Abstract
Pesticide exposure may affect semen quality and male fertility in humans. The aim of the present work was to elucidate the adverse effects of deltamethrin (Delta), a synthetic pyrethroid, on exposed male mice and their offspring. Adult male Albino/Swiss mice received deltamethrin (5 mg/kg) daily for 35 days and mated with untreated females to produce offspring. Classical measurements of ejaculate and sperm quality and testicular histopathological changes were assessed. Deltamethrin treatment affects sperm quality and quantity in the ejaculated semen of mice that had also markedly impaired libido as measured by indices of mating and fertility and number of pregnant females housed with male mice exposed to this pesticide. Exposure mice to deltamethrin significantly decreased their testosterone and inhibin B levels and affected reproductive performance. Testes of exposed mice showed marked histopathological alterations as compared to the control group. The mice exposed to 5 mg/kg body weight/day of deltamethrin showed severe alterations of the seminiferous tubules, sloughing of the germ cells, the vacuolization of germ cell cytoplasm, and the disruption of spermatogenic cells compared to the control group. Altered pregnancy outcomes were directly attributed to damage of sperm of male mice exposed to deltamethrin compared to the control group. We concluded that exposure to deltamethrin affected the reproductive system of male mice explored by altered total sperm density, motility, and morphology in mice spermatozoa.
Introduction
The increasing release of chemicals into the environment draws attention to a better understanding of their toxicity in human and animals. 1 Many studies suggest that environmental contaminants can damage human health. Small amounts of some pesticides cause death; others burn or irritate eyes and skin, 2 damage the nervous system, 3 disrupt hormone and immune systems, reduce ability to successfully reproduce and cause cancer. 4
Cases of acute pesticide poisoning account for significant morbidity and mortality worldwide, especially in the children. Annually, there are many people who suffer from pesticide-related health effects such as pyrethroids. 5,6 Using the synthetic pyrethroids has significantly increased as compared to organochlorine and organophosphorus insecticides due to their high bioefficacy, easy biodegradability, and photostability. 7,8 Previous studies observed an association between occupational exposure to pyrethroids and altered semen quality or sperm genotoxicity in terms of DNA damage and numerical chromosome aberrations in sperm among exposed workers. 9,10
Deltamethrin (Delta), a type II synthetic pyrethroid, is highly effective against a broad spectrum of insects. Delta is readily absorbed by the oral route. The pesticides persist in our environment. Residues of these chemical products, in many studies, are found on food or in the fish that live in contaminated waters. 11 Delta has been shown to have adverse effects on male and female reproductive system. 12 In recent years, endocrine disruptors may be found in many products—including pesticides. These chemical products can block the signaling systems of hormones. 13 The difficulty in assessing the risk of endocrine disruptors lies in their diverse mechanisms of action and in the potential for interaction between acting compounds in the body. The most studied aspect of endocrine disruptors is their ability to interact with receptors, especially those for estrogen. 14 In vitro studies have shown estrogenic responses to the pesticides deltamethrin, endosulfan and others. Different concentrations of these pesticides caused a cytotoxic effect in both the Chinese hamster ovary cells used for the androgenicity assay and the MCF-7 cells used for the estrogenicity assays. 15 Moreover, it was demonstrated that Delta induced testicular apoptosis, reduced the weight of reproductive organs and decreased the sperm parameters and plasma testosterone concentration. 16
The most common methods to collect sperm in the mouse are by electroejaculation 17 or artificial vagina; 18 both enable collection of whole semen with good quality that can be used for different purposes. Different anesthetic protocols have been reported in literature to perform electroejaculation in the cat, but the effects of xenobiotics on ejaculated sperm quality have never been studied. Recent study concluded that change of sperm by accessory gland proteins (Acps) secreted by male accessory glands affects the behavior of ejaculated sperm, possibly providing them with an advantage over epididymal sperm for fertilization of oocytes in vivo. 19 To the best of our knowledge, this is the first study to evaluate the effects of deltamethrin on the mouse-ejaculated sperm. This study aimed to evaluate endocrine-disrupting and cytotoxic potential of Delta on reproductive performance of male fertile mice.
