Abstract
Orellanine is a nephrotoxic toxin produced by some mushroom species of the Cortinarius genus, typically found in Europe and North America. The nephrotoxicity of Cortinarius orellanus is well known and was first recognized in the 1950s when this mushroom was identified as the cause of a mass poisoning in Poland. Typically, onset of symptoms is delayed for 1–2 weeks after ingestion. Some patients suffer mild gastrointestinal discomfort in the latency period before developing signs of renal impairment due to severe interstitial nephritis, acute focal tubular damage, and interstitial fibrosis. There is no specific antidote to orellanine poisoning. The mainstay of treatment is the prevention of secondary complications of kidney failure, adequate dialysis and, in the case of incomplete recovery, management of chronic renal insufficiency.
In this work, we aim to review about Cortinarius species, including epidemiological studies, chemical structure, toxicokinetics, toxic doses, mechanisms of toxicity, diagnosis, prognosis, and treatment options.
Introduction
Orellanine is a highly nephrotoxic bipyridine N-dioxide found in various mushrooms of the Cortinariaceae family, including fool’s webcap (Cortinarius orellanus) and deadly webcap (Cortinarius rubellus formerly named Cortinarius speciosissimus or Cortinarius orellanoides). 1,2 These two species are regarded as two of the world’s most toxic mushrooms and have claimed several lives in Europe and North America after being confused for edible mushrooms such as Cantharellus tubaeformis, Cantharellus cibarius, and the hallucinogenic Psilocybe genus. 3 –6 The nephrotoxicity of C. orellanus was first recognized in the 1950s when this mushroom was identified as the cause of a mass poisoning in Poland. 7 –9
The toxicity mechanism of orellanine is not yet fully understood. Chemically, orellanine resembles the bipyridyl herbicides diquat (1,1′-ethylene-2,2′-bipyridylium) and paraquat (1,1′-dimethyl-4,4′-bipyridylium; Figure 1), and some authors suggested a similar mono-electronic reduction mechanism (generation of a stable free radical) with strong prooxidant properties. 10 In accordance, experimental evidences have indicated that cytotoxic effects of orellanine are caused by the production of reactive oxygen species and subsequent oxidative stress. 11 –13

Chemical structures of paraquat (a), diquat (b) and orellanine (c).
Symptoms of orellanine poisoning are those resulting from renal damage (e.g. digestive signs, thirst, headache, muscle pain, flank pain, polyuria followed by oligoanuria) developing over several days or in some cases up to 2 weeks after intoxication. 14 Orellanine rapidly concentrates in the kidney and has been found in renal biopsy specimens up to 6 months after ingestion. 15 During the latent period, mild gastrointestinal disorders occur, which may be overlooked. 16 Accordingly, patients present themselves at the hospital only at the stage when renal failure has already developed. 17 The acute damage can be soft and brief or irreversible and even lethal. There are no specific antidotes for poisoning by Cortinarius species. 18 Treatment strategies include symptomatic treatment, hemodialysis, plasmapheresis, steroids, and N-acetylcysteine. 19 In the worst cases, only chronic hemodialysis or renal transplantation are effective. 20
The only few reviews that have outlined the main features of this unique type of intoxication date back to the 1980s and 1990s. 10,14,21 Due to the high toxic potential and the risks to amateur mycologists, the present review article attempts to present an update of the current knowledge concerning Cortinarius intoxications, namely the morphological characteristics of Cortinarius species, epidemiological studies, chemical structure, toxicokinetics, toxic doses, mechanisms of toxicity, diagnosis, prognosis, and treatment.
Materials and methods
All articles indexed to MEDLINE® and books were searched using the following key words: orellanine, mushroom poisonings, C. orellanus and C. rubellus, and acute renal failure.
