Abstract
Ethambutol (EMB) is conventionally used to treat tuberculosis and atypical Mycobacterium infections in combination with other antimycobacterial drugs. Eventually, EMB testicular toxicity has not been explored extensively yet. The aim of the study is to evaluate testicular toxicity of EMB. We explored the impact of EMB on male rats’ fertility, testosterone level and germ cells state, testicular pro- and anti-oxidant status and DNA damage, as well as identified EMB effects on cytochrome P-450 2E1 (CYP2E1) both with computer simulation and in vivo. We demonstrated that EMB administration to male rats decreased in epididymal sperm count (19%) and fertility index (53%). These events were accompanied by reduction in serum testosterone content (1.6 times) and appearance of spermatogenic epithelium damages. It was also found in testes the intensification of lipid peroxidation, decrease in reduced glutathione content and changes in DNA fragmentation. Additionally, computer simulation showed direct interaction of EMB with CYP2E1 active site and heme. On the top of this, we demonstrated that level of testicular CYP2E1 messenger RNA in EMB-treated rats was increased 8.7 folds and p-nitrophenol hydroxylase activity in testes rose three folds. As this shows, EMB-caused CYP2E1 induction, oxidative stress, and apoptosis in the testes contribute to inhibition of steroidogenesis enzymes and spermatogenesis disruption.
Introduction
To date, tuberculosis remains a global health problem; moreover, drug-resistant tuberculosis is a constantly growing threat, worldwide. 1 Initially, patients from countries with a known high incidence of resistant Mycobacterium tuberculosis strains, patients who had been treated previously, and patients with life-threatening tuberculosis, receive the same combination of isoniazid (INH), rifampin (RMP), and pyrazinamide (PZA), together with at least one additional medicine, which is ethambutol (EMB) and/or streptomycin. 2 It is well known that repeated exposure to anti-tuberculosis medication increases risk of various adverse drug reactions and toxicity dependent on age, gender, and life style. 3
We have previously reported that EMB, INH, RMP, and PZA in combination and INH alone caused an increase in the rate of thiobarbituric acid-reactive substances (TBARS) formation in rats testes and sperm, decrease in testis glutathione and protein SH-group contents, significant changes in DNA fragmentation, fatal reduction of male fertilizing capacity and fertility, and an increase in pre- and post-implantation embryo-lethality, increased content of testicular cytochrome P-450 isoenzymes (CYP2E1, CYP3A2, and CYP2C23) messenger RNA (mRNA) with simultaneous morphological and morphometric changes in spermatogenic epithelium. 4 –6
Taking into account our previous results referred to alterations of testicular CYP2E1 following INH and antitubercular drugs combination administration, it is important to note that this particular isozyme is localized in Leydig cells, where testosterone (TS) biosynthesis occurs. 7,8 Consequently, these structures, and their microenvironment damage as a result of CYP2E1-mediated processes presumably could be the causes of steroidogenesis inhibition and spermatogenesis disruption.
The current study has been devoted to EMB as it is commonly used for the treatment of tuberculosis and atypical Mycobacterium infections in combination with other antimycobacterial drugs to prevent or delay the emergence of drug resistance. 9
Beside the major adverse reaction of EMB as optic nerve toxicity, 10,11 others include dermatitis, pruritus, headache, malaise, dizziness, fever, mental confusion, disorientation, possible hallucinations, joint pain, and rarely anaphylactoid reactions. 12 Of note, there are two early reports regarding EMB to provoke loss of fertility and evident regressive lesions in the animals’ testes. 13,14 Thus, our interest in probable testicular toxicity of EMB and its ability to interact with CYP2E1 was encouraged by these findings and our previous data. Therefore, the aim of this study is to evaluate the testicular toxicity of EMB in the aspect of its effects on male rats fertility, TS level, and germ cells state, testicular pro- and antioxidant status, and DNA damaging effect, as well as to identify EMB effects on cytochrome P-450 2E1 (CYP2E1) with computer simulation and in vivo.
Materials and methods
Animals and treatment
The study was carried out on Wistar albino male rats with initial body weight (bw) 150–170 g (8–9 weeks old) and female rats with 150–170 g bw (9–10 weeks old) purchased from Biomodel Service (Kyiv, Ukraine). Animals were kept under a controlled temperature (from 22°C to 24°C), relative humidity of 40–70%, lighting (12-h light/12-h dark cycle), and on a standard pellet feed diet (“Phoenix” Ltd., Ukraine). Substance of EMB was supplied by the SIC “Borzhagovsky Chemical-Pharmaceutical Plant” CJSC, Ukraine.
