Abstract
Inhalant abuse is a globally prevalent health issue with particular concerns about substance-abusing pregnant women. In both animal models and clinical case reports of toluene exposure, the primary physiological outcome measure of prenatal inhalant exposure is low birth weight (BW). However, the effect of prenatal toluene exposure on animal BW varies widely in the literature. To clarify this effect and investigate possible design moderators of pup BW, a systematic review and meta-analytic techniques were applied to the existing peer-reviewed animal literature of prenatal and postnatal exposure models to the inhaled solvent toluene. Of 288 studies screened, 24 studies satisfied the inclusion criteria. Evaluation of these studies indicated that toluene exposure was negatively associated with pup BW (d = −0.39), with external inhaled concentration, route of administration, day of weighing, and toluene exposure magnitude moderating this association. Investigators doing animal studies should be cognizant of these factors before investigating the reproductive and developmental outcomes associated with prenatal and postnatal toluene exposure.
Introduction
The study of abused inhalants is as much a complex and multifaceted undertaking as exists in the world of substance abuse research. 1 Inhalants recognized to have abuse potential are found in thousands of registered products. 2 –5 While historically the misuse of inhalants was confined primarily to males, usage among females has increased to the point that at several ages, the abuse of volatile substances is now greater in females than in males of the same age. 6,7 As abuse of volatile substances is known to progress into adulthood, abuse by women in their reproductive years may also be increasing, 8 –10 elevating concerns of the potential detrimental effects of intentionally inhaled organic solvents on the developing fetus. 2,11 –15 Indeed, toluene-based solvents have been reported to be among the most frequently misused psychoactive substances during pregnancy. 16
While toluene does not carry the same stigma as reporting alcohol or illicit drug misuse during pregnancy, 2 it is highly lipid soluble, readily crosses the placenta, is absorbed into both fetal and maternal fat and brain tissue, and is a known teratogen. 17 –19 Indeed, the phrase fetal solvent syndrome is frequently used as a descriptor of the myriad of physical and behavioral effects that can develop following high levels of solvent exposure. 6,20 As with fetal alcohol syndrome, low birth weight (BW) is one of the most common physiological outcomes of prenatal solvent exposure in humans. 12,21,22 This makes BW a critical physiological marker for animal models of prenatal solvent exposure, alerting researchers to the effectiveness of their prenatal exposure technique. As such, it is a commonly reported descriptive statistic, if not necessarily the main outcome of the study as a whole. However, the effect of prenatal solvent exposure on BW is far from a standardized effect. Even studies with relatively similar inhaled concentrations report different impacts of solvent exposure on weight, suggesting the possible presence of a moderating variable. 23,24 Furthermore, some studies have reported no impact of exposure on BW, or even a trend toward an increase in BW following prenatal exposure. 25 –29 To date, there has been no recorded attempt to summarize the existing data on the impact of solvent misuse during pregnancy on fetal weight outcomes, despite the above evidence of discrepancies in the literature. Likewise, there has been no recorded attempt to analyze the differences in experimental design that may impact the outcome of prenatal solvent studies.
To address this gap, this review examined the existing animal literature on prenatal solvent exposure and applied quantitative meta-analytic techniques to the available data on toluene exposure-induced BW differences in non-primate mammals. As mentioned above, the field of volatile substance abuse research comprises many different substances including solvents, volatile anesthetics, nitrites, fuels, and so on.
3,4,30
However, this review limited itself specifically to toluene misuse for the simple reason that toluene is by far the most commonly studied abused inhalant. To that end, there are several proposed moderators that may explain the observed discrepancies. The expected effects of these moderators are detailed in the following hypotheses:
Method
Literature reviews (meta-analyses)
Peer-reviewed studies that included prenatal exposure of non-primate mammals to toluene were gathered via extensive literature searches. To be included, studies were required to give a measure of the weight of the offspring, whether live pups, fetuses, or neonates, along with some measure of variance. For this analysis, only published, peer-reviewed studies were considered. PubMed was the primary search engine used. A search of identical keywords in PsycINFO was also conducted to ensure a search representative of work in the field. Keywords used to find the relevant studies were as follows: inhalant + prenatal, toluene + prenatal, and gestational exposure + toluene. All sets of key words were used in both searches.
