Abstract
Sulfamonomethoxine (SMM) is widely used in the veterinary field in China. Although some clinical surveys have revealed that sulfonamide antibiotics cause adverse nervous system symptoms, the related mechanisms of maternal SMM exposure on the neurobehavioral development of offspring remain unclear. Here, we investigated the effects of perinatal SMM exposure on the physiological and behavioral responses of pubertal offspring mice and the underlying mechanisms. We randomly allocated pregnant mice into the groups treated with SMM at different doses and the saline-treated groups. Maternal mice were orally administered SMM daily from gestational day 1 to postpartum day 21. On postnatal day (PND) 22, the parameters of growth, endocrine hormones, and brain amino acid composition were assessed, as well as the brain transcript levels of key genes involved in the mammalian target of rapamycin (mTOR) signaling pathway. From PND 50 to 55, a battery of behavioral tests relevant to anxiety and memory were then administered. Analysis of the results indicated that the pups, particularly the pubertal female offspring, showed anxiety-like behavior. Moreover, the pubertal offspring showed cognitive impairments and fat accumulation. Furthermore, the relative mRNA expression of genes involved in the mTOR signaling pathway in females on PND 22 was elevated, whereas the expression of N-methyl-
Introduction
China produces and uses a very large amount of antibiotics. As a result, serious misuse or overuse of antibiotics is widespread in China. A survey estimated that the antibiotic usage in China in 2013 was approximately 162,000 tons, and that sulfonamides (SAs) accounted for 5% of the total of antibiotics used. 1 SAs are widely used in human therapies, veterinary medicine, farming, and aquaculture because of their advantage of broad-spectrum antibacterial activity, low cost, and ease of use. Owing to their abusive use by farmers, many SAs, and their metabolites can be detected in surface water and food. The European Union has set a series of maximum residue limits (100 µg/kg) in animal products. However, studies in China suggest that 75% of meat samples contain SAs at a total concentration greater than 100 μg/kg. 2 In addition, SAs degrade slowly and remain active in the environment for months or even years, eventually entering the human body directly or indirectly, which constitutes a health hazard. SMM exhibits the highest antimicrobial activity among various SAs, and it has been the most widely used antibiotic for therapeutic or prophylactic proposes in food-producing animal. Despite the extensive use and its widespread contamination, the toxic or neurotoxic effects of early life exposure to SMM are less known, and the molecular mechanisms remain to be elucidated.
A recent study shows that SMM exhibited the growth inhibition of freshwater Chlorella vulgaris at the dose of 5.9 mg/L, 3 indicative of the potentially adverse effect of SMM on aquatic organisms.
Previous experiments in rats receiving 270 mg/kg of SMM orally for 5 weeks have shown that a decrease of both serum triiodothyronine (T3) and thyroxin (T4) concentrations is observed, together with elevation of serum thyroid-stimulating hormone concentration; however, no changes are produced in monkeys at the same dose. 4 The result may indicate that SMM induces the endocrine disorders with species differences.
Another study in zebrafish embryos demonstrates that exposure to a low concentration of SAs results in significant deleterious effects on motor behavior, cardiac function, and embryo and larvae malformation. 5 These findings suggest that sulfa drug contamination of water may have a serious impact on the development of the central nervous system (CNS) of zebrafish. Given the effects of sulfa drug contamination in zebrafish, an aquatic organism, further investigation of sulfa drug contamination should be explored in mammals. Likewise, an interesting study indicates that the behavior and growth of the nematode progeny can be inhibited after prenatal exposure to SAs. 6
In humans, exposure to sulfa drugs during pregnancy may lead to birth defects, 7 suggesting the cautious use of SAs in early life. Some epidemiological studies have further demonstrated the long-lasting effects of early antibiotic exposure and found that antibiotic exposure in infants younger than 6 months is significantly associated with an increased body mass index later in life. 8,9 A retrospective cohort study demonstrates that sulfamethoxazole (SMZ)/trimethoprim (TMP) (odds ratio, 3.96 [95% CI, 2.42–6.49]) is associated with a higher rate of hypoglycemia, 10 suggesting that therapeutic courses of SAs antibiotic may yield a more limited effect on long-term metabolic outcomes.
The notable adverse effects associated with SAs in humans include serious neurological problems like nausea, headache, dizziness, hallucinations, and even psychosis. A recent research has shown that the risk of acute psychosis appears to increase with increases in the daily dosage of (SMZ/TMP) in HIV-infected patients. 11 In an immune-competent female adolescent, shortly after receiving her third dose of TMP-SMZ (160 mg of TMP and 800 mg of SMZ, twice per day); she develops an acute altered mental status with agitation as well as vivid visual and auditory hallucinations. 12 Also, an exciting experiment in vitro demonstrates that SMZ and other sulfa drugs are shown to be inhibitors of sepiapterin reductase, resulting in abnormal metabolism of phenylalanine, tyrosine, and tryptophan. 13
Taken together, whether SAs can result in a detrimental impact on anxiety and cognitive function remains unknown.
The mTOR signaling pathway is a convergence point for different extracellular stimuli, such as endocrine hormones, amino acid neurotransmitters, and growth factors, and regulates many integrated physiological functions of the nervous system, including energy metabolism, mood, memory storage, and cognition. Additionally, the N-methyl-
Therefore, the purpose of this article is to test the hypothesis that perinatal SMM exposure impacts the endocrine functions, further fat accumulation, and behaviors of mouse offspring later in life. As such, the potential mechanisms may be associated with the altered hormones, leading to the resultant alterations in brain amino acid neurotransmitters, transcriptional expressions of the NMDARs and mTOR pathway early in life. These results will provide important information for behavior toxicity risk assessment from exposure to SAs in mammals.
Materials and methods
Animals and treatment
Institute of Cancer Research (ICR) mice (8 weeks of age) were purchased from Beijing Vital River (China), whose foundation colonies were all introduced from Charles River Laboratories, Inc (Middlesex, Massachusetts, USA). The mice were housed at a controlled temperature (25°C) with a 12-h light/12-h dark cycle and free access to food and water. All of the animal experiments were performed in accordance with the guidelines for humane treatment established by the Association of Laboratory Animal Sciences and the Center for Laboratory Animal Sciences at Anhui Medical University (China).
Following a 1-week period of acclimation to the colony room, the males and females were paired for breeding. The time at which a vaginal plug was inserted was designated as GD 1.
SMM is commonly used as a veterinary antibiotic and not for human use in China. According to oral half of lethal dose of SMM (4680 mg/kg body weight) in mice 15 and its therapeutic dose (2000 mg/kg in feed) against mouse Isospora rivolta, 16 we randomly allocated all of the pregnant mice (n = 40) into one of the following four groups: three groups of mice were administered at low (10 mg/kg/day), intermediate (50 mg/kg/day) and high (200 mg/kg/day) doses of SMM, and normal saline-treated pregnant mice served as a control group. In addition, SMM and Sulfamethoxazole (SMX) have similar chemical structures, similar metabolites, share similar metabolic pathways, and have comparable toxicological profiles. Therefore, the lowest therapeutic dose level (23 mg/kg per day) for SMX in humans can be referenced. 17
In the SMM-treated groups, the dams received SMM sodium with a purity of 99% (CAS: 38006-08-5, Anhui HuaAo Biotechnology Co., Ltd.) by gavage daily from GD 1 to postpartum day 21. Within 24 h after birth, excess pups were removed such that 10 pups (five males and five females per litter) were kept per dam.
On postnatal day (PND) 22, each group had at least 4 litters with 10 surviving pups, and the pups were killed after anaesthetized with 10% chloral hydrate.
From PND 50 to 55, each group had at least 6 litters with 10 surviving pups, respectively. And then three batteries of behavioral tests were performed. On PND 56, all the pups were killed.
The body weights of the dams and pups were measured weekly during the experimental period.
Measurement of organ coefficients in mice
On PND 22, 40 pups (20 males and 20 females from 4 litters) in each group were allowed to fast for 12 h and then killed under anesthesia. According to the results of weight gain in mice and reported in the literatures, 5,18 SMM may be neurotoxic and cause liver and kidney damage. Therefore we collected the brain, serum, liver, kidney, and gastrocnemius from the pups. The tissues were washed in normal saline solution, sucked dry with normal filters, and then weighed. Organ coefficients (brain, liver, and kidney coefficients) were calculated as follows: organ coefficient = organ weight (g) × 1000/body weight (g).