The male rats were divided randomly into two groups: 1st—control (n = 6); 2nd—EMB administration (n = 6). EMB was suspended in 1% starch gel and was administered intragastrically by gavage. We used dose 155 mg/kg bw/day, which was recalculated from human dose 15 using the coefficient for conversion of human doses to animal equivalent doses based on body surface area (Guidance for Industry and Reviewers Estimating the Safe Starting Dose in Clinical Trials for Therapeutics in Adult Healthy Volunteers US. of Department of Health and Human Services, FDA, CDER, and CBER). For precise understanding of EMB contribution in antifertility effects, we used the same dose as in our previous study, when EMB was coadministered with other antituberculosis drugs. 4,5 The duration of EMB administration was 60 days according to the entire spermatogenesis cycle for rats (time of germ cell maturation in epididymis). The control group received only starch gel in corresponding volumes (5 ml/kg bw).
After 46 days of repeated EMB administrations, the males from both groups were mated with intact females at the ratio 1 male:2 females during 14 days (three oestrous cycles). During this period, the administration of EMB to male rats was continued.
According to generally accepted guidelines for the fertility study in laboratory rats, 16 the first day of pregnancy was established by vaginal cytology (the first day of sperm detection in vagina). Most males were mated within the first 5 days of cohabitation (i.e. at the females first available oestrus), but part of them demonstrated infertility. This fact was taken into account for evaluation of the effect of EMB on male fertilizing capacity, which was determined by the index:
The pregnancy was confirmed by necropsy. The females were killed under mild ether anesthesia via cervical dislocation on day 20 of pregnancy. Concentration of ether 80 µL per liter of volume of a container was used; time of induction took approximately 5 min.
Males were killed after 60 days (duration of spermatogenesis and time of germ cell maturation in epididymis) of experiment. Euthanasia was performed by the decapitation on the morning. The time of euthanasia was recorded. Testes were removed. Right testis was used for histological examination and left testis- for biomolecular investigations.
The study was carried out in accordance with the UK Animals (Scientific Procedures) Act, 1986 and associated guidelines, the European Communities Council Directive of November 24, 1986 (86/609/EEC), and approved by the Institutional Animal Care and Use Committee (approval number 01/12/09).
Measurement of total serum testosterone
Prior to euthanasia, blood samples from femoral vein were collected. Serum samples were separated and kept frozen at −70°C. Total TS levels were measured using DRC testosterone enzyme-linked immunosorbent assay kit (Germany) according to manufacturer’s instruction.
Spermatogenesis parameters evaluation
In all cases, the right testicle was used for the evaluation of morphologic and morphometric parameters and spermatogenesis indices. The tissue was fixed in Bouin’s solution (BS) for 6–8 h. After fixation with BS, picric acid was removed from the tissues by washing in 70% ethanol. Then the specimens were transferred to 70% ethanol and lithium carbonate to neutralize the picric acid in BS. The ethanol-lithium carbonate solution was changed three or more times until the yellow color of BS was almost depleted from the tissue. Then the tissues were dehydrated using graded ethyl alcohol and embedded in paraffin. Histologic sections (6 µm) were stained by hematoxylin and eosin. Histological examination was performed under a light microscope (400×).
The determination of the spermatogenic index in testicles was carried out according to four points system. It was based on the estimation of number of cell layers, types of cells, and the presence of late spermatids in the seminiferous tubules. The criteria were as follows: 1—only spermatogonia present; 2—spermatogonia and spermatocytes present; 3—spermatogonia, spermatocytes, and round (early) spermatids present with <5 late spermatids per tubule; 4—spermatogonia, spermatocytes, and round spermatids present with up to 25 late spermatids per tubule. 16 Spermatogenic index was calculated as a ratio of stages of spermatogenesis total to number of examined tubules. Two hundred seminiferous tubules per testis of each animal were observed by microscopy.
The obtained numerical data were recorded and processed with statistical methods, as described below. Simultaneously, occurrences of cells exfoliation (shedding of epithelial elements), desquamation epithelium (detachment) from tubule basal membrane, and presence of cell-free regions (“windows”) were taken into account.
The sperm count in epididymal suspensions was estimated as described by Chitra et al. 17 using Goryaev’s counting chamber and light microscope (200×).
