Abstract
Background:
Tobacco smoking is a serious threat to life and health of society. Among the most vulnerable to the toxic effects of tobacco smoke are foetuses and newborns. The objective of the research was to assess the impact of tobacco smoke exposure on oxytocin levels and biochemical oxidative stress parameters during pregnancy and after birth in an experimental model.
Methods:
In the experiment, exposure to tobacco smoke of gravid and non-gravid rats was monitored. A reliable biomarker of exposure – cotinine – was used in the process and it was determined by means of high-performance liquid chromatography with diode array detection, which ensured high analytical accuracy and precision. Determination of oxytocin was performed by means of enzyme-linked immunosorbent assay. The levels of selected oxidative stress parameters: total protein concentration, uric acid, trolox equivalent antioxidant capacity, protein S-nitrosylation and lipid peroxidation (thiobarbituric acid reactive substances) were measured by spectrophotometric methods.
Results and conclusions:
The effect of prenatal and postnatal exposure to tobacco smoke was a lower medium body mass of rat foetuses and pups. Oxidative stress during pregnancy, additionally intensified by tobacco smoke exposure, led to adaptive changes in properties of plasmatic antioxidant barriers. Moreover, the disturbance of oxidoreductive balance by tobacco smoke affects oxytocin fluctuations, what was observed in this study during lactation period. Therefore, women who smoke may breastfeed their children less frequently and for a shorter period.
Keywords
Introduction
Tobacco smoking is a serious threat to life and health of every society. It is estimated that 1 billion men and approximately 250 million women in the world smoke, and many of them during their reproductive years. According to the World Health Organization, smoking results in the death of nearly 6 million people annually, of whom 5 million are former smokers and over 600,000 are those who do not smoke but are exposed to second-hand smoke from others. 1 Tobacco smoke consists of many toxic compounds producing reactive oxidant species (ROS). 2,3 When concentrations of ROS exceed antioxidant capacities, this imbalance can cause systemic disorders development. 3,4 During pregnancy, it is particularly hazardous for both the mother and foetus.
Maternal smoking during pregnancy reduces foetal growth, both in humans and laboratory animals, and results in adverse health in later life. 5 Nicotine, a pyridine alkaloid, is responsible for the addictive and many of the toxic effects of tobacco. Foetal exposure to nicotine alters foetal heart rate variability and altered lung function. 6 Infants reveal similar disturbances in neural control of the cardiorespiratory system, with a heightened vascular tone, cardiac rate, and elevated blood pressure (compared to infants not exposed to nicotine). 7 Prenatal exposition to maternal smoking is related with an increased apnoea index in active sleep and a reduced arousal response. 8 Nicotine is easily excreted into the breast milk and rapidly absorbed in the neonate’s intestine and results in increased plasma multiline levels, which is related to both infantile colic with excessive crying and gastroesophageal reflux in infants. 9
Apart from the numerous lines of antioxidative defence (i.e. enzymes and vitamins), a peptide hormone produced by the hypothalamus, oxytocin, deserves particular attention in terms of safety of pregnancy. Oxytocin is a nonapeptide that acts as a neuromodulator. This hormone, synthesized in the neurons of the hypothalamus (paraventricular nucleus and supraoptic nucleus), is released into the systemic circulation, but also centrally, during vaginal stimulation and parturition, to act upon oxytocin receptors widely expressed through the central nervous system. 10 Suckling of the nipple is known to stimulate oxytocin release not only into the circulation but to cause milk ejection. 11 Oxytocin increases trust in human social interactions and is associated with prosocial behaviours such as maternal–infant bonding. Neurophysiologists have termed oxytocin as a ‘love and attachment’ hormone that promotes a unique emotional bond between the mother and her infant. 12,13
Oxytocin and nicotine have similarities. Both substances are related in fear reduction and stimulation of the brain ‘reward’ centres. Whilst tobacco smoke with nicotine enhances production of dopamine ‘pleasure neurotransmitter’, oxytocin exerts a similar response. Salamon employed oxytocin inhalation as a substitute for ‘cigarette lightening ritual’ and in the initial calming effects of smoking. Oxytocin, he claimed, could replace traditional nicotine replacement therapies for those seeking to stop smoking. 14 Cigarette smoke extracts containing nicotine have been shown to increase the contractile sensitivity of the preterm myometrium responsiveness to oxytocin by upregulating the expression of oxytocin receptors, and increases the risk of preterm delivery in women who smoke. 15 The role of nicotine and oxytocin in alternating maternal behaviours surrounding breastfeeding are largely unexplored.