On PND 56, mouse serum and organs such as brain, liver, and kidney were obtained in the above manner. After careful anatomy, abdominal fat was exposed and taken photos. In addition, the brain, liver, and kidney were removed, fixed with 10% neutral-buffered formalin, embedded in paraffin, sectioned at a thickness of 4 μM and then stained with hematoxylin and eosin (HE). The sections were examined by a blinded experienced pathologist under a light microscope (Nikon 80i; Nikon, Tokyo, Japan), and representative photographs of the organs were taken.
Determination of blood glucose, lipids, and serum hormones
On PND 22, serum from the killed mice (the above 4 litters) was collected, separated by centrifugation at 4°C, and stored at −80°C until utilized for measurements of T4, T3, insulin (INS), and INS-like growth factor-1 (IGF-1). Additionally, the fasting blood glucose levels in tail vein blood samples were measured using a hand-held Accu-check glucometer (Roche, Germany). On PND 56, serum (pups from 2 litters) was collected in the same manner and used to measure the concentration of the total cholesterol (CHOL), triglycerides (TG), high-density lipoprotein cholesterol (HDL), and low-density lipoprotein cholesterol (LDL).
The CHOL, TG, HDL, and LDL were measured using an automatic chemical analyzer 7020 (Hitachi, Tokyo, Japan). The serum concentrations of IGF-1 were measured using enzyme-linked immunosorbent assay kits (Wuhan Huamei Biotech Co., Ltd (China)) according to the manufacturer’s instructions. The serum INS was measured using 125I-based radioimmunoassay kits from the Beijing North Institute of Biological Technology (Beijing, China). The serum T4 and T3 were measured using chemiluminescence immunoassay kits (Roche) with the Cobas 4000 chemiluminescence immunoassay analyzer (Roche).
Behavioral tests
Behavioral testing was performed 48 h after the animals arrived in the facility. While a longer acclimatization period would be better for behavioral testing, this was the maximum time that was logistically possible to keep the animals in the same state. Prior to behavioral testing, mice were transferred into microisolator cages (five per cage). The animals’ behaviors during the tests were videotaped and scored using the “ANY-maze” video-tracking software (Stoelting Co., Wood Dale, Illinois, USA).
Open field
On PND 50, 20 pups (10 males and 10 females from 2 litters) in each group were employed in the open-field test. 19 The open-field apparatus was a black wooden box, 81 cm in length, 81 cm in width, and 28 cm in height, in an enclosed arena. The box floor was painted with 3-mm-wide white lines to form 16 equal squares (20 × 20 cm2) with a colored box (8× 5 × 3 cm3) in the center of the area. Illumination was provided by a 40-W white light bulb placed 2.80 m above the center of the field. Individual mice were placed in a corner square facing the walls and were allowed to explore for 5 min. The recorded parameters were total distance, latency to the first grid crossing, number of entries into the center zone, and time spent in the center zone. After each trial, the apparatus was cleaned with water containing detergents and then with 75% ethanol and dried with a tissue to avoid olfactory stimulation of subsequently tested animals.
Elevated plus maze
On PND 52, the same number of pups with the open field was employed. The elevated plus maze is a standard test for assessing anxiety-like behavior in rodents. 20 The maze consists of two opposite enclosed arms (30 cm in length, 5 cm in width, and 15 cm in height), two opposite open arms (also 30 cm in length, 5 cm in width, without edges), and a central arena (5 × 5 cm2). The whole apparatus was elevated 80 cm above the floor. Each mouse was successively placed in the central arena of the maze facing an open arm. The number of entries and the time spent in both the closed and open arms were recorded during a 5-min observation period. The maze was cleaned as described above for the open-field arena.
Morris water maze
On PND 50, using the same number of pups with the open field, the test was modulated as previously described. 21 The apparatus was a black pool with a diameter of 90 cm and a height of 45 cm. It was filled with water at room temperature (25 ± 1°C) to a depth of 25 cm and divided into four quadrants. The escape target was a black platform (diameter 10 cm), which was submerged 1 cm below the water surface. The testing procedure consisted of two parts: spatial training trials and a spatial probe test. In the spatial training trials, the animals were subjected to four trials per day of a place navigation task for five consecutive days. The latency to find the platform and the swimming speed were recorded. On the sixth day, a spatial probe test was conducted to test spatial memory. The parameters measured were swimming distance, swimming speed, number of entries into and swim time in the objective quadrant and entries into the island.
mRNA levels of genes involved in the mTOR pathway in the female brain
