Abstract
To provide support for future pharmacology and preclinical studies, we have established a stable nonhuman primate animal model to demonstrate the histopathological changes in the gastrointestinal tract following gamma ray irradiation. In this study, 12 healthy rhesus monkeys were divided into 2 groups (control and radiation groups). Animals in the radiation group were exposed to gamma rays (cobalt 60 source) at a dose level of 6.5 Gy total body irradiation bilaterally (i.e. 3.25 Gy on each side). Control animals were sham exposed using identical procedures. After a 5-day in-life observation period, gastrointestinal tract tissues (esophagus, stomach, duodenum, jejunum, ileum, colon, and rectum) were collected and fixed in 10% neutral-buffered formalin for subsequent hematoxylin and eosin and 5-bromo-2-deoxyuridine (BrdU) immunohistochemistry processing. The results showed that the esophagus was undergoing degeneration without obvious inflammatory changes, while the stomach and duodenum exhibited both degeneration and inflammation. From the jejunum to the rectum, late-stage inflammation with glandular regeneration, as well as a high-level BrdU labeling index, was present.
Introduction
Radiation, based on the spectrum of radiant energy, can be divided into two forms, nonionizing and ionizing. Ionizing radiation is far more harmful to living organisms due to its powerful penetrating ability and impact on double-stranded DNA, activating a number of DNA damage response and repair signaling cascades that control cell cycle arrest, DNA repair, and the cell’s fate. 1 Because of these characteristics, ionizing radiation has been considered an effective and commonly employed treatment (radiotherapy) in the management of more than half of human malignancies and also forms a major part of the treatment for about 40% of cured patients. 1,2
Earlier diagnosis may contribute to successful radiotherapy application. With the development of early detection techniques, such as gene analysis, early stages of tumors become detectable, which directly increases the identification of patients suitable for radiotherapy treatment. During treatment, not only target cells but also normal cells adjacent to the irradiated region will be inevitably affected by the radiation exposure and further bystander effects may occur. 3 In order to control and eliminate tumor cells, many patients are subjected to an increased threat of radiation injury. To solve this problem, different kinds of radioprotectors have been studied and reported. 4 –7 Among them, synthetic analogs such as amifostine, cysteine, cysteamine, and thiol compounds are well-known for their radioprotective activity, 8,9 but they were also found to induce serious side effects including nausea, vomiting, or even epidermal necrosis. 10,11 Unfortunately, to date, there is no radioprotector that can fulfill all criteria including high-level protective efficiency, acceptable route of administration, acceptable toxicity profile, acceptable stability profile, and compatibility with the wide range of other drugs. 12
Research into the possible pathogenetic mechanisms of radiation injury include oxidative stress-involved 13,14 pro-inflammatory cytokines, 15 DNA damage, 16 and dysregulated microRNAs. 17 Since more and more candidate agents are awaiting pharmacodynamic and preclinical studies to move forward to clinical trials, a reliable animal model is needed. The majority of relevant studies reported were conducted in an uncontrolled environment, and we could not reproduce the reported severity changes. Therefore, in order to control the quality and stability of data, this study was conducted in compliance with the good laboratory practice (GLP) regulations of US Food and Drug Administration (FDA), the China Food and Drug Administration (CFDA), and the Organization for Economic Co-operation and Development (OECD), aiming to establish a stable nonhuman primate (NHP) animal model and provide reliable histopathological support for subsequent drug screening and preclinical evaluation. In this study, we focused on the gastrointestinal (GI) tract which had been reported as one of the most important target sites (due to its rapid cellular turnover), where abnormal changes could be easily identified after radiation exposure. 18 We also employed 5-bromo-2-deoxyuridine (BrdU), a thymidine analog, with the capability of incorporating the DNA of S-phase cells, to identify and evaluate the proliferative activity of the epithelium in different segments of GI tract.
