Abstract
Cyclophosphamide (CYP) induces urothelial injury and causes excretion of cellular exudates at 24 h, followed by rapid restoration at 72 h. We investigated the role of urinary uroplakin II (UPII) levels in a CYP-induced cystitis model. For the purpose of this study, 10 controls and 26 CYP-injected female Sprague Dawley rats were killed at 24 h and 72 h postinjection. The vesical weight, severity of hematuria, and expression of UPII in the urinary bladder and urine were measured. CYP decreased the level of vesical UPII messenger RNA at 24 h, followed by rapid recovery at 72 h. Contrary to the negligible levels of urinary UPII and hematuria in controls, CYP treatment abruptly increased the excretion of urinary UPII at 24 h. The excretion had subsided at 72 h. Similarly, severe hematuria was observed at 24 h, with improvement at 72 h. However, some rats still exhibited hematuria at 72 h. CYP caused increase in vesical weight. The vesical weight at 24 h after CYP injection was negatively correlated with the vesical UPII level. Rats with significant hematuria demonstrated higher urinary UPII levels than those with insignificant hematuria. Vesical UPII could be an important barrier for early CYP-related injury, while the levels of urinary UPII may be associated with the severity of hematuria during dynamic periods in the urothelium.
Introduction
While cyclophosphamide (CYP) has been used as an anticancer drug or immunosuppressive agent, its main clinical limitation is that it induces cystitis and acute hemorrhage. 1 –4 In recent studies, we reported that animals injected with CYP had transient reduction the in expression of vesical uroplakins (UPs) at 24 h post CYP injection, followed by rapid restoration within 72 h after injection. UPs form a specialized barrier compartment in the urothelium, one of the tightest and most impermeable barriers in the body. 4 –7 The reduction of UP expression may create transient disturbance of barrier function in the urinary bladder, permitting the leaking of urine into the vesical submucosa, with consequent exposure to CYP metabolites in the blood vessels. 4 –6
The lower expression of UPs in the urinary bladder after CYP injection may theoretically be caused by decreased production in the urothelium and increased urinary excretion. Electron microscopic studies, immunochemical staining, and immunoblotting studies revealed that the expression of UPs is generally depressed along the urothelium upon CYP injection. 4 –6,8 Interestingly, we also found that much of the UPs stained on urothelial cells were detached from the bladder mucosa and excreted into vesical lumen. 4 Although the urinary loss of UP in urothelial cells may be an important clue for lower expression of vesical UPs, urinary excretion of UPs was not evaluated in the previous studies. 4 –6
To overcome CYP-related clinical complications, many researchers have used animals for studying the basic mechanisms of injury and regenerative processes in the urothelium. 4 –6,8 –10 To infer significance from the results of previous studies, some issues must be settled. First, objective parameters for evaluating the severity of vesical toxicity in CYP-injected animal models must be established. Second, specific samples need to be collected through noninvasive methods to examine the mechanisms of CYP-associated toxicity. Finally, the significance of hematuria must be emphasized. 11 Unfortunately, previous studies mainly evaluated the grade of vesical hemorrhage instead of the severity of hematuria in the CYP-induced rat cystitis model. 4 –6,9,10 Therefore, the objectives of this study were to investigate the expression of UPII in urine and the urinary bladder during the injury and repair period in the CYP-induced rat cystitis model. In addition, we also evaluated the correlations between the objective parameters for CYP-related toxicity and the expression of UPII in urine or the urinary bladder.
Materials and methods
All procedures were performed in accordance with the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The animal study protocol was approved by the Animal Care Committee of Dankook University.
