Abstract
Tetrabromobisphenol A (TBBPA) is used to protect a wide range of electrical and electronic equipment, consumer electronics and office and communication equipment from catching fire. TBBPA reacts covalently with other monomers becoming an integral part of the cross-linked molecular structure. This study was conducted to evaluate the subchronic toxicity of TBBPA administered by gavage daily for 13 weeks at 0, 100, 300, and 1000 mg/kg/day in male and female CD® rats. A 6-week post-treatment control and 1000 mg/kg/day recovery groups were included. TBBPA exerted no marked effect on the rate of mortality, clinical signs, body or organ weights, feed consumption, histopathology, urinalysis, ophthalmology, and neurological outcomes in a functional observation battery, motor activity, serum thyroid stimulating hormone, serum triiodothyronine, or other serum chemistries. Although differences were observed for bilirubin and alkaline phosphatase, the observed alterations were within the normal range and thus were neither biologically or toxicologically meaningful. The single thyroid-related parameter affected by TBBPA was a reduction in serum thyroxine levels, but the decrease was not of sufficient magnitude to induce other more sensitive indicators of thyroid perturbation. The No Observed Adverse Effect Level was at least 1000 mg/kg/day, the highest dose tested. Based on an upper bound aggregate exposure for adults estimated by the European Union, the margin of exposure is approximately 5000, suggesting that, for the endpoints examined in this study, exposure to TBBPA presents a reasonable certainty of no harm.
Introduction
Tetrabromobisphenol A (TBBPA), the most widely produced brominated flame retardant, is used to protect a wide range of electrical and electronic equipment from catching fire. The main application is in printed circuit boards or laminates, where it contributes to the fire safety of those items requiring flame resistant 4 protection (over 95% of printed circuit boards). TBBPA also is used in consumer electronics (audio & video systems, vacuum cleaners, and washing machines) and office and communication equipment (cell phones, copiers, computers, printers, and radios), as well as in wind turbines. As a ‘reactive’ flame retardant in printed circuit boards, it reacts with other monomers becoming an integral part of the cross-linked molecular structure of the circuit boards. TBBPA is also used as an ‘additive flame retardant’, mainly in acrylonitrile–butadiene–styrene plastics, as well as in light, rigid, and molded products such as electrical housings or piping. The developmental and reproductive and neurobehavioral toxicology of TBBPA has been extensively studied 1,2 and its general toxicity was recently comprehensively reviewed by Colnot et al. 3 The results reported in the present article were summarized by Colnot et al., but they have not been previously reported in detail.
This study was conducted to evaluate the subchronic toxicity of TBBPA administered by gavage daily for 13 weeks at 0, 100, 300, and 1000 mg/kg/day at a constant volume of 5 mL/kg/day in male and female CD® rats (10 animals/group). A 6-week post-treatment period recovery group was included in the control and high-dose groups (5 animals/group). This study had the same sponsor as the two-generation reproduction, fertility, and developmental neurobehavioral study reported by Cope et al. 1 and was conducted during the same time frame.
Methods
Test article
TBBPA was a composite of three commercial lots produced by Albemarle Corporation (Baton Rouge, Los Angeles, USA), Chemtura Corporation (Lafayette, Indiana, USA), and Israeli Chemical (Tel Aviv, Israel). Purity testing via high-performance liquid chromatography indicated that all three samples had the same purity (approximately 99%). The test article was used as received and no adjustment was made for purity. The vehicle, corn oil, was received in three lots from Spectrum Chemical Manufacturing Corporation (New Brunswick, New Jersey, USA). Other studies have shown that TBBPA is rapidly absorbed from the gastrointestinal tract when dosed in corn oil 4 or peanut oil. 5
Dosing formulations
Analytical work was conducted by KAR Laboratories, Inc. (Kalamazoo, Michigan, USA), in compliance with good laboratory practices. Homogeneity was assessed on test article suspensions at the low and high concentrations used during the first week of the study (20 and 200 mg/mL, respectively). Six samples (two top, two middle, and two bottom) were collected from each mix while stirring to assess for homogeneity. Refrigerated formulations of TBBPA in corn oil were determined to be stable for at least 14 days. Dosing formulations were analyzed for concentration and adjusted throughout the study to ensure the correct concentrations based on body weight.