Materials and methods
Animals and treatment
Twelve healthy rhesus monkeys (aged 4–5 years and weighing 4–5 kg) from Sichuan Greenhouse Biotech Co. Ltd (Certificate No.: SYXK (Chuan)2014-192, China) were randomly divided into two groups (control and radiation group), each containing six animals (three males and three females). All animals were housed singly in stainless steel monkey cages (XHL-4, Fengshi, China; dimensions 1590 × 800 × 2080 mm3), which allows monkeys to see others but unable to fight with each other. The animal room environment was strictly controlled and maintained at 18–24°C, relative humidity at 30%–70%, 8–15 times/hour air change, and 12-h light/12-h /dark cycle change. During the study, animals were supplied with Teklad laboratory primate diet (Harlan Laboratories, Inc., Madison, Wisconsin, USA) containing ≥25% protein, ≥4% fat, carbohydrates, vitamins, minerals, and fiber supplements; also, fresh fruits (disinfected with 0.4% 84 disinfectant) were provided twice a day, and all animals were allowed free access to drinking reverse osmosis water (AWL-1001-M, Aquapro International Company LLC., Dover, Delaware, USA). The study lasted for 12 days including a 7-day acclimation period and a 5-day clinical observation period after radiation exposure. When the 7-day acclimation period was completed, animals in the irradiation group were administered pentobarbital sodium (25 mg/kg) and exposed to gamma ray (cobalt 60 (Co60) source) at the dose level of 6.5 Gy total body irradiation (TBI) bilaterally (3.25 Gy on each side), while control animals were sham dosed, following the same procedures without receiving radiation exposure. Two hours prior to euthanasia at the end of the 5-day observation period, all surviving animals were injected intravenously (IV) with 50 mg/kg BrdU (Cat#B5002-1G, Sigma, St Louis, Missouri, USA) dissolved in sterile 1× Dulbecco’s phosphate-buffered saline. Animals were euthanized with pentobarbital (50 mg/kg, IV) and thoroughly exsanguinated. This study was approved by the Institutional Animal Care and Use Committee (IACUC) of Sichuan University, and all treatment procedures were in compliance with the animal welfare principles from the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC).
Tissue sample collection
Sections from all portions of the GI tract were evaluated in this study. In order to ensure consistency for each tissue to be evaluated, and to eliminate the influence of different regional distribution of epithelium and/or glandular numbers among individual animals, we precisely defined the trimming location for each 5 cm segment: esophagus, 5–10 cm cranial from the cardiac sphincter; stomach, 4–9 cm caudal from the cardiac sphincter along the greater curvature; duodenum, 3–8 cm caudal from the pyloric sphincter; jejunum, 5 cm cranial from the midpoint of the small intestine; ileum, 2–7cm cranial from the ileocecal valve; colon, 5–10 cm caudal from the ileocecal valve; and rectum, 3–8cm cranial from the anus. All tissues were fixed in 10% neutral-buffered formalin for 48 h. Tissues were then processed and stained with hematoxylin and eosin (H&E) for microscopic evaluation.
BrdU immunohistochemistry
Serial sections for all tissues (3–4μm thickness) were prepared and affixed to adhesive slides (Superfrost/Plus, Fisher brand, Waltham, Massachusetts, USA). After deparaffinization with xylene and rehydration with alcohol, sections were treated with 10 µg/mL proteinase K (Lot#0036B047, Amresco, Solon, Ohio, USA) for 20 min at room temperature. Endogenous peroxidase was blocked by incubating the tissue sections in 3% hydrogen peroxide in methanol for 20 min at room temperature. Then sections were incubated with 3% bovine serum albumin (ZLI-9027, ZSGB-BIO, China) for 20 min at 37°C and incubated with mouse monoclonal anti-BrdU antibody (ZM-0013, ZSGB-BIO, China) overnight at 4°C (1:100 dilution). Subsequently, horseradish peroxidase (HRP)-conjugated goat anti-mouse immunoglobulin G/HRP(PV-6002, ZSGB-BIO, China) were employed to provide specific binding to the primary antibody for 20 min at room temperature. Color development was achieved by incubation with a 3,3ʹ-diaminobenzidine (DAB) substrate kit (ZLI-9032, ZSGB-BIO, China) containing 0.02% 3,3ʹ-diaminobenzidine tetrahydrochloride and 0.02% hydrogen peroxide. Finally, slides were counterstained with hematoxylin for 1 min, cleared with xylene, and mounted in neutral resin.
BrdU-labeled cell counting
For semi-quantity analysis of BrdU-positive–labeled cells, images from the whole 5-cm-long mucosa of all tissues were taken using a digital camera attached to the microscope (BX51, Olympus, Japan). Then, manual counts for both the total number of epithelial cells and the BrdU-positive–labeled epithelium were performed by a single pathologist for consistency, followed by a review by a second pathologist. The labeling index (LI) was calculated according to the following formula.
