Abstract
Background:
Biliary lithiasis, or sludge, and nephrolithiasis have been reported as a possible complication of ceftriaxone therapy. However, no study related to cefotaxime-induced biliary pseudolithiasis or nephrolithiasis was observed in the literature. Therefore, we investigated the comparative formation of biliary pseudolithiasis and nephrolithiasis after cefotaxime and ceftriaxone therapies.
Methods:
The patients treated with ceftriaxone or cefotaxime were enrolled during the study period. Ultrasound imaging of the biliary and urinary tract was performed in all patients before and after the treatment. The patients with a positive sonographic finding at the end of treatment were followed up with monthly ultrasonography for 3 months.
Results:
The present study showed that abnormal biliary sonographic findings were demonstrated in 18 children (20.9%) treated with ceftriaxone, 13 (15.1%) had biliary lithiasis, 5 (5.8%) had biliary sludge and 1 (1.2%) had nephrolithiasis. Abnormal biliary sonographic findings were demonstrated in only four (5.9%) children treated with cefotaxime who had biliary sludge and only one (1.5%) had nephrolithiasis. It was observed that older age was at significantly higher risk of developing biliary sludge or stone formation. Receiver operating characteristic analysis was performed to determine the residual risk and analysis found that 4.5 years was the cut-off value for age.
Conclusions:
The present study is unique in the literature for reporting for the first time gall bladder sludge and nephrolithiasis associated with cefotaxime use. Therefore, patients treated with cefotaxime should be monitored for serious complications like patients treated with ceftriaxone. Nevertheless, if third-generation cephalosporin is used, cefotaxime is recommended to be used rather than ceftriaxone.
Introduction
Ceftriaxone and cefotaxime are third-generation parenteral cephalosporins with a wide spectrum of antimicrobial activity. Both drugs are effective against many gram-negative and gram-positive bacteria, though particularly so with gram negative. In addition, these are bactericidal, and their half-lives are long. This is why these antibiotics commonly are prescribed against serious infections such as meningitis; lower respiratory tract; complicated urinary tract; skin, bone and joint infections and bacteremias. 1
Biliary sludge, or biliary lithiasis, has been reported as a potential side effect of ceftriaxone therapy in the literature. 2 –6 Schaad et al. 2 first reported a sonographic finding of a ceftriaxone-induced transient formation of precipitations in the gall bladder in 1986. 2 Terms such as ‘biliary pseudolithiasis’ and ‘reversible cholelithiasis’ have been used to describe ultrasonographic (US) abnormalities in the gall bladder of patients treated with ceftriaxone. 2 –4 It has been considered that a high dose of ceftriaxone is accumulated in the gall bladder, where it is excreted as a calcium-binding complex. But sometimes, this complex may precipitate and cause pseudolithiasis if the ceftriaxone concentration exceeds resolution capacity. 5 Another rare complication of ceftriaxone therapy is nephrolithiasis, which was first reported by Shaad et al. in 1988, 3 and later it was reported in other studies. 7,8
These complications of ceftriaxone treatment, biliary pseudolithiasis and nephrolithiasis, are usually temporary upon discontinuation of ceftriaxone therapy and are asymptomatic. 3,5 –8 However, it has been reported that these complications sometimes may cause symptoms and even may develop more serious complications requiring surgical intervention. 4,9 –11 Therefore, it is important to notice these complications.
Different reports about the incidence of biliary pseudolithiasis and nephrolithiasis have been reported. 3 –8 However, no study related to cefotaxime-induced biliary pseudolithiasis or nephrolithiasis was observed in the literature. Therefore, we aimed to find which antibiotic is safer between ceftriaxone and cefotaxime with similar antimicrobial activity in terms of biliary pseudolithiasis and nephrolithiasis. To this end, we investigated comparatively the formation of biliary pseudolithiasis and nephrolithiasis after cefotaxime and ceftriaxone therapies.
Material and methods
This study was conducted prospectively in 154 children, 2 months to 18 years old, who were hospitalized with different types of infection at Yuzuncu Yıl University Hospital in Van City, Turkey, from November 2014 to January 2016. This study was approved by the Ethics Committee of Yuzuncu Yıl University.