The objective of this investigation was to assess the impact of tobacco smoke exposure on oxytocin levels and selected oxidative stress parameters (total protein concentration, uric acid (UA), trolox equivalent antioxidant capacity (TEAC), protein S-nitrosylation and lipid peroxidation) during pregnancy and after birth in an experimental model.
Materials and methods
Permission for the research was issued by The Local Ethics Committee for Animal Experimentation in Poznan, Poland (Act no. 53/2011 on 13 June 2011).
Animals
Wistar male and female 4-month old (sexually mature) rats bred in the Department of Toxicology, Poznan University of Medical Sciences, of mean body weight 232 g, were used in the experiment. The animals were kept in cages in standardized lighting conditions (12-h light/12-h dark cycle), humidity (50–60%) and temperature (20–22°C).
Experimental design
Sexually mature female and male rats were used in the experiment. Some of the female rats were enrolled in non-gravid groups. The remaining animals were selected for mating. Subsequently, during the oestrus phase, smear for microscopic detection of sperm was collected from females. After confirmation of sperm presence, the female was assigned to a specific group and kept in an individual cage until labour. Seventy-two female rats were enrolled in the study and divided into eight experimental groups. Control groups were formed from non-gravid and non-exposed females (NE-C) and non-gravid but tobacco smoke-exposed females (E-C). Experimental groups were created from gravid females non-exposed to tobacco smoke that were dissected 1 day prior to labour (NE1), 1–2 days post labour (NE2) and 2 weeks after labour (NE3). Moreover, experimental groups consisted of gravid rats exposed to tobacco smoke (E1, E2 and E3, respectively) that were divided similarly to non-exposed animals.
Exposure to tobacco smoke was performed in a toxicological chamber for 5 days a week, 6 h a day, during 3 weeks – throughout whole pregnancy. The chamber is a glass rectangular cuboid of 308 dm3 capacity, fitted with parallel tubing. There is a movable cover situated in the upper part and carbon monoxide (CO) detectors located in the lateral walls of the tank. Tobacco smoke is introduced into the chamber through a pipe, connected to perforated tubing. The air outlet vent is located on the opposite side. This tubing system provides an even and uniform distribution of smoke within the chamber. The CO concentration in the chamber reflected the smoke content in the inhaled air and was continuously monitored by a gas analyser, Infralyt 1110/1210 (infrared measurement), to maintain 1500 mg CO/m3 of air. The level of oxygen was established at 20 ± 0.5% of the air volume. The air in the chamber was changed 10 times per day. 5,16
Rat foetus and pups were weighed soon after birth. On scheduled days of experiment, dissection was performed. Ketamine in a dose of 40 mg/kg body weight with xylazine in a dose of 5 mg/kg body weight were administered intramuscularly for anaesthesia. Blood samples were collected into heparinized tubes and centrifuged at 3000 r/min for 10 min. Subsequently, plasma was obtained and frozen in −80°C for further analysis.
Reagents
Oxytocin (human, rat, and mouse) EIA Kit, Cat. # EK-051-01 was obtained from Phoenix Pharmaceuticals Inc. (Burlingame, California, USA). Uric acid ver.2 Roche/Hitachi cobas c systems from Roche Diagnostics GmbH (Mannheim, Germany). 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) (ABTS), 5,5′-dithiobis-(2-nitrobenzoic acid), 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (trolox), acetonitrile, albumin standard solution, ammoniumsulphamate, anhydrous sodium carbonate, copper sulphate, cotinine, deionized water, Folin–Ciocalteu reagent (FCR), glutathione, hydrochloric acid, hydrogen peroxide, mercury chloride, methanol, N-(1-naphthyl)ethylenediamine, n-butanol, norephedrine, phosphate-buffered saline (PBS), phosphoric acid, potassium hydrogen phosphate, potassiumpersulfate, sodium carbonate, sodium citrate, sodium hydrogen phosphate, sodium hydroxide, sodium nitrate, sulphosalicylic acid, sulphuric acid, thiobarbituric acid, trichloroacetic acid, and triethylamine were high-purity products, purchased from Sigma-Aldrich (St Louis, Missouri, USA) and local suppliers.
