneration of prostaglandin E2 (PGE2). Increased levels of COX-2 are detected in 85% of patients with CRC. Moreover, overexpression of COX-2 has been found to increase factors involved in angiogenesis and inhibits apoptosis pathway. 7 –9 Besides, previous studies have shown that PGE2 enhances the hypoxia-inducible factor-1α (HIF-1α) gene expression. HIF-1α plays a related role through the mechanisms by which PGE2 participates to the cancer development including angiogenesis, invasion, and chemoresistance. 10
Chemoprevention, by using aspirin and other nonsteroidal anti-inflammatory drugs (NSAIDs), is able to inhibit the development of colon cancer cells through blocking of COX activity, prostaglandin synthesis, and disruption of several signaling pathways such as extracellular signal-regulated kinases (ERK/MAPK) and nuclear factor kappa B (NF-kB). 11,12 However, long-term use of aspirin and other NSAIDs may be associated with some adverse effects such as gastric bleeding, platelet dysfunction, renal, and cardiovascular toxicity; thus, their prescription for chemoprevention should be regarded with caution. 13,14 In recent years, considerable efforts have been directed toward the other enzymes involved in the PGE2 synthase including prostaglandin E synthase (PGES) which is known as the terminal enzyme for the conversion of PGH2 to PGE2. 15,16 Three forms of PGES have been identified, namely cytosolic PGES (cPGES), microsomal PGES-1 (mPGES-1), and mPGES-2. cPGES and mPGES-2 are expressed constitutively, whereas mPGES-1 is induced by pro-inflammatory stimuli. 17 Clinical studies have displayed elevated levels of mPGES-1 within a number of cancers, comprising non-small cell lung cancer, stomach, endometrial adenocarcinoma, pancreas, prostate, and colon. 18,19
One of the promising resources of pharmaceutical agents is the use of natural compounds in plants. 20 Garcinol (camboginol) is a natural polyisoprenylated benzophenone derived from dried rind of the fruit Garcinia indica. 21 In medicine, garcinol is used for the treatment of several disorders including inflammatory and infectious diseases, obesity, diabetes, and ulcer. Garcinol has been represented to exhibit antioxidant, anti-inflammatory, and anti-microbial activities. 22 Anticancer and chemopreventive functions of garcinol have been reported in a wide variety of tumor cells, like leukemia, 23 colon cancer, 24,25 pancreatic adenocarcinoma cells, 26 breast cancer, 27 and head and neck squamous cell carcinoma. 28
To this date, the effect of garcinol on mPGES-1 in human colorectal adenocarcinoma cell line (HT-29) human colon cancer cell line has not yet been examined. In the present study, we investigated mechanistic details of anticancer and chemopreventive activity of garcinol on colon cancer cells.
Materials and methods
Cell culture
HT-29 cells, obtained from the Pasteur Institute (Tehran, Iran), were cultured in Roswell Park Memorial Institute-1640 medium (Gibco, Invitrogen, Carlsbad, California, USA) with L-glutamine, containing 10% (v/v) fetal bovine serum (FBS; Gibco, Invitrogen) and 1% penicillin/streptomycin (Gibco, Invitrogen). HT-29 cells were incubated in a humidified environment at 37°C and 5% carbon dioxide.
Cell viability assay
The inhibitory effects of garcinol on cell growth were determined by the analysis of viable cells using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Cells were seeded in 96-well plates at a density of 10,000 cells per well and then cells were treated with different concentrations of garcinol (0–50 µM; Sigma-Aldrich, St louis, Missouri, USA; purity ≥95.0%). After incubation for 24 h and 48 h, 10 µL of MTT solution (MTT; 0.05 g/mL; Sigma-Aldrich) was added to each well and incubated for 4 h at 37°C. The medium was discarded and 100 μL of dimethyl sulfoxide (Sigma-Aldrich) was added. The optical density of solubilized formazan was measured at 570 nm using an automatic microplate reader.