Animals
Thirty-six female Sprague-Dawley rats with body weights of about 180 g were randomly divided into three groups: one negative control and two CYP-injected groups (GR-24, killed at 24 h post CYP injection; and GR-72, killed at 72 h post CYP injection), with 10, 12, and 14 rats randomly allocated to the control, GR-24, and GR-72 groups, respectively. After anesthetizing with intramuscular injections of ketamine (15 mg/kg) and xylazine (5 mg/kg), the rats in each of the CYP treatment groups received an intraperitoneal injection of 150 mg CYP/kg (Sigma-Aldrich, St Louis, Missouri, USA) dissolved in 1 mL of distilled water. Rats in the control group received an intraperitoneal injection of 1 mL of distilled water as a sham. 4 –6
Rats in GR-24 were killed at 24 h postinjection with CYP, while the rats in GR-72 and the control group were killed at 72 h postinjection after injection with the same dosage of ketamine and xylazine listed above. Urine in the urinary bladder was evacuated by pressing on the lower abdomen and collected into a 1.5 mL tube. A 10 µL aliquot of the collected urine was then soaked in the block of occult blood on a dip strip (Uropaper-III, Shinyang Chemical, Seoul, Korea) and incubated for 30 s. Color change was recorded as negative (−), trace (±), 1+, 2+, and 3+ following the manufacturer’s instructions. For easy handling of the data, the severity of hematuria at 24 h postinjection with CYP was reclassified as insignificant for <2+ and significant for grades of ≥2+. Severity of hematuria at 72 h postinjection with CYP was also reclassified, with <1+ indicating insignificant hematuria, whereas grades ≥1+ on the dip strip were reclassified as significant hematuria.
Rat bladders were severed at the level of the bilateral ureterovesical junctions to harvest the fundus or detrusor portion. After removal of the remaining urine in the bladder with a cleansing tissue, the severed bladders were weighed. Each harvested bladder was then circumferentially cut into three equal pieces. Two-thirds of the portions were stored at −80°C for measurement of UPII messenger RNA (mRNA) and protein. The remaining one-third was fixed for 24 h in 4% paraformaldehyde for hematoxylin and eosin (H & E) staining and immunohistochemistry. Finally, all rats were killed by guillotine.
Determination of gene expression of UPII and β-actin by RT-PCR
The urine samples were centrifuged in 1.5 mL centrifuge tubes at 13,000 r/min for 10 min at 4°C. After discarding the supernatant, the sediments were resuspended with TE buffer (Tris–ethylenediaminetetraacetic acid) and recentrifuged at 13,000 r/min for 10 min at 4°C. After discarding the second supernatant, total RNA was extracted from the second pellets using Trizol reagent (Invitrogen, Carlsbad, California, USA) and chloroform, according to the manufacturer’s instructions. Total RNA was also extracted from each bladder specimen as described previously. 4 –6 The same polymerase chain reaction (PCR) primers for UPII and β-actin were used as in the previous reports. 5,6 The ImProm-II™ Reverse Transcription System (Promega, Madison, Wisconsin, USA) was used for preparation of complementary DNA, according to the manufacturer’s instructions. To convert the cycle threshold (C t) values from real-time (RT) PCR to relative concentration values, previously developed plasmid constructs for UPII and β-actin were used for standardization. 6 The materials and methods used for obtaining plasmid controls were described in our previous report. 6 To estimate mRNA expression, calibration curves were made from the measured fluorescence of dilution series of the control plasmids, as previously described. 6
Two microliters of the standard plasmid was added to a 25 µL reaction mixture containing forward primer (300 nM), reverse primer (300 nM), 0.5 unit of uracil-N-glycosylase (Sigma-Aldrich), and 2X QuantiTech SYBR Green PCR Master Mix (Qiagen GmbH, Hilden, Germany). RT-PCR was performed in a Rotor-Gene 6.0 (Corbett, Sydney, Australia), according to the manufacturer’s instructions. Amplification consisted of 2 min at 50°C for carryover prevention, 15 min at 95°C, and then 40 cycles of 10 s denaturation at 95°C, annealing at 52°C for 15 s, and extension at 72°C for 20 s. Melting curve analysis and the desired concentration value was achieved. Negative controls were also included. 6 The detected C t values were interpolated using equations calibrated from the standard curves of plasmid constructs for calculating of the mRNA concentration in each samples. 6 For normalization of the UPII expression in the samples, each UPII concentration was divided by the matched β-actin concentration.