Materials and methods
Pesticide qualification
Deltamethrin (98% purity) is a synthetic pyrethroid insecticide (C23H19Br23NO3). The chemical name of CAS is (1R, 3R)-3-2, 2-dbromovinyl-2,2-dimethyl-cyclopropane carboxylate de (S)-α-cyano-3 phenoxybenzyle, CAS registry number 52918-63-5, was used in the experiments. Delta was obtained from Sigma Chemical Co. (St Louis, France; D9315 SIGMA). All other chemicals used in this study were purchased also from Sigma Chemical Co.
Animals and dosage exposures
Swiss Albino male Adult mice, Mus musculus, aged (3 months old, 12–15% lifespan), 20 weighing 27 ± 3.0 g and of proven fertility (only those that were able to impregnate female were used in the experiments) were obtained from the Centre of Veterinary Research (Sfax, Tunisia), which have been used in his laboratory for more than 10 years. The source of mice has been Charles River, France, which we used since a long time.
Animals were examined for health status and acclimated to our laboratory environment for 1 week prior to the experimental assignment to ambient conditions (room temperature 23 ± 2°C, relative humidity 50 ± 10%, and 12-h light/12-h dark cycle). They were fed with a commercial pellet diet composed of protein, carbohydrates, fibers, vitamins, minerals and a small amount of fat and tap water ad libitum.
Mice were assigned and grouped according to approximately equal mean body weight (bw). Among the two groups (24 males/each), one was considered as control and received distilled water by gavage. The other group was given Delta regularly at 9.00 am by gavage at the dose level of 5 mg/kg bw/day (approximately ¼ DL50 for mouse 21 ) for 35 days. This dose was chosen based on other previous studies that we tested different concentrations of delta. Mortality was observed for concentration exceeding 5 mg/kg bw/day when we kept it. 22
The experimental protocol was performed according to the European convention for the protection of vertebrate animals used for experimental and other scientific purposes (Council Directive no. 2010/63/EU). Approval for these experiments was obtained under control of the Medical Ethics Committee for the Care and Use of Laboratory Animals of the Pasteur Institute of Tunis (approval number: FST/LNFP/Pro 152012).
Mating and fertility indices
After the 35th day, each treated male mouse, with continuation of daily doses, was co-housed with unexposed proven fertility (estrous phase), sexually receptive, female mice (one to two/box). Successful mating was confirmed by the presence of vaginal plug and/or sperm in the vaginal smear (the following morning between 9.00 and 10.00 h) and the day was counted as “0” day post-coitum and the following day as embryonic day 1.
23
Reproductive performance of parental males was summarized using the following indices: Male mating index (%): number of males inseminated females × 100/total number of males cohabited with females; Male fertility index (%): number of males proving their fertility (= number of pregnant females) × 100/total number of inseminated females with evidence of vaginal plug.
For females that failed to deliver a litter, this was considered as a sign of male infertility, whereas litter delivery indicated male fertility.
Clinical observations and body weight measurement
Each mouse in each group was observed twice daily (before and after exposure) for signs of clinical toxicity in the appearances of the skin and fur, eyes and mucous membrane, behavioral pattern, and mortality. Initial (weight of start point) and final (weight of end point) body weights were recorded. On day after fecundity and fertility test, male mice from each group were slightly anesthetized with ether and killed by decapitation to avoid stress conditions. The reproductive organs were dissected out and cleansed well and weighted accurately.