Real-time PCR
Total RNA was isolated by RNX-Plus solution (Sinaclone, Iran). Then, RNA concentration and quality were evaluated with NanoDrop. The cDNA was prepared with a kit (Thermo Scientific, Waltham, Massachusetts, USA) according to the manufacturer’s protocols. The reaction mixture was incubated 5 min at 25°C, 1 h at 42°C, and 5 min at 70°C. The reaction mixture for real-time polymerase chain reaction (PCR) contained 1 μL cDNA, 7 μL water, 10 μL SYBR Premix Ex Taq II Kits (Takara, Japan), and 1 μL of 10 pmol/μL specific primers (Table 1). Beta-Actin was used as an internal control gene. Thermal cycling was performed for 40 cycles of 15 s at 95°C, 30 s at 60°C, and 30 s at 72°C. Relative expression was reported by 2−ΔΔCT method analysis. 29
Primer sequences for qRT-PCR.

qRT-PCR: quantitative real-time polymerase chain reaction; mPGES-1: microsomal prostaglandin E synthase-1; HIF-1α : hypoxia-inducible factor-1α; MMP-2: matrix metalloproteinase-2; MMP-9: matrix metalloproteinase-9; VEGF: vascular endothelial growth factor.
PGE2 immunoassay
After the treatment period, supernatants were collected. The released PGE2 into the cell culture medium was measured by PGE2 enzyme-linked immunosorbent assay (ELISA) kit (Cayman Chemical) according to the manufacturer’s protocol.
Caspase 3 activity assays
The activation of intracellular caspases 3 was evaluated using caspase 3 colorimetric assay kit (Cambridge, Massachusetts, USA). After treatment, HT-29 cells were trypsinized, centrifuged at 1500 × g for 7 min, washed in ice-cold phosphate-buffered saline (PBS), and then lysed in lysis buffer. Then, after centrifuging at 10,000 × g for 1 min at 4°C, the supernatant was harvested. The protein concentration of the supernatants was measured by Bradford assay. Afterward, 200 μg proteins from each sample were added to 50 μL of reaction buffer containing dithiotreitol and caspase 3 substrate (DEVD-pNA) and incubated at 37°C for 1–2 h. The activity of caspases 3 in cell lysate was measured at 405 nm in the plate reader.
Sub-G1 DNA content assays
To determine the effect of garcinol on apoptosis, HT-29 cells were seeded in a 6-well plate at the concentration of 1 × 10 6 cells and then exposed to different concentrations of garcinol for 24 h. Cells were trypsinized, washed with PBS and fixed in ice-cold 70% methanol. Then, methanol was discarded by centrifuge and the cells were treated with RNase (0.5 μg/mL) at 37°C for 30 min. Staining was performed with propidium iodide (50 μg/mL) for 30 min. The proportion of dead cells in sub-G1 phase was detected using FACS caliber cytometer (Becton Dickinson, San Diego, California, USA).
Scratch test
Scratch test was performed to evaluate the in vitro cell migration and metastatic potential of HT-29 cells. HT-29 cells were seeded in 24-well plates at a density of 100,000 cells/well and cultured until they reached 80–90% confluence. Thereafter, the cell monolayers were scratched by sterile 100 μL pipette tips. The wounded monolayers were washed with PBS to remove debris and non-adherent cells. Cells were treated by 20, 40, and 60 μM of garcinol and incubated in serum-free media at 37°C for 48 h to allow cells to migrate into the wound area. Then, the images were recorded from three independent experiments for each set. In the next step, we converted the images to black and white and for each image, distances between one side of scratch and the other can be measured using Image J software (version 1.48). By comparing the images from time 0 to the last time point, the distance of each scratch closure on the basis of the distances was obtained. Quantification of the wounding area was performed using National Institutes of Health (NIH) Image J software.