For the search in PubMed, the keywords “inhalant + prenatal” returned 201 hits. Of these hits, eight studies were utilized for the meta-analysis. The remaining studies were disqualified because of using an inhalant other than toluene (e.g. nitrogen/nitrous oxide, ethanol, allergens, oxygen, cigarette smoke/nicotine, mercury, or various steroids), being in a language other than English, using a species other than non-primate mammals, or not reporting the mean BW of the offspring with variance. Additionally, studies were disqualified if the same animal species was not used in at least two published reports. The keywords “toluene + prenatal” yielded 59 hits, 12 of which were results overlapping with the original search. An additional 13 studies fulfilled the set requirements and were used for the meta-analysis. The remaining studies were discarded for the same disqualification reasons that were noted in the previous search. The last PubMed search performed used the keywords “gestational exposure + toluene.” This search yielded in 28 results, 3 of which did not overlap with the previous searches and did not meet the conditions for disqualification. A PsycINFO search was conducted using the same keywords and yielded overlapping results to those found in the PubMed search. There were a total of 24 usable studies at the end of the search process. These studies consisted of a total of 46 control-to-toluene comparisons with the comparisons differing only in the inhaled concentration of toluene. The inhaled concentration of toluene administered was considered to be the moderator for this meta-analysis.
Meta-analysis techniques
The software program DSTAT (Version 1.11, Lawrence Erlbaum Associates, Inc., Mahwah, New Jersey, USA) was used to analyze the data and to conduct the meta-analysis in accordance with the Hedges and Olkin model.
31
Briefly, an effect size statistic (a measure of deviance from the null hypothesis) was computed for each toluene-to-control comparison. For a fixed effects model, the effect size used for each toluene-to-control comparison (k) was a standardized Cohen’s d. This statistic was computed either by comparing mean pup weights from the toluene group to mean pup weights for the control group or, if the mean weights were not given, by converting whatever significance testing was used into an effect size measure. Standardized effect sizes were interpreted like z-scores, with further deviation from zero marking a larger effect. Because significance with effect sizes is often determined by deviation from a null hypothesis, 95% confidence intervals were computed for each d and were used in determining post hoc differences between moderator levels. When entering the data into DSTAT, any reported standard error-of-the-mean values were first converted to standard deviations using the formula:
Results
A summary of the study characteristics and effect sizes included in the meta-analyses is listed in Table 1. Outlier screening did not reveal any values exceeding the cutoff point of z = 3.29, the critical z-score for 0.1% of expected scores, commonly used as a rigorous estimate of univariate outliers. Therefore, all 24 cases were included in the analyses. The overall effect size was negative (d = −0.39), indicating that prenatal toluene exposure resulted in decreased BW. The 95% confidence interval (−0.42 to −0.35) does not include 0, indicating that the effect was significant.
Summary of study characteristics and effect sizes included in the meta-analysis.a

ppm: parts per million; PN: postnatal day; GD: gestational day.
aSignificant effect sizes are represented by 95% confidence intervals that do not include 0. Negative effect sizes indicate that toluene exposure resulted in decreased body weight. Exposure magnitude was computed by multiplying the concentration (in ppm) by the total exposure time (in minutes). The variable was divided into the following categories: low (L) = 0–900,000; medium (M) = 900,001–3,000,000; high (H) = 3,000,001–5,000,000; very high (VH) = 5,000,001–15,120,000. Due to sample size limitations, injections included both intraperitoneal and subcutaneous injections. If exposures continued after weighing, exposure range was truncated at the day of weighing.
Categorical moderator analyses
The results of the categorical moderator analyses are displayed in Table 2 and shown in Figures 1 to 5. As hypothesized in H1, inhaled concentration was found to be a significant moderator of BW, χ 2 (3, N = 39) = 479.24, p < 0.00001. The 95% confidence intervals for the four inhaled concentration categories examined did not overlap, indicating that each of the inhaled concentration levels produced a different effect on BW (Figure 1). The expected decreases in BW were observed for the three lower categories (0–500 ppm, 501–2000 ppm, and 2001–5000 ppm), with significantly greater decreases in BW for higher concentration categories (>5501 ppm). Unexpectedly, the highest dose category (7500 ppm and above) did not produce the greatest effect on BW. Although the effect size for this inhaled concentration level was significant, it was smaller than both the 2001–5000 ppm and 501–2000 ppm categories (Figure 1).
Effects of perinatal toluene exposure on weight taking into account the methodological characteristics.a

k: the number of effect sizes (the number of toluene-to-control comparisons in each study); Q b: between-subjects heterogeneity; Qw: within-subjects heterogeneity; ANOVA: analysis of variance.
aThese measures are analogous to the same measures for an ANOVA. The exposure magnitude categories were defined as follows: low = 0–900,000; medium = 900,001–3,000,000; high = 3,000,001–5,000,000; very high = 5,000,001–15,120,000.
bp < 0.0001.
cp < 0.00001.
dp < 0.001.

