Abstract
Amoxicillin (AMX) is one of the most commonly prescribed antibiotics for children, and childhood is the period to have the highest risk for toxicity cases including drug-induced adverse reactions. Some neurological adverse effects (anxiety, hyperactivity, confusion, convulsions, and behavioral changes) have been reported related to AMX treatment. In the present study, we aimed to determine the neurotoxic effects of AMX administration at clinically relevant doses in female juvenile rats. AMX was administered in single oral daily doses of 25 and 50 mg/kg for 14 days. According to our results, while AMX administration caused a significant increase in the immobility time of animals, swimming time of these animals significantly decreased. AMX administration significantly reduced the onset of pentylenetetrazole-induced convulsions. The serotonin levels of brain tissues in the AMX-administered groups were decreased significantly, which is thought to be related to depression. The glutamate levels in brain tissues increased significantly in AMX-administered groups, which is thought to be related to convulsion. Otherwise, superoxide dismutase and catalase activities were significantly decreased in brain tissues of AMX-administered groups. In conclusion, AMX administration triggered depression and shortened the time of the appearance of first seizure in juvenile rats. Also, altered brain neurotransmitter levels and increased oxidative stress observed in our study were thought to be the possible underlying mechanisms of AMX-induced neurotoxicity.
Introduction
For decades, clinicians have recommended the drugs for children, relying on data from adult clinical studies because appropriate pediatric safety or efficacy studies were lacking. The extrapolation of adult data to pediatric is often complicated because of the variability of the adverse effects and also pharmacokinetic and pharmacodynamic profiles in the pediatric population. 1 Young animals are considered to provide the best models for investigating effects on developing organs and systems. It is reported that juvenile animal studies when combined with successful pediatric clinical trials will be able to provide safe medications for children with acceptable benefit–risk profiles. 2
The
Till date, limited number of acute psychiatric adverse events (including seizure, encephalopathy, and tremor) related to AMX treatment have been reported in patients with no underlying psychiatric disorders. 11 –15 Furthermore, Food and Drug Administration have published a report that when AMX was coadministered with cephalosporin, this combination therapy might cause death by inducing seizures in a patient. 16
At this point, this study was designed to determine the effects of repeated AMX administration on neurologic functions at clinically relevant doses in juvenile rats whose blood–brain barrier is not fully developed. The levels of brain γ-aminobutyric acid (GABA), glutamate, dopamine, serotonin, adrenaline, and noradrenaline, which are endogenous mediators regulating mood and behaviors, were also determined for the purpose of identifying possible underlying mechanisms of behavioral test results. Additionally, oxidative stress induces abnormalities in different regions of brain, in cellular proteins, in membrane lipids, in DNA and RNA and it has been reported as one of the main mechanisms underlying neurodegenerative and neurobehavioral disorders. 17 –20 Therefore, brain glutathione (GSH), superoxide dismutase (SOD), catalase (CAT), and malondialdehyde (MDA) levels were measured in order to evaluate the contribution of oxidative stress to the possible AMX-induced behavioral effects.
Materials and methods
Materials
AMX was a kind gift from IE Ulagay-Menarini Group, Istanbul, Turkey. Brain SOD and CAT levels were determined by enzyme-linked immunosorbent assay (ELISA) kits from Cayman Chemical Company, Ann Arbor, Michigan, USA. Malondialdehyde (MDA) levels were assayed by ELISA kit from Oxis Biotech Inc., Tampa, Florida, USA. GSH,
An Agilent 1260 Infinity LC system (Waldbornn, Germany), used for the determination of the neurotransmitters, was equipped with a fluorescence detector, an autosampler, a column oven, and a binary pump. For the determination of the catecholamines, an coulometric detector with graphite carbon cells was used (Coulochem III, Thermo Fisher Scientific Inc., Waltham, Massachusetts, USA). Data acquisition and peak processing were performed with ChemStation for LC 3D systems, Rev. B.04.03. 16
Animals
Female Wistar rats weighing 60–80 g (approximately 21 days old) were obtained from Anadolu University Research Center for Animal Experiments. The rats were housed in a room with controlled temperature (24°C) for 12-h light/12-h dark cycle (lights on at 08:00 h). Temperature, sound, and light conditions of the laboratory were maintained during the course of the experiments. Animals were acclimatized to the laboratory environment for at least 48 h before the experimental session. The experimental protocol was approved by the Local Ethical Committee on Animal Experimentation of Anadolu University, Eskisehir, Turkey.