Three-dimensional tube formation
About 106 of human umbilical vein endothelial cells were mixed with 5 mg/mL dextran-coated cytodex 3 microcarriers bead (Amersham Pharmacia Biotech, UK). Beads with cells were shaken gently every 20 min for 3–4 h at 37°C. After incubating, beads with cells were transferred to a 6-well culture plates and were incubated for 18–20 h in 0.5 mL of Dulbecco’s modified eagle medium (DMEM) at 37°C. In this procedure, the microcarriers do not attach firmly to the surface of the culture plate. After this time, beads with cells were washed three times with PBS and were transferred to a sterilized 15 mL Falcon. Then, the beads were allowed to settle to the bottom of the falcon. The sediment was re-suspended in 10× DMEM and ice-cold collagen. The collagen/bead solution was added to wells of a 96-well tissue culture plates and was allowed to clot for 10 min at room temperature and then 1× DMEM (which contains 5% FBS) was added to each well and equilibrated with the collagen clot for 60 min at 37°C. Next, different concentrations of garcinol were (0–60 μM) added to the wells. Bead assays were monitored after 48 h. Branches formation was analyzed using NIH Image J software and are reported as percentages of control.
Statistical analysis
Data are reported as mean ± standard error of the mean. One-way analysis of variance and Tukey’s post hoc tests were used to determine the statistical significance of the differences between groups. A p < 0.05 was considered to be statistically significant.
Results
Effect of garcinol on HT-29 cells viability
The effect of various concentrations of garcinol on the proliferation of HT-29 cells was evaluated by MTT assay. Garcinol reduced the percentage of viable cells compared to the untreated cells, in a dose- and time-dependent manner. Garcinol exhibited significant cytotoxicity at concentrations of 10 μM for 48 h and 20 μM for 24 h (p < 0.001). The 50% inhibition concentration (IC50) values were determined as 41 μM and 27 μM after 24 h and 48 h, respectively. We next measured the effect of garcinol on