Immunohistochemical and histopathological evaluations
Bladders were fixed in 4% paraformaldehyde solution and embedded in paraffin. The paraffin blocks were then deparaffinized in xylene and alcohol. After antigen retrieval by heating the specimens, immunolabeling with anti-UPII polyclonal antibody (Santa Cruz Biotechnology, Santa Cruz, California, USA) was performed in 10% skim milk at 1:100 dilution. Rabbit anti-goat immunoglobin horseradish peroxidase (HRP; Zymed, South San Francisco, California, USA) at 1:500 dilution was used for the secondary antibody. Peroxidase activity was demonstrated with 3,3′-diaminobenzidine. The sections were counterstained with Mayer hematoxylin, dehydrated, and then mounted. The paraffin-embedded samples were stained with H & E. 4 –6
Western blotting
Whole bladder lysate was prepared in lysis solution. The amounts of loaded proteins for UPII were 30 µg each sample. Proteins were separated by 15% sodium dodecyl sulfate–polyacrylamide gel electrophoresis, followed by transfer to nitrocellulose membranes. The membranes were incubated with goat polyclonal primary antibody (UPII, 1:1000 dilutions; Santa Cruz Biotechnology) overnight at 4°C, followed by washing and incubation with the secondary antibody (donkey anti-goat immunoglobulin G HRP polyclonal antibody, 1:2500 dilution, Santa Cruz Biotechnology). After enhancement with luminol reagent (Santa Cruz Biotechnology) for 1 min, the membranes were exposed. Blot pictures were obtained using an LAS-3000 image reader (Fujifilm, Tokyo, Japan). 4 –6
Statistical analyses
All data were expressed as mean ± standard deviation. Significant differences in the levels of UPII and β-actin among experimental groups were determined by a nonparametric test (Kruskal–Wallis analysis for three groups and Mann–Whitney U test between two groups). The differences in UPII and β-actin gene expression levels between the group with significant hematuria and the insignificant group were also evaluated. Correlation analysis between vesical tissue weight and gene expression level was calculated with Pearson’s correlation coefficient. Two-sided statistical tests were used in all analyses. All analyses were performed using SPSS software for Windows (version 11; SPSS, Chicago, Illinois, USA). Statistical significance was considered when p values were less than 0.05.
Results
Vesical weight
Vesical weights in the control, GR-24, and GR-72 were 0.067 ± 0.013, 0.123 ± 0.032, and 0.106 ± 0.029 g, respectively. There were significant differences in vesical weight among the three groups (Kruskal–Wallis; p = 0.001; Figure 1(a)). While CYP-injected rats displayed significant increase in the vesical weight compared with the control, no significant differences were observed between GR-24 and GR-72 (Mann–Whitney U test; p = 0.202; Figure 1(a)).