Statistical analysis
SPSS 13.0 was employed to perform the statistical analysis of the LI using the Student’s t-test, and significant difference level between control and irradiation group was set at 5% (p < 0.05). Data are presented as mean ± standard deviation (SD).
Result
Esophagus
In the esophagus (Figure 1), control animals exhibited intact-stratified squamous epithelium. In irradiated animals, cells were loosely arranged with enlarged nuclei and prominent nucleoli, and duplication of the basal layer in some areas suggested hypertrophy/hyperplasia rather than a degenerative change. BrdU immunohistochemistry (IHC) results showed that the positive reactions were mainly deposited in the basal layer of the epithelium. Fewer positive-labeled cells were identified in irradiated animals than in control animals. The LI of epithelial cells in the irradiated (4.06 ± 1.12) and control (4.38 ± 0.77) groups were within the normal range, and no statistical significance was observed (p > 0.05). Thus, the morphologic suggestion of hyperplasia was not collaborated by the BrDU data.

Esophagus, stratified squamous epithelium, note basal layer (bar = 100 μm). (a) Control animal, H&E staining, ×200. Cells were tightly arranged with small nuclei and basophilic cytoplasm. (b) Irradiated animal, H&E staining, ×200. Cells were arranged loosely with enlarged nuclei and prominent nucleoli (bold arrow), and the cytoplasm of the epithelial cells were relatively more basophilic than that in control animals. Additionally, note the isolated and condensed nuclei distributed in the mucosa represent apoptotic cells. (c) and (d) BrdU IHC, ×200. Note the brownish yellow BrdU-positive-labeled cells are sporadically distributed in the basal layers (bold arrow). H&E: hematoxylin and eosin; BrdU: 5-bromo-2-deoxyuridine; IHC: immunohistochemistry.
Stomach
In the stomach (Figure 2), control animals exhibited intact overlying mucous cells on the luminal surface with normal nuclear-to-cytoplasmic ratio (N:C) of approximately 1:2, with clear gastric pits and regular arranged fundic glands containing chief cells, parietal cells, and basophilic neuroendocrine cells. However, in irradiated animals, both the population and cytoplasmic content of overlying mucous cells were obviously decreased, and the N:C ratio was about 1:1. In addition, epithelial shedding, stenotic or even atretic gastric pits, dilated fundic glands, glandular cell hypertrophy with decreased eosinophilic cytoplasmic granules, and mononuclear cell infiltration in the mucosa could also be identified in these animals. In the deeper mucosa, basophilic neuroendocrine cells undergoing atrophy may contribute to further dysfunction or dystrophy of the more superficial glandular cells. BrdU IHC results showed that the positive reactions were mainly located in the upper 1/3 part of the mucosa, the proliferative zone, and the LI of epithelium cells in irradiation group (4.93 ± 1.62) was slightly higher than that in control group (3.07 ± 1.20), however, no statistical significance existed (p > 0.05).

Stomach, fundic gland (bar = 100 μm). (a) Control animal, H&E staining, ×200. Note tightly arranged overlying mucous cells with abundant cytoplasm and basal-lined nuclei (bold arrow) as well as clear gastric pit (thin arrow) and normal fundic gland. (b) Irradiated animal, H&E staining, ×200. Note decreased population and cytoplasmic contents of overlying mucous cells (bold arrow), stenotic/atretic gastric pits (thin arrow), dilated fundic glands and glandular cell degeneration, and mononuclear cell infiltration (asterisk). (c) and (d) BrdU IHC, ×200. Note the brownish yellow cells, BrdU-positive–labeled cells, sporadically distributed in the upper 1/3 part of the mucosa (bold arrow). H&E: hematoxylin and eosin; BrdU: 5-bromo-2-deoxyuridine; IHC: immunohistochemistry.