caspase 3 activity. Garcinol boosted the caspase 3 activity compared to the control group. To further analysis and to assess the cell-killing effects of garcinol, apoptosis in HT-29 cells was evaluated by flow cytometric analysis, the extend of sub-G1 cell phase. Treatment of cells with garcinol increased the cell population in sub-G1.
Effect of garcinol on PGE2 generation
In this study, we examined the PGE2 production by ELISA. The level of PGE2 in garcinol-treated HT-29 cells was significantly lower than non-treated cells (Figure 1).
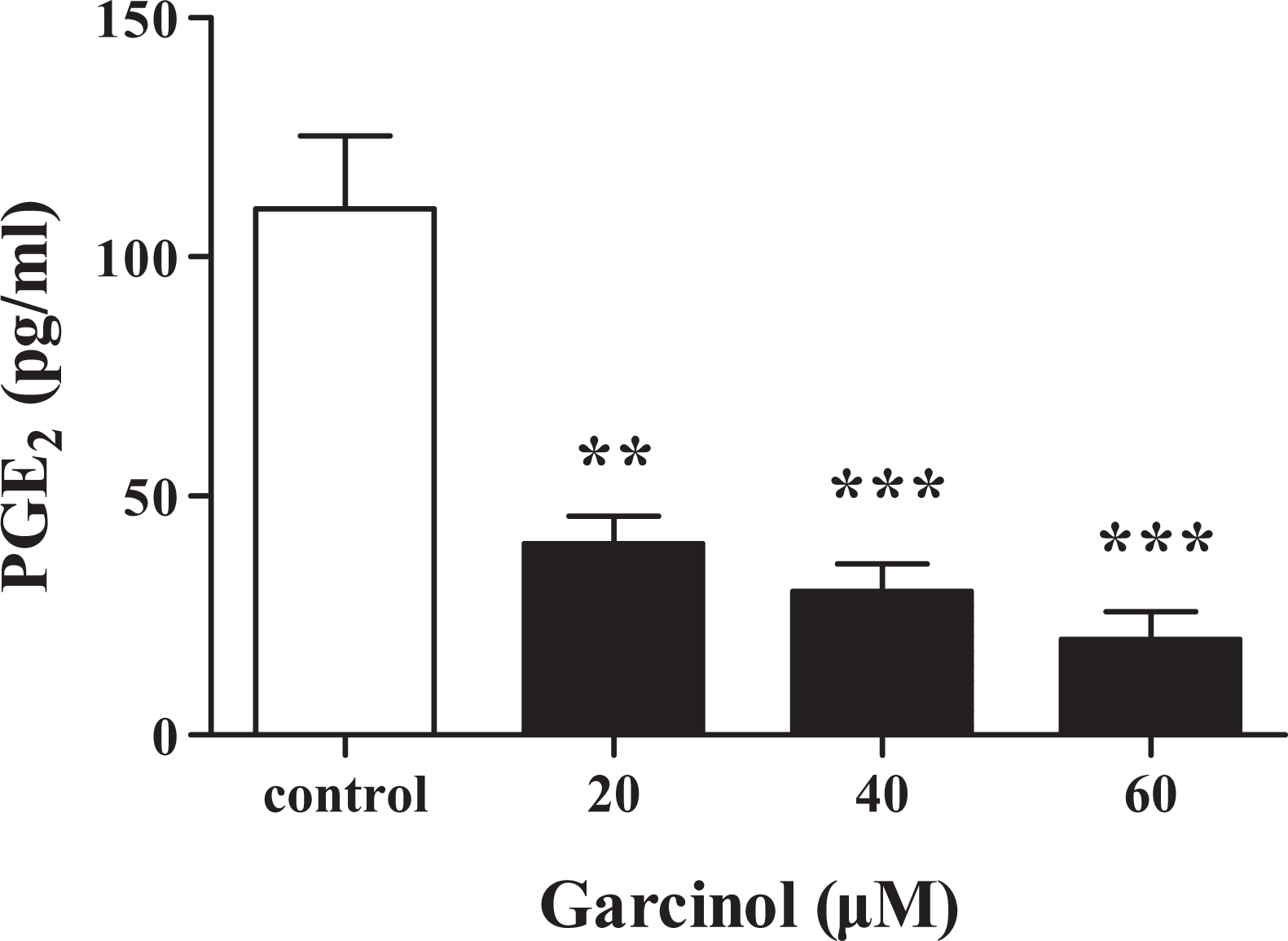
Effect of garcinol on PGE2 generation. Data are represented as mean ± SEM (n = 3). **p < 0. 01 and ***p < 0.001 versus control cells. PGE2: prostaglandin E2; SEM: standard error of the mean.
Effects of garcinol on expression of mPGES-1, HIF-1α, and CXCR4
The effects of garcinol on the gene expression of mPGES-1, HIF-1α, and C-X-C chemokine receptor type 4 (CXCR4) were performed using quantitative real-time PCR. As shown in Figure 2, expression of all these genes was effectively decreased by treatment with garcinol.

Effects of garcinol on expression of mPGES-1, HIF-1α, and CXCR4 mRNA. HT-29 cells were treated with garcinol (0, 20, 40, and 60 µM) for 24 h, and the expression of these genes was measured by real-time PCR. All data were normalized to levels of β-actin. ***p < 0.001 versus non-treated HT-29 cells. mPGES-1: microsomal prostaglandin E synthase-1; HIF-1α: hypoxia-inducible factor-1α; CXCR4: C-X-C chemokine receptor type 4; PCR: polymerase chain reaction; mRNA: messenger RNA.
Effect of garcinol on angiogenesis
To address the angiogenic effect of garcinol, we performed the tube formation assay as an in vitro model to evaluate the effect of garcinol on the capillary structure formation. As seen in Figure 3(a) and (b), garcinol-treated cells had inhibited sprout formation. We next examined the vascular endothelial growth factor (VEGF) gene expression in HT-29 cells. The amount of VEGF mRNA in garcinol-treated cell was significantly less than non-treated HT-29 cells (Figure 3(c)).