Cortinarius species
Cortinarius is the largest genus of fungi that forms mushrooms comprising over 250 species. These usually form mycorrhizae with trees and can be found in Northern Europe and in mountainous areas of Central Europe, 2 as well as in Northern America 22 at the end of summer or autumn. 22 C. rubellus species inhabit moist or wet conifer forest, particularly in moist acidic soil. 2
Cortinarius species have a partial veil composed of fine threads (cortina) connecting the margin of the cap to the stalk, particularly in young specimens (Figure 2(a)). As the mushroom ages, the cortina disappears, resulting in almost indistinguishable remains of the cortina (Figure 2(b)). 2 They also have an orange, purple, or greenish yellow-colored fruit bodies with characteristic rusty orange gills and a thick stalk with striae and bulbous base. 3 These are characterized by having a knob-like protuberance on the rust- to yellow–brown cap (2–12 cm/1–5 in) that is conical in shape on young species and more flattened on older specimens. The broad, elliptical spores yield a rust brown spore print. The cylindrical stalk is similar in color to the cap and stalk sometimes contains lemon yellow bands that represent the remains of the cortina. 2

(a) Cortina; (b) remains of cortina; (c) Cortinarius orellanus; (d) Cortinarius rubellus; (e) Cortinarius henrici; (f) Psilocybe semilanceata; and (g) Cantharellus cibarius.
Specifically, C. orellanus (Figure 2(c)) is a medium-sized gill mushroom that is characterized by a dome-shaped, dry, orange, red, brown-to-yellow brown cap; thick, well-spaced, orange brown gills; and a dry, yellowish to reddish brown stalk that is tapered below with some veil fibrils on the surface. The body of the mushroom is whitish to yellowish. Species in this group have rust brown spore prints and the spores are elliptic and distinctly ornamented. 23 C. rubellus (Figure 2(d)) is characterized by a cap rust brown to orange, often has a steeper and darker colored elevation at the top of the cap, and its surface is dry and slightly scaly. The cap diameter is typically 4–8 cm when fully expanded, and the margin is often slightly rolled down even in fully mature specimens. The gills are pale yellowish at first, becoming rusty brown as the spores mature. 24 It has been claimed that C. henrici (Figure 2(e)) and C. rainierensis are identical to C. rubellus. 25