Computer simulation of EMB and CYP2E1 interaction
Optimized computer model of human CYP2E1 spatial structure was used. 18 The spatial structure of human cytochrome P450 2E1 was created using web server Swiss Model (http://www.expasy.ch/swissmod/SWISSMODEL.html) as previously described. 19 Experimentally defined spatial structures of 4-methylpyrazole (PDB code 3E6I) and indazol (PDB code 3E4E) complexes 20 with human CYP2E1 were obtained from the database Protein Data Bank (http://www.rcsb.org).
Search of the enzyme active center was performed using Fred Receptor (version 2.2.3, OpenEye Scientific Software, Inc., Santa Fe, New Mexico, USA, http://www.eyesopen.com, 2005). Docking of heme molecule into the enzyme active site was performed with HEX 5.0 Software 21 using five-dimensional rotational correlations of shape and electrostatic expansions with following system energy minimization. The resulting structure was optimized in the aquatic environment with molecular dynamics method (computer simulation of the atoms and molecules movement) using NAMD Software. 22 Solvation of enzyme was performed using VMD Software; 23 solvation space was limited by protein size. Overall structure was surrounded by 6190 water molecules type TIP3P. 24 The calculation of molecular dynamics was performed under standard conditions in the microcanonical (NVE) ensemble at 37°C using force-fields Charmm35 25 within 10 ps.
For modeling of EMB and CYP2E1 interaction docking of this ligand to enzyme active center was carried out. Spatial structure of EMB was taken from PubChem data base (CID 14052). This spatial structure was optimized with ArgusLab Software (Mark Thompson and Planaria Software LLC) using molecular mechanics methods and Universal force field. The same software using Lamarkian Genetic Algorithm was applied for docking.
Evaluation of CYP2E1 mRNA expression
The expression of CYP2E1 mRNA in testes was determined by a reversed transcriptase polymerase chain reaction (RT-PCR). After collection of the testes samples (25 mg), they were snap frozen in liquid nitrogen, and stored at −80°C before RNA extraction. The isolation of total mRNA was carried out with a TRIZOL Reagent (Sigma-Aldrich, Inc., St. Louis, Missouri, USA). The integrity and concentration of RNA was analysed in a 2% agarose gel. First-strand complementary DNA (cDNA) was synthesized using a First-Strand cDNA Synthesis Kit (Fermentas, Germany) according to the manufacturer’s protocol. The reaction mixture contents for PCR, amplification protocol, and specific primers for the CYP2E1 gene were chosen according to Lankford et al. 26 The primer sequences were: sense, 5′-CTTCGGGCCAGTGTTCAC-3′ and anti-sense, 5′-CCCATATCTCAGAGTTGTGC-3′. RT-PCR with primers of β-actin sense, 5′-GCTCGTCGTCGACAACGGCTC-3′ and antisense 5′-CAAACATGAT CTGGGTCATCTTCT-3′ was carried out for internal control. All the primers were synthesized by “Metabion” (Germany). The MyCycler termocycler (BioRaD, Hercules, California, USA) was used for amplification. PCR products (CYP2E1-744 bp and β-actin-353 bp) were separated in a 2% agarose gel, stained with ethidium bromide, and visualized under a ultraviolet (UV) transilluminator (BioRaD). Data analysis was carried out with Quantity One Software (USA) and presented in relative units as CYP2E1 mRNA contents/β-actin mRNA ratio.
Immunohistochemical study of testicular CYP2E1
Immunohistochemical staining for testicular CYP2E1 was performed using 4 μm-thick sections of BS-fixed, paraffin-embedded sections. Briefly, tissue sections were dewaxed in xylene and were placed in water through graded alcohols. Antigen retrieval has been performed by microwaving slides in 10 mM citrate buffer (pH 6.2) for 30 min at high power, according to the manufacturer’s instructions. Human polyclonal antibody against CYP2E1 (Thermo scientific, Waltham, Massachusetts, USA) were used as primary antibodies. To remove the endogenous peroxidase activity, the sections have been treated with freshly prepared 1.0% hydrogen peroxide in the dark for 30 min at 37°C temperature. Nonspecific antibody binding was blocked by means of blocking serum. The sections were incubated for 30 min, at 37°C temperature, with the primary antibodies against CYP2E1 diluted 1:100 in phosphate-buffered saline (PBS) pH 7.2 then a triple washing with PBS follows. Anti-(rabbit IgG)–horseradish peroxidase conjugate (1:40,000 dilution) has been fulfilled for the detection of the CYP2E1 primary antibodies, then the sections were incubated for 20 min, at 37°C temperature. The reaction products were visualized with 3,3′-diaminobenzidine tetrahydrochloride. Immunohistochemical study of testicular CYP2E1 was performed in EMB-treated and vehicle-treated rats, respectively. Any intensity of reactivity for CYP2E1 in testes was considered positive. The proportion of testes staining positive was scored semiquantitatively as positive, focal/weakly positive, or negative.