Duodenum
In duodenum (Figure 3), control animals exhibited intact overlying columnar epithelium with abundant cytoplasm, and the N:C ratio was less than 1:2. Villi were long and isolated without obvious inflammatory changes. Intestinal gland epithelia were regularly arranged with basal nuclei. In irradiated animals, villi were shortened and blunted with increased N:C ratio (about 1:1) of the lining columnar epithelium, with focal necrosis at the top of villi and moderate mononuclear cell infiltration in the lamina propria. Intestinal glands were regularly and tightly arranged, but the glandular cells showed a more basophilic appearance with enlarged nuclei and prominent nucleoli. No obvious abnormal morphological changes could be identified in Brunner’s glands. BrdU IHC results showed that the positive reactions mainly affected glandular cells, and no significant difference (p > 0.05) was noted between the LIs in control (16.97 ± 5.98) and irradiation group (17.82 ± 7.85).
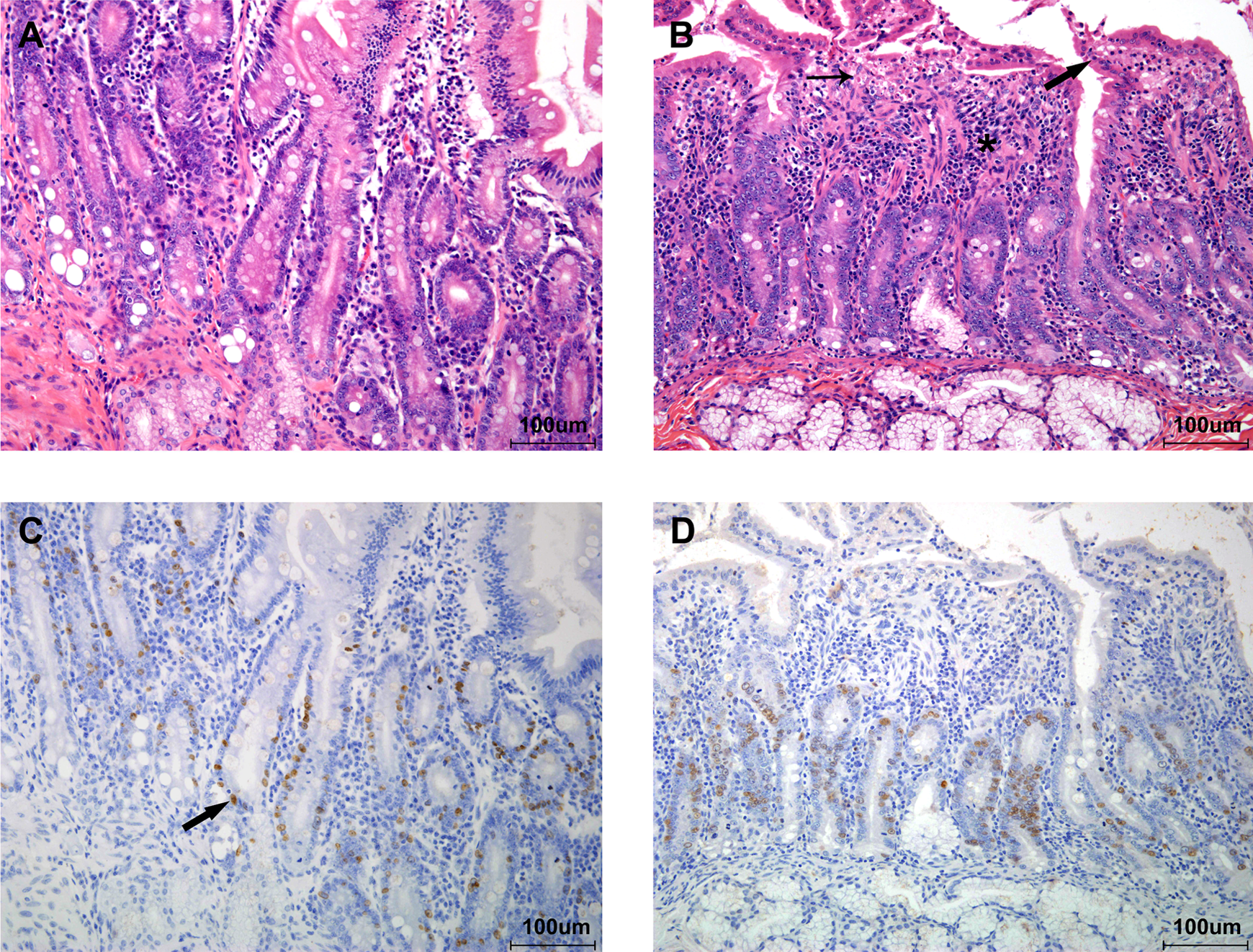
Duodenum, luminal mucosa (bar = 100 μm). (a) Control animal, H&E staining, ×200. Note intact mucosal epithelium with individual villi. (b) Irradiated animal, H&E staining, ×200. Note increased N:C ratio of the simple columnar epithelium (bold arrow), and the necrosis at the top of the villi which led to the shedding of lining epithelium in the same area (thin arrow). Also note the shortened and blunted villi associated with mononuclear cell infiltration in the lamina propria (asterisk). (c) and (d) BrdU IHC, ×200. Note the brownish yellow cells, BrdU-positive–labeled cells, extensively distributed in the mucosa (bold arrow). H&E: hematoxylin and eosin; BrdU: 5-bromo-2-deoxyuridine; IHC: immunohistochemistry; N:C: nuclear-to-cytoplasmic ratio.