Only some species of Cortinarius mushrooms, such as C. orellanus, C. rubellus, C. henrici, C. rainierensis and C. bruneofulvus contain the nephrotoxin orellanine. 26 –29 The content of orellanine was determined in dried fungus, varying greatly depending both on the mushroom portion considered and Cortinarius species. 30 The analysis of C. orellanus and C. rubellus species showed that the content of toxin (expressed on dry weight basis) was 9400 and 7800 mg/kg in caps, 4800 and 4200 mg/kg in stems, and 3100 and 900 mg/kg in spores, respectively. In mycorrhiza roots from C. rubellus, the orellanine content was 300 mg/kg. 30 In another study, the orellanine content in dried fungus was found to be about 14,000 mg/kg in C. orellanus and 9000 mg/kg in C. rubellus. 31
Epidemiological studies
Intoxications with the fungus C. orellanus have been described in literature. The toxicity of the mushroom C. orellanus was firstly discovered in 1957 when 135 habitants of Bygdosz (Poland) were intoxicated. 7 –9,32,33 Since then, most of poisoning reports have been described in Europe, 14 where mushroom foraging is popular, but cases were also described in North America and Australia. 5,14,34,35 In 1974, four cases of poisoning by C. rubellus occurred in Finland 36 ; further incidents were reported in Scotland, 37,38 Sweden, 39 Norway, 40,41 Italy, 42 Germany, 43 France 6,44,45 and Wales, United Kingdom. 46 In Austria, Franz et al. 47 reported a C. rubellus poisoning on a 28-year-old man who thought had eaten magic mushrooms. Indeed, nephrotoxic Cortinarius species are morphologically similar to mushrooms from the Psilocybe genus (Psilocybe semilanceata; Figure 2(f)), leading to mistakes by inexperienced hallucinogenic mushrooms hunters. 47 Likewise, misidentification of C. orellanus as C. cibarius (Figure 2(g)) may also occur. 2
Chemistry
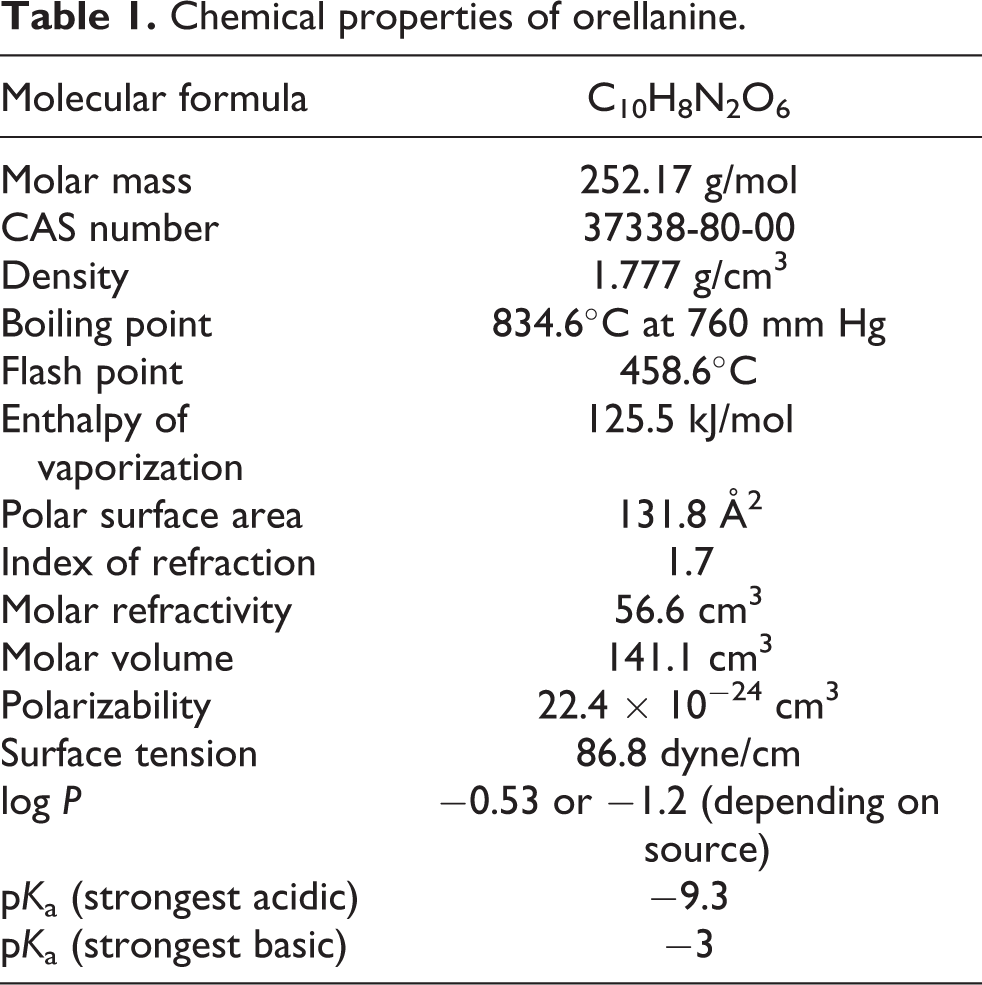
Table 1 presents chemical properties of orellanine. It is a bipyridine N-oxide (2,2′-bipyridine-3,3′,4,4′-tetrahydroxy-1,1′-dioxide) that exists as two tautomers 11,48 (Figure 1). The more stable tautomer is the amine oxide form. Orellanine is a colorless fine crystalline and navy blue-fluorescing compound that is stable at 150–160°C, decomposing slowly above this temperature and under ultraviolet (UV) light to the yellow nontoxic bipyridyl compound orelline by releasing oxygen. 27 An explosive degradation occurs at temperatures over 267°C. 27 Cooking temperatures, freezing, and drying do not reduce the orellanine content. 25,49 Orellanine is stable for many years in the intact mushroom. 10 Figure 3 demonstrates the reduction of orellanine to the nontoxic compound, orelline, via the intermediate orellinine. 27,49 –51 Orelline are bright yellow crystals, and orellinine is almost colorless.