Experimental groups
The study design was based on Japan Ministry of Health, Labour and Welfare guidelines (2012), the most recent guideline on juvenile safety studies, issued: “Guideline on the Nonclinical Safety Study in Juvenile Animals for Pediatric Drugs”. 21
The rats were assigned randomly into the following treatment groups: Control group: animals received distilled water orally for 14 days (
Behavioral tests
Elevated plus maze tests
Anxiety-related behavior of animals were assessed using the elevated plus maze test, as described previously.
25
The elevated plus maze consisted of two opposite open arms (50 × 10 cm2), crossed with two closed arms (50 × 10 cm2) with 40 cm high walls. The arms were connected by a central square (10 × 10 cm2), and the maze was elevated 50 cm above the floor. At the beginning of the test, each rat was placed on a central platform facing an open arm. The number of entries and the time spent in the closed and open arms during a 5-min test session were recorded by a blinded observer. As a parameter of anxiety level, percentage of number of entries into open arms and the percentage of time spent in the open arms for each animal were calculated.


Modified forced swimming test
The modified forced swimming test (MFST) was performed as described elsewhere. 26,27 Each rat was forced to swim in a glass cylinder. Twenty-four hours prior to the test, a pretest session was conducted for 15 min with no behavioral observation. During the test, swimming (horizontal movement on the surface of the water), climbing (upward-directed movements of the forepaws along the side of the cylinder), and immobility (movement required just to keep the head above the water) time of rats over a 5-s interval during 5 min were recorded using a stopwatch.
Activity cage tests
The spontaneous locomotor activities of rats were registered by the activity cage apparatus (Ugo Basile, No.7420, Varese, Italy). Interruptions of light beams to the photocells during horizontal and vertical movements of the rats were automatically recorded. 25,27
Rota-rod tests
Motor performances of rats were assessed by rota-rod tests as described previously. 28 Each rat was placed to the rota-rod apparatus (Ugo Basile, No. 47600, Italy), and latency to fall from the rotating mill was recorded as a criterion of motor coordination.
PTZ-induced seizures
To evaluate the seizure potential of animals, pentylenetetrazole (PTZ)-induced seizure test was performed. PTZ at a dose of 90 mg/kg was administered to the rats to induce seizures. The animals were observed immediately after PTZ injection for a period of 30 min. The latency (seconds) to the first seizure was recorded. 29
Following the behavioral tests, the animals were killed by decapitation with a guillotine, brain tissues were dissected and were frozen as quickly as possible in liquid nitrogen.
The brain tissues were washed with phosphate-buffered saline (PBS), pH 7.4. They were diluted at a ratio of 1:20 (w:v) with PBS solution and were homogenized. The homogenates were centrifuged at 10000
The supernatants were used to determine GABA, glutamate, serotonin, dopamine, adrenaline, noradrenaline, and GSH levels with a standard analysis method by high-performance liquid chromatography (HPLC) and CAT, SOD, and MDA levels by ELISA kits.
Determination of brain neurotransmitter levels
Determination of GSH, glutamate and GABA
Compounds were measured as their OPA/βME derivatives according to Lunn and Hellwig. 30 Derivatization was performed by mixing 9 μL of derivatization solution (5 mM OPA and 2 mM βME in 0.1 M borate buffer; pH 9.3) with 12 μL microdialysate and by holding 1 min before injection. The procedure was performed automatically by an autosampler. The derivatives were detected at the excitation wavelength of 230 nm and the emission wavelength of 450 nm. The analytical column was Zorbax, Extend-C18 (150 × 3 mm2, particle size 3.5 μm). A gradient elution consisting of solvent A: (methanol:acetonitrile:40 mM potassium phosphate buffer; pH 6.7; 20:2:78) and solvent B: (methanol:acetonitrile:40 mM potassium phosphate buffer; pH 6.7; 50:10:40) was used. The gradient elution was: 0–4 min, 0% B; 4–15 min, linear from 0% to 100% B; 15–20 min, holding 100% B; 20–25 min, linear from 100% to 0% B; 25–28 min, initial conditions (0% B) for equilibration of the column. Efficient and symmetrical peaks were obtained at 30°C at a flow rate of 0.6 mL min−1 with a sample injection volume of 20 μL after the derivatization procedure.