The patients, treated with ceftriaxone (intravenously, 100 mg kg−1 day−1, divided into two equal doses) 12 and cefotaxime (intravenously, 150 mg kg−1 day−1, divided into three or four equal doses) 12 for infection like pneumonia, pyelonephritis, lymphadenitis, bacterial meningitis, bacteremia, gastroenteritis and peritonitis, were enrolled during the study period. Age, sex, blood test results and received medication of patients were recorded. Ultrasound imaging of the biliary and urinary tract was performed in all patients before treatment, and patients with biliary sludge, gallstones and nephrolithiasis in the first ultrasound imaging were not included. The second US was performed at the end of ceftriaxone or cefotaxime therapy. The patients with a positive sonographic finding at the end of treatment were followed up with monthly US for 3 months. The patients with a positive sonographic finding also were confirmed by another radiologist.
US findings in patients treated with ceftriaxone and cefotaxime were divided into separate groups. The patients with abnormal US findings were then compared with the patients with normal US findings in terms of age, sex, duration of therapy and blood test results.
Results
This study included 154 children (76 girls and 78 boys) between 2 months and 18 years old. Cases treated with ceftriaxone and cefotaxime were divided into two groups based on treatment. Ceftriaxone-treated group included 86 children (43 girls and 43 boys) and the mean age of patients was 4.77 ± 4.91 years. The number and mean age of patients in the cefotaxime-treated group were 68 (33 girls and 35 boys) and 4.53 ± 4.50 years old (Table 1). The types of infection were pneumonia, pyelonephritis, lymphadenitis, bacterial meningitis, bacteremia, gastroenteritis and peritonitis.
Comparison of ceftriaxone and cefotaxime groups in terms of age and gender.
This study showed abnormal biliary sonographic findings in 18 (20.9%) children treated with ceftriaxone, 13 (15.1%) had biliary lithiasis, 5 (5.8%) had biliary sludge and only 1 (1.2%) patient had nephrolithiasis. On the other hand, abnormal sonographic findings were demonstrated in only four (5.9%) children treated with cefotaxime who had biliary sludge and only one (1.5%) patient had nephrolithiasis (Table 2). All of them were asymptomatic, and none of the patients had complications. The types of infections in patients with complications are bacteremia found in nine patients, bacterial meningitis in four, pneumonia in four and pyelonephritis in five patients. The infection in patients with nephrolithiasis was bacteremia.
Comparison ceftriaxone and cefotaxime groups in terms of the incidence of abnormal sonographic findings.

Cases treated with ceftriaxone and cefotaxime were divided into two groups and compared in terms of ultrasound findings, age and gender. These two groups did not differ in terms of age and gender (Table 1). Abnormal biliary ultrasound findings were significantly more in the ceftriaxone-treated group (Table 2). There were four patients with biliary sludge in the group treated with cefotaxime, and as this condition has never been reported previously in the literature, it was an interesting finding. To determine risk factors, cases with and without abnormal biliary ultrasound findings were divided into two groups and compared in terms of ultrasound findings, age, gender and laboratory tests. The two groups did not differ in terms of gender and laboratory tests (Tables 3 and 4). However, the older children were at significantly higher risk of developing biliary sludge or stone formation (Table 3). Receiver operating characteristic (ROC) analysis was performed to determine the residual risk revealed the age cut-off of 4.5 years (Figure 1). Also abnormal sonographic findings can be seen in Figure 2.
Comparison of patients with and without abnormal biliary sonographic findings in terms of age, treatment durations and gender.

Comparison of patients with and without abnormal biliary sonographic findings (mean ± SD) in terms of laboratory parameters.

AST: aspartate aminotransferase; ALT: alanine aminotransferase GGT: gamma-glutamyl transpeptidase; BUN: blood urea nitrogen.

ROC analysis of patients with and without abnormal biliary sonographic findings in terms of age. Area under curve: 0.706; p: 0.002. Cut-off age: 4.5 years; sensitivity: 70%; specificity: 40%. ROC: receiver operating characteristic.