Biochemical assays
Samples of testes was homogenized in 0.05 M Tris-hydrochloride, pH = 7.4 buffer with a teflon-glass Potter-Elvehjem homogenizer, and aliquots of crude homogenate were used for ascorbate- and nicotinamideadenine dinucleotide phosphate (NADPH)-induced TBARS rate formation investigations. 27
The contents of reduced glutathione in testes homogenates were determined as described by Sedlak and Lindsay. 28
Microsomal fraction of testes was obtained by the method of Kamath et al., 29 and aliquots were kept frozen at –70°C until needed.
p-Nitrophenol (PNP) hydroxylase activity—a selective enzyme marker for CYP2E1 was determined in microsomal fraction of testes according to the method of Koop. 30
Microsomal proteins contents were determined with Total Protein Kit, Micro Lowry, Onishi & Barr Modification (Sigma-Aldrich).
DNA isolation
DNA from rat testes was isolated as described previously. 31 Briefly, tissue was homogenized and digested in digestion buffer (100 mM sodium chloride, 10 mM Tris-hydrochloride, 25 mM ethylenediaminetetraacetic acid (EDTA), pH 8, and 0.5% sodium dodecyl sulfate) and freshly added 0.1 mg/mL proteinase K (Sigma-Aldrich) (1:1.2 mg/ml) with shaking at 50oC for 15 h. RNA was degraded by incubation of the samples with 1–100 mg/mL thermostable RNAse H for 1.5 h at 37°C. DNA was extracted with an equal volume of phenol-chloroform-isoamyl alcohol (25:24:1) and centrifuged for 10 min at 1700 × g. The DNA was precipitated by adding 0.5 vol 7.5 M ammonium acetate and 2 vol 100% ethanol to the aqueous layer; samples were separated by centrifugation at 1700 × g for 5 min, rinsed with 70% ethanol, and air-dried. The pellet was dissolved in Tris/Borate/EDTA (TBE) buffer (10 mm Tris-HCl and 1 mm EDTA, pH 8), and then were fractionated through 2% agarose gels (50–60 V; 3.5 h). After electrophoresis gels were stained with ethidium bromide and visualized under a UV transilluminator (Bio-Rad). Electrophoresis data analysis was carried out with Quantity One Software (USA).
Statistical analysis
Numerical data are represented as means ± SE and were compared using unpaired Student’s t test to find out the level of significance between control and experimental rats. Differences were considered statistically significant at p < 0.05. Statistical analyses were performed using the Excel Statistics 2007 (Microsoft, Redmond, Washington, USA).
Results
Testosterone level and fertility
EMB administration significantly influenced the total TS level (Figure 1). The level of total TS in EMB-treated group was lower 1.6 folds as compared with control.

Total TS level in male rats post-EMB treatment (n = 6). **p < 0.01 in comparison with control. EMB: ethambutol.
Along with the decrease in TS level, a reduction in EMB-treated males’ fertility was recorded. These data are presented in Table 1.
Male rats’ fertility index following EMB treatment.

EMB: ethambutol.
The fertility index in the group treated with EMB was almost three folds lower as compared to the control group.
Spermatogenesis parameters evaluation
Administration of EMB caused a development of destructive changes in spermatogenic epithelium in rats. It is seen from the data summarized in Table 2 that spermatogenic index at experimental group was significantly decreased in comparison with control group. Primary spermatogenesis cell population also was affected, namely mitotic activity was inhibited and number of spermatogonia in testes tubules sections was decreased. In addition, the number of cells at XII stage of spermatogenesis (characterizing primary spermatocytes meiotic division processes) was 1.8 times lower than in control.
Parameters of spermatogenic epithelium in testes of EMB-treated rats (n = 6).

EMB: ethambutol.
a p < 0.001 in comparison with control.
b p < 0.05 in comparison with control.
Beside the above-mentioned quantitative changes, qualitative changes of spermatogenic epithelium were also present in the seminiferous tubules of EMB-treated rats (Table 2, Figure 2).