Determination of adrenaline, noradrenaline, dopamine, and serotonin
Adrenaline, noradrenaline, dopamine, and serotonin were analyzed with ion-pair liquid chromatography using an electrochemical detector. The analytical column (C18, 50 × 3 mm2, 3 μm particle size) and the isocritic mobile phase (0.03 M monosodium phosphate pH 4.11, 2.4 × 10−3 M sodium dodecyl sulfate, 6 × 10−5 M ethylenediaminetetraacetic acid, 7 % methanol) were used for chromatographic separation. The flow rate of the mobile phase, column temperature, and the ejection volume of the samples were 0.8 mL min−1 and 35°C and 10 μL, respectively. For the monitoring of the compounds, an electrochemical coulometric detector with graphite carbon cells was used. A guard cell was placed just before autosampler in the HPLC system in order to decrease the background noise and to increase sensitivity. The working cell and the guard cell were applied with 400 mV and 600 mV oxidation potentials, respectively. Under the analysis condition, retention time of the adrenaline, noradrenaline, dopamine, and serotonin were recorded as 2.12, 3.19, 8.05, 10.02, and 13.23 min, respectively.
Biochemical measurements
The supernatants were assayed for CAT, SOD, and MDA activity according to the manufacturer’s instructions.
Statistical analysis of data
The data used in statistical analyses were acquired from 10 animals for each group. Statistical evaluation of the experimental data was performed using GraphPad Prism 4.03 software (GraphPad Software, San Diego, California, USA). Experimental data were analyzed by one-way analysis of variance, followed by Tukey’s test. Experimental results were expressed as mean ± standard error of mean. Differences between given sets of data were considered as significant when the
Results
Effects of AMX in elevated plus maze tests
Figure 1 illustrates the effects of the oral AMX treatments on anxiety-related parameters of rats in the elevated plus-maze test. AMX administrations did not significantly change the POAE and PTOA values of the rats at any of the tested doses (25 or 50 mg/kg; Figure 1).

Effects of the AMX (25 and 50 mg/kg) administration on the (a) POAE% and (b) PTOA% in the elevated plus maze test. Values are given as mean ± SEM. One-way ANOVA, post hoc Tukey’s test. AMX: amoxicillin; POAE = number of open arm entries/number of total arm entries) × 100; PTOA = (time spent in the open arm/time spent in total arms) × 100; ANOVA: analysis of variance; SEM: standard error of mean.
Effects of AMX in MFSTs
The effects of 25 and 50 mg/kg AMX administrations on the immobility, swimming, and climbing time of rats in the MFST are shown in Figure 2. AMX treatments caused a significant increase in the immobility time of animals. Furthermore, swimming time of these animals significantly decreased after 25 and 50 mg/kg doses of AMX administrations with respect to control group. On the other hand, AMX administrations did not significantly change the climbing durations of rats. There was no statistical difference between the AMX-administered groups (Figure 2).

Effect of the AMX (25 and 50 mg/kg) administration on the (a) immobility time, (b) swimming time, and (c) climbing time of rats in the MFST. *
Effects of AMX in the activity cage tests
Figure 3 shows the effects of the AMX treatments on spontaneous locomotor activities of rats in the activity cage test. Application of AMX at 25 and 50 mg/kg doses caused a significant decrease in the numbers of both horizontal and vertical locomotor activities compared with the control group. There was no statistical difference between the AMX-administered groups (Figure 3).

Effects of the AMX (25 and 50 mg/kg) administration on the (a) number of horizontal movement and (b) number of vertical movement in the activity cage test. *
Effects of AMX in the rota-rod tests
Effects of AMX on motor coordinations of rats in the rota-rod tests are demonstrated in Figure 4. AMX treatments did not change the falling latencies of the rats from the rotating mill at none of the applied doses (Figure 4).