Jejunum
In jejunum (Figure 4), control animals also had intact overlying columnar epithelium with low-level N:C ratio (less than 1:2), and villi were long and isolated with regularly and closely arranged intestinal glands. In irradiated animals, shortened villi as well as decreased mucosal thickness could be identified, and a decreased population of lining epithelium with an increased N:C ratio (about 1:1) was noted. Moreover, glandular cells were tightly arranged, with increased height of single cells, resulting in slightly enlarged intestinal glands. Slightly dilated central lacteals were also apparent. Similar to the duodenum, BrdU IHC-positive reactions in the jejunum mainly affected glandular cells; however, the LI of glandular cells in irradiation group (29.41 ± 8.22) was significantly higher (p < 0.01) than that in control group (11.01 ± 3.63), indicating increased proliferative activity. However, based on the relatively normal glandular structure with a single layer of cells, the correct diagnosis is regeneration.

Jejunum, luminal mucosa (bar = 100 μm). (a) Control animal, H&E staining, ×200. Note intact overlying epithelium with normal mucosa and isolated villi. (b) Irradiated animal, H&E staining, ×200. Note increased N:C of the simple columnar epithelium (bold arrow) and decreased mucosal thickness. Also, glandular cells were tightly arranged, and central lacteals were dilated (asterisk). (c) and (d) BrdU IHC, ×200. Note the brownish yellow cells, BrdU-positive–labeled cells, extensively distributed in the mucosa (bold arrow). H&E: hematoxylin and eosin; BrdU: 5-bromo-2-deoxyuridine; IHC: immunohistochemistry; N:C: nuclear-to-cytoplasmic ratio.
Ileum
In ileum (Figure 5), control animals exhibited intact overlying columnar epithelium with goblet cells, and intestinal glandular cells were regularly arranged with basal nuclei, while in the basal part of the gland crypt, clusters of eosinophilic cells, the Paneth cell, known to be important in immunity and host defense, could be identified and gut-associated lymphatic tissue (GALT) with germinal centers and numerous lymphocytes were also present. In irradiated animals, a decreased population of the lining epithelium was noted, while an increased population and cytoplasmic contents of overlying goblet cells could be identified. Intestinal glandular cells were regularly arranged, but showing a more basophilic appearance with enlarged nuclei and prominent nucleoli; intestinal glands were crowded in contrast to those loosely arranged in control animals. Microscopically, Paneth cells had disappeared, and there was depletion of lymphocytes in GALT. BrdU IHC-positive reactions were prominent in the glandular cell area. The LI of the glandular cells in the irradiation group (20.44 ± 5.85) was significantly higher (p < 0.01) than that in control group (5.97 ± 2.03). Meanwhile, no positive BrdU-labeled lymphocytes in GALT could be identified in irradiated animals.