Reproductive outcome data
Females were then separated and allowed to gestate to term. The morning on which the parturition was complete was designated as postnatal day (PND) 0. All the pups were examined on PND 0 to determine the number of alive and dead pups. All the live pups were individually counted. 23
Evaluation of sperm characteristics
Electroejaculation
After the sexual cohabitation and fecundity test period, the electrical stimulator (EP-2 Electroejaculator; Beltron Instruments, Colorado, USA) consists of a sine wave rheostat transformer, possessing variable output of 0–6 V at 60 Hz. The ejaculate was obtained from mice by applying two successive series of electrical stimuli, which delivered via a bipolar rectal probe. A probe was covered throughout its length with a polyethylene sheath, except for 3 mm sections at the tip and 7 mm from the tip, which were cut out to expose a stainless steel and copper electrode, respectively. The rectal probe was lubricated with gel and inserted 2–3 cm into the rectum of mouse. Two stimuli were administered at each voltage setting, up to a maximum of 5 V, or until a coagulum was detected at the tip of the glans penis, whichever occurred first. The combined fluid and coagula resulting from the two sets of electrical stimuli were regarded as the ejaculate. 17
Semen analysis
Immediately after collection, semen was kept at 35°C in water bath for evaluation. Due to the small volume, the semen was subsequently diluted in 9 volumes of phosphate buffered saline 1× solution.
Evaluation of spermatozoa motility, count, viability, and abnormalities
A 10 µL sample was taken for the determination of sperm concentration and percentage sperm motility. The sperm concentration was determined with a Neubauer hemocytometer. 24 The percentage motility was determined by the progressive and nonprogressive movements of sperm observed under a compound microscope.
Assessment of live and abnormal spermatozoa was performed using eosin–nigrosine staining. Unstained (intact) and red-colored (with damaged membranes) spermatozoa were counted under the microscope using 1000× objectives and oil immersion. Sperm viability was defined as the percentage of intact cells, as per the procedures described in the Laboratory Manual of World Health Organization. 25 To evaluate the spermatozoa abnormalities, the spermatozoa were classified according to the Wyrobek and Bruce criteria. 26 Sperm cells with normal morphology and cells presenting abnormalities in head, midpiece, and tail were assessed. At least 200 sperm cells were observed in each slide, under 1000× magnification.
Biochemical assays
After the biophysical semen parameters were evaluated, the semen samples were centrifuged at 3000g for 20 min at 4°C to separate out seminal plasma. Initial hydrogen ion concentration (pH) of semen samples was determined immediately after collection using a pH paper (Universal indicator pH 0–14 Merck, Merck KgaA, 64271 Darmstadt, Germany). The remainder of each diluted ejaculate was used to determine the fructose,
Fructose
The level of fructose of the samples was determined using the colorimetric method that each semen sample was deproteinized with 63 µM of zinc sulfate and 0.1 M sodium hydroxide and centrifuged at 8000g for 5 min. Indole reagent and concentrated hydrochloric acid were added to supernatant and heated for 20 min at 50°C.The calibration curve was prepared using fructose as standard. The reaction was measured spectrophotometrically at 470 nm. 27
Evaluations of serum hormones in mice
Blood samples were collected and serum were separated and stored at −20°C for the measurement of testosterone and inhibin B levels.
Testosterone level
Serum testosterone was measured by enzyme-linked immunosorbent assay method using commercial kit (testosterone enzyme-linked immunosorbent assay (ELISA) kit (Neogen Product #402510; Lansing, Michigan, USA)). Briefly, 50 µL of samples or standards were placed on testosterone antibody-coated plate, followed by the diluted enzyme conjugate. The mixture was incubated for 1 h at room temperature, during which competition for binding sites took place. The plate was washed with buffer (WA2). Bound enzyme conjugate was detected by coloring agent (KBlue) in the kit. Absorbance readings at 650 nm of the samples and standards were compared to obtain quantitative results. Standard curve was plotted and the concentration of sample was estimated from curve. The intra-assay coefficient of variation was 3.6%, the interassay coefficient of variation was 6.0%, and the sensitivity was 0.029 ng/mL.