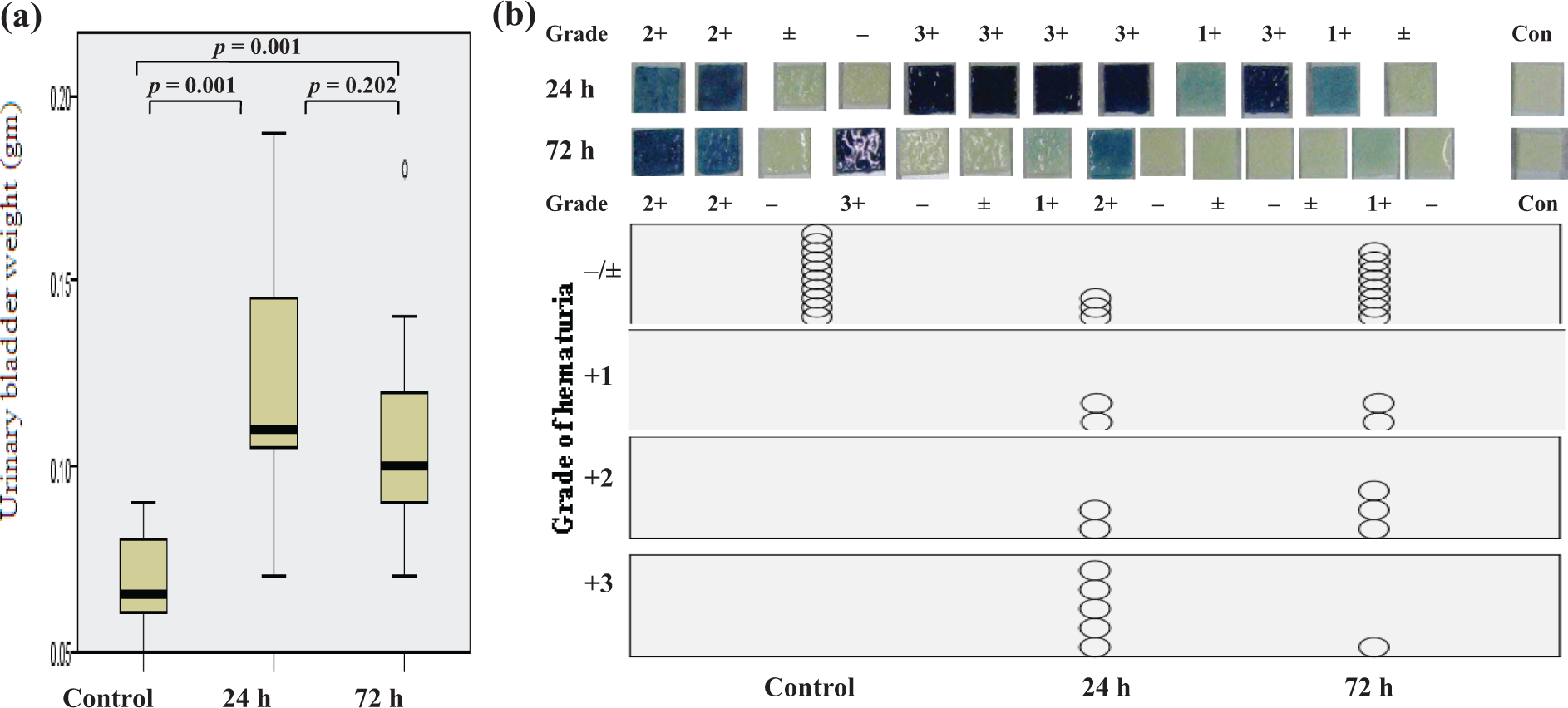
(a) Significant difference in vesical weight among the three groups (Kruskal–Wallis, p = 0.001). While the CYP-injected rats revealed significant increase in urinary bladder weight compared with the control, no definitive differences in vesical weight were observed between the CYP-injected groups (Mann–Whitney U test, p = 0.20 (a)). The control revealed no hematuria on the blood block of a dip strip. Hematuria may be more severe at 24 h postinjection with CYP, followed by improvement at 72 h (b). However, the CYP-injected rats revealed various grades of hematuria in a time-dependent and object-specific manner (b). Outliers were shown as circles or stars outside the box. CYP: cyclophosphamide.
Grade of hematuria on dip strip
The control group did not display hematuria on the blood block of the dip strip. Hematuria was generally more severe at 24 h postinjection with CYP but improved at 72 h postinjection (Figure 1(b)). However, the CYP-injected rats had various grades of hematuria that were time dependent and object specific (Figure 1(b)).
Histopathological evaluation
The single injection of CYP caused initial extensive vesical injury, which was followed by a repair process. However, there were not only individual or regional variations in destructive injury at 24 h postinjection with CYP but also variations in the regenerative process at 72 h postinjection (Figure 2(a) and (b)). Immunochemical reaction with anti-UPII antibody revealed that there was a significant decrease or loss of UPII expression along the urothelium at 24 h postinjection with CYP as well as a significant cellular loss through the bladder cavity. However, the expression of UPII was restored at 72 h. There were very few cellular exudates in the vesical cavity (Figure 2(d)).

Histological analysis of cross sections of representative cases (a, b); hematoxylin–eosin, ×12.5). The CYP-injected rats showed individual or regional variations in regenerative period at 72 h after CYP injection (×12.5). The upper left figure (a) revealed improved cystitis findings, while the upper right figure (b) revealed submucosal edema and hemorrhage. (c, d) Immunohistochemical staining with anti-UPII antibody. (c) At 24 h after CYP injection, exfoliated urothelial cells in the urinary bladder and decreased UPII expression along the urothelium were observed (×200). (d) The UPII expression was completely recovered. No detached urothelial cells could be found in the vesical cavity at 72 h (×12.5). Scale bars represented actual size. CYP: cyclophosphamide; UPII: uroplakin II.