The results from our preliminary experiments demonstrated more severe impairments of anxiety-like behavior in the female pups later in life. Therefore, at weaning, female brains were rapidly dissected, frozen rapidly in liquid nitrogen, and stored at −80°C. Total RNA was extracted using TRI-zol Regent (Molecular Research Center, Cincinnati, Ohio, USA) according to the manufacturer’s instructions. A ultraviolet spectrophotometer was used to estimate the RNA concentration based on measurements at 260 and 280 nm, and the RNA was then transcribed into cDNA (A3500, Promega, Fitchburg, Wisconsin, USA). The expression of mRNA was measured by qRT-PCR using the RealStar Power SYBR mixture (GenStar BioSolutions Co. Ltd., Beijing, China), and the levels were determined through the 2−ΔΔCT method. The measured key genes: glyceraldehyde-3-phosphate dehydrogenase (GAPDH), mechanistic target of rapamycin (mTOR), thymoma viral proto-oncogene 1 (Akt1), thymoma viral proto-oncogene 3 (Akt3), phosphatidylinositol 3-kinase, catalytic, alpha polypeptide (Pik3ca), phosphatidylinositol 3-kinase, regulatory subunit p85 alpha (Pi3k3r1), eukaryotic translation initiation factor 4E binding protein 1 (Eif4ebp1), ribosomal protein S6 kinase, polypeptide 1 (Rps6kb1), INS-like growth factor I receptor (igf1r), glutamate receptor, metabotropic 1 (mGlurR1), glutamate receptor, ionotropic, NMDA2A (NR2A), N-methyl-
Primers and annealing temperature for RT-PCR.
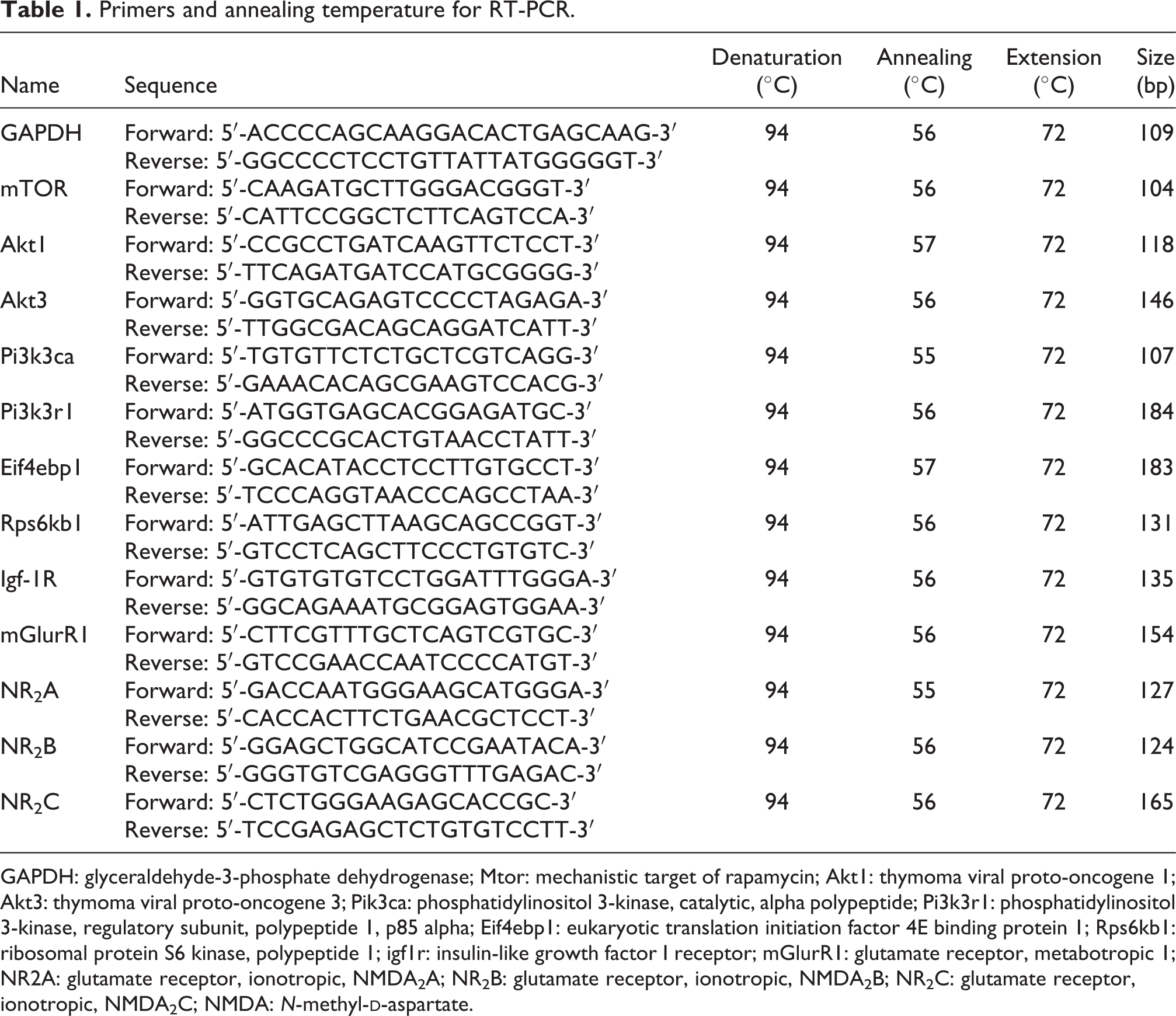
GAPDH: glyceraldehyde-3-phosphate dehydrogenase; Mtor: mechanistic target of rapamycin; Akt1: thymoma viral proto-oncogene 1; Akt3: thymoma viral proto-oncogene 3; Pik3ca: phosphatidylinositol 3-kinase, catalytic, alpha polypeptide; Pi3k3r1: phosphatidylinositol 3-kinase, regulatory subunit, polypeptide 1, p85 alpha; Eif4ebp1: eukaryotic translation initiation factor 4E binding protein 1; Rps6kb1: ribosomal protein S6 kinase, polypeptide 1; igf1r: insulin-like growth factor I receptor; mGlurR1: glutamate receptor, metabotropic 1; NR2A: glutamate receptor, ionotropic, NMDA2A; NR2B: glutamate receptor, ionotropic, NMDA2B; NR2C: glutamate receptor, ionotropic, NMDA2C; NMDA: N-methyl-
Determination of amino acids in the female mouse brain
On PND 22, the female brain (0.10 g, 2 litters) was homogenized on ice in 0.5 mL of hydrochloric acid solution (0.01 mol/L), vortexed lightly for 30 s, and centrifuged at 3000g at 4°C for 5 min. The supernatant was mixed with 0.4 mL of sulfosalicylic acid (10%), allowed to stand for 15 min, and centrifuged at 10,000g for 10 min at 4°C. Subsequently, the final supernatant was filtered using a 0.22-μm membrane. Finally, the filtrate was separated using a five-buffer system on an automatic amino acid analyzer (A300, membraPure, Germany). Amino acid levels in the brain tissues were calculated using the modified version of software supplied by the manufacturer (A300, membraPure). The free amino acids were divided into four groups: essential amino acids (EAAs), nonessential amino acids (NEAAs), branched-chain amino acids (BCAAs), and aromatic amino acids (AAAs). The EAAs measured were lysine (Lys), methionine (Met), isoleucine (Ile), leucine (Leu), tryptophan (Trp), phenylalanine (Phe), histidine (His), valine (Val), and threonine (Thr); the NEAAs measured were taurine (Tau), aspartic acid (Asp), serine (Ser), glutamic acid (Glu), glycine (Gly), alanine (Ala), g-amino butyric acid, cysteine (Cys), tyrosine (Tyr), and arginine (Arg); the AAA levels were calculated as the sum of the values of Phe, Trp, and His levels; and the BCAA levels were calculated as the sum of the values of Val, Leu, and Ile levels. The amino acid levels were calculated as nanomoles per gram of the brain tissue.
Data analysis
All of the data are expressed as the mean ± SEM. The data from the Morris water maze were analyzed through repeated-measures two-way analysis of variance (ANOVA). Other data were analyzed using a one-way ANOVA. Specific post-hoc comparisons between groups were performed using Fisher’s protected least significant difference t tests. The significance level was set at p < 0.05. All of the statistical analyses were performed using the Statistical Package for Social Sciences (SPSS, version 12.0).
Results
Impact on maternal and offspring body weight
Because most of the offspring mice were born between GD 17 and 20, maternal body weight data were obtained before GD 16 to obtain uniform data. As shown in Figures 1(a) and (b), no significant differences were observed in maternal weight between the control and exposed groups during gestation and location (p > 0.05). In addition, there were no obvious differences in female pup body weight (Figure 1(d), p > 0.05) between the control and treated groups, although the high-dose-treated group showed an elevated trend after PND 28. Notably, the male offspring body weight in the high-exposure group was strikingly heavier than that of the control groups on PND 28 and 56 (Figure 1(c), p < 0.01 and p < 0.001, respectively). Subsequently, we also found that the abdominal fat content was significantly higher in the high-dose-treated males than in the nontreated males (see Figure 2). All the aforementioned results show a more pronounced effect of exposure on lipid metabolism in male mice.
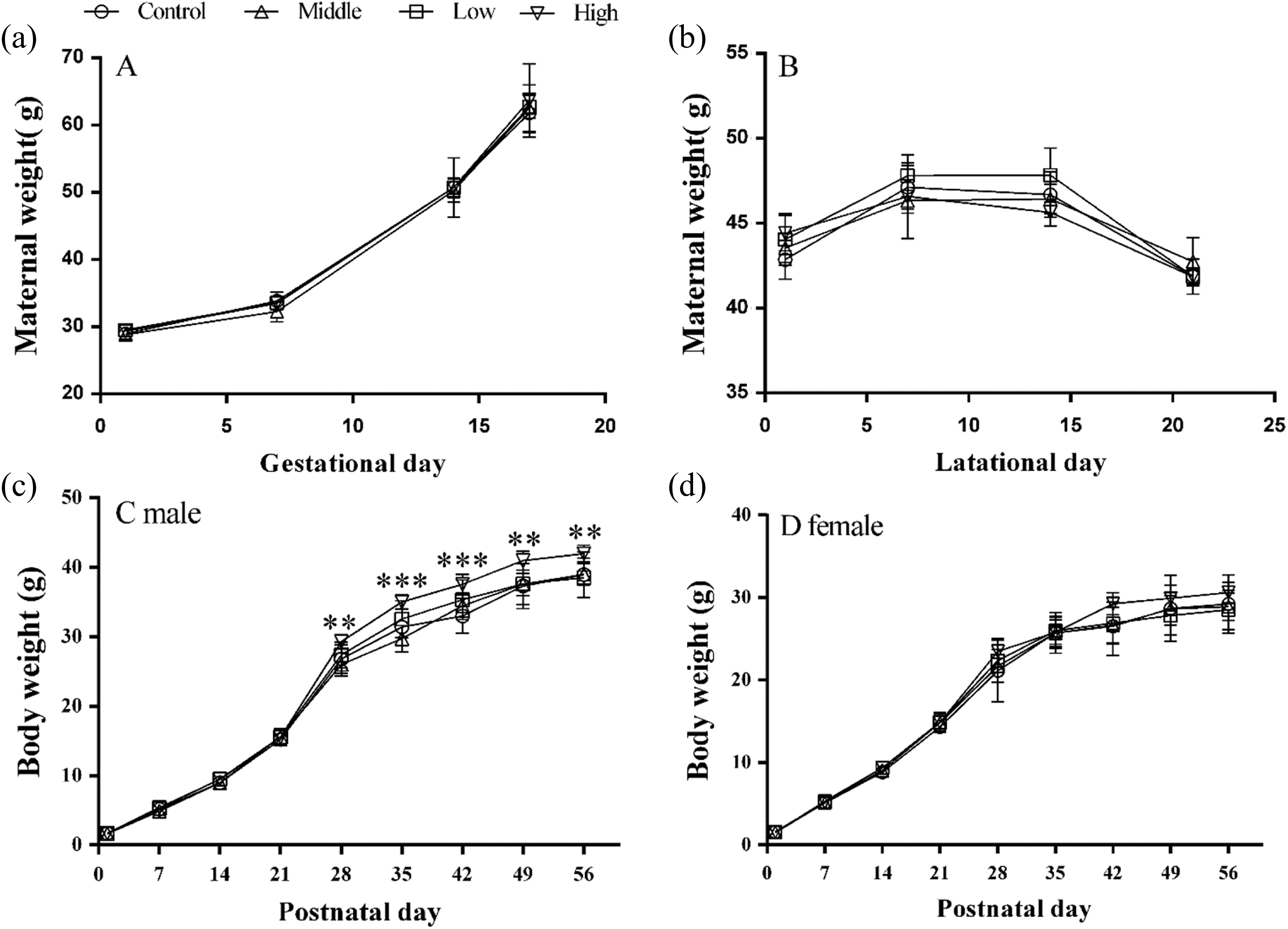
Effects of perinatal SMM exposure on maternal and pups’ body weight. Pregnant and lactational mice were orally administered with SMM at different doses (low: 10 mg/kg/day; middle: 50 mg/kg/day; high: 200 mg/kg/day), the normal saline-treated pregnant mice served as controls. Maternal body weight (a) was examined on GD 1, 7, 14, and 16. The maternal body weight (b) was weighed during lactation. The pups’ body weight in males (c) and females (d) were examined on PND 1, 7, 14, 21, 28, 35, 42, 49, and 56. Data were expressed as mean ± SEM. **p < 0.01, ***p < 0.001: means difference between high-dose group and controls. SMM: sulfamonomethoxine; GD: gestational day; PND: postnatal day.
