Abstract
The fungicide carbendazim (CBZ) and insecticide chlorpyrifos (CPF) are currently applied together by farmers for the control of pests. Here, we investigated the impacts of 7 days oral co-exposure to 10 mg/kg body weight of CPF and 50 mg/kg body weight of CBZ on selected oxidative stress and antioxidant biomarkers in the liver, kidney, and spleen of female rats. The results showed that while the body weight gain and relative organ weights were not significantly affected after separate exposure to CPF and CBZ, there was a significant decrease in the body weight gain with concomitant increases in the relative kidney and spleen weights of rats treated with the mixture. Also, CPF and CBZ co-exposure significantly increased the levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), urea, and creatinine (p < 0.05) when compared with the groups treated with CBZ or CPF alone and the control. The significant decreases in both antioxidant enzymes activities and nonenzymatic antioxidant level following individual administration of CPF and CBZ to rats were intensified in the co-exposure group (p < 0.05). Additionally, the marked increases in the levels of oxidative stress indices in liver, kidney, and spleen of rats treated with CPF or CBZ alone were intensified in the co-exposure group (p < 0.05). Histopathologically, co-exposure to CPF and CBZ exacerbates their individual effects on the liver, kidney, and spleen. These findings showed that co-exposure to CPF and CBZ in rats elicited more severe oxidative damage on the liver, kidney, and spleen of the rats, indicative of an additive effect compared to CPF or CBZ alone and as such, may pose a greater environmental risk to humans.
Introduction
Exposure to pesticide mixtures continues to create serious challenges to the natural environment and human health. 1,2 An increasing number of studies have demonstrated the concurrent presence of different pesticides in fruits and vegetables commonly consumed by humans. 3 –5 Organophosphate pesticides are the major class of agricultural chemicals commonly used in controlling a wide range of pests in field crops, fruits, and vegetables. 6 Chlorpyrifos (CPF; O,O-diethyl O-3,5,6-trichloropyridin-2-yl phosphorothioate) is a colorless to white crystalline solid organophosphate insecticide. Due to the risks posed by CPF to human health, the US Environmental Protection Agency in the year 2000 placed restriction to its domestic applications. In spite of this, CPF is still widely used as an insecticide in agricultural practices. 7 In fact, CPF is currently used for the maintenance of public recreational areas. 8 The contamination of air and water courses, which are about 15 miles from the site of CPF application, has been attributed to its potential for long-range environmental transport. 9 Exposure to CPF is known to produce numerous detrimental effects such as neurotoxicity, genotoxicity, teratogenicity, and endocrine disruption in both humans and animals. 10 –13
Carbendazim (CBZ; methyl-2-benzimidazole carbamate) is a systemic broad-spectrum fungicide currently used in the control of various foliar diseases on arable crops such as cereals, fruits, flowers and flower bulbs, sugar, and fodder beet. 14,15 Exposure to CBZ occurs via inhalation of fumes during residential and occupational applications as well as ingestion of contaminated water, fruits, and vegetables. 16 Exposure to CBZ adversely impacts the environment and potentially human health due to its persistence in soil and tissues such as skin, adipose tissue, gonads, and liver. 17,18 CBZ exposure elicited detrimental effects in rats on vital organs including liver, kidney, and testis thereby resulting in functional and structural damage. 19 –21
Most often, people are exposed to a mixture of chemicals rather than to single chemicals. The nature of coexistence of numerous xenobiotics in complex environmental samples including fruits, food, and water is widely reported. 22 –25 Chemicals occurring as complex mixtures have the potential for interactions and may consequently cause toxicological effects which are difficult to predict based on single chemical toxicological data. 26,27 Chemical–chemical interactions following exposure to chemical mixtures could lead to a stronger effect (additive or synergistic) or a weaker effect (antagonism or inhibition). 28 Moreover, because children are commonly susceptible to the adverse effects of pesticides, there is increasing public health concern about the need for identification and risk assessment of exposure to mixtures of chemicals. 29,30
The present study aimed at investigating the nature of toxicity following co-exposure to fungicide CBZ and insecticide CPF which are commonly applied together by farmers for control of pests and improvement of farm productivity. To achieve this aim, prepubertal female Wistar rats were exposed to CPF and CBZ separately or in combination. Subsequently, endpoints including markers of hepatic and renal damage as well as antioxidant enzyme activities, oxidative stress parameters and histopathology of the liver, kidney, and spleen were evaluated.