Degenerative changes in rats’ testes. Hematoxilin and eosin; A—Proper structure of control group seminiferous tubules, ×400; B—exfoliation (ex) of epithelial cells into the lumen of seminiferous tubule (EMB-treated rat), ×400; C—tubule with loss of spermatogenic epithelium—“windows” (w) (EMB-treated rat), ×400; E—desquamated (d) (EMB-treated rat); E—spermatogenic epithelium became thin (th) (EMB-treated rat), ×400; F—separation of tubules, edema (EMB-treated rat), ×200. EMB: ethambutol.
It was observed 2.8 folds increasing of epithelial cells desquamation in rats’ testes (Table 2). Although, noticeable degenerative changes in testes such as epithelium exfoliation into the lumen of seminiferous tubules, desquamation of epithelium, and presence of cell-free regions (windows) were detected in substantial quantity at the experimental group, they didn’t reach the level of statistical significance due to high inter-individual differences (Table 2, Figure 2). In the testes of EMB-treated rats separation of tubules and edema were observed too (Figure 2).
Therefore, it can be concluded that our results indicate considerable spermatogenesis impairment, which, in turn, causes lowering of sperm count in EMB-treated animals 19% (Figure 3).

Epididymal suspension sperm count in rats following EMB treatment (n = 6). **p < 0.01 in comparison with control. EMB: ethambutol.
Computer simulation of EMB and CYP2E1 interaction results
The most energetically efficient complex of CYP2E1 with EMB was selected during protein-ligand docking (Table 3, Figure 4).
Interaction energy (ΔG gibs) and amino acid residues, which can interact with ligand.

EMB: Ethambutol.

Location of EMB in active center of CYP2E1 (green color). Amino acids residues, which can interact with ligand, are shown. EMB: ethambutol; CYP2E1: cytochrome P-450 2E1.
Computer simulation showed that EMB can directly interact with CYP2E1 active site and heme (Figure 5). Enzyme–EMB binding energy approaches to protein–substrate interaction level (Table 3).

Average rate of CYP2E1 mRNA expression, (n = 6) (panel A) and CYP2E1 enzymatic (PNP hydroxylase) activity, n = 6 (panel B) in testes of EMB-treated rats. Representative electrophoregrams of CYP2E1 (744 bp) and reference-gene β-actin (353 bp) RT-PCR products are shown in panel C. *p < 0.05 in comparison with control; ***p < 0.001 in comparison with control; CYP2E1: cytochrome P-450 2E1; PNP: p-Nitrophenol; EMB: ethambutol; RT-PCR: reversed transcriptase polymerase chain reaction.
Evaluation of testicular CYP2E1 mRNA and protein levels, and its enzymatic activity
RT-PCR was performed to evaluate the effect of EMB administration on CYP2E1 transcriptional activation in testes. The expression of CYP2E1 mRNA in EMB-treated rats’ testes was increased by 8.7 folds in comparison with control group (Figure 5(a) and (c)).
We also determined the PNP hydroxylase activity as a selective enzyme marker for CYP2E1. The significant elevation (three folds) of CYP2E1 enzymatic activity in the testicles of EMB-treated rats is seen from Figure 5(b).
The evaluation of the testicular CYP2E1 level was performed using immunohistochemical analysis. The immunoexpression of CYP2E1 in testes was confirmed by the presence of brown staining. Figure 6 illustrates broad expression of CYP2E1 in EMB-treated rats’ testes that was significantly higher than in control group (Figure 6).