Ileum, luminal mucosa (bar = 100 μm). (a) Control animal, H&E staining, ×200. Note intact epithelium with goblet cells and isolated villi as well as Paneth cells (thin arrow). (b) Irradiated animal, H&E staining, ×200. Note decreased population of overlying columnar epithelium and increased population of enlarged goblet cells (bold arrow), basophilic glandular cells with enlarged nuclei and prominent nucleoli, crowded intestinal glands, and the disappearance of Paneth cells. (c) Control animal, BrdU IHC, ×200. Note the brownish yellow cells, BrdU-positive–labeled cells, sporadically distributed in the mucosa (bold arrow). (d)Irradiated animal, BrdU immunohistochemistry, ×200. Note significantly increased BrdU-positive–labeled glandular cells. (e) Control animal, H&E staining, ×200. Note abundant lymphocytes located in GALT with prominent germinal center (asterisk), and the upper right small figure was showing enormous BrdU-positive lymphocytes. (f) Irradiated animal, H&E staining, ×200. Note depletion of lymphocytes in GALT with the disappearance of germinal center (asterisk). Meanwhile, the upper right figure was indicating negative reaction for BrdU IHC in the same location. H&E: hematoxylin and eosin; BrdU: 5-bromo-2-deoxyuridine; IHC: immunohistochemistry; GALT: gut-associated lymphatic tissue.
Colon
In colon (Figure 6) of control animals, the intact overlying epithelial cells were regularly arranged with basal nuclei and a low N:C ratio (less than 1:2), while intestinal glands were loosely arranged with diffuse distribution of lymphocytes in the lamina propria. Sporadic goblet cells were also present. In irradiated animals, the overlying epithelial cells were decreased in number with an increased number of goblet cells, and the N:C ratio of the lining epithelium, except for the goblet cells, was increased to 1:1. Basophilic luminal mucus contents with some residue sticking to the surface were also notable. The glandular cells had a basophilic appearance with enlarged nuclei and prominent nucleoli. Crypt dilation could also be identified, and the population of lymphocytes in the lamina propria was obviously decreased in contrast to that in control animals. BrdU IHC-positive reactions were mainly present in the glandular cells, and the LI of the glandular cells in irradiation group (30.98 ± 9.83) was significantly higher (p < 0.01) than that in control group (3.08 ± 1.15), indicating active proliferation.

Colon, luminal mucosa (bar = 100 μm). (a) Control animal, H&E staining, ×200. Note intact overlying epithelium with regularly arranged intestinal glands, and abundant lymphocytes in the lamina propria. (b) Irradiated animal, H&E staining, ×200. Note increased N:C of the lining epithelium (bold arrow) and increased goblet cells (thin arrow); intestinal glands with enlarged nuclei and prominent nucleoli as well as decreased lymphocytes in the lamina propria. Additionally, mucous contents were sticky to the luminal surface (asterisk). (c) Control animal, BrdU IHC, ×200. Note the brownish yellow cells, BrdU-positive–labeled cells, sporadically distributed in the mucosa (bold arrow). (d) Irradiated animal, BrdU IHC, ×200. Note significantly increased BrdU-positive–labeled glandular cells. H&E: hematoxylin and eosin; BrdU: 5-bromo-2-deoxyuridine; IHC: immunohistochemistry; N:C: nuclear-to-cytoplasmic ratio.
Rectum
In rectum (Figure 7), control animals exhibited intact overlying epithelial cells with interspersed goblet cells, while glandular cells were regularly arranged and abundant lymphocytes were distributed in the lamina propria. In irradiated animals, the overlying epithelial cells were decreased in number with increased goblet cells, and the intestinal glands were also decreased in size with enlarged basophilic nuclei and prominent nucleoli, and decreased numbers of lymphocytes with increased fibrous tissue in the lamina propria. BrdU IHC result showed that the positive reactions were mainly present in the glandular cells (similar to the previous intestinal sections). The LI in the irradiation group (23.16 ± 8.97) was significantly higher (p < 0.01) than that in control group (7.82 ± 2.81), also, indicating cellular proliferation.

Rectum, luminal mucosa (bar = 100 μm). (a) Control animal, H&E staining, ×200. Note intact overlying epithelium with interspersed goblet cells and regularly arranged intestinal glands as well as abundant lymphocytes in the lamina propria. (b) Irradiated animal, H&E staining, ×200. Note slightly increased goblet cells (bold arrow), and intestinal glands with enlarged nuclei and prominent nucleoli. Dilated crypts were notable (thin arrow), and the population of lymphocytes was decreased accompanied by increased fibrous tissue in the lamina propria (asterisk). (c) Control animal, BrdU IHC, ×200. Note the brownish yellow cells, BrdU-positive–labeled cells distributed in the mucosa (bold arrow). (d) Irradiated animal, BrdU IHC, ×200. Note significantly increased BrdU-positive–labeled glandular cells. H&E: hematoxylin and eosin; BrdU: 5-bromo-2-deoxyuridine; IHC: immunohistochemistry.