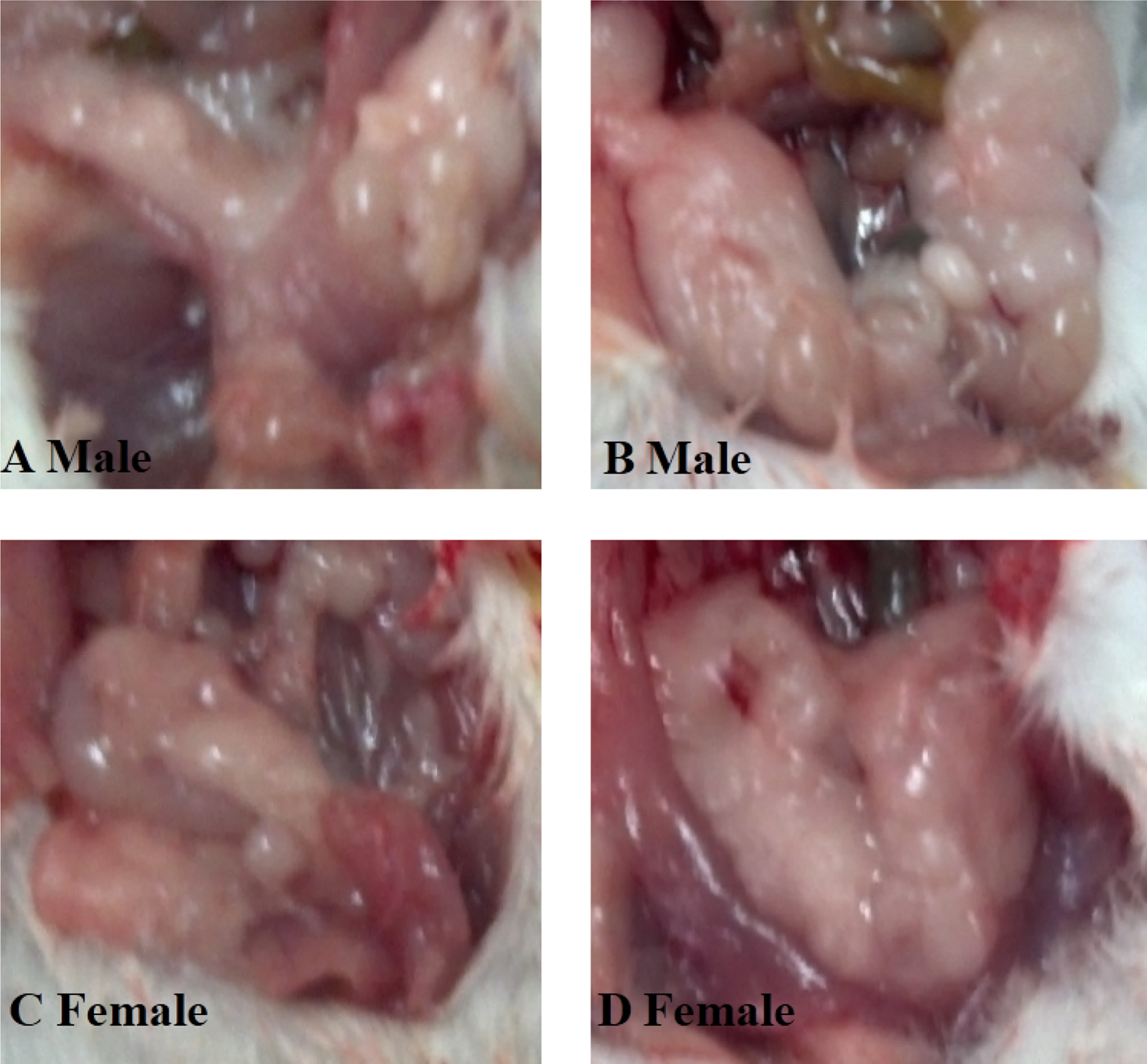
Performance of abdominal fat in offspring mice on PND 56. (a) Males in control groups. (b) Males in the high-dose group. (c) Females in control groups. (d) Females in the high-dose group. PND: postnatal day.
Organ coefficients of the offspring mice
As shown in Figure 3, on PND 22, the male brain coefficients for the low-dose-treated group (Figure 3(a), F = 3.852, p < 0.05) and the female brain coefficients for the low- and middle-dose-treated groups (Figure 3(a), F = 17.473, p < 0.001) were markedly decreased compared with the controls, suggesting a more profound impairment of female brain development. Notably, no obvious dose–response relationship was observed for the parameters. A study has shown that nonlinear pharmacokinetics of SMM occurred after a high dose (100 mg/kg) of SMM in pigs, which might explain the nonlinear pharmacodynamics in our findings. 22 The liver coefficients of females in the high-dose-treated group were significantly reduced (Figure 3(b), F = 3.426, p < 0.05), and this parameter was also reduced, although not observably, in males. Compared with the controls, the kidney coefficients in both males (Figure 3(c), F = 6.594, p < 0.01) and females (Figure 3(c), F = 3.119, p < 0.05) that received the high-dose SMM treatment were significantly decreased. However, the gastrocnemius coefficients showed no significant differences regardless of treatment (Figure 3(d), p > 0.05). Together, these results show that early life SMM exposure impairs the development of the brain, kidney, and liver in offspring.
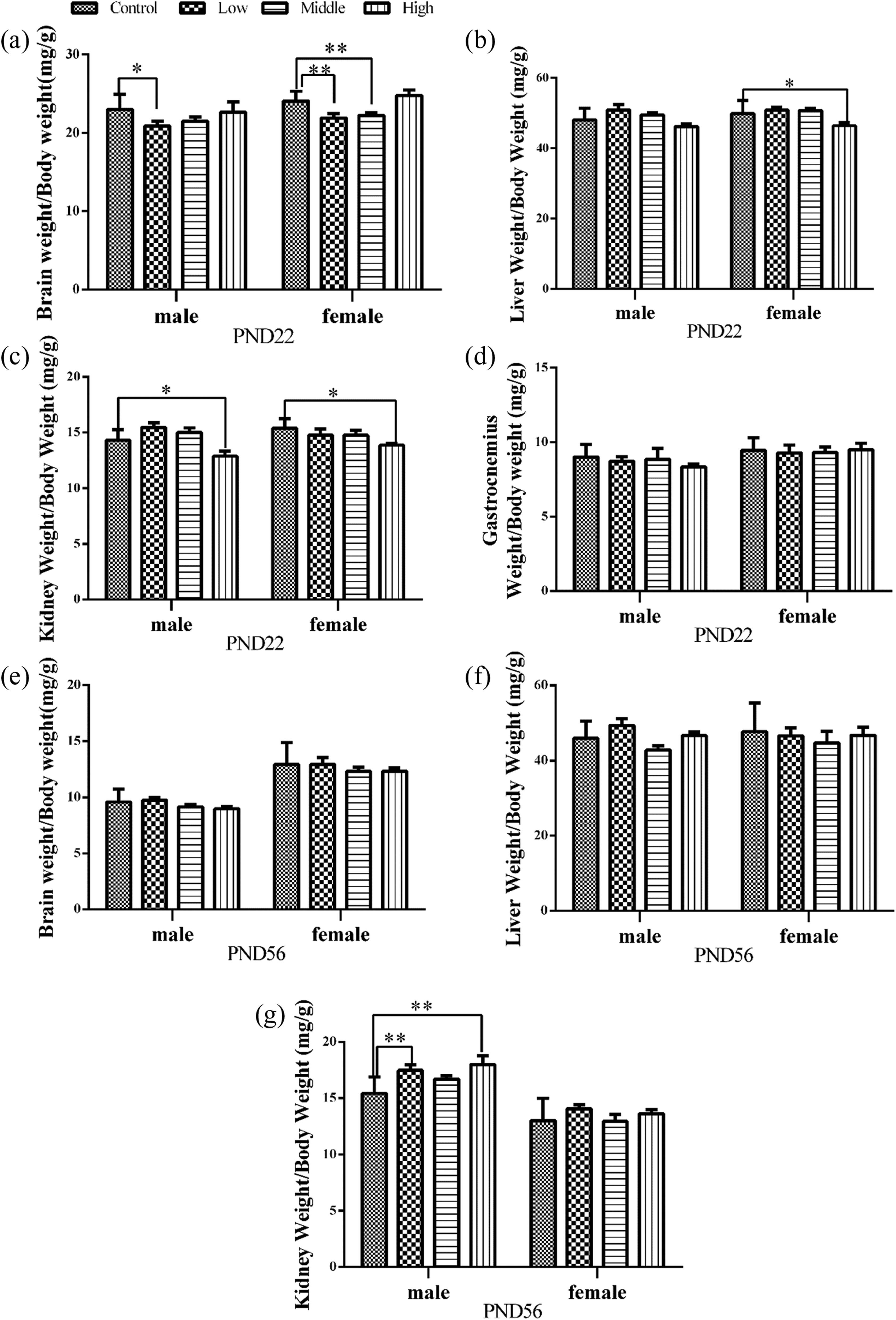
Effects of perinatal SMM exposure on organ coefficients in mice offspring. On PND 22, in pups, the brain (a), liver (b), kidney (c), and gastrocnemius coefficients (d) were measured, respectively. In addition, on PND 56, the brain (e), liver (f), and kidney coefficients (g) were also examined. Data are expressed as mean ± SEM. *p < 0.05, **p < 0.01: as compared with controls. SMM: sulfamonomethoxine; PND: postnatal day.