Inhibin B level
The inhibin B ELISA (Abnova; catalog no. KA1683; Taiwan) is an enzymatically amplified three-step “sandwich” assay. Briefly, 100 µL of each standard, positive control, and sample were added to the wells of a microplate. After 2.5 h incubation on a shaker, the microplate was washed four times and 100 µL of prepared HRP-streptavidin solution was added to each well and incubated for 45 min. The plate was washed with buffer. Then, Add 100 µL tetramethylene benzidine one-step substrate reagent to each well. The plate was incubated for 30 min at room temperature. Finally, 50 µL of stop solution was added to each well. The degree of enzymatic turnover of the substrate was determined by dual wavelength absorbance measurement at 450 nm.
Histopathological examination
The left testis was removed and fixed in 10% formaldehyde solution for 24 h. Samples were then dehydrated and embedded in paraffin. Sections 5 µm thick were performed in a microtome and stained with Mayer’s hematoxylin and eosin. For testicular toxicity, approximately 200 circularly sectioned seminiferous tubules for each mouse were assessed microscopically.
Statistical analysis
The results obtained are expressed as mean values ± SD. Values of p < 0.05 were considered significant. Comparisons were made between control and treatment groups using Student’s t test for normally distributed values. All statistical analyses were performed using the SPSS 13.0 software.
Results
Clinical observations and body weight measurement
During the exposure duration, all male mice survived until the end of the experimental period in treated and control group. One male mouse showed a swelling in the testicle, one pregnant female mouse also died just before delivery. No significant changes were observed in body weight and weight of reproductive organs during treatment (Table 1).
Effect of deltamethrin on body and reproductive organ weight after treatment in Swiss albino mice.a

aData are presented as mean ± SD (standard deviation); bw: body weight.
Evaluation of reproductive performance quality
Mating and fertility indices and reproductive outcome data
In comparison with control group, the mating and fertility indices in male mice exposed to Delta for 5 weeks at 5 mg/kg bw/day were significantly (p < 0.001) decreased (Table 2).
Characteristics of electrolejaculated semen.a

bw: body weight.
aData are presented as mean ± SD. Number of males which used for mating (n = 24).
bMating index (%): number of males inseminated females×100/total number of males cohabited with females.
cFertility index (%): number of males proving their fertility (=number of pregnant females) × 100/total number of inseminated females with evidence of vaginal plug.
Significant reduction was observed in the number of pregnant females (p < 0.05), number of litter and number of live fetuses in untreated females that mated with males exposed to 5 mg/kg bw/day, as compared to controls (Table 3).
Clinical Findings in the pregnant mice after cohabitation with exposed male mice to deltamethrin.

PND: postnatal day; bw: body weight.
aData are presented as mean ± SD.
Semen analysis
Treatment with Delta decreased (p < 0.001) semen ejaculate volume initial pH and libido as compared to control (Table 2). Administration of 5 mg/kg bw/day of Delta to male mice resulted in a decrease in counts in ejaculated spermatozoa (p < 0.05) and a reduction of sperm motility (p < 0.001), while the number of dead sperms significantly increased (Table 2). Delta displayed significant varying changes on semen analysis. No correlation was observed between fertility and fructose,
Evaluations of reproductive hormones
The testosterone levels were decreased in the animals treated with 5 mg/kg bw/day of Delta (p < 0.01; Figure 1). Similarly, the level of inhibin B (Figure 1) showed a significant decrease (p < 0.01) after exposure to Delta compared to control group.

Testosterone and inhibin B levels in mice after exposure to deltamethrin. Values are expressed as means ± SD. **p < 0.01.
Histological finding
Testicular histopathology showed the effect of Delta on spermatogenesis (Figure 2). The testes of control mice showed the presence of normal testicular architecture and regular seminiferous tubular morphology with normal spermatogenesis and the presence of primary and secondary spermatocytes, spermatids, and spermatozoa (Figure 2(a)). The mice exposed to 5 mg/kg bw/day of Delta showed severe alterations of the seminiferous tubules. The affected tubules were lined by fewer spermatogenic cells. In addition, vacuolization in Sertoli cells and loss of germ cells were also found compared to the controls group. Testicular damage in treated mice group of Delta resulted in atrophy of seminiferous tubules and subsequently an increase of the interstitial space between these (Figure 2(b)).