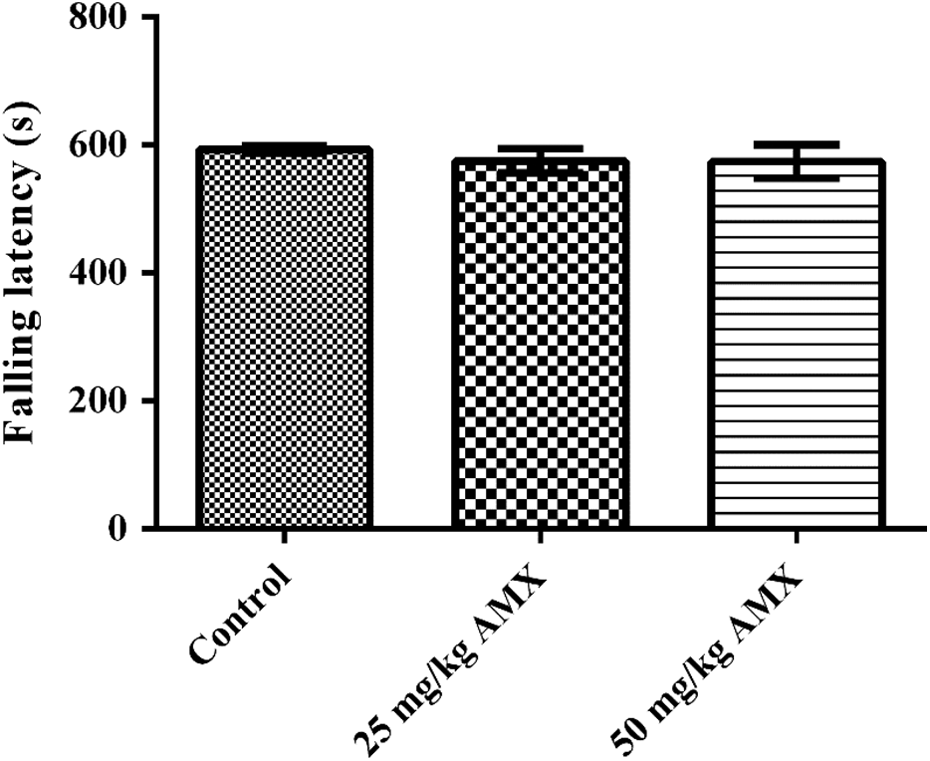
Effects of the AMX (25 and 50 mg/kg) administration on falling latencies of rats in the Rota-rod test. Values are given as mean ± SEM. One-way ANOVA, post hoc Tukey’s test. AMX: amoxicillin; SEM: standard error of mean; ANOVA: analysis of variance.
Effects of AMX on PTZ-induced seizures
Figure 5 shows the effects of the oral AMX treatments on PTZ-induced seizures. 25 mg/kg and 50 mg/kg AMX administration significantly reduced the onset of PTZ-induced convulsions compared with control group. There was no statistical difference between the AMX-administered groups (Figure 5).

Effects of the AMX (25 and 50 mg/kg) administration on the latency to the first seizure in the PTZ-induced seizure test. *
Neurotransmitter content in the brain
The effect of 25 and 50 mg/kg AMX administrations on the dopamine, noradrenaline, adrenaline, serotonin, glutamate, and GABA levels measured in the brain homogenates is presented in Figures 6 and 7.

Effects of the AMX (25 and 50 mg/kg) administration on the brain (a) adrenaline, (b) noradrenaline, (c) dopamine, and (d) serotonin content of rats. *