The mean effect size for each inhaled concentration category is shown along with the 95% confidence interval. Only studies in which toluene was administered through inhalation were included in this analysis. Inhaled concentration was found to be a significant moderator. The effect size became greater with increasing dosage level with the exception of the 7500 ppm and above category. Significant group differences are indicated by nonoverlapping confidence intervals.

The mean effect size for each exposure magnitude category is shown along with the 95% confidence interval. Exposure magnitude was computed by multiplying the inhaled concentration (in ppm) by the total exposure time (in minutes). Only studies in which inhalation was the method of administration were included in this analysis. The categories represent the following values: low = 0–900,000; medium = 900,001–3,000,000; high = 3,000,001–5,000,000; and very high = 5,000,001–15,120,000. This variable was found to significantly moderate the effect of toluene on body weight. The very high magnitude category was found to have the largest effect on body weight, followed by the high category. The medium and low categories resulted in comparatively lower effect sizes. Significant group differences are indicated by nonoverlapping confidence intervals.

The mean effect size for each exposure period category is shown along with the 95% confidence interval. Exposure time was not found to be a significant moderator.

The mean effect size for each species is shown along with the 95% confidence interval. Species used was found to significantly moderate the effect of toluene on body weight. Whereas weight decreased as a result of toluene exposure in rats and rabbits, the opposite pattern was observed for mice. Significant group differences are indicated by nonoverlapping confidence intervals.
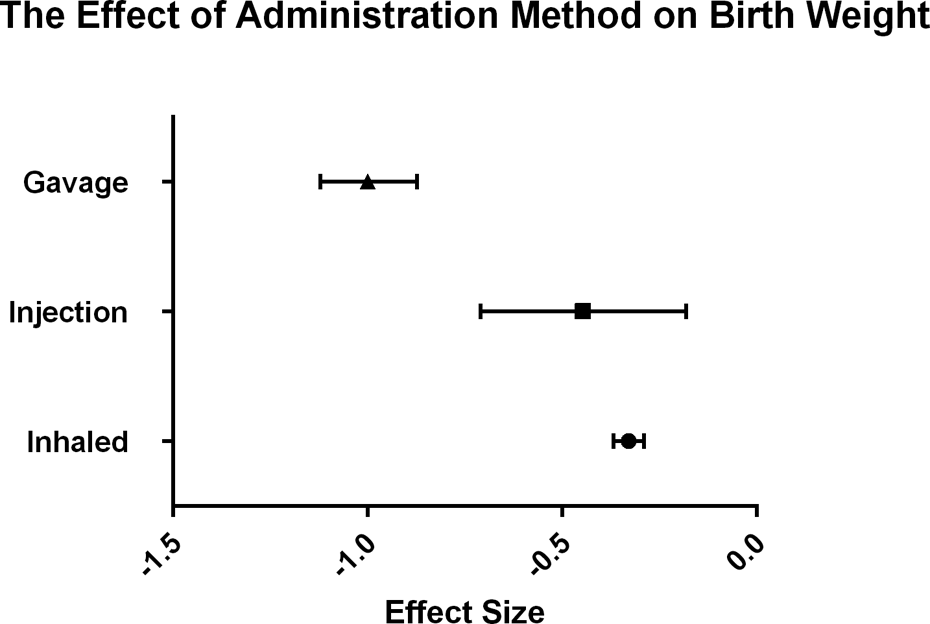
The mean effect size for each toluene administration method is shown along with the 95% confidence interval. Method of administration was found to be a significant moderator. The effect size was greater for studies using gavage compared with studies using injection and inhalation. Significant group differences are indicated by nonoverlapping confidence intervals.
As hypothesized in H2, exposure magnitude was also found to be a significant moderator of BW, χ 2 (3, N = 36) = 178.32, p < 0.00001, with greater exposure magnitudes producing greater decreases in BW (Figure 2). Excluding the “low” magnitude, all other exposure magnitudes (medium, high, and very high) resulted in significant negative effects on BW. The confidence intervals for the “high” and “very high” magnitudes did not overlap with any of the other categories, indicating that these exposure magnitudes resulted in the greatest decreases in BW (Figure 2).