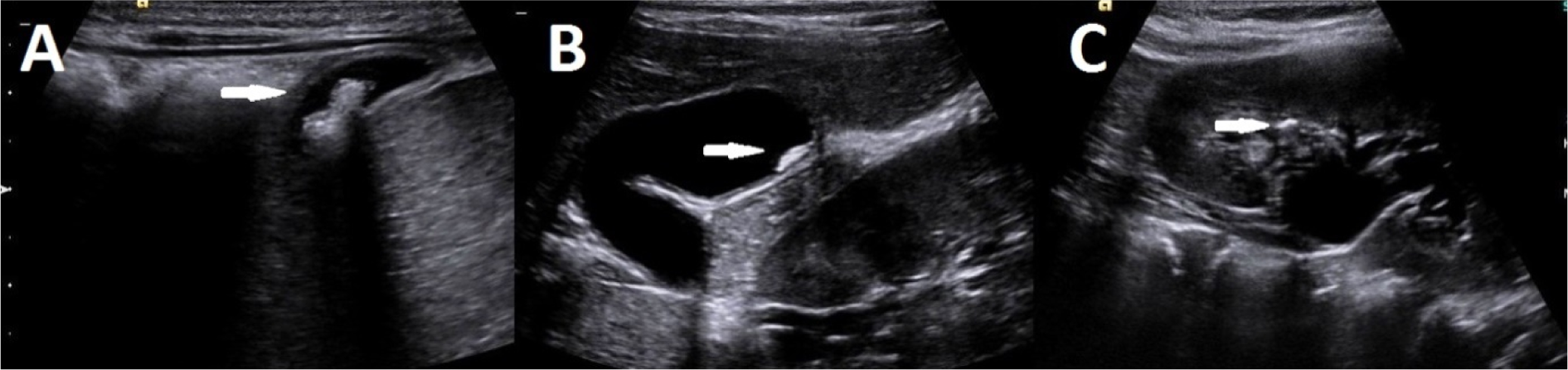
Ultrasonographic images of patients with biliary lithiasis, biliary sludge, and nephrolithiasis. (a): Biliary lithiasis; (b): biliary sludge; (c): nephrolithiasis.
Discussion
Biliary pseudolithiasis and nephrolithiasis are two complications of ceftriaxone treatment. This study aimed to reveal the frequency of these complications in children treated with ceftriaxone or cefotaxime. The frequency that abnormal gall bladder sonograms included biliary sludge or biliary lithiasis was 20.9% in the ceftriaxone group and 5.9% in the cefotaxime group. Additionally, nephrolithiasis frequency was 1.2% in the ceftriaxone-treated group and 1.5% in the cefotaxime-treated group. None of the cases were symptomatic and no complications were seen with biliary sludge, gallstones and nephrolithiasis.
The study outcomes are concordant with some of the literature. Although there are many studies about ceftriaxone-induced pseudolithiasis and nephrolithiasis, there is not enough study about the frequency of these complications, and there are conflicting results. 3 –8 There are many publications about ceftriaxone-induced pseudolithiasis and nephrolithiasis, but according to our information, there are no studies about cefotaxime outcomes about these complications in the literature, so our study is unique in filling the gap.
Schaad et al. first reported a case of an 18-year-old male who developed a ceftriaxone-associated transient formation of precipitations in the gall bladder in 1986. 2 Also a study was first published about ceftriaxone-induced nephrolithiasis by Schaad in 1988. In that study, 37 children were treated with ceftriaxone, and all 16 developed pseudolithiasis. One had concurrent biliary and urinary lithiasis, manifesting renal colic and urinary obstruction. 3 In a prospective study that was published in 2006 by Biner et al., 27 (17%) of 156 children who were administered ceftriaxone had abnormal gall bladder sonograms, 16 (10%) had biliary lithiasis, 11 had biliary sludge (7%) and one developed nephrolithiasis. Of all children, five who had pseudolithiasis became symptomatic. 4 Acun et al. found gall bladder and urinary tract precipitations in 5 of 35 ceftriaxone-treated children. Of those patients, three had biliary pseudolithiasis, one had biliary sludge and one had biliary pseudolithiasis and urinary bladder sludge. The patients who had biliary pseudolithiasis with urinary bladder sludge and biliary sludge had abdominal pain, nausea and vomiting. Three children remained symptom-free. 5 Another study published by Murata et al. in 2015 found pseudolithiasis incidence in 60 children (18.3%) who were treated with ceftriaxone, and none of the patients had complications. 6
The predisposing factors of ceftriaxone-induced biliary pseudolithiasis remain unknown. 6 However, risk factors are defined in the literature, including high doses of ceftriaxone (>100 mg kg−1), short infusion time (<30 min), co-therapy with other nephrotoxic drugs, associated kidney disease, dehydration, hypoalbuminemia and calcium-containing infusions. 13 Also, it was reported that older age increases the risk of biliary pseudolithiasis. 4
Ceftriaxone binds to serum albumin and 85–95% of the drug given intravenously passes the tissues. Approximately, 55% of the drug is excreted by the kidneys and 45% is excreted by the gall bladder. 14 The ceftriaxone drug concentration in the gall bladder is 20–150 times higher than the serum concentration. If the ceftriaxone concentration exceeds resolution capacity, it precipitates with binding to calcium ions. 15