Toxicological and biochemical assays
Cotinine plasma extraction was performed using the method of solid phase extraction described by Lozano et al. 17 The concentration of this compound was determined by means of high-performance liquid chromatography with diode array detection (HPLC-DAD).
Determination of oxytocin was performed by means of enzyme-linked immunosorbent assay ( protocol for catalog # EK-051-01 oxytocin).
Quantification of total protein concentration was performed with the use of Lowry et al.’s method based on a sensitive reaction of peptide bonds and aromatic amino acids with FCR. 18 In vitro enzymatic colorimetric test carried out by Roche/Hitachi cobas c system was used to assess UA levels (UA ver.2 Roche/Hitachi cobas c systems, Roche Diagnostics GmbH, D-68305). Trolox equivalent antioxidant capacity (TEAC) was measured according to Re et al.’s method. During assessment, the ability of the studied substance to reduce ABTS radical was compared to the standard (trolox). 19 Protein S-nitrosylation was evaluated with the use of sulfanilamide and N-(1-naphthyl)ethylenediamine in a reaction described by Bonina et al. 20 Lipid peroxidation (detection of thiobarbituric acid reactive substances (TBARS)) was assayed with a fluorescent product of thiobarbituric acid reaction. 21
Statistical analysis
A statistical analysis was performed by Statistica PL version 10 (StatSoft). A comparison between control and experimental groups for variables that are not normally distributed or without equal variances was performed via Kruskal–Wallis test. For variables with normal distribution and equal variances, an analysis of variance test for unrelated samples was used.
A comparison between exposed and non-exposed groups was also made for variables that are not normally distributed using the Mann–Whitney test and for variables with normal distribution and equal variances through the agency of Student’s t test for unrelated samples. The t test with independent estimates of variance was used for variables with normal distribution but without equal variances.
Results
Plasma cotinine levels
Measurements of cotinine concentration in exposed animals from group E2 set against exposed control group E-C show a 50.08% decrease. In the group of animals 1 day before delivery exposed to tobacco smoke, cotinine concentration was 18% lower than in the control group, but these differences were not statistically significant. The difference between the two groups exposed to tobacco smoke – 1 day before delivery and 1–2 days after delivery – is a result of extending the elimination of this xenobiotic. For animals exposed to tobacco smoke 14 days after delivery, all the determined concentrations were below the limit of quantification. The results of cotinine concentration are shown in Figure 1.

Plasma cotinine levels. (a) Statistically significant difference compared with the control group (exposed rats).
Plasma oxytocin levels
An analysis of plasma oxytocin concentrations (Figure 2) in non-exposed animals 1 day prior to delivery (NE1) shows a 38.70% decrement in comparison to non-gravid and non-exposed control animals (NE-C). In gravid non-exposed animals from group (NE2) it was 37.25% lower than in non-exposed control group (NE-C). A comparison of the concentration determined among gravid rats, from exposed group E3 with non-exposed (NE3) and exposed (E1 and E2) groups demonstrated a 45.71%, 40.93% and 43.92% decrease, respectively. The difference between gravid, exposed group of rats (E3) and non-gravid, exposed control (E-C), was also observed (63.55% decrease).