Contrary to our third hypothesis, exposures occurring in either the prenatal or postnatal developmental periods resulted in significant decreases in BW with no differences between developmental periods, χ 2 (1, N = 46) = 1.11, p = 0.92 (Figure 3). As hypothesized in H6, the species used for determining toluene’s effects was also found to be a significant moderator of BW, χ 2 (2, N = 46) = 389.61, p < 0.00001. While toluene exposure resulted in decreased BW in rats and rabbits with a greater effect observed for rats, toluene exposure resulted in increased BW for mice (Figure 4).
Finally, as hypothesized in H5, the route of toluene administration was found to be a significant moderator of BW, χ 2 (2, N = 46) = 99.49, p < 0.00001. While all administration methods resulted in significant decrease in BW, as seen in Figure 5, gavaging toluene resulted in greater effects on BW than inhaling toluene, while toluene injection and inhalation produced similar effects.
Continuous moderator analyses
Although we hypothesized that both day of weighing (H6) and total exposure time (in minutes; H7) would impact effect size, only day of weighing was found to be significant, Z = −3.96, p < 0.0001, with later days of weighing resulting in smaller BW effect sizes (data not graphed).
Associations among moderators
The results of the correlations between inhaled concentration, exposure time, and day of weighing are displayed in Table 3. As shown, only the correlation between inhaled concentration and exposure time was significant, r(73) = −0.69, p < 0.001, indicating that as inhaled concentration increased, exposure time tended to be shorter.
Correlations among inhaled concentration, exposure time, and day of weighing.

aSignificance at the 0.001 level.
The results of the Fisher’s exact tests examining the associations among the remaining moderators are listed in Table 4. Because all the postnatal studies utilized injection as the route of administration, analyses were not conducted to test the association between exposure type and exposure magnitude, inhaled concentration, or exposure time. Exposure magnitude, inhaled concentration, and exposure time applied only to studies in which inhalation was the route of administration. Likewise, associations between route of administration and these three moderators were not computed because inhalation was the only route that yielded values for exposure magnitude, inhaled concentration, and exposure time.
Fischer’s exact tests assessing associations among moderators.a

aResults are not reported for associations among exposure type, exposure magnitude, concentration, and exposure time because all postnatal studies utilized injection as the route of administration. Exposure magnitude, concentration, and exposure time only applied to studies in which inhalation was the route of administration. Likewise, associations among route of administration, exposure magnitude, concentration, and exposure time are not presented because inhalation is the only route that yields values for these three moderators.
bp < 0.01.
cp < 0.0001.
dp < 0.001.
As presented in Table 4, the species used was significantly associated with exposure magnitude, inhaled concentration, exposure time, and day of weighing. Examination of the cross tabulations revealed that studies with mice and rabbits tended to have smaller exposure magnitudes as compared to those with rats. The cross tabulations for inhaled concentration and exposure time revealed that this pattern was due to the use of lower inhaled concentrations in mouse and rabbit studies rather than lower exposure times. The exposure time for all rabbit studies was 4680 min, whereas exposure times for rat and mouse studies spanned from 195–7560 min to 360–6120 min, respectively. For day of weighing, the cross tabulations revealed that the majority of rat and mouse studies weighed pups on postnatal day 1 (PN1), whereas the rabbit studies tested on PN29.
Exposure type was significantly associated with day of weighing and route of administration. The cross tabulations revealed that studies that utilized a postnatal exposure measured weights on PN9 and later, whereas the day of weighing for prenatal studies ranged from gestational day 17 (GD17) to GD29, with the majority of weight being obtained on PN1. Additionally, all postnatal studies used injection as their route of administration, while prenatal studies used all three routes.
Finally, route of administration was significantly associated with day of weighing. Studies in which the route of administration was inhalation obtained weights primarily on PN1, with some studies testing earlier than this with others testing later. Studies that utilized injections tested only on PN1 and later. The day of weighing for studies in which the method of administration was gavage were spread out fairly evenly from GD19 to PN21.
Discussion
This systematic review and meta-analysis of the animal literature have demonstrated that higher prenatal toluene exposure during pregnancy is associated with a greater risk of delivering low BW pups. While the main effect of inhaled toluene concentration on pup BW followed expectations, the reversal of the weight loss trend at the highest inhaled concentrations was surprising. Indeed, this result was contrary to expectations and may be a result of the action of a second moderator. This is juxtaposed by the nonsignificance of the total amount of time that the exposure lasted, which supports the results described by Bowen et al. (2005) suggesting that it is the magnitude of exposure, not time, that induces the greatest BW changes. The enigma of the high-inhaled concentration reversal is clarified somewhat by the significant result of the inhaled concentration–magnitude moderator (represented by the concentration in parts per million × the total exposure time for all exposures in minutes). The use of this compound moderator is supported by the fact that inhaled concentration and exposure times were significantly correlated, indicating that the variables are related to one another. When expressed as a product, it appears that the real impact of toluene on pup BW is not expressed in terms of simply the external dose of toluene the animal is administered, nor how often or how long the administration occurs, but rather a combination of the two. Instead, the evidence suggests it is some balance between high inhaled concentration and length of exposure that produces the greatest BW reduction.