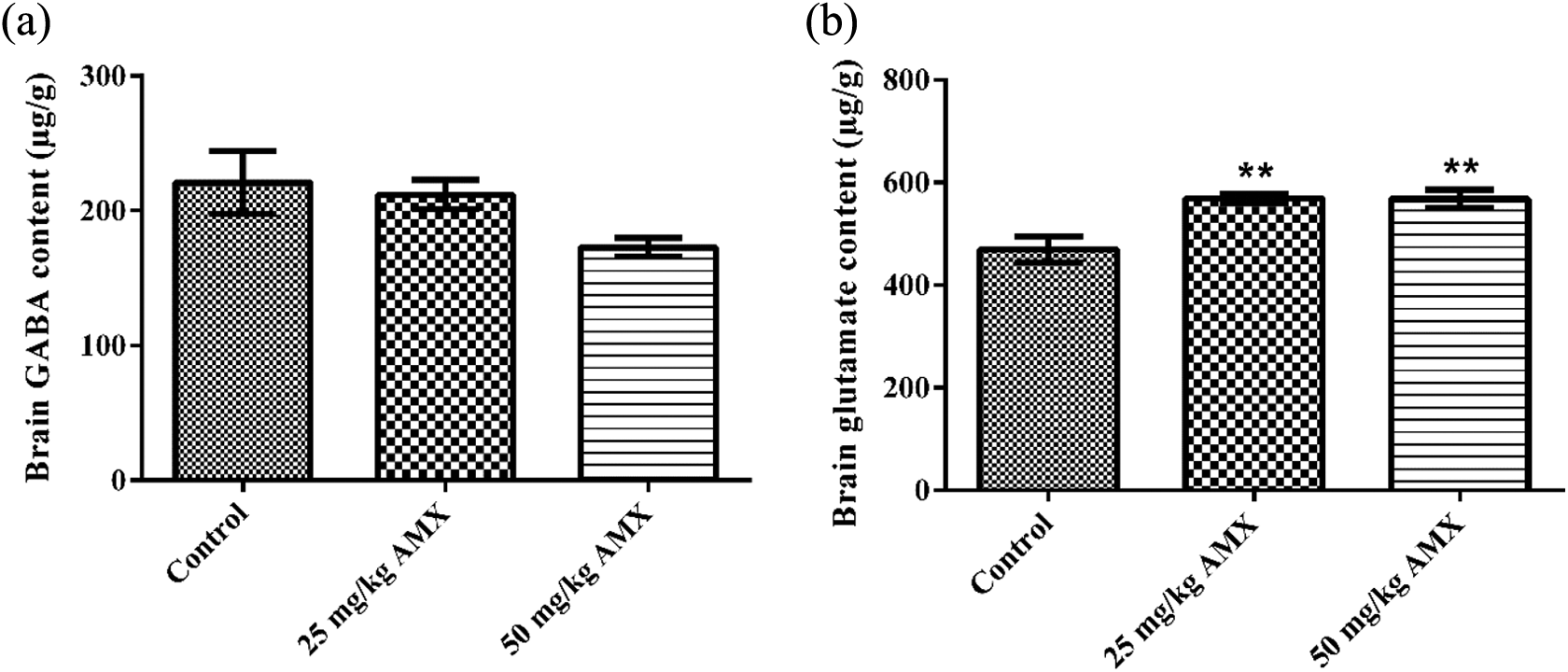
Effects of the AMX (25 and 50 mg/kg) administration on the brain (a) GABA and (b) glutamate content of rats. **
AMX administration caused no alteration in the brain dopamine, noradrenaline, and adrenaline levels compared to their respective control groups (Figure 6). HPLC analysis exhibited that serotonin levels of brain homogenates displayed significant decreases, 48.66% and 52.94% in 25 and 50 mg/kg AMX-treated groups compared with the control group, respectively (Figure 6). Glutamate levels in brain homogenates of animals treated with 25 and 50 mg/kg AMX showed significant increases of 21.27% and 21.12%, respectively, when compared with the control values (Figure 7). On the other hand, there was no significant difference in the brain GABA content of animals treated with AMX (Figure 7).
Biochemical measurements
MDA level
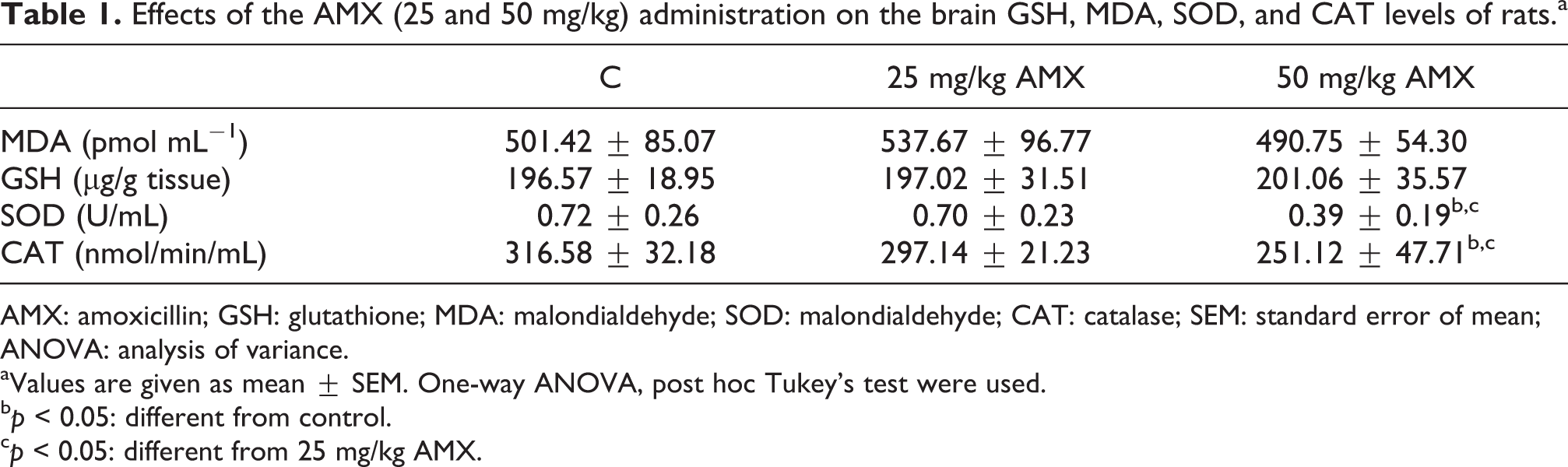
There was no significant difference in brain MDA levels of AMX-treated groups compared to the levels of control group. Also, no significant differences were found between the two treatment groups (Table 1).