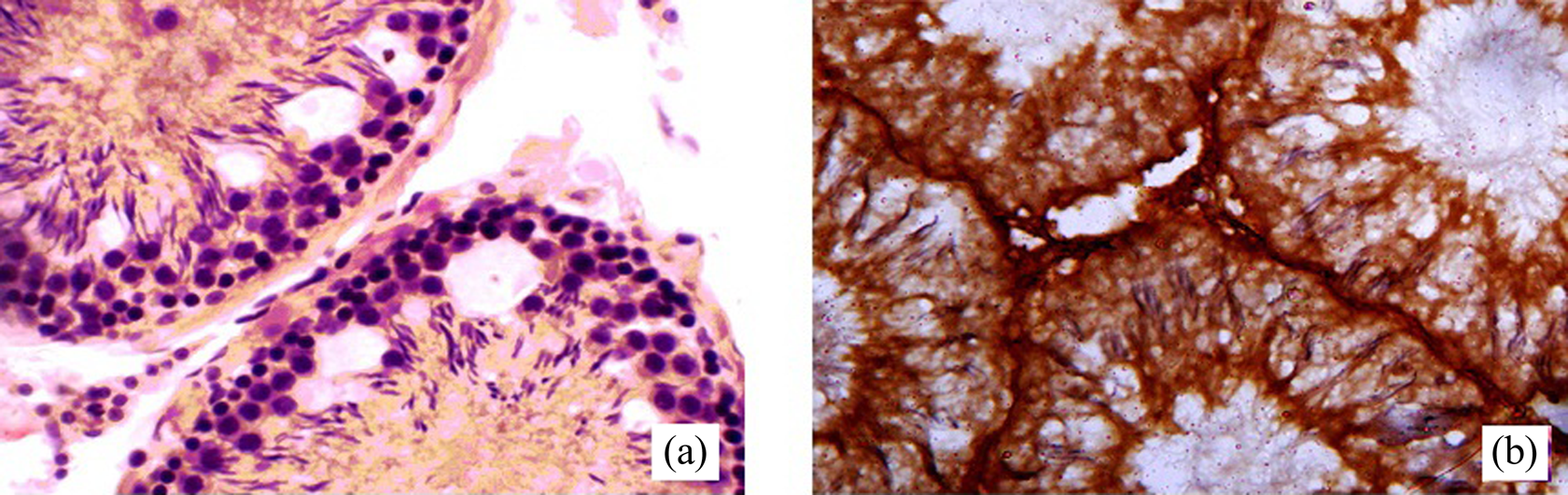
Immunohistochemistry analysis of CYP2E1: A—negative reaction in testis of control rat; B—strongly positive reaction in testis of EMB-treated rat. CYP2E1: cytochrome P-450 2E1; EMB: ethambutol.
Pro- and anti-oxidant parameters in testicular tissues
In order to explore the impact of EMB on the pro- and anti-oxidant state of the rat’s testes, the parameters of lipid peroxidation and reduced glutathione content were evaluated. The significant increase in level of endogenous lipid peroxidation in testes of EMB-treated rats was detected. The rate of ascorbate- and NADPH-induced formation of TBARS was substantially elevated 1.4 and 2.4 folds, respectively, in comparison with control. Along with that, the reduced glutathione content was on 13% less than in control group (Table 4).
Pro- and antioxidant parameters in testicular tissues of EMB-treated rats (n = 6).

EMB: ethambutol; TBARS: thiobarbituric acid-reactive substances; NADPH: nicotinamideadenine dinucleotide phosphate.
a p < 0.05 in comparison with control.
b p < 0.001 in comparison with control.
c p < 0.01 in comparison with control.
DNA fragmentation level in testes
The testicular DNA fragmentation was studied for apoptosis detection. EMB-exposed rats showed substantial intensification of apoptosis in testicular cells as compared with control (Figure 7).

DNA fragmentation level in EMB-treated rats’ testes. Panel A—electrophoregram of DNA fragmentation. Panel B—relative level of different molecular weight DNA fragments. Analyses were carried out using the Quantity One Software. EMB: ethambutol.
The level of testes DNA fragmentation of EMB-treated rats was significantly higher than in controls. Namely, we detected two main fractions of high-weighted DNA fragments over 1000 bp. Among this five fractions of DNA fragments below this level (1000, 900, 800, 700, and 400 bp) were detected. Single fraction of low-weighted DNA fragments was 20 bp. In control, we observed only two fractions of DNA fragments: one—with weight over 1000 bp; and one—low-weighted (20 bp).
Discussion
Despite some early experimental evidence of EMB gonadal toxicity, 13,14 to date, just little emphasis has been placed on profound examination of these effects and probable involved mechanisms. We suppose that the results represented in this article could clarify the biochemical and molecular changes at testicular levels in response to EMB exposure.
After analyzing our results, first, the definite evidences of the spermatogenesis impairment in EMB-treated male rats can be seen. For instance, the decrease of spermatogenic index and spermatogonia number confirms depression of spermatogenic cells activity. In addition, we have detected intensification of germ cells exfoliation into the lumen of the seminiferous tubules, as evidence of loss of their adhesion with Sertoli cells or shearing of Sertoli cells cytoplasm. 32 We also demonstrated that alterations of spermatogenesis along with low TS level have led to a reduction in spermatozoids production. Following EMB administration the total sperm count was markedly reduced and as a result the fertility index for experimental males was three times lower than in control group.
We have previously shown that EMB, INH, RMP, and PZA coadministration to male rats led to increase in CYP2E1 mRNA content in testes, 5 as well as CYP2E1 mRNA and enzymatic activity in liver. 33 Notably, the ability of INH to induce CYP2E1 in the liver has been proved in many studies. 34 –36 In addition, our recent experiments demonstrated induction of testicular CYP2E1 following INH administration. 6 Important to note, that interactions between other anti-tuberculosis drugs and CYP2E1 have not been investigated yet extensively.