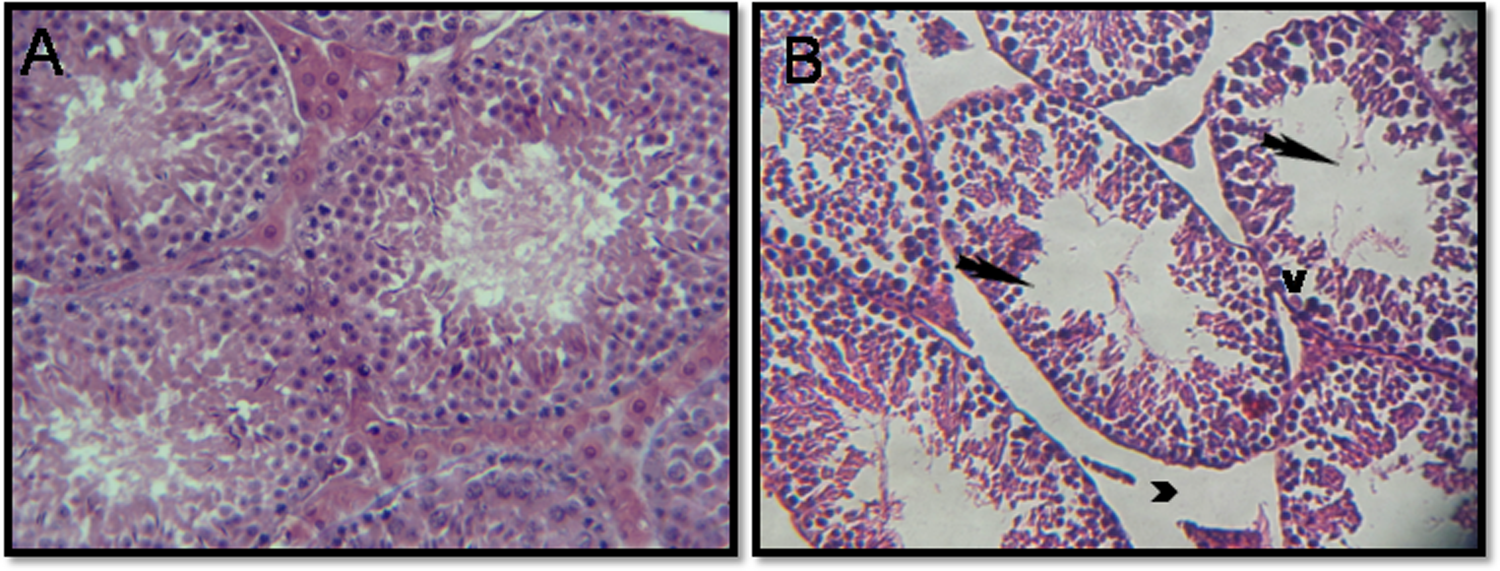
Testicular sections of control mice (part A), which show normal spermatogenesis: Note the normal cell arrangement in the seminiferous tubules. The interstitial spaces also appear normal (×400 H&E). Testicular sections of mice treated with 5 mg/kg/day of Delta (part B): Note the atrophic seminiferous tubules and subsequently an increase of the interstitial space between these (head arrow). Sloughing of germ cells into tubular lumen (arrow) and vacuolization of germ cell cytoplasm (V) and the disruption of spermatogenic cells (×400 H&E).
Discussion
Xenobiotics and environmental contaminants such as pesticides are known to induce a broad spectrum of toxicological effects and biochemical dysfunctions constituting serious hazards to health. Semen quality was deteriorated after Delta administration (Table 2). Our results are in agreement with reported studies.
30
Previous study showed that exposure to lambda-cyhalothrin caused sexual dysfunction in male rats.