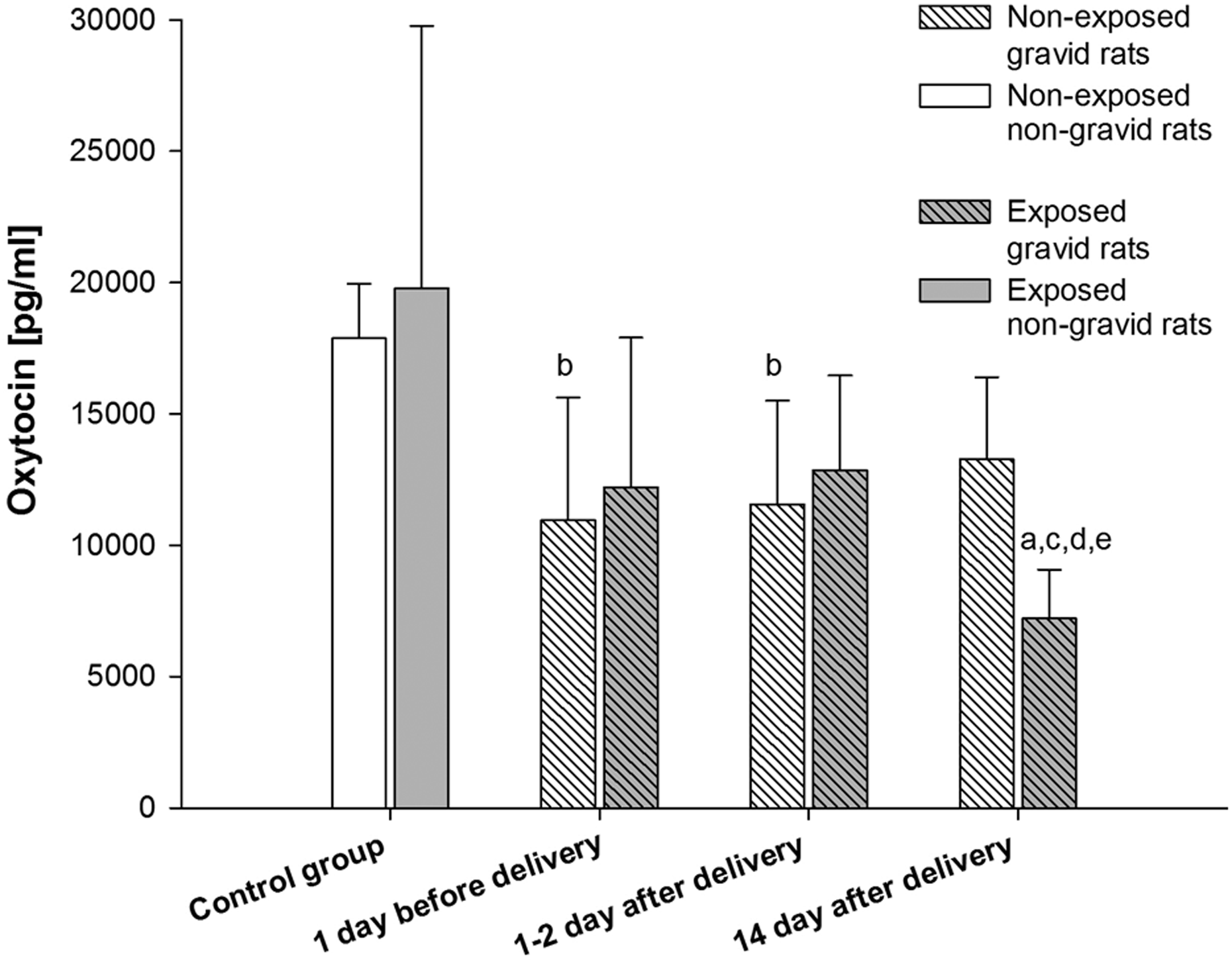
Plasma oxytocin levels. (a) Statistically significant difference compared with the control group (exposed rats). (b) Statistically significant difference compared with the control group (non-exposed rats). (c) Statistically significant difference compared with the non-exposed rats at the same time intervals. (d) Statistically significant difference compared with 1 day before delivery group (exposed rats). (e) Statistically significant difference compared with 1–2 days after delivery group (exposed rats).
Oxidative stress parameters levels
The levels of total protein concentration, UA, TEAC, protein S-nitrosylation and lipid peroxidation (TBARS) were measured.
Total protein plasma concentration (Figure 3) decreased by 14.22% in E3 group (50.18 ± 12.49 mg/mL), in comparison to exposed animals from the control group (58.50 ± 4.64 mg/mL).

Plasma total protein levels. (a) Statistically significant difference compared with the control group (exposed rats).
In plasma of rats from gravid and exposed group (E2), a 59.36% increase in UA concentration (Figure 4) was observed (6.8 ± 1.9 μg/mL), in comparison to non-gravid and the exposed control group (4.3 ± 1.3 μg/mL). The parameter was also 49.99% higher in group E3 (6.4 ± 4.1 μg/mL), in comparison to E-C group (4.3 ± 1.3 μg/mL).

Plasma uric acid levels (UA). (a) Statistically significant difference compared with the control group (exposed rats).
Another analysed parameter was TEAC (Figure 5). The levels in E2 group (5.52 ± 0.66 μM/mg of protein) were elevated by 13.07% when compared to exposed control group (4.88 ± 0.55 μM/mg of protein). A 29.45% increase in the parameter value was also observed in rats from E3 group (6.32 ± 0.84 μM/mg of protein), in comparison with exposed control group (4.88 ± 0.55 μM/mg of protein) and a 29.77% increase in relation to NE3 group (4.87 ± 0.55 μM/mg of protein).