Effects of the AMX (25 and 50 mg/kg) administration on the brain GSH, MDA, SOD, and CAT levels of rats.a
AMX: amoxicillin; GSH: glutathione; MDA: malondialdehyde; SOD: malondialdehyde; CAT: catalase; SEM: standard error of mean; ANOVA: analysis of variance.
aValues are given as mean ± SEM. One-way ANOVA, post hoc Tukey’s test were used.
b
c
GSH level
Brain GSH levels did not change significantly in AMX-treated groups compared to the control group. Also, no significant differences were found between the two treatment groups (Table 1).
SOD level
Brain SOD levels were found to be significantly decreased in 50 mg/kg AMX-treated group compared with the control rats. Also, the decrease in 50 mg/kg AMX-treated group was significantly different compared to 25 mg/kg AMX-treated group (Table 1).
CAT level
CAT activity was found to be significantly lower in 50 mg/kg AMX-treated rats compared with the control rats. Also, the decrease in 50 mg/kg AMX-treated group was significantly different compared to 25 mg/kg AMX-treated group (Table 1).
Discussion
Adverse drug reactions can severely affect children when compared to adults and lead to significant morbidity in children. In a previous study, antibiotics were reported to represent the mostly associated drug group with adverse reactions (67%) in children. These antibiotics were vancomycin, cloxacillin, AMX, ampicillin, meropenem, ciprofloxacin, and cefixime. 31 At this point, the present study was designed to determine the neurotoxic effects of repeated amoxicillin administration at clinically relevant doses in juvenile rats.
MFST is a very useful test to predict depression levels in rodents and also allow to make correlations between analyzed behaviors and different neurotransmitter systems. 32,33 In MFST, increase in immobility time indicates a depression-like response, whereas decrease in immobility time points out an antidepressant-like effect. 26 In our study, AMX administration significantly decreased the swimming time, while the duration of immobility increased without any change in the climbing durations of juvenile rats in MFST. Prolonged immobility and shortened swimming time was observed by AMX administration, which indicated a depression-like behavioral response. In MFST, climbing behaviors of rodents were widely associated with catecholaminergic system, whereas swimming behaviors were associated with serotonergic system of the brain. 26,32 Matching to our results at this point, the serotonin levels in the groups which received AMX significantly decreased. On the other hand, AMX administration did not have any significant effect on the levels of noradrenaline and dopamine.
The elevated plus maze test depends on tendency of rodents toward dark, enclosed spaces (approach) and their unconditioned fear of heights/open spaces (avoidance). The anxiety behavior of rodents was assessed by evaluating POAE and PTOA values. 20,34 Anxiolytic agents increase POAE and PTOA parameters in this test; however, anxiogenics decreases both of these parameters. 35 In our study, we determined that AMX did not affect POAE and PTOA parameters of animals. These findings showed that an anxiety-like or anxiolytic-like behavior did not develop in the AMX-treated groups. As a matter of fact, GABA, noradrenaline, and dopamine levels, which are associated with anxiety disorders, 36,37 did not change significantly in AMX-treated groups compared with the control group. Results of the HPLC analysis, indicating unchanged GABA, noradrenaline, and dopamine levels, supported the data obtained from elevated plus maze tests.
The findings obtained from the activity cage test showed that the horizontal and vertical locomotor activities of the groups that received AMX were significantly decreased. Generally, increased dopaminergic neurotransmission results in increased locomotor activity, and decreased dopaminergic neurotransmission causes declined locomotor activity. 38 –40 However, in our study, decreased locomotor activity of rats could not be explained as a consequence of any change in dopaminergic neurotransmission. It may be considered that repeated administration of AMX might affected dopaminergic neurotransmission by causing an alteration on receptor level or on post receptor processes. The exact mechanism of this effect should be clarified with further detailed studies.