UPII and β-actin mRNA expression in urine
Expression of UPII mRNAcould not be detected in the urine from the control rats. In contrast, all of the GR-24 and GR-72 rats had significant UPII expression in the urine (log-transformed value of 2.73 ± 1.07 for the GR-24 and 1.57 ± 1.01 for GR-72). The urinary UPII excretion was significantly higher at 24 h postinjection with CYP but decreased at 72 h (Mann–Whitney U test, p = 0.015; Figure 3(a)). Similar results were obtained for urinary β-actin excretion (log-transformed value of 2.80 ± 0.44 for GR-24 and 2.09 ± 0.34 for GR-72). The urinary β-actin excretion was significantly higher at 24 h post CYP injection, with lower values at 72 h (Mann–Whitney U test, p = 0.001; Figure 3(b)). Normalized UPII expression (uroplakin II expression/the matched β-actin expression in log-transformed value) levels in urine were 0.96 ± 0.31 for GR-24 and 0.73 ± 0.44 for GR-72 (Mann–Whitney U test, p = 0.145; Figure 3(c)).

Urinary UPII and β-actin excretion using real-time polymerase chain reaction and normalized urinary UPII expression by β-actin expression. Urinary UPII mRNA expression was not detected in the control. (a) Urinary UPII excretion was significantly higher at 24 h postinjection with CYP and decreased at 72 h (Mann–Whitney U test, p = 0.015). (b) Similarly, the urinary β-actin excretion was significantly higher at 24 h postinjection with CYP and lower at 72 h (Mann–Whitney U test, p = 0.001). (c) However, the normalized UPII expression levels in the urine did not significantly differ (Mann–Whitney U test, p = 0.145). CYP: cyclophosphamide; UPII: uroplakin II; mRNA: messenger RNA.
Vesical UPII and β-actin expression
Log-transformed vesical expression values of UPII were 5.73 ± 1.10 for the control, 2.92 ± 2.43 for GR-24, and 4.81 ± 2.51 for GR-72 (Kruskal–Wallis, p = 0.028; Figure 4(a)). The CYP injection induced a significant reduction of the vesical UPII expression at 24 h (Mann–Whitney U test, p = 0.011), which was restored at 72 h (Mann–Whitney U test, p = 0.036; Figure 4(a)). However, the log-transformed vesical expression values of β-actin were 4.07 ± 0.64 for control, 2.64 ± 1.54 for GR-24, and 3.62 ± 1.58 for GR-72. No statistical significance could be found in vesical β-actin expression among the three groups (Kruskal–Wallis, p = 0.71; Figure 4(b)). The normalized UPII levels in the urinary bladder tissue did not differ significantly different among the three groups (Kruskal–Wallis, p = 0.118). Overall, the protein expression of UPII was significantly decreased at 24 h but restored at 72 h postinjection with CYP. However, individual variation in UPII protein expression at 24 h postinjection with CYP was observed (Figure 4(c)).

Statistical significance in vesical UPII expression among the three groups (Kruskal–Wallis, p = 0.028). (a) CYP injection induced a significant reduction of vesical UPII expression at 24 h (Mann–Whitney U test, p = 0.011), which was restored at 72 h (Mann–Whitney U test, p = 0.036). (b) However, no statistical significance in vesical β-actin expression and the normalized UPII level could be found among the three groups (Kruskal–Wallis, p = 0.71 in β-actin and p = 0.118 in normalized UPII level). The protein expression of UPII was significantly decreased at 24 h and restored at 72 h postinjection with CYP. (c) However, individual variation in UPII protein expression was observed at 24 h after CYP injection (inset box). Arrow indicates UPII protein expression. Arrowhead represents β-actin as a housekeeping gene expression. M: marker; CYP: cyclophosphamide; UPII: uroplakin II.
Relationships between CYP-induced vesical toxicity and gene expressions
Vesical weight
Rats in the GR-24 group showed a significant negative correlation between the vesical UPII mRNA expression and the matched vesical weight (r 2 = 0.419, p = 0.023). A significant negative correlation between normalized vesical UPII mRNA expression and the matched vesical weight was also found in these rats (r 2 = 0.353, p = 0.042). However, no significant correlation between vesical weight and β-actin expression in urinary bladder or urine was found (Figure 5).