Materials and methods
Chemicals
Technical-grade CPF from Milenia Agrociências S.A., Paraná, Brazil was used for this study. CBZ (97% pure), epinephrine, glutathione (GSH), thiobarbituric acid, 1-chloro-2,4-dinitrobenzene, hydrogen peroxide (H2O2), and 5′,5′-dithiobis-2-nitrobenzoic acid were procured from Sigma Chemical Co. (St. Louis, Missouri, USA). All other reagents were of analytical grade and were obtained from the British Drug House (Poole, Dorset, UK).
Experimental animals and research design
Forty female Wistar rats (4 weeks old, 90 ± 4 g) obtained from the Department of Biochemistry, University of Ibadan, Ibadan, Nigeria, were used for this study. The rats were housed in plastic cages placed in a well-ventilated rat house, provided rat feed and water ad libitum and subjected to natural photoperiod of 12-h light/12-h dark cycle. All the animals received humane care according to the criteria stated in the “Guide for the Care and Use of Laboratory Animals” prepared by the National Academy of Science (NAS) and published by the National Institute of Health. The experiment was performed in accordance with the US NAS guidelines and approval by the University of Ibadan Ethical Committee. The research design involves treatment of the rats with the chemicals for 7 days as indicated below: Group 1: Control rats orally treated with 2 ml/kg of corn. Group 2: Rats orally treated with 10 mg/kg CPF dissolved in corn oil.
31,32
Group 3: Rats orally treated with 50 mg/kg CBZ dissolved in corn oil.
19
Group 4: Rats orally treated with the mixture of CPF (50 mg/kg) and CBZ (10 mg/kg) dissolved in corn oil.
Twenty-four hours following the last treatment, the overnight-fasted rats were killed by cervical dislocation, and blood was collected by retro-orbital bleeding. Plasma samples were separated from blood cells by centrifugation at 3000 × g for 10 min. Subsequently, plasma samples were stored frozen at −20°C until the liver and kidney function indices were determined.
Determination of liver and kidney function markers
The liver and kidney function indices were assessed using commercially available kits from Randox Laboratory Limited (UK). Plasma activities of aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were determined according to the method described by Reitmann and Frankel. 33 The levels of plasma urea and creatinine were estimated according to the method of Jendrassik and Grof. 34
Estimation of liver, kidney, and spleen oxidative stress and antioxidant markers
The liver, kidney, and spleen of rats were homogenized in 50 mM Tris–HCl buffer (pH 7.4) containing 1.15% potassium chloride. The homogenate from each of the organs was subsequently centrifuged at 12,000 × g for 15 min at 4°C to obtain the post mitochondria fraction, and the supernatant was collected for biochemical estimations. Protein concentration was determined according to the method of Lowry et al. 35 Superoxide dismutase (SOD) activity was determined according to the method described by Misra and Fridovich, 36 while catalase (CAT) activity was determined using H2O2 as a substrate according to the method described by Clairborne. 37 Reduced GSH level was determined at 412 nm according to the method described by Jollow et al., 38 while glutathione-S-transferase (GST) activity was determined based on the method of Habig et al. 39 H2O2 generation was measured according to the method described by Wolff. 40 Lipid peroxidation was quantified as malondialdehyde (MDA) according to established method, 41 and the result was expressed as micromoles of MDA per milligram protein. In addition, the level of nitric oxide (NO) was determined in the samples by the method of Green et al. 42 while that of myeloperoxidase (MPO) was estimated using the method described by Granell et al. 43
Microscopic examinations of liver, kidney, and spleen
Samples of liver, kidney, and spleen from rats in each group were processed for histology according to standardized procedure. 44 Briefly, the tissues were fixed in 10% phosphate buffered formalin (PBF) for 3 days. The PBF solution was changed daily to minimize shrinkage and prevent the occurrence of artefacts. Following dehydration procedures, the samples were embedded in paraffin. Sections of 4–5 µm were cut by a microtome and stained with hematoxylin and eosin. All slides were coded before examination under a light microscope (Olympus CH; Olympus, Tokyo, Japan) by pathologists who were blinded to control and treatment groups. Photomicrographs were taken with a Sony DSC-W 30 Cyber-shot (Sony, Tokyo, Japan).