Experimental design
Animals
The study was conducted in compliance with the U.S. Department of Agriculture’s Animal Welfare Act (9 CFR Parts 1, 2, and 3). 6 The Guide for the Care and Use of Laboratory Animals was followed. 7
The study protocol met or exceeded study guidelines by the U. S. Environmental Protection Agency (Health Effects Test Guidelines OPPTS 870.3100: 90-Day Oral Toxicity in Rodents) and the Organisation for Economic Co-operation and Development (test no. 408: Repeated Dose 90-day Oral Toxicity Study in Rodents). 8,9 The study protocol was approved by the Institutional Animal Care and Use Committee.
Male and female CD® (Crl: CD (SD) IGS BR) rats (6-weeks old on arrival; body weight of males 128–156 g and 110–151 g for females) from Charles River Laboratories (Portage, Michigan, USA) were used. During a 15-day acclimation period, rats were observed daily for adverse clinical signs and given a detailed clinical examination prior to selection for the study. Rats considered suitable were weighed prior to treatment and randomized, by sex, into treatment groups using a standard, by weight, block randomization procedure. The body weight range of individual animal in each group did not exceed ±20% of the mean for the group.
Rats were housed individually in suspended, stainless steel wire mesh-type cages in a room where temperature and relative humidity were maintained between 19.4°C and 23.9°C and 28–70%, respectively. Fluorescent lighting provided illumination for approximately 12 h per day. Diet (meal lab Diet® Certified Rodent Diet #5002, PMI Nutrition International, Inc. (St Louis, Missouri, USA)) and tap water were available ad libitum.
Dose levels and duration of treatment
Doses of 0, 100, 300, and 1000 mg/kg/day (gavage) were based on data from previous studies 1 that indicated that the limit dose of 1000 mg/kg/day would not produce excessive toxicity. 10 Animals were treated for 13 consecutive weeks (once/day, 7 days/week). Recovery animals in the control and high-dose groups were not treated during a 6-week post-treatment recovery period.
Body weight
Body weights were measured and recorded within 3 days of arrival (day 15), at least once prior to randomization (day 1), and weekly during the study, through the day of necropsy.
Feed and water consumption
Feed consumption was measured and recorded weekly. Water consumption was not measured.
Clinical pathology: Hematology, clinical chemistry, and urinalysis
Animals had free access to water but were allowed to fast overnight prior to sample collection. Blood samples (approximately 3 mL) were collected via the orbital sinus after carbon dioxide (CO2) anesthesia from all main study rats at termination, but not at the beginning of the study. Blood was also collected from high dose and control animals following the 6-week recovery period but not at the beginning of the study.
At termination of the main group and at 4 weeks into the 6-week recovery period (but not at week 6 of recovery), fasting animals (control and 1000 mg/kg/day groups) were placed in stainless steel metabolism cages for approximately 16 h to collect urine for urinalysis (volume, specific gravity, and pH).
Thyroid hormone analyses
Blood (approximately 2 mL/animal) was collected from all study animals after 4.5 weeks (33 days) of treatment, after 13 weeks, and following the 6-week recovery period for analysis of thyroid stimulating hormone (TSH), 3,5,3′-triiodothyronine (T3), and thyroxine or 3,5,3′5′-tetraiodothyronine (T4) levels. Blood was collected from the orbital sinus into vials without anticoagulant and the serum collected and stored frozen (approximately −20°C) until sent frozen via overnight carrier to Lab Corp, Research Triangle Park (North Carolina, USA), for analysis via electrochemical luminescence immunoassay.
In-life examinations
All animals were observed at least twice a day, 7 days a week, for morbidity, mortality, signs of injury, and availability of feed and water. Mortality or other signs of toxicity were recorded.
A detailed clinical examination of each study animal was conducted once prior to the start of treatment and once during each study week. The examinations included, but were not limited to, evaluation of the skin, fur, eyes, ears, nose, oral cavity, thorax, abdomen, external genitalia, limbs and feet, mucous membranes, occurrence of secretions, excretions, and autonomic activity (e.g. lacrimation, piloerection, and pupil size) as well as evaluation of respiration and central nervous system effects including tremors and convulsions.
Assessment of neurotoxicity
Examinations were conducted according to the Standard Operating Procedure (SOP) of the Testing Facility titled ‘Abbreviated Functional Observation Battery to Meet Office of Prevention, Pesticides and Toxic Substances Guidelines’. This assessment consisted of (1) neurobehavioral examinations conducted once prior to the start of treatment, then weekly throughout the study, and weekly for the first 4 weeks of the 6-week recovery period; (2) a functional observation battery (FOB) prior to the start of the study and at week 12; and (3) a motor activity evaluation at study week 12.