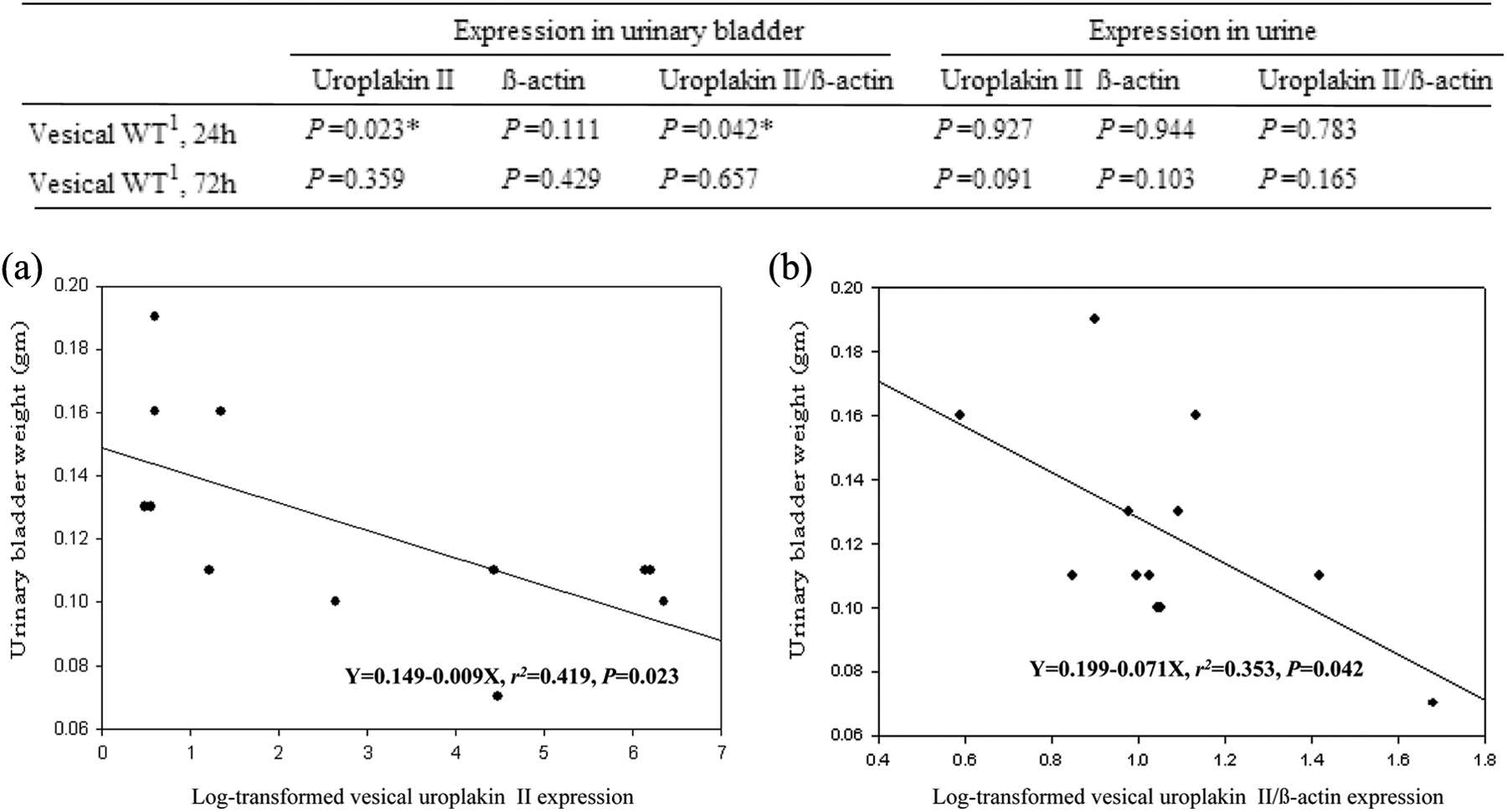
At 24 h postinjection with CYP, the rats showed a significant negative correlation between vesical UPII mRNA expression and the matched vesical weight (a). Similarly, rats revealed a significant negative correlation between normalized vesical UPII mRNA expression and the matched vesical weight (b). However, no significant correlation between the vesical weight and the β-actin expression in urinary bladder or urine could be found (upper table). *p < 0.05. WT: weight; CYP: cyclophosphamide; UPII: uroplakin II; mRNA: messenger RNA.