Chemical structures of orellanine, orellinine, and orelline.
Chemical properties of orellanine.
The stereochemistry of orellanine was confirmed by X-ray crystallography. 52 In crystal conformation, the planes of the two pyridyl rings are nearly perpendicular, making the molecule chiral. However, orellanine isolated from mushroom is an optically inactive racemic mixture, and this is probably due to the low rotational barrier to racemization. 53
In 1962, Grzymala 32 was the first to demonstrate experimentally the nephrotoxicity of C. orellanus and was the first to isolate orellanine on biological analysis. Dehmlow and Schulz 54 were the first to report the successful synthesis of orellanine and orelline. Orellanine was synthesized in nine steps from 3-amino pyridine. Orelline has also been synthesized from 2-bromo-3-hydroxypyridine. 55
As referred, when placed under UV light, orellanine is, at first, navy blue fluorescent, and after several minutes shifts to bright turquoise fluorescent due to a rapid photochemical reaction. 56 This photochemical decomposition has been shown to proceed by stepwise loss of the N-oxides to give first the mono N-oxide orellinine and, subsequently, orelline. 27
Orellanine, orellinine, and orelline are soluble in dilute sodium hydroxide, ammonium hydroxide, and dimethyl sulfoxide; slightly soluble in methanol; and practically insoluble in most organic solvents and water. 10,58 Orellanine, contrary to orelline, is well soluble in alkaline solutions.
Toxicokinetics
Little is known about the toxicokinetics of orellanine in humans, especially during the first 24 h after ingestion. 57,31 In animal studies, kidneys excrete detectable quantities of orellanine only for the first 24 h after exposure. 31 This is consistent with the observations in human cases of orellanine poisoning, where the toxin is not detected in urine samples collected from 2 to 18 days after ingestion. 59,60 Typically, clinical samples are not obtained until several days after ingestion due to the delayed onset of symptoms. Indeed, this has led to misdiagnosis of the poisoning since the victim often does not associate the symptoms with a meal taken several days before. For this reason, no information is available regarding human plasma levels of orellanine attained shortly after exposure. 59
Based on the ingestion of a toxic dose of 100–200 g of mushrooms containing 0.1% orellanine, and assuming complete absorption and full plasma distribution, maximal peak plasma levels of the toxin is expected to range from 130 to 260 µmol/L. 7,30 Based on limited testing, the clearance of orellanine from the blood is probably rapid (i.e. <2–3 days). 25
The continued presence of orellanine in renal cortex after hemodialysis, and its persistent presence in this tissue for several months in the absence of detectable orellanine in urine or blood, suggests that the toxin is sequestered in the kidney in a poorly exchangeable form. 59
Toxic doses
Table 2 resumes the toxic doses of orellanine and dried Cortinarius homogenates for different species. Several studies on the toxicity of C. rubellus in rats were carried out by Nieminen and collaborators. 63 –65 Authors have shown that rats appear to be resistant to Cortinarius poisoning and a genetic variability was suggested since females were more resistant. 66 In another study, 67 23% of the animals showed total resistance to toxins, irrespective of dose, whereas in others the observed renal lesions were dose dependent.
Toxicity of orellanine and dried Cortinarius homogenates.

SC: subcutaneous; PO: per os; IP: intraperitoneal; LD50: median lethal dose.
Source: Adapted from the work by Carter et al. 62
Clinical data indicate that humans appear to be more sensitive to toxic effects than mice and rats. The ingestion of only two to three mushrooms (3 mg of toxin that corresponds to 0.04 mg/kg for human of 70 kg) seems to be enough to make the victim dependent on dialysis for the entire life. 45,68 Similarly, in humans, the greater susceptibility of males to Cortinarius toxicity has been described by several authors. 14,37 Herrmann and colleagues 1 described a lethal dose of fresh C. rubellus between 29 g and 227 g for humans weighing about 70 kg.