Statistical analysis
Statistical analysis was carried out using one-way analysis of variance to compare the experimental groups followed by Bonferroni’s test to identify significantly different groups (SPSS for Windows, version 17). The value of p < 0.05 was considered statistically significant.
Results
Influence of treatment with CPF and CBZ, separately and in combination, on body weight and relative organ weights of rats
Table 1 presents the body weight and the relative liver, kidney, and spleen weights of rats treated with CPF and CBZ separately or as a mixture for 7 consecutive days. Separate administration of CPF or CBZ did not affect the final body weight and the relative liver, kidney, and spleen weights in all the treatment groups when compared with the control. However, rats co-treated with CPF and CBZ showed significant decrease in the final body weights, whereas the relative organ weights were significantly increased compared with the control (p < 0.05). Furthermore, CPF as well as CBZ treatments significantly decreased the body weight gain of the experimental animals when compared with the control. Co-exposure of rats to CPF and CBZ significantly decreased the body weight gain of the treated rats compared with the control, CPF alone, and CBZ alone groups (p < 0.05).
Body weight and relative organ weights (g/100 g bw) of rats following exposure to chlorpyrifos, carbendazim, and their mixture for 7 consecutive days.

CPF: chlorpyrifos; CBZ: carbendazim; bw: body weight. Values are expressed as mean ± SD of eight rats.
a p < 0.05 versus control.
b p < 0.05 versus CPF only.
c p < 0.05 versus CBZ only.
Hepatic and renal function indices of rats after treatment with CPF and CBZ for 7 days
The data on the hepatic and renal function parameters in rats treated with CPF and CBZ separately or in combination for 7 days are presented in Table 2. The activities of AST and ALT were significantly increased following separate oral administrations of rats to CPF and CBZ. This effect was more pronounced in the CPF and CBZ co-exposure group. Whereas AST activity was significantly (p < 0.05) increased by 61%, 150%, and 205%, ALT activity was significantly increased by 116%, 116%, and 278% in rats treated with CPF alone, CBZ alone, and co-exposure group, respectively, when compared with the control (p < 0.05). Administration of CPF and CBZ separately caused significant increases in plasma urea and creatinine levels. These increases were intensified in the rats treated with CPF and CBZ co-exposure. Urea level was significantly (p < 0.05) increased by 122%, 99%, and 201%, while creatinine level was elevated by 81%, 52%, and 111% in rats treated with CPF alone, CBZ alone, and co-exposure group, respectively, when compared with the control (p < 0.05).
Liver and kidney function indices following exposures of rats to chlorpyrifos, carbendazim, and their mixture for 7 consecutive days.

CPF: chlorpyrifos; CBZ: carbendazim; AST: aspartate aminotransferase; ALT: alanine aminotransferase. Values are expressed as mean ± SD of eight rats.
a p < 0.05 versus control.
b p < 0.05 versus CPF only.
c p < 0.05 versus CBZ only.
Antioxidant status of the liver, kidney, and spleen of rats after exposure to CPF and CBZ for 7 days
Effects of separate administration of CPF, CBZ, and their co-treatment on antioxidant defense system and biomarkers of oxidative stress are presented in Figures 1 to 4. Exposure to CPF alone and CBZ alone resulted in significant decrease in antioxidant enzymes and GSH level, but caused significant elevations in the inflammatory and lipid peroxidation biomarkers in the liver, kidney, and spleen when compared with the control (p < 0.05). Specifically, SOD activity in all investigated organs decreased significantly following individual administration of CPF and CBZ and was intensified in the co-exposure group. Moreover, while the hepatic CAT activity was not significantly affected in all treatment groups, there were significant decreases in kidney and spleen CAT activities following individual administration of CPF and CBZ and the decreases were intensified in the co-exposure group. Similarly, the significant decrease in the GST activity and the reduction in GSH level in rats separately treated with CPF and CBZ was intensified in all investigated organs of the co-exposed rats. Conversely, the significant increases in MPO activity and the marked elevation in the levels of NO, H2O2, and MDA in rats separately administered with CPF and CBZ were intensified in the exposure group (p < 0.05).