The neurobehavioral examination occurred outside of the home cage and was conducted according to the testing facility’s SOP that was designed to meet appropriate guidelines of the U.S. Environmental Protection Agency’s Office of Prevention, Pesticides and Toxic Substances. Observations included changes in the level of activity, gait and posture, reactivity to handling or sensory stimuli, altered grip strength, and stereotypes or bizarre behavior (e.g. self-mutilation and walking backwards). Modifiers were included when necessary to describe the location, size, shape, color, or other characteristics.
The categories evaluated as part of the FOB are summarized in Table 1.
Categories evaluated as part of the FOB.

FOB: functional observation battery.
Motor activity was measured in male and female rats from all dose groups during study week 12 in a Digiscan® Animal Activity Monitor, Omnitech Electronics, Inc. (Columbus, Ohio, USA). Recovery animals were not evaluated. The activity chamber was 16 × 16 × 12 inches and was equipped with a computer analyzer. Activity was recorded for three consecutive 10-min intervals allowing for examination of both exploratory and acclimation activity levels. Movement was recorded by 16 photocell sensors; each in one vertical and two horizontal planes. Two planes were used to record horizontal activity and intersected at right angles to form a grid pattern. The third plane was located above the first two and recorded vertical activity. The activity chambers were calibrated just prior to each testing interval. The overhead lights in the test room were turned off and the animals were acclimated in their home cages for 10 min before being transferred to the activity chambers. The following activities were recorded: (1) horizontal, (2) vertical, (3) total distance (cm), and (4) stereotypic activity.
Postmortem evaluations
Macroscopic
At termination of the study period, all animals scheduled for necropsy were euthanized with CO2 and a postmortem examination overseen by a board certified veterinary pathologist was performed. Animals found dead were stored at 4°C until necropsied.
Following euthanasia, each rat was exsanguinated by cutting the dorsal aorta and examined carefully for external abnormalities, including masses. The skin was reflected from a ventral midline incision taking care not to open the thoracic cavity. Subcutaneous abnormalities were identified and correlated with antemortem findings when appropriate.
The abdominal, thoracic, and cranial cavities were examined and the organs were removed, examined, and where required, fixed in 10% neutral buffered formalin. The pituitary was fixed in situ. Ten percent neutral buffered formalin was infused into the lung via the trachea and into the urinary bladder.
Organs were trimmed of fat and connective tissue and weighed. Paired organs were weighed together. The thyroid/parathyroid gland and pituitary were weighed following fixation; a combined weight of the thyroid gland with bilateral parathyroid glands was obtained.
Organ weight ratios were calculated (relative to body and brain weights).
Microscopic
Organs and tissues including any gross lesions were fixed in 10% neutral buffered formalin. Hematoxylin–eosin-stained samples from control and 1000 mg/kg/day male and female animals were examined by a board-certified veterinary pathologist. The following tissues/organs were examined: adrenal glands (cortex and medulla), artery (aorta), bone (femur and sternum), bone marrow (femur and sternum), brain, esophagus, eye (including optic nerve and retina), epididymis, heart, kidney, large intestine (cecum, colon, and rectum), larynx, liver, lung, lymph node (mandibular, mediastinal, and mesenteric), nasal tissue, nerve (sciatic), pancreas, parathyroid gland, pharynx, pituitary gland, salivary gland (mandibular), skeletal muscle (biceps femoris), skin, small intestine (duodenum, ileum, and jejunum), spinal cord (cervical, lumbar, midthoracic, spleen, stomach (glandular and nonglandular), thymus gland, thyroid gland, teeth, trachea, and urinary bladder; females only: mammary gland, ovary, skeletal muscle (diaphragm), skin (subcutis), uterus (including cervix), and males only: prostate gland, seminal vesicle, and testis.
Statistical analysis
Group pairwise comparisons (all end points except FOB (except continuous data), leukocyte counts, and urinalysis)
For each specified end point and for all collection intervals, Levene’s test 11 was used to assess homogeneity of group variances. If the test were not significant (p > 0.01), Dunnett’s test 12 was used to compare each group receiving test article with the control group. If Levene’s test were significant (p < 0.01), comparisons with the control group were made using Welch’s test 13 with a Bonferroni correction. Results of all pair-wise comparisons are reported at the 0.05 and 0.01 significance levels. All end points were analyzed using two-tailed tests unless indicated otherwise.