Mechanism of toxicity
Orellanine and orellinine have similar toxicity, but orelline proved to be nontoxic. 69 The mechanism of toxicity is not yet fully understood. It was shown that the toxin inhibits the synthesis of macromolecules such as proteins, RNA, and DNA 14,25,70 ; promotes a noncompetitive inhibition of the activity of alkaline phosphatase, γ-glutamyl transpeptidase, and leucine aminopeptidase 71 ; and interrupts the production of adenosine triphosphatase, 69 namely at the proximal tubular brush border, compromising utilization and reabsorption of peptides, polysaccharides, and other molecules.
Moser 72 showed that C. orellanus toxin inhibits DNA-dependent activity of RNA polymerase B from rat liver (eukaryotic cell) and DNA-dependent activity of RNA polymerase from Escherichia coli. Later, Richard and co authors 70 pretreated rat liver microsomes with orellanine and then exposed rabbit reticulocyte lysate to this mixture. Only the inhibition of protein synthesis was observed, while direct addition of untreated orellanine or only microsomes did not. This suggests that the inhibition of protein synthesis is due to a metabolite of orellanine.
Orellanine inhibits pinocytosis in Amoeba proteus and inhibits growing of both slime mold Discyostelium discoideu and E. coli at 80 µM. 69 The large spectrum of toxic effects in plants, animals, and microorganism suggests that the target is likely to be a cellular process found in both prokaryotes and eukaryotes. 49
Rapior et al. 29 suggested that the Cortinarius toxicity is caused by metabolites with the isoxazolium core derived from the photochemical rearrangement of orellanine (called the phototoxicity mechanism of orellanine). In accordance, authors found that orellanine purified in the dark and administered to laboratory animals show low toxicity, while the one extracted in the light induces a toxic response (Figure 4). These intermediates can bind covalently with numerous proteins in the body, leading to organ damage.

Phototoxicity mechanism of orellanine.
As already referred, orellanine chemically resembles the pyridine herbicides diquat (1,1′-ethylene-2,2′-bipyridinium) and paraquat (1,1′-dimethyl-4,4′-bipyridinium), and some authors suggested a possible mono-electronic reduction mechanism (generation of a stable radical) for its toxicity mechanism. 10,13 The consequence is obvious: production of free radicals and therefore oxidative stress similarly to paraquat. 73,74 In opposite to paraquat, this proposed intracellular toxic mechanism with nicotinamide adenine dinucleotide phosphate plus H+ depletion would need a long time to damage the cells to the degree of inevitable necrosis, according with the delayed toxic effects observed in this type of mushroom poisoning. 75
However, this model has been criticized since orellanine has a much more negative redox potential than paraquat and diquat. 20,76 Noteworthy, these authors observed the formation of a radical form of orellanine induced by near UV (370 nm) at physiological pH under aerobic or anaerobic conditions. 20 This apparently stable radical, identified as ortho-semiquinone anion radical (Figure 5), was also generated using biological oxidizing agents (e.g. cytochrome c and nicotinamide adenine dinucleotide) or enzymatic systems such as tyrosinase/oxygen (O2) and peroxidase/hydrogen peroxide (H2O2). 13,50 Indeed, it is the ortho-semiquinone anion radical that is responsible for superoxide radical production. At the renal spot of bioaccumulation of the toxin, a quick change of small portions of orellanine rapidly cycling from its oxidized to its reduced form might be sufficient to support continuous production of reactive oxygen species, which will induce oxidative stress. This process may lead to a large oxygen consumption, which might create hypoxic conditions, as well as the dramatic depletion of renal glutathione and ascorbate levels consequently making cells more susceptible to oxidant damage. 12,13,50 Corroborating this hypothesis, orellanine-treated animals did experience increased oxidative stress, which is indicated by increase in both plasma levels of ascorbyl radicals and protein oxidation in renal tissue. 11

Redox cycling of orellanine.