Activities of SOD and CAT in the liver, kidney, and spleen of rats following oral treatment with CPF, CBZ, and their mixture for 7 consecutive days. Values are expressed as mean ± SD of eight rats. a p < 0.05 versus control; b p < 0.05 versus CPF only; c p < 0.05 versus CBZ only. CPF: chlorpyrifos; CBZ: carbendazim; SOD: superoxide dismutase; CAT: catalase; SD: standard deviation.

Activity of GST and GSH level in the liver, kidney, and spleen of rats following oral treatment with CPF, CBZ, and their mixture for 7 consecutive days. Values are expressed as mean ± SD of eight rats. a p < 0.05 versus control; b p < 0.05 versus CPF only; c p < 0.05 versus CBZ only. CPF: chlorpyrifos; CBZ: carbendazim; GST: glutathione-S-transferase; GSH: glutathione; SD: standard deviation.

Levels of H2O2 and LPO in the liver, kidney, and spleen of rats following oral treatment with CPF, CBZ, and their mixture for 7 consecutive days. Values are expressed as mean ± SD of eight rats. a p < 0.05 versus control; b p < 0.05 versus CPF only; c p < 0.05 versus CBZ only. CPF: chlorpyrifos; CBZ: carbendazim; LPO: lactoperoxidase; SD: standard deviation.

Level of NO and MPO activity in the liver, kidney, and spleen of rats following oral treatment with CPF, CBZ, and their mixture for 7 consecutive days. Values are expressed as mean ± SD of eight rats. a p < 0.05 versus control; b p < 0.05 versus CPF only; c p < 0.05 versus CBZ only. CPF: chlorpyrifos; CBZ: carbendazim; NO: nitric oxide; MPO: myeloperoxidase; SD: standard deviation.
Histopathological observations of liver, kidney, and spleen of rats treated with CPF and CBZ for 7 days
Figures 5 to 7 depict the representative photomicrographs of the liver, kidney, and spleen from the experimental groups. The results of microscopic examination of the organs are summarized as follows. The liver, kidney, and spleen of control rats appeared structurally and functionally normal without any visible lesion (Figures 5(a), 6(a) and 7(a)). In the liver, CPF treatment resulted in congestion of vessels and mild periportal infiltration by inflammatory cells, whereas CBZ-treated rats showed focal area of necrosis and the presence of inflammatory cells. Co-exposure to CPF and CBZ caused hepatic necrosis and diffused Kupffer cell hyperplasia (Figure 5(b) to (d)). Moreover, administration of CPF alone caused congestion of renal vessels and focal areas of peritubular and periglomerular infiltration by inflammatory cells, whereas CBZ alone caused mild congestion and neutrophilic cellular infiltration of renal vessels. Co-exposure to CPF and CBZ caused renal necrosis and vacuolation (Figure 6(b) to (d)). The spleen of rats treated with CPF alone showed normal architecture, whereas CBZ-treated rats showed neutrophilic infiltration. CPF and CBZ co-exposure caused splenic architectural degeneration characterized with multifocal cellular infiltration (Figure 7(b) to (d)).