We have demonstrated the significant increase of CYP2E1 mRNA and protein levels in testes, as well as marked activation of CYP2E1-catalyzed oxidation of PNP. This clearly indicates the testicular CYP2E1 induction following the long-term EMB administration to male rats in therapeutic dose. It is agreed with recent data that showed the EMB-related increase in CYP2E1 mRNA expression and enzymatic activity in rat’s liver. 37
The ability of EMB to cause an increase in testicular CYP2E1 mRNA and protein contents, as well as its activity, probably, could be achieved by different physiological mechanisms at the levels of transcription, mRNA stabilization, translation, and enzyme stabilization. 38 Apparently, a mechanism involved in CYP2E1 induction may depend on the drug dose, duration of action, and/or route of administration. 39
As for the EMB-induced increase of testicular CYP2E1 protein level and its enzymatic activity in our experiments, we could assume a mechanism, which is partially similar to such for ethanol (CYP2E1 protein stabilization). 40,41
Our computer simulation data demonstrate that EMB directly interacts with CYP2E1 active site and heme. Such location of EMB is typical for CYP2E1 substrates, but ligand interacts with heme by its hydroxyl group, and thus it could not be metabolized. Enzyme-EMB binding energy approaches to protein–substrate interaction level. This makes it possible to assume that EMB could be a weak CYP2E1 competitive inhibitor. These results are in accordance with recently published results of in vitro study with pooled human liver microsomes. 42 In these experiments, Lee et al. have shown strong inhibitory potential of EMB against CYP2E1, based on the half maximal inhibitory concentration values using liquid chromatography–electrospray ionization tandem mass spectrometry. In addition, inhibition of CYP2E1 was not increased by preincubation with EMB and β-NADPH, suggesting that the EMB-induced CYP inhibition may not be metabolism-dependent. According to results obtained from kinetic analysis the authors supposed that the inhibition of CYP2E1 by EMB was apt to a competitive inhibition model. 42
Overall, we can assume that EMB interacts with CYP2E1 in distinct manners. On the one hand, the computer stimulation and another in vitro data 42 suggest EMB ability to inhibit CYP2E1, and on the other hand, our and another in vivo experiments data 37 have given the compelling evidence that EMB can induce this particular isoform. Differences in the ability of EMB to inhibit or induce the CYP2E1 could be explained by discrepancies between in vitro and in vivo experimental conditions. However, this phenomenon needs to be profoundly investigated.
Nevertheless, data on EMB and CYP2E1 interplay is controversial; this allows us to suppose involvement of this isoform in EMB toxicity mechanisms. CYP2E1 is known to be one of the most active CYPs in producing reactive oxygen species (ROS), such as the superoxide anion radical and hydrogen peroxide. In the presence of iron catalysts, it produces powerful oxidant, such as the hydroxyl radical. 43
It is known that ROS play a central role in sperm physiology, such as sperm maturation and capacity, but unusual ROS production is associated with defective sperm function. Owing that delicate balance between ROS production and recycling is essential for spermatogenesis the excessive generation of seminal ROS can cause male infertility. 44
Based on our results demonstrating rise in ascorbate- and NADPH-induced formation of TBARS in testicles of EMB-exposed animals, we suppose that EMB-mediated CYP2E1 induction could contribute to an imbalance of oxidative processes and antioxidant defense in the testicular tissues. Our results on pro-oxidant action of EMB are in compliance with other authors’ data suggesting the ability of EMB to induce an oxidative stress in myocardial tissue. 45 We speculate that shift of the equilibrium toward oxidizing conditions can manifest not only in relation with CYP2E1, but also due to EMB and its metabolites ability to form complexes with zinc 46 which is involved in numerous aspects of cellular metabolism. 47 Nair et al. 48 have been demonstrated that in rats zinc deprivation resulted in increased sensitivity of testes and epididymis to oxidative stress, as a consequence of increased ROS generation and/or decreased zinc-dependent antioxidant processes. In addition depletion of zinc may enhance DNA damage via impairments of DNA repair mechanisms. 49
Reduced glutathione (GSH) is probably one of the most important nonenzymatic antioxidant present in testicular cells. Its significant functions in spermatogenesis and the reproductive process have been reported. 50 Enzymes involved in GSH production are critical to the cells’ ability to protect themselves against oxidative stress. 51 In testicular tissues, it has been found, several isoforms of glutathione peroxidases that use GSH as a source of electrons to reduce hydrogen peroxide to water. They are localized in the mitochondria, nucleus and acrosomal domain of differentiating spermatozoa. 52 Thus, EMB-induced reduction of testicular glutathione level can lead to germ cells integrity disruption.