Oubrahim and colleagues 28 proved the formation of a characteristic ortho-semiquinone radical anion (SQ·) derived from orellanine in a system containing horseradish peroxidase and H2O2. Since peroxidative oxidation proceeds via two one-electron oxidation steps, this enzyme can oxidize two molecules of orellanine to the corresponding semiquinone during one enzymatic cycle or one molecule of orellanine to the corresponding quinone by two successive monoelectronic oxidation steps. The ortho-semiquinone form of orellanine was also generated by the enzymatic system tyrosinase/O2, which proceeds via a two-electron oxidation. In this case, the generation of the ortho-semiquinone radical likely occurs due to the equilibrium between orellanine and its corresponding quinone. During oxidation of orellanine in renal tissue, ortho-semiquinone likely accumulates, whatever the mode of enzymatic oxidation may be (one or two electrons). The ortho-semiquinone, as well as the quinone, could participate in a variety of reactions including covalent binding to biological compounds leading to cell damage. In addition, when oxidation of orellanine by horseradish peroxidase/H2O2 occurred in the presence of reducing agents of biological interest, such as ascorbic acid or glutathione, the formation of ascorbyl or glutathionyl radicals was observed.
Although all authors agree that orellanine is the main toxin, Cortinarius species also contain cyclic decapeptides (cortinarin A, B, and C) that produce renal damage. 77,78 Cortinarin A and B (not C) were found to be nephrotoxic in animal studies, 78 but the role of these decapeptides in causing the Orellanus syndrome or even their existence has been questioned by several authors. 79
Diagnosis
The diagnosis of orellanine poisoning can be clinical, mycological, or toxicological. 1,3,51
Clinical diagnosis
Orellanine poisoning is characterized by a long latent period. Typically, the onset of symptoms is delayed from 2–4 to 14 days after ingestion. 14 The higher the quantity of mushrooms consumed, the shorter the latent period. 62 There is a significant variation on individual responses to poisoning, with the degree of renal injury ranging from mild and transient to severe and irreversible. 59
Some patients suffer mild gastrointestinal discomfort in the latency period before developing signs of renal impairment. Table 3 shows signs, symptoms, and analytical findings related to orellanine poisoning. The gastrointestinal phase (also known as prerenal phase) is primarily characterized by vomiting and nausea, and, less often, abdominal pain, asthenia, chills, burning sensation in the mouth, thirst, headache, myalgia, anorexia, and diarrhea. 10,14,15,21,25
Signs, symptoms, and analytical finding in intoxications by orellanine.

Clinical signs may spontaneously disappear in some patients, leaving the poisoning unnoticed; in others, the signs become more intense and are accompanied by neurological manifestations (e.g. paresthesia, taste impairment, cognitive disorders, and dizziness), lumbar pain, and anuria requiring hospitalization. 14,15,21
Renal failure may develop several days to weeks after initial symptoms due to orellanine sequestration in the kidney. The incidence of renal failure varies from 30% to 70% of patients, and 30–65% need temporary dialysis. 14 Renal involvement is the other peculiarity of this poisoning and is mainly characterized by lumbar and flank pains, intense thirst, oliguria, polyuria, proteinuria, hematuria, and leukocyturia. 10,14,21,23,80 The blood balance shows an increase in urea and creatinine levels. 21,23 Differential diagnosis with other nephrotoxic compounds such as oxalates crystals found in certain plants (e.g. genus Philodendron and Dieffenbachia) and ethylenoglycol found in antifreeze products is relatively easy. In these poisonings, signs and symptoms are evident during the first hours after exposure and circumstantial evidences can help diagnosis. 81
Nephrotoxicity is characterized by an early and severe interstitial fibrosis, interstitial edema, and tubular epithelial necrosis. 10,21,51,80 Short and colleagues 37 have shown that when initial biopsy was made until 2–3 weeks after poisonings, specimens exhibited pronounced focal tubular damage with tubulorrhexis, cast formation, and severe interstitial edema with patchy infiltration of lymphocytes, plasma cells, and some polymorphs. The glomeruli showed only slight mesangial cell reaction. In the later biopsy specimens (obtained 7–8 weeks after poisoning), the mild glomerular reaction was still present, but the major features were tubular dilatation and cellular atrophy with groups of apparently normal tubules between damaged areas. In both cases, the interstitial edema was much less in the later specimens, but there was early fibrosis between the damaged tubules. A mild mononuclear inflammatory cell infiltration was present. Immunofluorescence studies showed no significant deposition of immunoglobulin, complement, or fibrin in glomeruli, but immunoglobulin G, immunoglobulin A, and fibrin were isolated from tubular casts. 37 Therefore, the existence of renal alterations as a consequence of some immune reaction cannot be excluded.