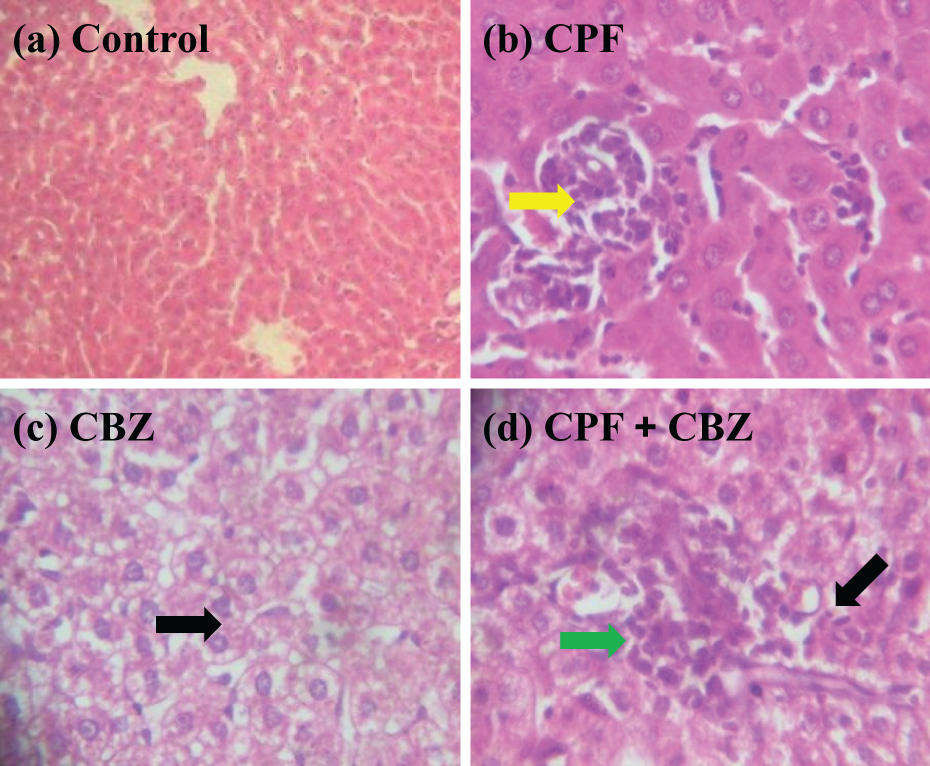
Photomicrographs of the liver. (a) Control liver showing normal architecture. (b) Liver of rats treated with CPF showing mild periportal infiltration by inflammatory cells (yellow arrow). (c) CBZ-treated rats showing focal area of hepatic necrosis (black arrow). (d) Rats liver exposed to mixture of CPF and CBZ showing hepatic necrosis (black arrow) and diffused Kupffer cell hyperplasia (green arrow). Original magnification: ×200. CPF: chlorpyrifos; CBZ: carbendazim.

Photomicrographs of the kidney. (a) Control rat showing normal renal architecture. (b) Rats treated with CPF showing congestion of renal vessels (blue arrow) and focal area of peritubular and periglomerular infiltration by inflammatory cells (green arrow). (c) CBZ-treated rats showing mild congestion (blue arrow) and neutrophilic cellular infiltration of renal vessels (green arrow). (d) Rat kidney exposed to mixture of CPF and CBZ showing neutrophilic cellular infiltration (green arrow), necrosis (green chevron), and vacuolation (black chevron). Original magnification: 200×. CPF: chlorpyrifos; CBZ: carbendazim.

Photomicrographs of the spleen. (a) Control rat showing normal splenic architecture. (b) Rats administered CPF showing normal splenic architecture. (c) CBZ-treated rats showing neutrophilic infiltration (black arrow). (d) Spleen of rat treated with mixture of CPF and CBZ showing architectural degeneration characterize with multifocal cellular infiltration (black arrow). Original magnification: ×200. CPF: chlorpyrifos; CBZ: carbendazim.
Discussion
Chemicals may interact in several ways leading to synergistic, antagonistic, or additive effects. 28,45 The co-occurrence of CPF and CBZ in the environment, and the potential toxicity of their co-exposure, are of great importance to human risk assessment. 46,47 The present study aimed at characterizing the effects of co-exposure of prepubertal rats to CPF and CBZ. The choice of the concentrations of CPF and CBZ were based on doses used in previous studies. 19,31,32
The present study demonstrated that while the body weight gain and relative organ weights were not significantly affected following separate exposures to CPF and CBZ, there were obvious deleterious effects on the body weight gain and relative organ weights in the rats co-exposed to these compounds when compared with the control. Co-exposure to CPF and CBZ caused a significant decrease in the body weight gain, but increased the relative kidney and spleen weights.