Log transformation
Historical data indicate that leukocyte counts (total and differential) are not normally distributed; therefore, a log transformation was performed. The transformed data were then analyzed as described previously in the group pairwise comparisons.
Rank transformation
For each specified end point and for all collection intervals for urinalysis, a rank transformation was performed on these data. The transformed data were then analyzed using Dunnett’s test to compare each treatment group with the control group. All end points were analyzed using two-tailed tests.
Results
Mortality and clinical findings
Six females (two control animals and four rats in the 1000 mg/kg/day group) died or were euthanized in extremis. The mortality/morbidity was related to dosing and was not treatment related. On the first day of treatment, one male in the 300 mg/kg/day group was euthanized due to excessive weight loss; this animal was replaced.
No mortality occurred as a result of treatment with TBBPA. The low occurrence of salivation in the 300 and 1000 mg/kg/day groups, possibly related to residual test article in the oral cavity, was not considered toxicologically meaningful because salivation was not seen in the 1000 mg/kg/day animals during the recovery period. Other clinical findings in the TBBPA-treated groups occurred at low incidence and were considered unrelated to treatment because they were spurious and not dose related.
Neurobehavioral findings
No adverse effects of treatment with TBBPA were evident in the weekly neurobehavioral evaluations. During the recovery period, a slight increase in grid activity counts in the 1000 mg/kg/day males in comparison to controls was observed. This was considered incidental and not toxicologically meaningful since this effect was not evident during the treatment period and not present in females.
Functional observational battery
No adverse effect of treatment with TBBPA was evident from the FOB evaluations.
Locomotor activity
No changes were observed in females at any time or dose. In the case of males, the only statistically significant changes occurred in the 1000 mg/kg/day males (Table 2). Total distance traveled for these males was statistically lower than controls over the 0–10-min interval and over the entire 0–30-min trial. There was considerable variability in this parameter as evident from the large standard deviations in all groups (control and treated) at each time interval and the lack of clear dose–response relationships in any of the intervals. In the absence of a similar response on total activity for the other two evaluation intervals (10–20 and 20–30 min), this change was considered spurious and not toxicologically meaningful. Vertical activity in the 1000 mg/kg/day males was statistically lower than controls during the 10–20 min interval and over the entire 0–30-min trial (data not shown). This change in vertical activity in the 1000 mg/kg/day males for this period, although statistically significant, was not considered toxicologically meaningful in the absence of a similar response for the remaining trial intervals (i.e. 0–10 and 20–30 min). As with the total distance traveled, the data were highly variable and no clear dose–response relationships were evident in any of the intervals.
Total locomotor activity in male rats.

aSignificantly different from control (p < 0.05).
In the absence of an effect on locomotor activity in the main study animals, these evaluations were not conducted for the recovery animals.
Body weight and feed consumption
TBBPA had no effect on body weights or feed consumption.
Ophthalmoscopic examinations
Ophthalmoscopic examination revealed no TBBPA treatment-related effects.
Clinical pathology
Clinical chemistry
With the exception of total bilirubin and serum alkaline phosphatase (ALP), all clinical chemistry values were within normal ranges for this species and not different from concurrent control values. After 13 weeks of dosing, total bilirubin values (mg/dL) were statistically higher than control mean values (males: 0.14 ± 0.05; females: 0.13 ± 0.05) in males in the 1000 mg/kg/day dose (0.34 ± 0.024; p < 0.01) and in females in the 300 (0.19 ± 0.03; p < 0.05) and 1000 mg/kg/day (0.2 ± 0.06; p < 0.01) groups. Mean serum ALP levels (U/L) after 90 days of dosing in the female 1000 mg/kg/day (98.9 ± 49.47) group were statistically higher than that of the control mean values (58.4 ± 28.46; p < 0.05; females: 47.7 ± 8.15 in the 100 mg/kg/day group; 62.6 ± 20.92 in the 300 mg/kg/day group). Serum bilirubin and ALP levels in control and treated groups of both sexes were comparable after the recovery period. In the absence of any corresponding elevation in liver weights, liver weight/body weight, or liver weight/brain weight, as well as no histopathological changes, these clinical chemistry elevations were not considered to be toxicologically significant.