On PND 56, the brain coefficients (Figure 3(e), p > 0.05) were slightly decreased in the SMM-treated groups with respect to the control, but the differences were not significant. Additionally, all liver coefficients (Figure 3(f), p > 0.05) of the treated groups were not significantly different from those of the controls. In contrast, in male offspring, the kidney coefficients of the low- and high-dose-treated group were significantly increased compared with the corresponding coefficients of the control group (Figure 3(g), F = 4.501, p < 0.01). The pubertal findings indicated that SMM exposure in early life caused long-term injury of the brain and kidney in mice.
Organ histopathology of the offspring mice
The histopathological alterations of brain, kidney, and liver tissues on PND 56 are shown in Figure 4. In brain tissues, the cell outlines in the control groups (Figure 4(a) male, Figure 4(g) female) were clear, and the structure was compact. Exposure to the high dose of SMM (Figure 4(b) male, Figure 4(h) female) resulted in neuronal cell edema and nuclear shrinkage in the CA3 region of the hippocampus. Analysis of the kidney tissues revealed that a high dose of SMM (Figure 4(d) male, Figure 4(j) female) resulted in vacuolar dystrophy of renal tubule cells in the medulla of the kidney and more severe changes, such as necrosis. In contrast, no tissue damage was observed in the control groups (Figure 4(c) male, Figure 4(i) female). HE staining showed differences in the liver tissue structure and lipid accumulation between the high-dose-treated and control groups of both males and females. The livers of the control mice (Figure 4(e) male, Figure 4(k) female) had well-organized structures. Hepatic sinusoids were clearly visible, and hepatic cords were neatly arranged and distributed radially around the central veins. The analysis of the high-dose-exposed males (Figure 4(f)) and females (Figure 4(l)) revealed that the hepatocytes of males showed more severe damage characterized by cell necrosis, lipid deposition in the cytoplasm, and vacuolization.

Histological alteration of the brain, kidney, and liver in control and high-exposed pups on PND 56. Hematoxylin and eosin stain (×200). —(a to f): male offspring (a: the brain in control group, b: the brain in the high-dose group, c: control of the kidney, d: the high-dosed kidney, e: control group liver, f: high-dose group liver). (g to l) Female offspring ((g) nondosed brain, (h) the high-exposed brain, (i) kidney in controls, (j) the high-exposed kidney, (k) liver in controls, (l) liver in the high-dose group). PND: postnatal day.
Serum hormones in offspring mice
On PND 22, we examined the blood glucose, INS, IGF-1, T3, and T4 levels in serum. These data are shown in Figure 5. The concentration of glucose in females in the high-dose-treated group was significantly lower compared with that of the control group (Figure 5(a), F = 7.085, p < 0.01). The contents of male serum INS in the middle- and high-dose-treated groups were increased substantially compared with the levels observed in the control animals (Figure 5(b), F = 4.187, p < 0.05). In addition, the concentrations of INS in all the females treated with SMM were markedly higher than those of the control group (Figure 5(b), F = 6.018, p < 0.01). However, in male and female offspring, SMM exposure did not induce a significant difference in the serum IGF-1 concentrations (Figure 5(c), p > 0.05). The T3 content in females belonging to the middle-dose-treated group was significantly decreased (Figure 5(d), F = 3.224, p < 0.05) compared with the content found in the control group, whereas no significant differences were found in the concentrations of T4 in both males and females (Figure 5(e)). In contrast, the rats receiving 270 mg/kg of SMM showed a decrease in the serum T4, but no change in T3. 4 The reason for the differing T3 and T4 responses may be explained in the further experiments.

Effects of perinatal SMM exposure on blood glucose and serum hormones in pups. Blood glucose (a), insulin (b), IGF-1 (c), T3 (d), and T4 (e) were checked in pups on PND 22. TG (f), CHOL (g), HDL (h), and LDL (l) were examined in offspring on PND 56. Data are expressed as mean ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001: as compared with controls. SMM: sulfamonomethoxine; IGF-1: insulin-like growth factor-1; T3: triiodothyronine; T4: thyroxin; PND: postnatal day; TG: triglyceride; CHOL: cholesterol; HDL: high-density lipoprotein cholesterol; LDL: low-density lipoprotein cholesterol.
On PND 56, we detected the concentrations of serum TG, CHOL, HDL, and LDL. The serum concentrations of CHOL (Figure 5(g)), HDL (Figure 5(h)), and LDL (Figure 5(i)) in SMM-treated males were increased compared with the controls, but the difference was not statistically significant. Surprisingly, the serum contents of TG, CHOL, HDL, and LDL in the middle-dose-treated females were significantly decreased compared with those of the nontreated group (Figure 5(f) to (i), p < 0.05 or 0.01).
Exploration and anxiety of offspring mice
In the open-field test, as shown in Figure 6, the lower total distance (Figure 6(a)) was obtained in females at the high dose (F = 4.876, p < 0.01). In addition, the latencies of the first grid cross (Figure 6(b)) of males from the low- and middle-dose groups (F = 2.864, p < 0.05) and females from the middle-dose group (F = 5.387, p < 0.01) were markedly increased compared with the controls. The numbers of entries into the central area (Figure 6(c)) were significantly decreased for the males in the low- and middle-dose-treated groups (F = 5.079, p < 0.01). A similar effect on female pups was found for the middle-dose-treated group (F = 5.154, p < 0.01). Consistent with these results, the time spent in the central area (Figure 6(d)) was markedly shortened for the females in the middle-dose-treated group (F = 4.108, p < 0.05).