Effect of garcinol on angiogenesis. (a) Capillary sprouting of HUVECs in (I) medium with 2% FBS (II) 20 µM, (III) 40 µM, and (IV) 60 µM garcinol-treated HUVECs were monitored after 48 h. (b) Change in capillary length determined by NIH image. (c) HT-29 cells were treated with or without garcinol for 24 h and the gene expression of VEGF were measured by real-time PCR. Data reported as mean ± SEM (n = 3); ***p < 0.001 versus normal cells. HUVEC: human umbilical vein endothelial cell; FBS: fetal bovine serum; VEGF: vascular endothelial growth factor; PCR: polymerase chain reaction; SEM: standard error of the mean.
Effect of garcinol on migration of HT-29 cells
To detect the effect of garcinol on the migration ability of HT-29 cells, the scratch test was performed. Our results (Figure 4(a) and (b)) exhibited that after treatment of cells with garcinol, scratches were wider than the untreated cells due to reducing the number of cells migrated from the wound edge. In addition, real-time PCR analysis showed that the expression of matrix metalloproteinase-2 (MMP-2) and matrix metalloproteinase-9 (MMP-9) were remarkably decreased in garcinol-treated HT-29 cells (Figure 4(c)).

Effect of garcinol on HT-29 cells migration. (a) Representative image of HT-29 cells migration in various concentrations of garcinol at zero hour and then subsequently again 48 h later. (b) The scratched areas were quantified in three random fields in each group. (c) HT-29 cells were treated with garcinol (0–60 µM) for 24 h and the MMP-2 and MMP-9 gene expression were measured by real-time PCR. Data represented as mean ± SEM. **p < 0.01 and ***p < 0.001 versus non-treated cell. MMP-2: matrix metalloproteinase-2; MMP-9: matrix metalloproteinase-9; PCR: polymerase chain reaction; SEM: standard error of the mean.
Discussion
Over the last years, chemoprevention has emerged as an appealing approach for perturbation of cancer initiation and progression. 30 Various compounds have been identified as chemopreventive agents such as minerals, vitamins, phytochemical, and synthetic substances. 31
The aim of this study was to investigate the effect of garcinol treatment on molecular mechanism involved in apoptosis and migration of HT-29 cells. Cells were treated with different concentrations of garcinol and then the expression levels of mPGES-1, HIF-1α, and genes that are involved in the invasion (MMP-2 and MMP-9), angiogenesis (VEGF), and migration (CXCR4) were evaluated. In addition, we analyzed the PGE2 level, caspase 3 activity as well as the migration of HT-29 cells and tube formation in vitro.
PGE2, the product of COX-2, is one of the main actors in cancer initiation and progression through cell proliferation, angiogenesis, migration, and apoptosis evasion. Most of these effects are performed by PGE2 receptor. 32 mPGES-1 is the terminal and crucial enzyme involved in the generation of PGE2. Previous studies have shown the overexpression of mPGES-1 in several cancers including colon, lung, and gastric adenocarcinoma. 33,34 Recent data suggest that the mPGES-1 is an attractive target in the cancer therapy by allowing more specific and selective regulation of the PG pathway. 35
Our result showed that treatment of HT-29 cells with garcinol significantly decreases the expression of mPGES-1, leading to the reduced production of PGE2. This finding is in agreement with results reported by Koeberle in which garcinol inhibited the activity of mPGES-1 in cell-based and cell-free assays. 21 The inhibitory effect of garcinol, moreover, on the generation of PGE2 in human pancreatic cancer cell lines (BxPC-3) has been reported recently. 26
In the current study, we examined the effect of garcinol on cell death in HT-29 cells. The MTT assay revealed that garcinol significantly decreased cell growth in a time- and dose-dependent manner. By flow cytometry, garcinol treatment of cells increased apoptotic cells at sub-G1 phase. In addition, garcinol induced cell death in HT-29 cells by caspase 3 activation.