31
Fertility was related to ejaculate volume, sperm counts and the number of motile spermatozoa per ejaculate (Table 2). Past studies demonstrated reduced semen quality in men occupationally exposed to various pesticides
32
and in animals.
18
The decline in ejaculate volume, initial pH sperm concentration, and increased reaction time can be partly attributed to the deltamethrin-induced reduction in testosterone levels (Figure 1). The effects of pesticides on spermatogenesis may be mediated through their effects on hormonal balance. The seminal plasma mediates the chemical function of the ejaculate. Thus, biochemical evaluation of seminal plasma is an important criterion for assessing fertility and diagnosing male reproductive disorders.
33
The sugar composition of seminal plasma has also been correlated with fertility, mainly due to its importance to spermatozoa energy production. Fructose and glucose are essential for adenosine triphosphate production and motility of spermatozoa. In the present study, no correlations were found between fertility and fructose and citric acid levels, respectively. However, evaluation of
Mice treated with Delta had also markedly impaired libido as measured by indices of mating and fertility and number of pregnant females housed with male mice exposed to this pesticide. Ratnasooriya et al. 31 reported that lambda-cyhalothrin exposure of male rats may induce anti-libido effects probably via brain GABAergic and/or dopaminergic systems.
Pesticide has been proven to alter steroidogenic hormones, 36 which result in impairment of the reproductive physiological mechanisms. 37 Testosterone is the major driver of male reproductive development and function and suppression of its levels within the adult testis shuts down spermatogenesis (the process by which mature sperm cells are formed) and induces infertility. 38,39 Inhibin B, produced by the seminiferous epithelium, suppresses follicle-stimulating hormone (FSH) secretion of the pituitary and plays an important role in the feedback regulation of the pituitary–gonadal axis. 40 Perturbations in this axis can be caused by stress induced by pesticide exposure. 41 The main function of dimeric inhibin (both A and B) is the negative control of FSH secretion. 42 In the mouse, inhibin might have an intratesticular action, for example, in the control of Sertoli and Leydig cell neoplastic proliferation, 43 but there is no evidence of any clinically relevant, intratesticular inhibin action so far .The present study shows that exposure of male mice to Delta is associated with decreased serum testosterone and inhibin B levels. These effects on testosterone were attributed to a decrease in serum luteinizing hormone (LH), since LH serves as a normal stimulus for the secretion of this steroid from the testicular Leydig cells. Similar to the effects of other insecticide, 44 it is probable that the production of gonadotropins has been affected by Delta in male mice by disruption of the hypothalamic–pituitary–gonadal axis and could be due to sensitivity of neuroendocrine neurons in the anterior hypothalamus. 45 Because testosterone stimulates the production of inhibin B, decreased testosterone levels result in a reduction of inhibin B concentration, which further induces FSH and LH secretion. 46 Inhibin B is produced by the Sertoli cells. Its concentration is a reflection of the amount of these cells and could be a marker of spermatogenesis. This study has shown that the value of inhibin B may be used as a marker of spermatogenesis, both qualitatively and quantitatively. Previous studies have shown the relationship between inhibin B levels and concentrations of ejaculated spermatozoa. 47 Once full spermatogenesis is reached, changes in inhibin B levels reflect mainly the status of germ cell proliferation and development and depend only secondarily on FSH. 48
Conclusion
Based on these results, this study concluded that Delta exhibits a central role in affecting the male reproductive system. Delta exposure leads to a significant reduction in serum testosterone and inhibin B concentrations, which has caused a drastic decrease in ejaculated sperm motility, count and viability. These sperm parameters are the key indices of male fertility, which are the prime markers in testicular spermatogenesis. Delta may have a harmful effect because of its potential to modify the male hormone profile as a function of the type of pesticide used as well as the magnitude of exposure.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present work was supported by Direction Générale de la Recherche Scientifique et Technique-Tunisie grants (DGRST). Appui à la Recherche Universitaire de base UR11ES70).