Notably, intracellular GSH levels are known to play a key role in the regulation of ROS-mediated apoptosis. 53 Our results regarding escalation of DNA fragmentation in testes of EMB-treated rats are in good accordance with our previous data, which demonstrated presence of epigenetic effects of other antitubercular medicines in vivo, 31 and with another author’s results on EMB-stimulated cell death and inhibition of normal DNA synthesis in vitro. 54 Such changes could correlate with changes in levels of tumor necrosis factor alpha, interleukin-10, and nitric oxide production, and caspase-1 activation. 55 Differences in DNA-fragmentation processes between control and EMB groups could be caused by changes in effectiveness of DNA-fragmentation processes. 56 Taking into account that apoptosis plays an important regulatory role in testicular homeostasis and production of high quality sperm, its dysregulation in germ cells due to external disturbances, such as exposure to certain chemotherapeutic agents, 57 can contribute to reduced fertility and impact on the health of future progeny. 58,56 Increased apoptotic degeneration in testes may cause irreversible changes in the germ cells associated with decreased epididymal sperm concentration, motility, and fertility index which contributes to the low efficiency of spermatogenesis. 59
It appears, therefore, that revealed by us, EMB-caused CYP2E1 induction, oxidative stress, and apoptosis at the level of the testes could be the reasons of steroidogenesis enzymes inhibition and disruption of the capacity of the germinal epithelium to differentiate normal spermatozoa. 60,61 Our results on testicular CYP2E1 induction following EMB administration could be of importance because this process is localized in Leydig cells, 7,8 which provide the synthesis of androgens necessary for the maintenance of spermatogenesis and extra-gonadal androgen actions in mammals. 62 Consequently, these structures, and their microenvironment damage by free radicals as a result of EMB-mediated CYP2E1 induction, could be one of the reasons of steroidogenesis enzymes inhibition and spermatogenesis disruption. It is known that CYP2E1 is an effective generator of hydrogen peroxide, 63 and it acts directly on rat Leydig cells to diminish TS production by inhibiting cytochrome P450 side chain cleavage enzyme (P450scc) activity and steroidogenic acute regulatory (StAR) protein expression. 64 Moreover, recently it has been reported, that ROS signaling-mediated c-Jun upregulation suppresses the expression of steroidogenic enzyme genes by inhibiting Nur77 transactivation (one of the major transcription factors that regulate the expression of steroidogenic enzyme genes), resulting in the reduction of testicular steroidogenesis. 65
On the other hand, we assume that well-known zinc-chelating effect of EMB and its metabolite 46 also can contribute to TS deficiency development. For instance, another ultrastructural study in zinc-deficient rats’ testes have revealed several apoptotic features such as wavy basement membrane, displaced nuclei, chromatin condensation, plasma membrane blebbing, nuclear membrane dissolution, loss of inter-Sertoli cell junctional complexes, and intercellular bridges and deformed mitochondria. 59 The association between zinc concentrations in organism and TS levels has also been demonstrated in human males. 66 The fact that lowered level of TS can affect Sertoli cells function and negatively influence spermatogenesis is of importance. 67
Our findings also prove the prominent role of EMB in male reproductive toxicity of antituberculosis drug combination, which has been revealed by us previously. 4 In the light of our present and previous observations, it seems warranted to conclude that the critical examination of both laboratory animals’ and epidemiological data is required.
Conclusion
Our data clearly shows that EMB has testicular toxicity, even being administered in therapeutic dose. Notably, it is possible to state that EMB has an ability to modify CYP2E1 mRNA expression and enzymatic activity in rats’ liver and testes. These events are accompanied by imbalance of testicular pro-/antioxidant system, changes in levels and character of DNA fragmentation, as well as spermatogenic epithelium damage, TS inhibition, decrease of sperm number, and loss of fertility in comparison with untreated animals. However, future experiments are necessary to address the precise mechanisms of EMB gonadal toxicity. Particularly the links between zinc-chelating effect of EMB and its ability to provoke spermatogenesis disorders needs to be explored in depth.
Footnotes
Acknowledgments
The authors would like to thank Yevhen Kuzenko for immunohistochemical studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