Plasma trolox equivalent antioxidant capacity (TEAC). (a) Statistically significant difference compared with the control group (exposed rats); (c) Statistically significant difference compared with the non-exposed rats at the same time intervals.
In the case of S

Plasma levels of S-nitrosylated proteins. (a) Statistically significant difference compared with the control group (exposed rats); (c) Statistically significant difference compared with the non-exposed rats at the same time intervals; (e) Statistically significant difference compared with 1-2 days after delivery group (exposed rats); (f) Statistically significant difference compared with 14 day after delivery group (exposed rats); (h) Statistically significant difference compared with 1-2 days after delivery group (nonexposed rats).
TBARS levels (Figure 7) in gravid females exposed 1 day prior to delivery (E1) reduced by 62.64% in relation to non-exposed from the same period of time (0.68 ± 0.58 ng/mg of protein vs. 1.82 ± 0.52 ng/mg of protein). A comparison of exposed group – E2 (1.12 ± 0.91 ng/mg of protein) and non-exposed group – NE2 (2.43 ± 0.70 ng/mg of protein) showed a decline in the parameter value by 53.91%. Levels of TBARS were 135.39%, 194.19%, 570.59% and 307.14% higher in animals from E3 group (4.56 ± 1.23 ng/mg of protein) than in rats from the exposed control – EC (1.94 ± 0.63 ng/mg of protein), NE3 (1.55 ± 1.13 ng/mg of protein), E1 (0.68 ± 0.58 ng/mg of protein) and E2 group (1.12 ± 0.91 ng/mg of protein), respectively.
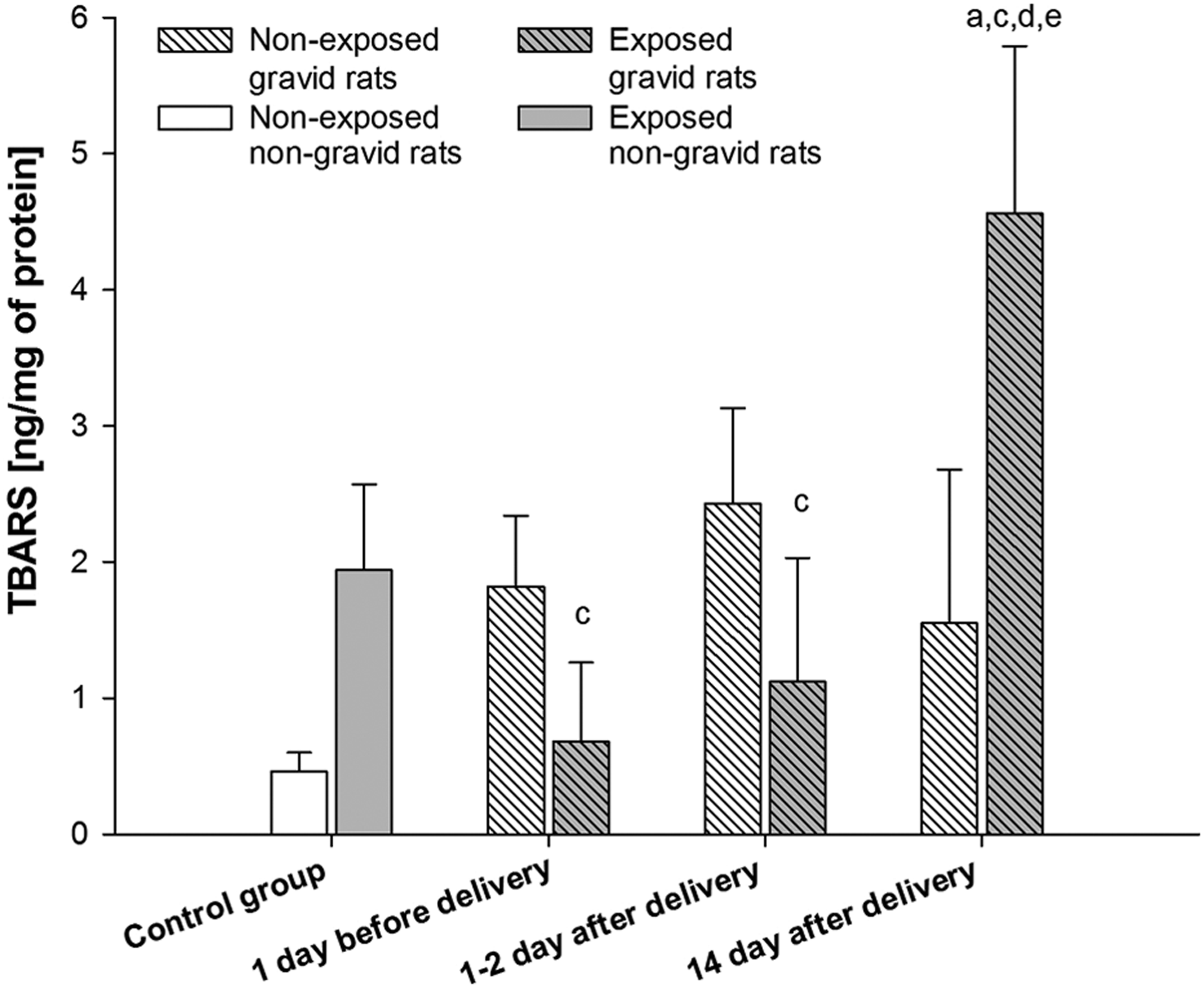
Plasma levels of thiobarbituric acid reactive substances (TBARS). (a) Statistically significant difference compared with the control group (exposed rats); (c) Statistically significant difference compared with the non-exposed rats at the same time intervals; (d) Statistically significant difference compared with 1 day before delivery group (exposed rats); (e) Statistically significant difference compared with 1-2 days after delivery group (exposed rats).
Foetal/pup body weight
The body weight of rat foetuses and pups from mothers which were exposed (E1, E2 and E3 groups) demonstrated a decrease of value by 77.04%, 14.16% and 38.70% compared to non-exposed animals (groups NE1, NE2 and NE3).
Discussion
During pregnancy, various adaptive changes occur in the mother’s organism. 22,23 Higher energy and oxygen requirements contribute to disturbance of pro-oxidative/antioxidative balance, resulting in potential damage of the organs. 24,25 Both the foetus and the mother are to a certain extent vulnerable to the negative effects of free radicals, especially ROS and reactive nitrogen species (RNS). 25
Furthermore, oxidative processes, naturally enhanced in the gestation period, can be additionally intensified by the use of some xenobiotics. For example, maternal tobacco smoking is a source of active oxidizing agents that release free radicals harmful for health. 26,27
However, natural defence systems of the body are mobilized in pregnancy for protection of the developing foetus. Apart from the numerous lines of antioxidative defence, the neurohypophysial peptide oxytocin deserves particular attention in terms of safety. 28 –31