Table 3 (total bilirubin) and Table 4 (ALP) summarize the clinical chemistry-related parameters in male and female rats.
Treatment-related clinical chemistry responses (total bilirubin) in rats.

aSignificantly different from control (p < 0.05).
bSignificantly different from control (p < 0.01).
cCompared to recovery controls.
Treatment-related clinical chemistry responses (Serum ALP) in rats.

aSignificantly different from control (p < 0.05).
bCompared to recovery controls.
Hematology
A few spurious results were seen, but overall, there were no hematological alterations attributed to TBBPA treatment. Platelet counts were slightly lower (20.9%) in males at 1000 mg/kg/day at termination; but not at the lower doses, at earlier time points (males: 1194.9 ± 173.66, 52 K/mm3 in the 100 mg/kg/day group; 1049.9 ± 206.95 52, K/mm3 in the 300 mg/kg/day group; and 944.2 ± 163.81 52, K/mm3 in the 1000 mg/kg/day group versus control 1141.3 ± 116., K/mm3), at recovery or in females at any time or dose. Therefore, the slight variation in platelet counts at this single time point in male rats was considered not to be the result of treatment.
Table 5 summarizes the hematology-related (platelet counts) parameters in male and female rats.
Treatment-related hematology responses in rats.

aSignificantly different from control (p < 0.05).
bCompared to recovery controls.
Serum thyroid hormone values
No treatment-related changes in mean plasma T3 or TSH were seen at any dose or time in either males or females. In contrast, mean plasma T4 levels were decreased at all dose levels in both males and females at termination of the study (Figure 1 for males; Figure 2 for females). Mean T4 levels (ng/dL) were statistically lower than control (day 33: 4.96 ± 0.84; terminal: 5.09 ± 0.80) in the 100 (day 33: 3.66 ± 0.88; terminal: 3.27 ± 0.67), 300 (day 33: 3.42 ± 0.71; terminal: 2.61 ± 0.87), and 1000 (day 33: 3.39 ± 0.55; terminal: 3.09 ± 0.91) mg/kg/day male dose groups at days 33 and 90 (p < 0.01), respectively. Mean T4 levels were also statistically lower than control values (4.27 ± 0.96) in females in the 100 (3.31 ± 1.08), 300 (3.24 ± 0.85), and 1000 (3.33 ± 0.84) mg/kg/day dose groups at day 33 (p < 0.01). Mean T4 levels in all female dose groups were unchanged relative to the control mean values at day 90. Following the recovery period, T4 levels in males at 1000 mg/kg recovered to levels comparable to controls, whereas the T4 levels in females at 1000 mg/kg were lower relative to controls, although they were comparable to controls at the 90-day time point.
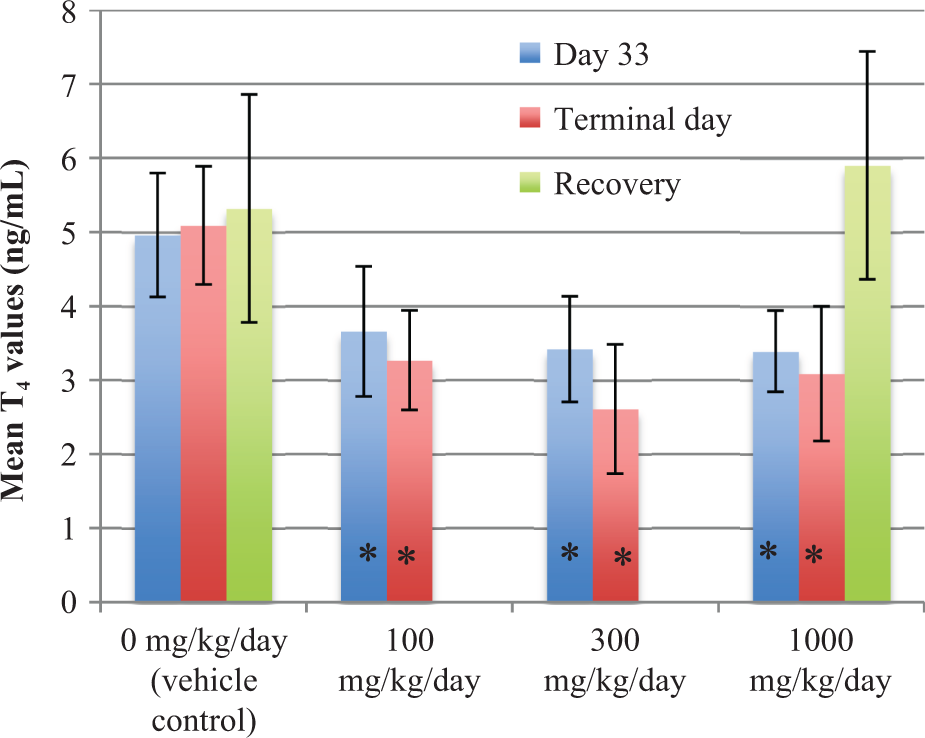
Mean T4 levels in male animals (+/− SD). *p < 0.01: Significantly different from control.