In poisoning due to C. rubellus, most cases have not been admitted to hospital before 8–14 days after the mushroom ingestion and then the clinical picture is that of an acute renal failure. 10 It is estimated that 30–45% of individuals who ingest nephrotoxic Cortinarius mushrooms develop acute renal failure. 14 Of these, half usually recover the renal function and half progress to chronic renal insufficiency and require maintenance hemodialysis or kidney transplant. 59 Some patients may become asymptomatic and the renal injury is only identified by biochemical tests. 46 In a study of 26 patients with nephrotoxicity secondary to Cortinarius mushroom ingestion, the incidence of end-stage renal failure requiring dialysis and renal transplantation was approximately 8%, 6 whereas 9 of 22 (41%) Swedish patients developed end-stage renal disease after ingesting mushrooms from Cortinarius species. 15
Liver injury has also been observed based on increase of transaminases and bilirubin levels, hepatomegaly, hepatalgia, and from lipoidosis and necrosis lesions evidenced by histological analysis, 8,33 but most of the studies ruled out liver involvement. 14
Worth mentioning, orellanine is currently being tested as a potential treatment for metastatic renal cancer based on its highly selective toxicity to renal cells. 82
Mycological and toxicological analyses
Several analytical methods were developed for the analysis of both mushrooms and biological samples, such as serum and renal tissue. The ferric–orellanine reaction could be useful for both mycologists and medical personnel for demonstrating whether an unknown Cortinarius specimen contains orellanine or not. Schumacher and Hoiland, 10 have proposed a rapid qualitative test to detect orellanine in mushrooms. A fresh or dried mushroom is crushed in five volumes of water and filtered after 10 min at room temperature. The filtrate is then mixed with an equal amount of 3% ferric chloride hexahydrate dissolved in 0.5 N hydrochloric acid (HCl). The presence of orellanine is suspected if a dark gray–blue color ink appears. However, other authors described an immediate change of the yellowish liquid to purple–red or violet. 83
Thin-layer chromatography can be used for separation prior to proof for the presence of toxins under UV light. A simplified procedure for detecting orellanine is as follows: a small piece of dried fungus is crushed and extracted in 50% ethanol, allowed to stand for 15 min at room temperature, and then the extract is applied to a silica gel plate and chromatographically developed by n-butanol:acetic acid:water (3:1:1). After drying, the plate is sprayed with 2% ferric chloride in 0.5 N HCl. Orellanine is visible as navy blue spot from the application point (retention factor of 0.25–0.5), orellinine as dark blue spot, and orelline as light blue spot. 10,22 Other analytical procedures are available such as electrophoresis 28 and high-performance liquid chromatography (HPLC) with photodiode array, 30 electrochemical, 84 and UV 85 detection. In these last two methods, reversed-phase ion-pair HPLC with phosphate-containing eluent20 and amide and C18 columns with phosphoric acid as the eluent10 were employed. Orellanine has also been detected in single MS mode using electron impact 84 and electrospray ionization. 1,30
In a renal biopsy specimen, orellanine can be detected up to 6 months after poisoning by performing a thin-layer chromatography technique. 51,60,86 Orelline, the di-reduction metabolite, has also been found in renal biopsies in cases of orellanine poisoning and possibly originates from either the mushroom itself or from extra- or intrarenal metabolism of orellanine. 59
Since orellanine is rapidly concentrated in the kidney, it will not be detected in urine, blood, and dialysis fluids at the time when the first symptoms appear. 87 Toxin in plasma can be only detected for up to 2 weeks after ingestion. 53,86 However, Andary and colleagues 86 were able to detect a plasma orellanine concentration of 20 µmol/L in a sample obtained from a patient 9 days after eating two mushrooms, and they used hemodialysis to effectively clear the circulating toxin. The toxin is not detected in urine samples collected as early as 2 days after ingestion and as late as 18 days. 51