In the presented study, attention was focused on the fact that the nicotine addiction can impair the protective function of oxytocin and, as a result, increase negative effects of oxidative stress in the mother and the foetus.
Since it is established that oxytocin plays a key role in delivery and lactation, its concentration was monitored in experimental animals, both exposed and non-exposed to tobacco smoke, 1 day prior to delivery (date calculated on the basis of insemination time), 1–2 days after delivery and 2 weeks after delivery. 32
An evaluation of the correlation between tobacco smoke and the plasma oxytocin levels was possible due to a simultaneous determination of cotinine (the main tobacco smoke exposure biomarker). A marked decrease in cotinine concentration was observed in rat plasma: by the end of exposure period it had declined by half and after 2 weeks it was no longer present in animals’ blood. These results are concordant with t 0.5 of cotinine amounting to approximately 17 h. 33
Many scientists emphasize that smoking reduction and cessation give beneficial health outcomes within only a few days (stabilization of blood pressure, enhancement of respiration parameters, and improvement of heart functioning). 34 –36 Desisting from smoking is extremely relevant in pregnancy. Smoking during the foetal development period can result in respiratory, cardiovascular and nervous system impairment, and an increased incidence of sudden infant death syndrome. 37 Diminution foetal in body weight and length is a common consequence, which has also been observed in this study. 36,38 In all the animal groups exposed to the tobacco smoke lower average masses of foetuses and newborn pups in comparison to the corresponding groups of non-exposed animals were observed. The most profound difference (77.04%) was noticed between the groups consisting of gravid rats exposed to tobacco smoke and gravid females non-exposed to tobacco smoke (all animals dissected 1 day prior to labour). It is worth mentioning that in the groups of pups fed by their mothers for 2 weeks after delivery the difference in body masses was also significant: it was 38.70% lower in the pups of exposed dams than in the pups of those non-exposed. At this point, it is relevant to quote the data, suggesting that this process can be impeded in the mothers that smoked during pregnancy and lactation. 39
Studies on an animal model show that a reduced level of oxytocin affects the onset of maternal behaviour. 40 In human studies, in turn, women who smoked during pregnancy breastfeed their children less frequently and for a shorter period than non-smoking women. 41 Nicotine raises circulating adrenaline significantly, which modulates the release of oxytocin. This may result in a reduction of oxytocin, thus interfering with milk ejection in women who smoke. 42 Changes in oxytocin levels during pregnancy are highly correlated with sex hormones activity, especially with a concentration of oestrogens and progesterone. The latter is essential for pregnancy maintenance. Its concentration increases with the course of pregnancy, repressing contraction-associated agents, such as oxytocin. 43 This fact was reflected by the outcomes of the presented research, namely by a lower concentration determined in plasma of animals 1 day prior to delivery, in comparison to the control group.