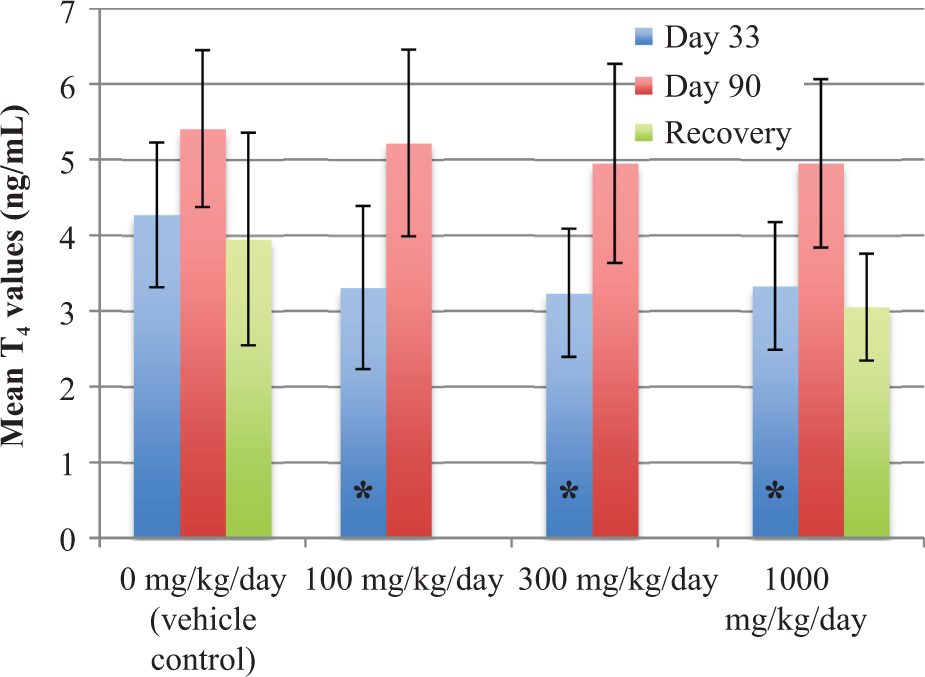
Mean T4 levels in female animals (+/− SD). *p < 0.01: Significantly different from control.
Table 6 (male rats) and Table 7 (female rats) summarize the thyroid-related parameters.
Treatment-related thyroid responses in male rats.

NA: not applicable; ND: not determined per study protocol; WNL: all animals within normal limit.
aNo statistical significance observed.
bSignificantly different from control (p < 0.01).
cCompared to recovery control.
Treatment-related thyroid responses in female rats.

NA: not applicable; ND: not determined per study protocol; WNL: all animals within normal limit.
aNo statistical significance observed.
bSignificantly different from control (p < 0.05).
cCompared to recovery control.
Urinalysis
There were no alterations in the urinalysis.
Postmortem study evaluations
Organ weights
No statistically significant, treatment-related changes were noted in females either at the end of dosing or following the recovery period. A statistically significant decrease in absolute mean spleen weights occurred in the 300 mg/kg/day (0.63 ± 0.117 g) and 1000 mg/kg/day (0.61 ± 0.059 g) males compared to controls (0.74 ± 0.102 g) and in mean spleen/brain wt % × 10−1 (3.08 ± 0.373 g at 1000 mg/kg/day compared to controls 3.64 ± 0.447 g) at the end of treatment (13 weeks). However, because no microscopic differences were noted in spleens between the vehicle control groups and the 1000 mg/kg/day groups, these differences were considered to be spurious and unrelated to TBBPA treatment.