Data acquired from the AMX-treated animals in rota-rod tests were not significantly different with respect to control animals. Absence of alterations in the rota-rod activity of juvenile rats indicates AMX administration did not change the motor coordination of animals or induce a nonspecific effect at neuromuscular junction level. Therefore, it can be concluded that, in this study, the findings of the behavioral tests were not affected by any alterations in the motor performance of the rats in this study.
Effects of AMX administration on seizure potentials of animals were assessed by PTZ-induced acute convulsive model. PTZ is a chemical agent that has been widely used in experimental models for inducing seizures. 41 A significant decrease in the latency to the first seizure-induced by PTZ in the juvenile rats with AMX administration was an important neurotoxicity finding in our study. It has been reported that, in a child, seizure-characterized encephalopathy syndrome occurred with AMX treatment. 14 As a general knowledge, GABA is the most important inhibitory neurotransmitter in the central nervous system and also one of the key Neurotransmitters in epilepsy disease. In contrast, glutamate is an important excitatory neurotransmitter in the brain and also plays an important role in the development of epilepsy. Moreover, previous studies have underlined the fact that the imbalance of GABA/glutamate in the brain is a very important factor in convulsions. 42,43 In a study, which investigated the basic mechanisms of epileptogenic potential of penicillins in mature animal brain by microdialysis, it was observed that all of the excitatory (glutamate and aspartate) and inhibitory neurotransmitter levels (GABA, glycine, and taurine) were increased in the interstitial cerebrospinal fluid after focal application of penicillin. 44 In another study, the excitatory responses to glutamate and the inhibitory responses to GABA in neurons were found to be decreased during penicillin-induced epileptiform activity. 45 At this point, in our study, as an important finding, the glutamate levels in brain homogenates increased significantly in AMX-treated groups, whereas GABA levels did not change significantly. Therefore, it can be emphasized that the induction of glutaminergic neurotransmission is effective in shortening the latency to the first seizure.
Normally, cells can tolerate moderate oxidative status by their defense system and restore the oxidant/antioxidant balance. Several antioxidant enzymes, such as SOD, CAT, and glutathione peroxidase scavenge reactive oxygen species accumulated in the cells to maintain cellular homeostasis and prevent injury. However, in the conditions when the oxidative load exceeds the antioxidant capacity, the oxidant/antioxidant imbalance occurs, and the result is oxidative stress leading to cellular damage. 46 –49 Oxidative stress is determined by the increased levels of oxidation products like MDA and/or decreased levels of antioxidant enzymes such as CAT, SOD, glutathione peroxidase in cells. Also, increased oxidant/antioxidant ratios like MDA/GSH, MDA/SOD, and so on are indicators of an increased oxidative stress. 47 –49 In the present study, it was shown that AMX-associated depression-like behavior and decreased latency to the first seizure in juvenile rats were accompanied with oxidative stress in brain tissues. These effects were reflected by the decreased activities of SOD and CAT causing higher oxidant/antioxidant ratios in brain tissues of high dose AMX-administered rats compared with the control rats. The central nervous system is extremely vulnerable to oxidative damage since it is rich in oxidizable substrates and has high oxygen tension and limited amount of antioxidant capacity. There are increasing evidences, both in experimental and clinical studies, suggesting that oxidative stress plays an important role in the pathophysiology of depression 18,50,51 and epilepsy. 52 –54 There are numerous reports indicating disturbed equilibrium between oxidative processes and antioxidant defense in the progression of seizures and depression. 17,19,20,51 Also, in our study, AMX-induced oxidative stress in brain tissue in juvenile rats, whose blood–brain barrier is not fully developed, may be one of the possible underlying mechanisms for observed depression-like behavior and decreased latency to the first seizure.
In conclusion, it has been shown that the AMX treatment triggered depression and decreased latency to the first seizure in juvenile rats. It was also determined that AMX induced oxidative stress and damaged the protective antioxidant defense. As a result of the frequent and widespread use of AMX, clinicians should be aware of its neurotoxicity, particularly in patients who have risk factors for neurotoxic effects. Additional studies such as retrospective analysis of neurotoxic effects in patients under AMX treatment are needed before concluding whether AMX is safe to use in children.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