Rapid decrease in enteral nutrition, long-term hunger and weight loss are defined as risk factors. These factors, especially rapid decrease in enteral nutrition, reduce gall bladder contractions and bile excretion, and these factors may cause pseudolithiasis. 6 It is likely that the same mechanism is valid with other bile-excreted drugs. Reduced bile excretion proceeds increased concentration of these drugs, causing precipitation and pseudolithiasis. Of the given cefotaxime dose, 40% binds to serum proteins and 60% is excreted by the kidney. 1 Cefotaxime and its metabolites are classified as mildly bile-excreted agents. 16 We hypothesized that if the risk factors described above are existent, cefotaxime also may cause gall bladder sludge or biliary lithiasis. Therefore, in our study, we involved investigation of cefotaxime-induced biliary sludge and found it in four patients for the first time in the literature.
If we consider the risk factors described above, the patients who had abnormal sonographic findings of the biliary tract, biliary sludge (or biliary lithiasis), and those who did not have, compared by ignoring which antibiotic was administered. Age was the major difference among the groups, as was found in the study by Biner at al. 4 ROC analysis of age risk revealed that 4.5 years was the cut-off age (sensitivity 70% and specificity 40%). According to the ROC analyses, biliary sludge development risk increased at older than 4.5 years of age.
Administered medication is determined as a predisposing factor for nephrolithiasis. 17,18 Nephrolithiasis may occur because of precipitation of inorganic salts as well as genetic/metabolic disorders that causes stone modulator dysfunction. 17 Also, long-term and high-dose treatment with some drugs that are predisposing urinary calculi may cause nephrolithiasis. These lithogenic agents are classified into two groups: inducing metabolic abnormalities (e.g. hypercalciuria, hypocitraturia and urine acidity) or some agents that make crystals in the urine because of high concentration and low solubility (e.g. uric acid nephropathies). 19 In the literature, ceftriaxone is recognized as a lithogenic drug. 7,8,18 Chutipongtanate and Thonqboonkerd’s study suggest that the normal range of a ceftriaxone dose may become precipitated with calcium ions in the urine, even under physiologic condition. They suggest that ‘tubular occlusion’ and ‘crystal-cell adhesion’ may have a prominent role in ceftriaxone-induced nephrolithiasis. 18 Two prospective studies showed that a 7-day course of normal ceftriaxone treatment develops renal calculi in 1.4–7.8% of patients. 7,8 Although we do not know the exact predisposing renal calculi mechanism of this drug, age and genito-urinary abnormalities may increase the risk of developing urinary calculi. 7,8,20
Nearly, all of the patients with ceftriaxone-associated calculi seem to remain asymptomatic, 7,8 but some of them may present with abdominal pain and vomiting as well as urinary obstruction leading to anuria and uremia. 11,20
In this study, there were two patients with nephrolithiasis. One of them was administered high doses of ceftriaxone therapy, the other was treated with cefotaxime as well. These two patients remained asymptomatic and, in the follow-up, the calculi disappeared. We did not find hypercalciuria or hyperuricosuria in the urine analysis. Urinary pH in patients were acidic, and urine samples were izostenuriac.
In conclusion, there are many publications about ceftriaxone-induced pseudolithiasis or nephrolithiasis. However, according to our information, there were no studies about cefotaxime outcomes for these complications in the literature. Therefore, this study is unique for reporting, for the first time, gall bladder sludge and nephrolithiasis associated with cefotaxime use. Therefore, patients treated with cefotaxime should be monitored for serious complications like patients treated with ceftriaxone. Nevertheless, if third-generation cephalosporin is used, cefotaxime is recommended to be used rather than ceftriaxone.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