The only other statistically significant organ weight change following 13 weeks of dosing was an increase in mean epididymis/body weight in the 300 mg/kg/day males. This difference was also considered spurious as it was not dose related and the mean absolute epididymis weights across all the groups were essentially the same. There was a statistically significant elevation in mean kidney/brain weight at 1000 mg/kg/day in the male recovery group that also was not considered to be treatment-related.
Macroscopic
There were no test article-related macroscopic observations noted in either sex at terminal or recovery necropsy.
Microscopic
There were no test article-related microscopic findings either in male or female animals at the high dose (1000 mg/kg/day) at terminal euthanasia. Thus, tissues from the mid and low dose terminal euthanasia animals and from recovery animals were not evaluated. Microscopic examination of organs from recovery animals was not conducted as terminal euthanasia did not indicate any TBBPA treatment-related effects or target organs. Gross lesions from recovery animals were examined microscopically and indicated changes typical of rats of this strain and age and not treatment-related.
Discussion
The only TBBPA treatment-related response was a non-dose dependent decrease in serum T4, evident in both males and females at all dose levels. No microscopic changes were seen in the thyroid nor did treatment affect thyroid weight or the serum levels of the biologically active T3. The reduction in serum T4 levels was not accompanied by evidence of other toxicity or adverse effects. Functionally, the decrease in the pro-hormone T4 is a decrease in the reserve pool of the pro-hormone rather than a decrease in the thyroid active T3. 14,15 Moreover, TSH levels were not significantly affected by TBBPA treatment. Cope et al. 1 described a multigenerational reproductive study in CD (Crl: CD (Sprague-Dawley) IGS BR) rats in which TBBPA was administered by gavage at 0, 10, 100, or 1000 mg/kg body weight/day. It is of interest that TBBPA did not alter TSH levels or thyroid histopathology in the parental and F1 generation but decreased T3 levels in the 1000 mg/kg males in the parental generation and T4 levels in the 100 mg/kg/day males and in the 1000mg/kg/day males and females in the parental generation, and in the 100 mg/kg/day and 1000 mg/kg/day males and females in the F1 generation. 1
In a National Toxicology Program 3-month study, TBBPA at 500 and 1000 mg/kg reduced T4 levels in F344 rats with no change in T3, TSH, or thyroid histopathology. TBBPA did not markedly alter thyroid hormone levels or tissue histopathology in mice at 3 months or 2 years. 16 In the subsequent 2-year study in Han rats, TBBPA did not affect thyroid histopathology. 16 This is important because TSH levels and thyroid gland histopathology are considered to be more sensitive indicators of thyroid status than T3 or T4 serum concentrations. 17
The observation of a decrease in serum T4 is consistent with a report showing TBBPA to displace T4 competitively from human transthyretin (TTR), a major serum T4-binding protein, in vitro. 18 Under this scenario, the portion of serum T4 displaced from its binding site may be available for metabolism and elimination, thereby leading to a decrease in circulating levels of serum T4. However, despite the high in vitro TBBPA-TTR binding affinity (10.6 times that of T4), no effects were noted on plasma total T4 or free T4 in pregnant rats and fetuses or on maternal total T4 levels in dams treated with TBBPA. 19 The authors suggested that this may be due to the rapid debromination of TBBPA in vivo and failure to an effective in vivo tissue concentration comparable to the shown to competitively displace T4 from TTR. This may be expected as data show that following an oral dose of 5 mg/kg body weight [14C]-TBBPA from day 10 to day 16 of gestation, only 0.83% of the administered dose was detected in maternal animals and 0.34% in the fetuses.
An alternative explanation for the observed decrease in T4 in our study may be the TBBPA-mediated induction of glucuronidation of T4 and subsequent elimination.
The induction of thyroid hormone metabolizing enzymes in the rat is a complex, yet to be fully understood phenomenon. Experiments have indicated that elevated hepatic T4 glucuronidation may not mediate the increase in TSH observed after enzyme induction. Hood and Klaassen 20 observed that treatment with phenobarbital, pregnenolone-16α–carbonitrile, 3-methylcholanthrene, and Aroclor 1254 (commercial polychlorinated biphenyl (PCB) mixture) enhanced glucuronidation of T4 and decreased serum T4. However, only phenobarbital and pregnenolone-16α–carbonitrile increased both T3 glucuronidation and TSH levels. In contrast, 3-methylcholanthrene and PCB did not increase T3 glucuronidation or TSH. 20,21 Although TBBPA may be acting like 3-methylcholanthrene or PCB, such enzyme induction is usually reflected in hepatic hypertrophy and increased liver weights, neither of which was observed in this study. It is possible that the potency of TBBPA with regard to enzyme induction is too low to result in overt hepatic changes.