Ahmad et al. showed that garcinol promotes caspase-mediated apoptosis through inhibition of NF-kB activity in human breast cancer MCF-7 and MDA-MB-231 cells. 36 Moreover, reactive oxygen species (ROS)-induced mitochondrial membrane damage plays a main role in apoptosis. In this regard, a recent study has published that garcinol induced the ROS generation in multiple cell lines. The elevated ROS levels resulted in increased Bax/Bcl2 ratio, activation of caspase 3/9, downregulation of c-FLIP, upregulation of DR4 and DR5, and other anti-apoptotic proteins. 22,37
The current results reveal that garcinol significantly decreases the expression of HIF-1α and CXCR4 mRNA in HT-29 cells. These results are consistent with previous reports demonstrating that PGE2 increases the HIF-1α levels in human prostate cancer cells (PC-3 ML), gastric carcinoma cells, A549 cells, human breast adipose stromal cells, and human colon carcinoma cells (HCT-116). 38 –42
HIF-1α is not only stabilized by hypoxia but also increased by various compounds such as growth factors, hormones, cytokines, nitric oxide, and inflammatory mediators including PGE2. It seems likely that induction of HIF-1α expression is mediated through binding of PGE2 to the EP1 receptor and activation of Phosphatidylinositol 3-kinase /Akt/mammalian target of rapamycin (PI3K/Akt/mTOR) and MAPK extracellular signal-regulated kinase/extracellular signal-regulated kinase (MEK-ERK) signaling pathway. 41,43
Chemokines and chemokine receptors play important roles in cancer growth, angiogenesis, metastasis, and modulation of tumor microenvironment. 44 The pervious study indicated that PGE2-EP3/EP4 signaling enhances the expression of SDF-1/CXCR4 which confirms our results. 45
In this study, we observed that treatment of HT-29 cells with garcinol decreases the expression of VEGF mRNA and in vitro tube formation. Angiogenesis is an essential process in tumor growth and metastasis. Among the factors involved in angiogenesis, VEGF has been known as an important signaling protein. VEGF binds to VEGF receptors and stimulates signaling pathway that increases the tumor cell proliferation and migration and induces degradation of the extracellular matrix. 46 Previous studies have shown that PGE2 is able to increase VEGF expression via a variety of signaling pathways: ERK2/JNK1 signaling pathway in rat gastric microvascular endothelial cells, 47 cAMP-dependent pathway in PC-3 cells, 48 HIF-1α-dependent pathways in HCT-116 cells, 41 and Epidermal growth factor receptor/Mitogen-activated protein kinases (EGFR-MAPK) signaling pathways in gastric cancer cells line (MKN28). 49
Other major factors implicated in angiogenesis are proteolytic enzymes. MMPs have an important role in tumor spread and invasion by digestion of several components of the extracellular matrix. 50 Recent studies have suggested a crucial role for MMP-2/9 in invasion and metastasis in gastric cancer SGC7901 cell ovarian cancer cell. 51,52
Then, we evaluated the expression of MMP 2/9 and cell migration following treatment with garcinol and found that MMP-2/9 and cancer cell migration were affected by garcinol. MMP-2 expression has increased in the pancreas, colon, skin, and prostate cancer. 53 Early studies identified that PGE2 stimulates MMP-2 expression via EP1 receptor in human cholangiocarcinoma cells 54 and human lung cancer cells. 55
In addition to the signaling pathways mentioned above, it seems that PGE2-induced HIF-1α plays a vital function in the VEGF, CXCR4, and MMP-2 expressions. It is observed that these genes are potential transcription targets for HIF-1α. 56
In summary, here we provided evidence that garcinol suppresses the proliferation, angiogenesis, migration, and invasiveness of HT-29 cells mainly by inhibiting CXCR4, VEGF, and MMP2/9 expression and promoting the apoptosis. This activity is correlated with the mPGES-1/PGE2/HIF-1α pathways. Therefore, mPGES-1 is considered as an attractive target for anticancer drugs such as garcinol in order to perform colorectal cancer chemoprevention.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from Hamadan University of Medical Sciences.