Histogram for LI
Figure 8 shows the LI for control and irradiated animals in each sections of the GI tract. In control animals, the LI in small intestines were higher than those in other GI tract sections, with the peak value presented in duodenum, which is almost consistent with the normal physiological distribution. However, in irradiated animals, the LI were increased in both small and large intestines, and the LIs from jejunum to rectum were significantly higher than those in control animals (p < 0.01).
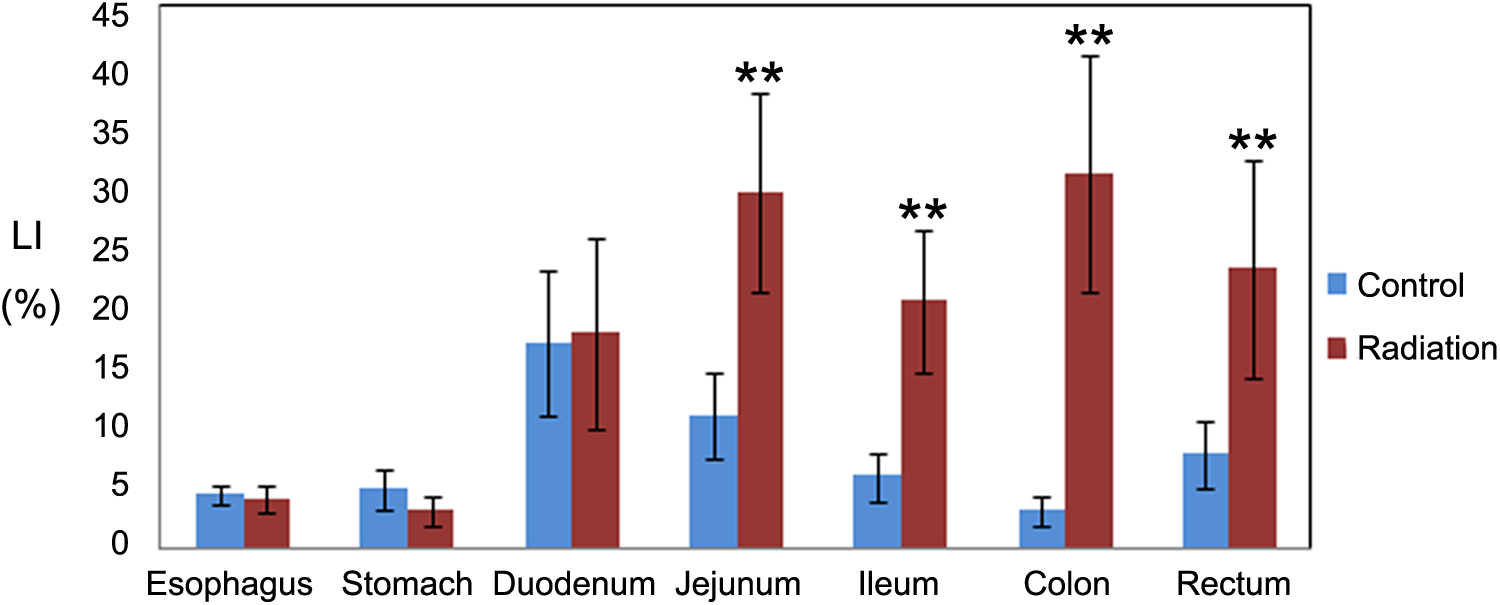
BrdU LI changes noted in different segments of GI tract. N = 6; **p < 0.01. Compared with control group, the LIs from jejunum to rectum were significantly increased (p < 0.01), while no statistical significance (p > 0.05) were noted from esophagus, stomach, or duodenum in the irradiated group. BrdU: 5-bromo-2-deoxyuridine; LI: labeling index; GI: gastrointestinal.