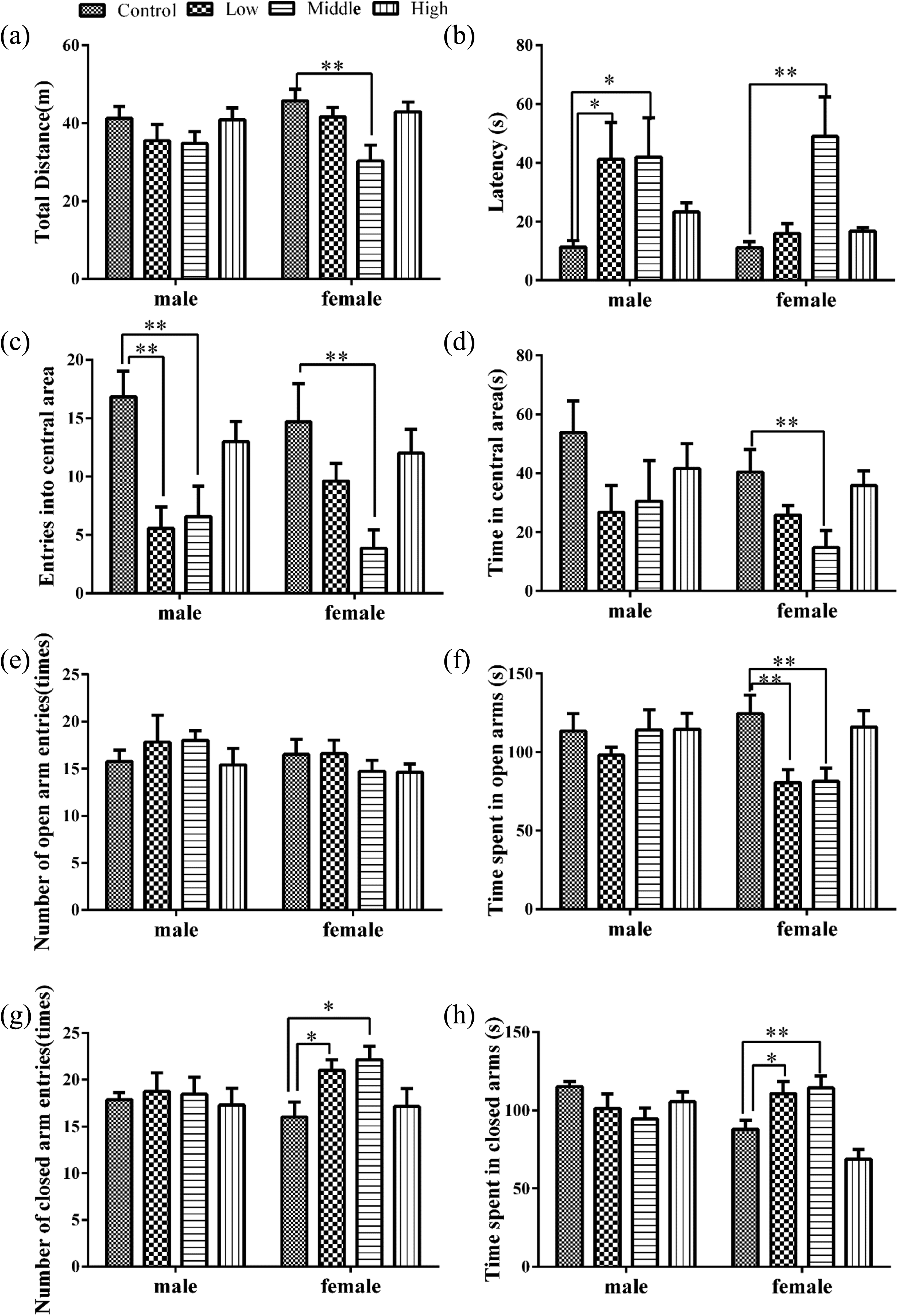
Effects of perinatal SMM exposure on anxiety and exploration activities in pubertal pups. Open field: total distance (a), latency to the first grid crossing (b), number of entries into central area (c), and time in central area (d). Elevated plus maze: number of entries to the open arms (e), time spent on the open arms (f), number of entries to the closed arms (g), and time spent on the closed arms (h). Data are expressed as mean ± SEM. *p < 0.05, **p < 0.01: as compared with controls. SMM: sulfamonomethoxine.
The elevated plus maze revealed no significant effect on the numbers of entries into the open arms for both males and females (Figure 6(e)). However, the time spent in the open arms (Figure 6(f)) was substantially lower for females from the low- and middle-dose-treated groups compared with the controls (F = 5.421, p < 0.01). The numbers of entries into the closed arms (F = 3.500, p < 0.05) and the time spent on the closed arms (F = 12.476, p < 0.001) obtained for the females of the low- and middle-dose-treated groups were significantly greater than those of the control groups but not observably altered for males (Figures 6(g) and (h)). These results demonstrated significant alterations in the exploration and anxiety behaviors of mice as a result of perinatal treatment with SMM. In particular, SMM had a greater impact on exploration and anxiety in female pups.
Performance on spatial learning and memory of offspring mice
During the training days, the mean latency for finding the escape platform and the swimming speed were measured to assess spatial learning abilities. As shown in Figure 7, male pups in the high-dose-treated group exhibited a significantly longer swimming latency compared with those of the control groups on the fourth day (Figure 7(a), p < 0.05). However, in female pups, there were no significant differences compared with the controls during the training days (Figure 7(b)). In addition, the swimming speed of the high-dose-treated group was significantly slower than that of the controls for both males (Figure 7(c), p < 0.05 or p < 0.01) and females (Figure 7(d), p < 0.05 or p < 0.01).

Effects of perinatal period SMM exposure on learning performance during the 5-day training period. Morris water maze was performed in pups from PND 50 to PND 55. (a) Escape latency in male mice. (b) Escape latency in female mice. (c) Swimming speed in males. (d) Swimming speed in females. Data are expressed as mean ± SEM. *p < 0.05, **p < 0.01: means difference between high-dose group and controls. SMM: sulfamonomethoxine; PND: postnatal day.
The results of the probe trial (in which the hidden platform is removed) are shown in Figure 8. The swimming distance (Figure 8(a)) of the high-dose group was significantly decreased for both males (F = 9.301, p < 0.01) and females (F = 8.009, p < 0.01). Similarly, the swimming speed (Figure 8(b)) of males (F = 9.912, p < 0.01) and females (F = 7.904, p < 0.01) was significantly reduced with respect to the controls. The number of entries into the target quadrant (Figure 8(c)) was also decreased in the high-dose-treated group for both males (F = 5.057, p < 0.01) and females (F = 3.599, p < 0.05). In addition, our results showed that the time spent in the target quadrant (Figure 8(d)) and the numbers of entries into the island (Figure 8(e)) were not significantly different between males and females. However, the number of entries into the island for the treatment groups tended to decrease.

Effects of perinatal SMM exposure on learning and memory performance on the testing day. The Morris water maze test was performed on PND 55. (a) Swimming distance. (b) Swimming speed. (c) Entries into target quadrant. (d) Time in target quadrant. (e) Entries into island. Data are expressed as mean ± SEM. *p < 0.05, **p < 0.01: as compared with controls. SMM: sulfamonomethoxine; PND: postnatal day.
mRNA levels of the mTOR pathway in female pup brains
Given that females showed more severe behavior dysfunctions, the effects of maternal SMM exposure on the mRNA levels in the offspring brain were evaluated in only females on PND 22. The mRNA levels of key genes involved in the mTOR pathway are presented in Table 2. There was no significant difference in the mRNA levels of brain Akt3, Eif4ebp1, and mGlurR1, while significantly increased mRNA levels of IGF-1 R, Pi3k3ca, Pi3k3r1, Akt1, Mtor, and Rps6k1 in the brain were observed between the control and treated pups whose mothers were exposed to SMM during the perinatal period. Moreover, the mRNA level of NDMA receptor subunits NR2B and NR2C were decreased significantly in the SMM-treated group. Interestingly, the mRNA level of NR2A was elevated in females that received the middle dose of SMM compared with the controls. We confirmed that SMM exposure led to changes in the transcriptional levels of genes related to the mTOR and NDMAR subunits. The NR2B is also predominant in the early brain, whereas the expression of NR2B mRNA was more decreased by SMM exposure in the present study. This may, in part, account for less memory abilities later in life. However, the precise role of NR2B in learning and memory remains unclear due to a lack of selective pharmacological compounds.