Physiological levels of oxytocin have to be maintained by progesterone at a congruently low concentration during the whole period of pregnancy (for its proper development) until the imitation of labour, when its concentration rapidly raises. At the end of parturition the hormone stabilizes at an average concentration and increases again only at the time of lactation, which is correlated with maternal milk secretion.
As is well known, smoking affects female sex hormones, that is, oestrogen and progesterone, which control the course of gestation. 44 Significant differences in the oxytocin concentrations in groups exposed and non-exposed to tobacco smoke have been reported. A comparison of oxytocin concentrations between exposed and non-exposed groups of animals over the same period of time provides surprising information. One day prior to labour and 1–2 days after delivery, these differences were not statistically significant, oxytocin concentration notwithstanding the exposure time was comparable.
An interesting phenomenon is, however, a marked reduction in the oxytocin concentration in the mothers from the exposed group, observed 2 weeks after exposure. It is lower by more than 45% in comparison to non-exposed group at the same time. Furthermore, the concentration of oxytocin in exposed animals was significantly reduced as compared to the values marked the day before and 1–2 days after birth (by almost 41% and 44%, respectively). This unusual dependence may result from the fact that during the pregnancy as well as the labour, in response to large amounts of free radicals present in tobacco smoke, there was a mobilization of protective factors in rat organisms, maintaining oxytocin levels. However, this effect was not observed at the time of lactation.
Many researchers suggest that the occurrence of oxidative stress during pregnancy is highly probable. 24,25,45,46 One of the evaluated parameters in this research was the determination of protein damage by ROS and RNS. An analysis of the measurements of total protein concentration in plasma suggests that exposure to tobacco smoke contributed to a slight reduction in the concentration of plasma proteins in all groups of animals, whereas in the group of animals dissected 2 weeks after delivery, the difference compared to the exposed control group was statistically significant (14%). In animals not exposed to tobacco smoke, there was no statistically significant difference in total protein levels.
A common effect of intensified exposure to free radicals is an increased activity of antioxidant enzymes and an increment of low-molecular weight antioxidants, caused mainly by an augmented synthesis of enzymatic proteins. This defence enhancement against free radicals is adaptive and is even considered by many researchers for the testimony of oxidative stress occurrence. 47 This situation was observed in the case of UA concentration and the analysis of TEAC (differences between gravid and non-gravid animals, exposed to tobacco smoke).
UA (physically occurring mainly in the form of urate) is oxidized by strong oxidizing agents (usually by peroxide and hydroxyl radicals), which leads to urate anion free radical. 48 It is relatively non-reactive and does not react with radical oxygen. Acid molecule oxidation leads to the formation of allantoin. UA reacts also with peroxynitrite and is the main factor responsible for the total antioxidant capacity of blood plasma. 49 A significantly elevated level of UA in rats exposed to tobacco smoke, determined in 1–2 days and 2 weeks after delivery, compared to the exposed but non-gravid group, indicate the mobilization of plasma antioxidant defence during the postpartum and pups feeding period.
Measurements of TEAC showed a similar relationship. In rats from the postpartum groups, TEAC levels were apparently higher than those of the control groups of animals. Moreover, in the group of animals, which were being during lactation period and after the end of tobacco exposure, adaptive processes, expressed by elevated TEAC levels, were significantly augmented in comparison with the group not exposed to tobacco smoke.