Discussion
Radiation therapy has been widely employed to treat and control various kinds of cancers 19 ; however, the threat of radiation injury is still important. A recent study has cited experimental and epidemiologic evidence showing the linkage between low-dose ionizing radiation exposure and the development of solid cancers and leukemia. 20 To solve the problem, the search for compounds that can reduce the deleterious effects of radiation are of interest both for therapeutic radiation for cancers and for accidental or terrorism-related exposures. 21 There are a number of agents that can be used to protect people from radiation injury, such as nitroxides, polyphenols, tocols, ethyl pyruvate, superoxide dismutase mimetics, amifostine and other thiols, 22 melatonin and its homologues, 23 and other free radical scavengers. In addition, checkpoint kinase inhibitors, 24 inhibitors of c-Abl 25 and modulators of apoptosis 26 have also been found to possess radioprotective capabilities. 27 However, the majority of these agents still have some degree of toxicity. Thus, we intended to establish an NHP model for further drug screening, aiming to find a safe agent with definite radioprotective effect. In this study, we mainly focused on the effects of ionizing radiation on the GI tract, which has been widely reported as one of the major targets of radiation because of its high radiosensitivity. 28 We also employed BrdU to identify the proliferative changes of the GI in both control and irradiated primates. Additionally, this is also the first time, in compliance with CFDA, FDA, and OECD GLP regulations, to provide detailed information about radiation-induced morphological changes in each segment of GI tract and should give further support to histopathological evaluation in relevant preclinical studies.
In this study, the histopathological results showed that the entire GI tract in the radiation group was markedly impaired after radiation exposure. The esophagus had degenerative changes, and the overlying epithelium was composed of squamous cells-containing enlarged nuclei and apparent centrally located nucleoli. As stated in a previous report, 29 patients receiving radiotherapy may develop esophagitis. However, we didn’t find any evidence in the esophagus inflammation in our study, which may be due to the different TBI levels. Meanwhile, the stomach had complex changes involving both inflammatory and degenerative changes. The overlying epithelium was atrophied with elevated N:C ratio, and epithelial shedding with irregularly arranged residue cells were also identifiable, and inflammatory cell infiltration beneath the luminal epithelium in the lamina propria reflected the inflammatory response to the mucosal damage. The overlying mucous cells are known to play a defensive role by secreting bicarbonate-rich mucus that coats and lubricates the gastric surface and constructing a mucus barrier against luminal acid and other chemical insults; once the barrier is gone, the stomach would become vulnerable. 30 Aside from the direct impact of radiation exposure on the fundic glands, in the present study, the overlying epithelial injury probably provided another possible way for fundic gland degeneration, which was characterized by parietal/chief cell hypertrophy with decreased eosinophilic cytoplasm granules. The upper small intestine (duodenum and jejunum) exhibited similar changes, such as shortened villi and crypts, as well as decreased mucosal thickness, which were generally consistent with those found in a previous study. 31 In addition, we also found prominent inflammatory changes in the duodenum, associated with focal necrosis and blunted villi. Unlike in the duodenum and jejunum, the ileum was more likely to show proliferative changes with increased crypts and goblet cells, which are known to play an important role in constructing the first line of defense against potential physical and chemical injury caused by ingested food or bacteria in the GI tract. 32 Moreover, lymphocyte depletion was identified in the GALT of the ileum in the radiation group; as previously reported, this is likely a direct effect of radiation exposure. 33 In the colon and rectum, goblet cells were also increased in number, and increased myxoid content could be identified in the lumen of irradiated animals compared with the control animals, which has been considered as a protective reaction to prevent further injury to the mucosa.
The BrdU IHC result, as shown in Figure 8, showed that the LIs of the epithelium in the lower intestinal segments were undergoing tremendous changes. Significant increased LIs (p < 0.01) were noted in jejunum, ileum, colon, and rectum compared with the corresponding control data, indicating elevated proliferative activity in these organs. Although these changes were considered as regenerative changes in this study, more attention should be paid to these organs due to their higher risk of tumor development.
Conclusion
After 6.5 Gy gamma ray exposure, the esophagus underwent epithelial degeneration, while the stomach and the duodenum were involved in glandular epithelium degeneration and inflammation. From jejunum to rectum, late-stage inflammation characterized by increased regenerative glands dominated the major pathological changes. Besides, goblet cells proliferation in ileum, colon, and rectum were notable and may play an important role in protective reaction.
Footnotes
Acknowledgments
We’d like to give our sincere thanks to technicians from histopathology department of National Chengdu Center for Safety Evaluation of Drugs for their excellent work. We also thank Dr Peter Mann from EPL for his professional interpretation and revision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All the costs were funded by the authors.