Regardless of the reason for the decline in T4, the decrease was not of sufficient magnitude to alter mean serum TSH or T3 levels, thyroid histopathology, thyroid weight, or other parameters more indicative of thyroid pathology (e.g. body weight, etc.). Thus, the decrease in serum T4 levels is not considered adverse, especially when the World Health Organization International Programme on Chemical Safety definition of adverse effect is considered: Change in the morphology, physiology, growth, development, reproduction, or life span of an organism, system, or (sub)population that results in an impairment of functional capacity, an impairment of the capacity to compensate for additional stress, or an increase in susceptibility to other influences.
22
Further, understanding the relationship of the observations in the present rat study to potential effects in humans requires consideration of the marked differences in how thyroid hormones are transported. For example, the plasma T4 half-life in rats is 12–24 h. 17 This short half-life of T4 in the rat is due to its transport by relatively low affinity TTR (predominant thyroid hormone binding protein in the rat), and thus this species is sensitive to perturbations in T4 levels. In contrast, in humans, although TTR is present, circulating T4 is bound primarily to thyroxin binding globulin, a high affinity binding protein not present in rodents. As a result, the half-life of T4 in humans is 5–9 days, rendering it less responsive to agents that might act to lower its circulating level. 17 Thus, TBBPA is unlikely to have any effect on T4 levels in humans. To the best of our knowledge, no attempt has been made to correlate serum thyroid hormone levels with exposure to TBBPA in humans.
The neurotoxicity findings are consistent with the observations of a critical review of currently available TBBPA neurotoxicity data by Williams and DeSesso 2 and the recent report from Cope et al. 1 showing no adverse effects in rats exposed to up to and including 1000 mg/kg/day (the highest dose tested) on neurodevelopment, neuromotor functions, learning, memory, and neurobehavioral endpoints.
Given the above, the no observed adverse effect level (NOAEL) in this rat 90-day oral toxicity study with TBBPA was at least 1000 mg/kg/day, the highest dose tested. No effects on mortality, clinical signs, body or organ weights, histopathology, urinalysis, ophthalmology, FOB, motor activity, serum TSH, serum T3, or serum chemistries were observed. A small but statistically significant difference was observed for bilirubin and ALP but neither was considered to be biologically or toxicologically meaningful or adverse. Serum T4 levels were decreased in treated animals, but the decrease was not of sufficient magnitude to induce adverse effects.
Conclusions
TBBPA had no effect on mortality, clinical signs, body or organ weights, histopathology, urinalysis, ophthalmology, FOB, motor activity, serum TSH, serum T3, or other serum chemistries. Differences were observed for bilirubin and ALP, but neither was biologically or toxicologically meaningful. The single thyroid-related parameter affected by TBBPA was serum T4 levels, but the decrease was not of sufficient magnitude to induce other more sensitive indicators of thyroid perturbation.
The NOAEL was at least 1000 mg/kg/day, the highest dose tested. Considered with the upper bound aggregate exposure for adults estimated by the European Union, the margin of exposure for TBBPA is greater than 5000, suggesting, for the endpoints examined in this study, that this exposure presents a reasonable certainty of no harm.
Footnotes
Acknowledgment
The authors thank the members of the American Chemistry Council’s North American Flame Retardant Alliance, Albemarle Corporation, Chemtura and ICL-IP, for financial support in preparing this manuscript as well as Study Director Raymond E Schroeder (MPI Research, Inc.).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The American Chemistry Council’s North American Flame Retardant Alliance (NAFRA) funded the study and preparation of the manuscript. Both TGO and AWH serve on NAFRA’s Science Advisory Council and receive compensation for doing so. TGO does occasional paid scientific analysis and legislative testimony on behalf of ACC. WD is paid by Science Strategies, LLC, for her time on the project. AWH serves as editor for the Americas for Human & Experimental Toxicology.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The American Chemistry Council’s North American Flame Retardant Alliance (NAFRA) commissioned the study, reviewed, and approved the study protocol and funded the study that was conducted at MPI Research, Mattawan, Michigan, USA. The authors independently interpreted the data and wrote the manuscript that was reviewed by NAFRA. NAFRA did not alter the authors’ conclusions.