Aminotransferases (ALT and AST) are biomarkers enzymes of early acute hepatic damage because their serum or plasma activities apparently increase due to loss of hepatocyte structural integrity and leakage. 48 In the present study, individual administration of CPF and CBZ caused liver damage evidenced by the significant increase in these biomarkers of hepatic toxicity in the rats. The observed intensification in the AST and ALT activities in the co-exposure group may reflect additive effects of these chemicals on these markers of hepatic damage. Moreover, elevated plasma urea levels are indicative of decrease in reabsorption at the renal epithelium, whereas elevated plasma creatinine levels suggest kidney dysfunction, primarily in the glomerular filtration rate. 49 The renal damage caused by separate exposures of rats to CPF and CBZ was intensified in the co-exposure group as evidenced by the escalation in the levels of renal function indices namely urea and creatinine above the control and CBZ-treated rats.
The first line of protection of cell against oxidative damage is attributed to the mutual action between SOD and CAT. While SOD accelerates the conversion of endogenous cytotoxic superoxide radicals to H2O2, CAT converts the noxious peroxide radicals into water and oxygen. 50 The present investigation demonstrated that apart from hepatic CAT activity, there were significant decreases in SOD and CAT activities in all the investigated organs following separate administrations of CPF, CBZ and their mixture to rats. However, the diminution in the activities of these antioxidant enzymes was intensified in rats simultaneously exposed to CPF and CBZ, suggesting additive inhibition of their defensive activity against cellular oxidative damage in the rats.
GSH is an effective reductive nonenzymatic antioxidant which provides secondary line of defense against intracellular harmful effects of free radicals and peroxides generated by oxidative stress. 51 GST protects cellular macromolecules against noxious chemicals and oxidative damage by conjugating toxic electrophilic molecules with GSH. 52 –54 The direct involvement of GST, a phase-II metabolizing enzyme, in the detoxification mechanism of pesticides has been previously reported. 55 The reductions in the GSH level and GST activity in the liver, kidney, and spleen suggest GSH overutilization and inhibition of GST-mediated detoxification process in the treated rats.
The damaging effects of H2O2 molecules have been linked with their direct oxidizing properties and the indirect involvement in the generation of more harmful species like hydroxyl radicals and hypochlorous acid. 56 The present study showed that exposure to CPF, CBZ, and their mixture induced oxidative damage evidenced by the significant elevations in the levels of H2O2 and MDA, a biomarker of lipid peroxidation in the investigated organs.
Individual administration of CPF and CBZ to rats resulted in significant increases in MPO activity and NO level in all the tested organs. This effect was exacerbated in the tested organs of the rats treated with a mixture of CPZ and CBZ. Indeed, NO is well known to be a pro-inflammatory mediator that induces inflammation owing to excessive generation by inducible NO synthase (iNOS) in abnormal situations. 57 This elevated level of NO could increase the chemical reaction of NO with superoxide anion to generate a more deleterious nitrite anion which subsequently damages the liver, kidney, and spleen of experimental rats as shown in this study. 58
MPO possesses cytokine-like properties and has been reported to activate neutrophils. 59 MPO uses H2O2 to produce hypochlorite through a process well known to generate reactive oxygen species and consequently tissue damage. Several previous reports have demonstrated that an increase in MPO activity following tissue infiltration by mononuclear cells resulted in inflammation. 57 The depletion in the antioxidant status, elevated levels of inflammatory markers, and lipid peroxidation, were accompanied by hepatic, renal, and splenic oxidative damage in rats separately treated with CPF and CBZ, which was intensified in the co-exposure group. The histopathological findings in rats co-exposed to CPZ and CBZ, showed marked architectural degeneration characterized by hepatic necrosis and diffused Kupffer cell hyperplasia, renal necrosis and vacuolation, and splenic lesion with multifocal cellular infiltration. This further confirms the toxicity of CPZ and CBZ mixture in the rats. This study supports the findings of Palanikumar et al. 60 who reported alterations in the activities of selected antioxidant enzymes in milkfish Chanos chanos model exposed to CPF and CBZ.
In conclusion, the present results showed that CPF and CBZ co-treatment elicited greater detrimental impacts than the single treatment groups due to the additive adverse effects on hepatic, renal, and splenic functions of the rats. The combined toxicity of CPF and CBZ is attributable to increased oxidative stress and inflammation in the treated rats.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