The nonsignificance of the developmental period (pre- or postnatal) suggests that the timing of the exposure is irrelevant so long as it occurs during the early maturation stages. As the first week of postnatal development in these models is considered analogous to the human third trimester, this suggests that the effects of prenatal toluene exposure on BW may not be limited to third trimester exposure. This report did not draw distinctions between when during gestation exposure began (e.g. between the analogues for first and second trimester development), as most prenatal studies shared similar start and stop points for exposure. Likewise, the significance of administration route suggests that oral toluene resulted in more BW decreases than either injection or inhalation. However, this result is somewhat complicated by the finding that the day on which the animal was weighed does influence the effective difference between weights. Because exposure period is confounded by route of administration (all postnatal exposures were injections), these results should not be over-interpreted.
Perhaps the most surprising result was that the species tested was a significant moderator of BW. This effect appears to be solely driven by the studies using mice in that mice did not lose weight, and most of the mouse studies reviewed demonstrated some level of weight gain as compared to control animals. This finding has not been suggested before and would indicate that animal models of toluene exposure are not universal with respect to the species used. However, results from the Fischer’s exact tests examining the relationships between the moderators offer the explanation that, in general, the mouse studies had shorter exposure times and lower inhaled concentrations as compared to the rat studies. Another possible explanation for the positive weight gain observed in these effect sizes may be the result of toluene-enhancing appetite in mice but not in rats. However, it should be noted that the rabbit studies also tended to have lower inhaled concentrations and exposure times, and yet their effect sizes were commensurate with the rat studies. Further analyses of the mouse data were limited by the small number of effect sizes (k = 8), which rendered any more complex moderator analyses impossible.
It should be noted that this analysis and review did not make use of any quantitative estimation techniques of internal toluene concentration and only utilized variables reported in the studies themselves. The use of physiologically based pharmacokinetic (PBPK) modeling to estimate blood concentrations of substances following exposure is a well-established method and several validated toluene PBPK models exist. 51 Future studies should seek to apply this computational estimation technique to the results displayed herein.
This review was a comprehensive summary of the developmental toxicity studies with respect to BW in animals exposed to a variety of toluene concentrations and durations. While it would not be appropriate to suggest that researchers restrict themselves to a single toluene concentration (as providing concentration–effect curves is a standard part of pharmacology and toxicology), it would be sensible to indicate that some consensus be arrived upon as to the length of time such exposures should last. Likewise, the results of this review suggest that the body of literature may not be uniform with regard to animal species, and that care should be taken when interpreting findings as universal.
There are two important experimental manipulations that need to be performed to validate the findings of this review. First, research should directly examine the effect of exposure time and inhaled concentration on BW. Fully understanding the nature of the interplay between the two requires experimental control and statistical manipulations not possible in this analysis. Second, future research should endeavor to utilize an experimental procedure to deduce whether the species used truly does impact BW, or if this finding is an artifact of some unforeseen moderator. Specific attention should be paid to exposure magnitude (higher doses for longer periods of time) to insure that the results are comparable to the rat studies.
The most obvious limitation of this study is that it is a meta-analysis, which is by its very nature hypothetical. No results obtained through this study are truly considered valid until supported by direct experimental manipulation. In addition, these results may be subject to revision should the available sample increase, as the size of the body of literature was limited with only 46 effect sizes spaced across fewer studies.
In conclusion, this review was a comprehensive summarization of the reproductive and developmental toxicity in animal studies associated with toluene. Empirical evidence from these studies demonstrated a strong negative association between prenatal and postnatal toluene exposure and BW in rats and rabbits at multiple doses and routes of exposure. While there are still gaps in our understanding of the reproductive toxicity of toluene that need to be addressed by further human and animal studies, we conclude that intentionally inhaling organic solvents, particularly those containing toluene, may be a potential threat to the developing fetus. With toluene-based solvents being among the most frequently misused psychoactive substances during pregnancy, 16 future studies should seek to experimentally validate the effects identified in this analysis as well as utilize additional estimation techniques (including PBPK models) to validate these findings.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