Effects of perinatal period SMM exposure on relative mRNA expression of the brain mTOR pathway in female offspring.a
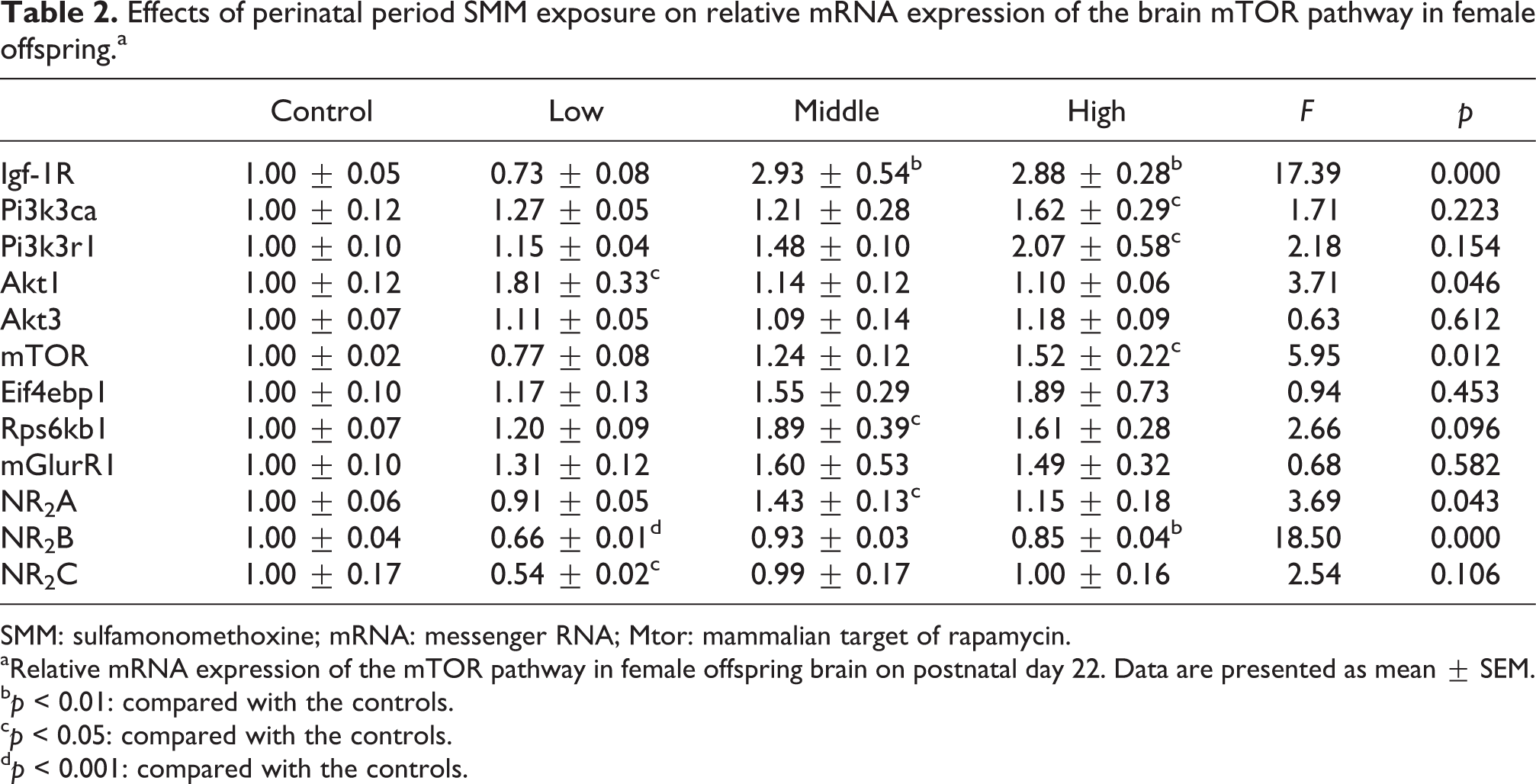
SMM: sulfamonomethoxine; mRNA: messenger RNA; Mtor: mammalian target of rapamycin.
aRelative mRNA expression of the mTOR pathway in female offspring brain on postnatal day 22. Data are presented as mean ± SEM.
bp < 0.01: compared with the controls.
cp < 0.05: compared with the controls.
dp < 0.001: compared with the controls.
Amino acid levels in the female offspring brain
Given the more severe behavioral impairments in females, we further investigated the effects of maternal SMM exposure on the brain amino acid levels in female pups on PND 22, and the results are presented in Table 3. Notably, the concentrations of Tau, which is essential for the development and function of the CNS, were significantly reduced in the middle- and high-dose-treated groups. Additionally, we found that the level of the excitatory Glu was significantly elevated in the high-dose-treated group. The level of Gly, an inhibitory neurotransmitter, was observably reduced in the high-dose-treated group. In addition, the concentration of α-aminoadipic acid was significantly increased in the high-dose-treated group compared with the control group. Moreover, we found that the content of camosine was elevated, while the content of anserine was reduced in the low- and middle-dose-treated groups. The levels of the AAAs, EAAs, Tyr, and Phe were also decreased in the middle- and high-dose-treated groups, albeit not significantly, compared with the controls.
Free amino acids profiles in the brain of female offspring mice on PND 22 after maternally exposed to SMM (PND 22, nmol/g wet weight, n = 6).

SMM: sulfamonomethoxine; PND: postnatal day; Tau: Taurine; Ser: serine, Asp: asparagine; Glu: glutamic acid; α-AAA: α-aminoadipic acid; Gly: glycine; Ala: alanine; Cit: citrulline; Val: valine; Met: methionine; Ile: isoleucine; Leu: leucine; Tyr: tyrosine; Phe: phenylalanine; g-ABA: γ-amino butyric acid; His: histidine, Trp: tryptophan; Car: camosine; Ans: anserine; Om: omithine; Lys: lysine; NH4: ammonium chloride; EOHNH2: ethanol amine; Arg: arginine; Pro: proline, EAA: essential amino acid; AAA: aromatic amino acids; BCAA: branched chain amino acid; NEAA: nonessential amino acid. Data were presented as mean ± SEM.
ap < 0.05: compared with the control.
bp < 0.01: compared with the control.
cp < 0.001: compared with the control.
Discussion
Puberty in mammalian species is a period of rapid dynamic changes in endocrine functions and tissue morphology. Therefore, it is not surprising that early life exposure to a variety of pharmaceutical and environmental compounds has been shown to alter the timing of pubertal development.
Most clinicians are familiar with mild adverse drug reactions that are typically associated with antimicrobials. However, the toxic effects of sulfa drugs on the CNS are often underreported, and the mechanism(s) may be complex or obscure. The present study investigated the effects of maternal SMM exposure during the perinatal period on behavioral and physiological responses in offspring mice and the possible mechanisms of SMM-induced neurotoxicity.
In this study, the high-dose SMM induced a significant increase in the body weight of male offspring 1 week after cessation. In addition, the abdominal fat content was higher in the groups treated with a high dose of SMM compared with the controls. Recent research demonstrates that antibiotic exposure (penicillin, cloxacillin, cephalexin, cefadroxil, and erythromycin) during the first year of life is associated with an increased risk of being overweight and central adiposity in preadolescent boys, but no evidence of a dose–response relationship is observed between infant antibiotic exposure and childhood overweight outcomes. In boys, infant antibiotic exposure was associated with a 5-fold increased risk of overweight at age 12, whereas there was no association in girls. 23 Similar gender-specific associations were found in our study. Although the precise mechanisms are poorly understood, it is widely accepted that growth promotion by antibiotics is mediated through interaction with the gut microbiota, because antibiotic-enhanced growth is not seen in microbiota-free animals. In mice, early life subtherapeutic antibiotic treatment can enhance fat mass and alter the levels of metabolic hormones, the hepatic metabolism, and the macrobiotic composition. 24
Another study also indicates that mTOR activation contributes to obesity by enhancing excess fat deposition in white adipose tissue, liver, and muscle, which in turn promotes INS resistance. 25 Indeed, INS and IGF-1 enhance mRNA translation in neurons, possibly through mTOR. 26 Therefore, the reported findings may at least partly explain the elevated weight gain and altered metabolism observed in the SMM-treated animals in this study.
On PND 22, the female liver coefficient was significantly decreased compared with that of the control group, demonstrating that SMM may induce liver damage. In support of our results, a previous study also found that antibiotic-induced perturbation of the gut microbiota aggravates cholesterol accumulation and liver injury in rats fed a high-cholesterol diet. 27 Additionally, the kidney coefficients on PND 22 or PND 56 and pathological observations have shown that early life SMM exposure induces renal injury later in life, suggesting a long half-life for SMM. An experimental study in rabbits also showed that sulfa drugs cause a deterioration of renal function by affecting both the reabsorption and excretion functions of the renal tubule. 18 At weaning, the brain coefficient was decreased, indicating that early life SMM exposure may also impair brain development.
A previous study shows that modulation of the gut microbiota through antibiotics improves INS signaling in mice fed a high-fat diet. 28 Additionally, SAs can also influence the composition and metabolic capabilities of the gut microbiome. 29 Probably, SMM-induced gut microbiome dysbiosis, resulted in the altered lipid metabolism. In the current study, on PND 22, the female blood glucose level was markedly declined in the high-dose group, which likely resulted from a significant increase in serum INS that was induced by SMM. The decreased blood glucose level is consistent with the other report. 10
We did not find significantly altered levels of serum IGF-1, although the mRNA level of Igf-1 R was observably increased, an effect that was likely activated by INS. Consistent with our findings, SAs can induce hypoglycemia along with an elevation in the INS levels in patients with chronic renal failure. 30 Thyroid hormones play a vital role in the process of early growth and development of most organs, particularly the brain. Our present results show that SMM induces a lower T3 in females with reduced brain coefficients. In addition, research has also shown no changes in thyrotropin, T3, or T4 levels or thyroid weight for rats administered up to 160 mg/kg of sulfamethazine for 4 weeks. 31 This finding suggests a species difference between mice and rats with regard to sensitivity to SMM.