Light microscopy has also been useful to highlight characteristic renal histopathological findings such as acute tubular injury and interstitial edema and invasion of inflammatory cells with interstitial nephritis. 87
Prognosis
The chance of full recovery from orellanine poisoning depends on the amount of ingested toxin, age, and general health of the individual patient, probably preexisting subclinical renal disease, and the time at which therapy is started. 22
Holdmdalh 88 proposed a method for estimating the prognosis in individual cases. This method is called the “Cortinarius nephron toxicity prognostic index (CNT)” and is based on two parameters generally available. The parameters used are the serum creatinine level before treatment and the former days after ingestion of the mushrooms. It is calculated from the formula CNT = (y + 316)/X × 104, where y is the serum creatinine and X the number of elapsed days. CNT index <1.1 indicates a good prognosis; CNT between 1.1 and 2.1 indicates “intermediate” prognosis (probably chronic renal failure with serum creatinine higher than 200 µmol/L) and >2.1 a poor outcome with end-stage renal failure requiring renal replacement therapy.
Grzymala
8,33
established a relationship between the duration of the latent period and poisoning severity. In his series, the latent period was:
10–17 days in patients presenting with thirst, burning sensation in the mouth, and polyuria (mild intoxication); 6–10 days in patients presenting with digestive disorders, polyuria or oliguria, hematuria, and leucocyturia, but no significant renal impairment; 2–3 days in patients with acute renal failure and death rate of about 50%.
Treatment
There is no specific antidote to orellanine poisoning, treatment being mainly supportive care as well as the use of hemodialysis as needed. Prolonged monitoring of renal function is necessary because of the slow resolution of kidney dysfunction. 2
Emesis or gastric lavage might, in theory, be indicated if the patient is seen earlier than 6 h after ingestion. 69
Extracorporeal hemoperfusion, hemodialysis, and plasmapheresis are techniques used to remove the toxin from circulation but should only be considered if the patient is seen within 1 week after ingestion. 22,42,51,89 –91 Beyond that period, the use of hemodialysis depends only on the need to support renal function. 2 About half of the patients requiring dialysis did not recover kidney function. 14
There are also reports of cases where the use of corticosteroids, N-acetylcysteine, and selenium allowed clinical improvement. 3,90 N-Acetylcysteine is a glutathione donor and antioxidant, 13 and selenium is an essential component of several major metabolic pathways including immune and antioxidant defense systems. 92
It has been emphasized that forced diuresis could rather accelerate and amplify the nephrotoxic process since it possibly accentuates the accumulation of the toxins in the kidney and therefore it is not recommended. 10,69
Complete recovery of renal function is attained only in 30% of the poisoned patients with the majority of patients healing with fibrosis and a variable loss of renal function, and 20–40% patients requiring long-term renal replacement therapy. 6,90 Therefore, renal transplantation has been considered, but should not be performed too early in the course of illness. The median time for transplant is 9–10 months after presentation, wherein appears safe without risk of further toxicity from orellanine. 14,15
Concluding remarks
The incidence of mushroom poisoning greatly varies all over the world depending on local traditions, lifestyle, nutritional factors, climate, and the occurrence of wild mushrooms. Orellanine poisoning is mostly accidental and the result of a mix-up between edible and toxic fungi. Only by exception, mushroom poisoning has a suicidal or homicidal cause. Confusion with many nephrotoxic Cortinarius species occurs every year throughout Europe and North America. This poisoning is associated with high morbidity and exceptional mortality. Treatment of orellanine poisoning includes gastrointestinal decontamination and general symptomatic and supportive care. Specific antidotes do not exist. Further knowledge of orellanine toxicokinetics and toxicity mechanisms is still necessary to be able to propose a proper treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: RDO acknowledges Fundação para a Ciência e a Tecnologia (FCT) for his Investigator Grant (IF/01147/2013).