Hematuria
Contrary to the negligible levels of hematuria seen in controls, CYP treatment induced various grades of hematuria. Generally, the hematuria was severe at 24 h postinjection with CYP but improved at 72 h. Rats with significant hematuria demonstrated higher Urinary UPII mRNA (UUM) levels than those with insignificant hematuria (p = 0.003 at 24 h and p = 0.013 at 72 h). Urinary UPII levels also showed significant association with the severity of hematuria after normalization with β-actin (p = 0.005 at 24 h and p = 0.013 at 72 h). Although the vesical weight was not different between the groups with significant and insignificant hematuria at 24 h postinjection, significant difference was observed at 72 h postinjection. Vesical UPII expressions did not differ between the two groups of hematuria.
Discussion
We have reported the role of UPs as a specialized barrier compartment in the urothelium. The reduction of UP expression early in the injury period creates transient disturbance of barrier function in the urinary bladder, while the restored UP expression during the regenerative period may occur emergently to seal the urinary leakage into the vesical submucosa and stop the exposure of the underlying tissue or blood vessels to toxic metabolites of CYP. 4 –6,8,12
Many researchers have demonstrated the role of UPs in the urothelium via immunohisochemical staining or electron microscopic examinations in the CYP-induced cystitis model. 4 –6,8,12 We added biochemical results in our previous reports by demonstrating that CYP decreased UP mRNA and protein expressions in the urothelium of the urinary bladder. 4,5
Interestingly, during the early injured period, we found that many UP-stained urothelial cells were detached from the bladder mucosa and excreted into the vesical cavity (Figure 2(c)). 4 The excreted cells were obviously lost into the urine. Although the urinary loss of UPs in urothelial cells must be an important factor for evaluating the UP-associated mechanisms of the barrier system, its significance was not given attention in our previous studies. 4 –6 Furthermore, urine can be an ideal noninvasive specimen for studying the UP-mediated barrier mechanism in the urothelium. However, very few studies have used urine samples for the evaluation of barrier mechanisms.
Submucosal edema, mucosal detachment or necrosis, and hemorrhage are the typical cystitis findings in animal experiments after CYP injection. 4 –6,9,13 However, the morphologic findings differ from animal to animal and from region to region within the same animal. 4 –6,14 Because of individual or regional variation in the severity of the urinary bladder architecture after CYP injection (Figures 2(a) and (b) and 4(c)), the observation of severely injured lesions in a limited area cannot provide an objective or representative evaluation of toxicity. To overcome the limitation of this subjective parameter, we used whole vesical weight as the ideal parameter for evaluating the severity of cystitis in the animal model because it is not a focal, but an entire portion for evaluating the CYP-related toxicity. In addition, while the ordinal value has customarily been used for evaluating the severity score in animal cystitis models, 6,9 the scale value of the vesical weight provides another merit for statistical analysis.
The main clinical issues faced by CYP-injected patients are symptoms of extensive cystitis and hematuria. The significance of hematuria should be focused on because hematuria is a big issue in CYP-treated patients. 3,11,15 Unfortunately, many researchers evaluated the severity of hemorrhage in the urinary bladder instead of hematuria. 4 –6,9,10 However, there is no decisive proof that the grade of submucosal hemorrhage indeed serves as direct evidence of the severity of hematuria in CYP-induced toxicity experiments. For this reason, the severity of hematuria must be evaluated instead of hemorrhage because hematuria is an important factor for evaluating the major complications in CYP-induced toxicity.
Gene expression analyses must be endorsed with equal numbers of cells in each experimental group. However, since CYP induces rapid urothelial loss into the urine, controlling the number of cells in each sample is very difficult. Therefore, the raw data must be normalized with internal reference genes to get accurate and reliable readings of target gene expression in a specific experiment. 16 –18 Usually, “housekeeping genes” such as β-actin and glyceraldehyde-3-phosphate dehydrogenase can be chosen as reference genes because they theoretically display constant expression during the dynamic molecular conditions. We used β-actin as the housekeeping gene to normalize the UPII gene expression, as significant differences in vesical β-actin expression were not found among the three groups in this CYP-induced cystitis model (p = 0.71, Figure 4(b)).