Up to now, neurotoxicity has not been directly evaluated for SMM. Through a series of behavioral tests, we found that perinatal SMM exposure increases anxiety-like behaviors in mouse offspring, particularly in females. Simultaneously, spatial learning and memory in the offspring were impaired. Moreover, we found that perinatal SMM exposure activated the PI3K/Akt/mTOR pathway and increases the transcription of two downstream targets of mTOR signaling, namely, p70S6 K and 4EBP1. These results demonstrate a link between abnormal behaviors and the mTOR pathway.
Although many previous studies demonstrate that antibiotic-induced severe neurotoxicity to other model organism, few data are available on neurotoxic mechanisms from the perspective of the mTOR pathway.
Therefore, precise regulation of mTOR activity to prevent hypo- or hyper-activation is necessary for optimal cognitive function throughout life. This equilibrium is also necessary during neuronal development, as pathological activation of mTOR deregulates excitatory/inhibitory balance at synapses.
Thus we propose that the mTOR signaling pathway integrates the enhanced INS signal and serves as an adaptive regulator of cell metabolism, growth, proliferation, and survival. Furthermore, hyper-activation of the mTOR is recently reported to be an important cause of intellectual disability in mouse models. 32 Previous research provides evidence for the negative effects of mTOR signaling on motor function, showing that administration of rapamycin, an mTOR inhibitor, prevents the development of dyskinesia in a mouse model of Parkinsonism. 33 The link between activation of mTOR signaling and behavioral abnormalities has also been supported by our findings.
Interestingly, some recent reports indicate that blockade of mTOR signaling also exerts a negative impact on neuronal activity. For example, a behavioral study reveals a rapamycin-induced increase in anxiety-related behaviors in rats. 34 Similarly, prenatal rapamycin treatment of mice adversely impacts early developmental milestones and functions in adult animals, which results in anxiety-like behaviors during both early development and adulthood. 35 These results indicate that the inhibition, but not activation, of mTOR signaling leads to enhanced depression and anxiety-like behaviors. Thus, deregulation of the mTOR signaling pathway may result in neurobehavioral impairment.
The mTOR signaling pathway is activated by growth factors (such as IGF-1 and INS), as well in dendrites by the stimulation of group I mGIuRs (metabotropic glutamate receptors). 36 In the current study, on PND 22, we detected an increase in the concentration of serum INS and in the mRNA levels of IGF-1 R and mGIuR1 (Figure 5B, Table 2). These results suggest a mechanism of SMM-induced impairment that involves activation of the mTOR pathway and indicate that activation of the mTOR pathway in early life likely acts as an initial compensatory response to SMM exposure, resulting in a long-term negative impact on CNS development. Therefore, we speculate that the altered physiology and behavior in this study is due to activation of mTOR pathway.
Moreover, we selected the NMDAR subunits NR2A, NR2B, and NR2C for examination, as the receptors are known to play an important role in synaptic development and plasticity, learning and memory. Prior research has demonstrated that NMDAR activation will dampen mTORC1 over-activity, 37,38 suggesting the existence of a molecular and functional link between the NMDAR and the mTOR pathway. Sulfasalazine blocks the NMDA response by reducing the number of NMDA channels available for activation. 39 Our results indicate that maternal exposure to SMM during the perinatal period alters NMDAR subunit expression in the mouse brain, which weakens spatial memory. In addition, research has shown that the NMDAR antagonist ketamine can increase mTOR pathway signaling.
In addition, the results from other experimental models suggest that exposure to xenobiotic may interfere with the expression of the NMDAR subunits NR2A and NR2B during brain development. 40,41 Consistent with this idea, the administration of ketamine to pregnant rats causes depression- and anxiety-like behaviors and impaired memory in the offspring and further causes NR2A receptor subunit upregulation, 42 which is similar to our results. NR2B has been implicated in the modulation of synaptic function in learning and memory processes, and reduced expression of NR2B has been observed in connection with cognitive impairment and neuropathology, 43,44 in line with our findings in the present study. At 3 weeks old, the NR2A and NR2B expression in the developing rat brain decrease to adult levels, whereas the NR2C remains unchanged. Research shows that IGF-1 can inhibit NR2B receptors and activate the PI3K-AKT-mTOR pathway, 38 and the reduced NR2B may be related to the elevated gene levels of IGF-1 R in our study.
Amino acid neurotransmitter systems also play a major role in neuropathology. A previous study suggests that the behavioral effects may be related to the changes in amino acid neurotransmitter in the brain including Asp, Glu, and g-ABA. 45 Notably, in our study, the level of Glu was significantly elevated in females in the high-dose-treated group. Because of its role in synaptic plasticity, Glu is involved in cognitive functions such as learning and memory. In the brain, excess Glu can accumulate outside cells, causing calcium ions to enter cells via NMDAR channels, leading to neuronal damage. In addition, Tau is an effective neurons-protective agent and plays a role in the modulation of anxiety and locomotors activity. 46 Thus, in the present study, the reduced Tau in the SMM-treated group may exacerbate Glu-induced excitotoxicity, leading to neuronal damage. Furthermore, camosine and anserine are protective against neurotoxicity, 47 and the carnosine and anserine changes obtained in the present study indicate that SMM exposure induces neurotoxicity in female mice.
Previous research shows that SAs interfere with the biosynthesis of neurotransmitters like serotonin and dopamine in cultured human cells, 13 which can account for their reported neurological side effects. Whereas the levels of both tyrosine and tryptophan (precursors of dopamine and serotonin) in mouse brain show no significant differences in the current study and the discrepancy seem to be a consequence of methodological differences between the in vivo and in vitro responses to SMM exposure. In addition, it is possible that the impact on tyrosine and tryptophan contents in the brain is different between SAs.
A recent study shows that β-Diketone antibiotics (DKAs), including ofloxacin, ciprofloxacin, enrofloxacin, doxycycline, chlortetracycline, and oxytetracycline, significantly increase zebrafish shoaling behavior resulting from an anxiety-like state at 6.25 mg/L, but decrease zebrafish social cohesion possibly due to an autism-like state at 25 mg/L; the authors also show that after long-term exposure (3-month DKAs exposure at 6.25, 12.5, and 25 mg/L), low concentrations of DKAs (6.25 mg/L) may cause antianxiety behavior, while higher DKAs concentrations (25 mg/L) can lead to anxiety-like behavior with no significant dose–effect relationship, 48 and the result obtained in this investigation is similar to our findings shown in Figure 6.
In rats, the ciprofloxacin antibiotic is administered in single oral daily doses of 20 and 50 mg/kg for 14 days, induces depression-like and anxiety-like behaviors with decreases in the brain serotonin and γ-amino-butyric acid levels only in the 50 mg/kg. 49
Our study has some limitations. First, we did not use tests to characterize important variables, such as early life microbe-host metabolic interactions. Second, our study did not measure signaling proteins related to the mTOR pathway or the levels of mRNA related to the mTOR pathway in adolescence. Third, because the SMM-induced anxiety-like behavior was more severe in female offspring, we only analyzed the levels of mRNA expression and amino acids in female offspring mice, likely leading to the loss of some useful information. Thus, additional experiments should be performed to directly address these issues. Finally, our studies were not implicated in the microbiome–gut–brain axis of substantial relevance to mood and behavior.
In summary, our results show that maternal exposure to different doses of SMM during the perinatal stage promotes weight gain and fat accumulation and influences the behavioral functions of the offspring mice later in life. These outcomes may originate from SMM-induced early life neuroendocrine impairments and aberrant transcription of mTOR signaling molecules in the brain.
Footnotes
Authors' note
Qiang Zhang and Dan Zhang contributed equally to this work.
Acknowledgements
We are grateful for the help provided by Professors De-xiang Xu and Fang-biao Tao and their lab groups. We also thank Associate Professor Jin-fang Ge for her help with performing the behavioral assay experiments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the National Natural Science Foundation of China (81202209), the Young Talents in College of Anhui Province (2012SQRL075ZD) and the Scientific Research of BSKY from Anhui Medical University (XJ201112 and XJ201116).