Owing to pregnancy, redox imbalance may lead to alterations in the direction of many structures and molecules. 50 An example is nitric oxide (NO), which under physiological conditions is an important signal factor. During severe oxidative stress conditions, the beneficial properties of NO change to harmful ones. 51 This situation also occurs in the case of tobacco smoke exposure. The number of free radicals in cigarette smoke is approximately 1.015 for 1 swig. 52 NO is relatively little reactive, but is oxidized to a much more reactive nitrogen dioxide. The ‘nitrosative’ stress resulting from excessive production of toxic nitrogen oxides may lead to thiols S-nitrosylation or post-translational modification of cysteine residues in proteins. Those mechanisms of action cause functional disruption of proteins, mitochondrial dynamics, protein folding in the ER or transmission of signals contributing to synaptic damage, neurodegeneration and even cell death. 53 –55
In this study, results showed a large decrease in the nitrosylated proteins concentrations in animals from the exposed groups that were necropsied 1–2 days after birth and 2 weeks after birth (by 100% and 133%, respectively), compared to the antenatal group. Within a group of animals that were exposed to tobacco smoke during pregnancy, protein S-nitrosylation levels determined 2 weeks after birth were about 52% lower than in the exposed but non-gravid control group. Probably the increases reported in TEAC and UA concentrations in the postnatal groups, particularly in those exposed to cigarette smoke, could be an adaptive response to the described redox imbalance and enhanced free radical production. This antioxidant mobilization of lactating rats (up to 14 days after exposure) was reflected in lower protein S-nitrosylation.
Bodily defence mechanisms activated during the experiment in response to cigarette smoke proved highly effective in the case of proteins protection. Similarly, this can be seen in the analysis of lipid peroxidation among animals exposed to smoke at the moment of birth. The groups analysed for concentrations of TBARS the day before and 1–2 days after birth showed a significant inhibition of peroxidation (expressed by more than two times lower TBARs concentration, compared to non-pregnant animals at the same time intervals). However, this situation is quite different in animal subjected to autopsy 14 days after birth that were exposed to smoke during pregnancy. TBARS concentration in this case was 135% higher than in the exposed animals from the control group. Moreover, the level of peroxidation was almost 200% higher than in the non-exposed group.
Among groups exposed to tobacco smoke, the TBARS levels were 570% and 307% higher in plasma of animals subjected to the autopsy 2 weeks after delivery than those subjected to autopsy 1 day before and 1–2 days after delivery, respectively. This fact may be connected with oxytocin level during this period. The concentration of oxytocin 2 weeks after birth was significantly reduced in exposed animals as compared to the values marked on the day before and 1–2 days after birth (by almost 41% and 44%, respectively). This may confirm antioxidant properties of oxytocin during prenatal period.
On the other hand, exposure to tobacco smoke is associated with gradual formation of free radicals during fatty acid oxidation, as a chain reaction. The modification and lipid structures damage can be process extended and deepening over time process. Malondialdehyde, which is a product of polyunsaturated fatty acids degradation detected during a TBARs analysis, represents the subsequent effects of lipid peroxidation associated with cell damage and decay. 56 The effects on lipid membranes degradation after tobacco smoke exposure may therefore appear after a certain time, which will contribute to additional deactivation of antioxidant defence mechanisms and manifest, among others, as the lower oxytocin concentration, even in the absence of a stimulus (e.g. tobacco smoke).
Conclusions
Cotinine (nicotine metabolite) was an adequate tool in the assessment of exposure of experimental and control groups. The most distinct effect of exposure was lower median body mass of rat foetuses and pups in groups of dams that inhaled tobacco smoke. This fact confirms the need to refrain from smoking in order to provide proper development of the foetus and newborn.
Oxidative stress, naturally increased in the gestation period, can be additionally intensified by tobacco smoke. Consequently, during pregnancy, adaptive changes may occur in the properties of plasmatic antioxidant barriers.
Moreover, by disturbance of oxidoreductive balance, tobacco smoking affects the endocrine system of the female, which may result in oxytocin fluctuations. This was manifested especially during lactation period in this study. Therefore, women who smoke during pregnancy may breastfeed their children less frequently and for a shorter period after delivery.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research project was developed and carried out in cooperation with School of Medicine at Loma Linda University (Loma Linda, California, USA) and Laboratory of Environmental Research, Department of Toxicology and Department of Neonatal Infections at Poznan University of Medical Sciences (Poznan, Poland).