While the normal turnover rate of the urothelium is very slow, CYP-injected rats were previously found to undergo rapid urothelial turnover. 8 The findings of negligible expression of urinary UPII in the control rats and increased levels of expression in CYP-injected rats are not surprising. The urinary loss of UPII was inversely correlated with the vesical UPII expression (Figures 3(a) and 4(a)). However, for the normalized urinary UPII expression, indicating the UPII expression in each urothelial cell, differences were not observed between GR-24 and GR-72 (Figure 3(c)).
In the early injured period, the weight of the urinary bladder correlated well with the expression of vesical UPII and the normalized vesical UPII value (Figure 5). However, this correlation in urinary UPII expression was not observed during the dynamic period (24 h and 72 h) or in vesical UPII expression at 72 h. These findings suggest that urothelial loss or UPII leakage through the urine may not be an important factor for determining vesical edema during the experiment. On the other hand, conserving the expression of the UPII gene in the urothelium during the early injury period might be an important factor for determining the vesical weight.
Many researchers have overlooked the significance of hematuria in animal experiments. They usually evaluated the CYP-related urological toxicity according to the grade of submucosal hemorrhage instead of the severity of hematuria in the CYP-induced rat cystitis model. However, hematuria is a big issue in CYP-treated patients. While many patients report transient gross hematuria, massive hematuria from a few patients that cannot be controlled by conservative treatments must be treated with more aggressive methods, such as selective embolization or even cystectomy. 11,15,19
In contrast to the negligible levels of hematuria in control rats, CYP induced various grades of hematuria on a case-by-case basis (Figure 1(b)). Generally, the hematuria tended to be severe at 24 h postinjection with CYP but improved at 72 h. Since the first day after CYP injection is a critical point in both clinical and animal study, 20,21 we divided the grade of hematuria into significant and nonsignificant levels. Significant hematuria was defined as those at grades ≥2+ on the dip strip at 24 h after CYP injection and those at grades ≥1+ on the dip strip at 72 h postinjection with CYP. In the early injured period, the vesical weight did not differ according to the severity of hematuria. This finding may be due to the fact that high levels of CYP metabolites in the urine at 24 h could induce severe destructive changes in the urothelium, subsequently inducing generalized hematuria. 20,21 In contrast, the significant hematuria group displayed heavier vesical weight than the nonsignificant hematuria group at 72 h.
The significant hematuria group consistently excreted higher levels of urinary UPII expression during the early destructive and late regenerative stages. Furthermore, some of the rats at 72 h still displayed excretion of hematuria during this regenerative phase. Additionally, the normalized UPII level in urinary exfoliated cells at 72 h was still higher in the group with significant hematuria (1.060 ± 0.454) than in the nonsignificant group (0.496 ± 0.259). For these reasons, we can presuppose that some rats with poor recovery in the regenerative phase, 72 h postinjection with CYP, presented with significant hematuria that was associated with urinary UPII excretion (Table 1).
Association between UP II expression and significant hematuria in CYP-induced rat cystitis model.

CYP: cyclophosphamide; WT: weight; UPII: uroplakin II; SD: standard deviation.
aData are expressed as log-transformed mean ± SD values.
b p < 0.05.
In conclusion, we investigated the role of UPII expression in the urine and the urinary bladder in a CYP-induced rat cystitis model. Vesical UPII could be an important barrier for early CYP-related injury. In addition, urinary UPII levels were associated with the severity of hematuria during dynamic periods (24–72 h) in the urothelium. In near future, the UP-associated molecular mechanisms in urinary exfoliated cells should be examined to improve the understanding of hematuria in the CYP-injected rat cystitis model. In addition, further investigation into the role of UP for cytoskeletons in the exfoliated urothelial cells is also needed.
Footnotes
Authors’ Note
HP and GL designed the experiment, collected the samples, extracted DNA from the samples, and performed real-time polymerase chain reaction. YSK designed the experiment and collected the samples from experimental animals. All the three authors performed the analysis of the data and contributed to writing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Research Fund of Dankook University in 2015.
