Abstract
Drug-induced agranulocytosis is a rare haematological disorder considered as severe adverse drug reaction. Due to its low incidence, the number of studies are low and the variability of clinical features and presentation in hospitalized patients is rarely described. Awe performed an observational, transversal and retrospective study in the haematology and toxicology unit in a tertiary hospital located in Spain (Valencia) (1996–2010) in order to assess its incidence, the drugs involved, the management and outcomes of drug-induced agranulocytosis. Twenty-one cases of agranulocytosis were retrieved. All of them presented severe and symptomatic agranulocytosis (fever and infection). The most common drug associated with drug-induced agranulocytosis was metamizole administration but other drugs belonging to different pharmacological classes as well (carbimazol, sulfasalazine, bisoprolol, itraconazole, amitryptiline, ketorolac and claritomicine+cefuroxime). No differences between sex and age were found in relationship with the manifestations or course of agranulocytosis. In contrast, a significantly negative association was found between age of patients and the percentage of increase in neutrophil count. Administration of human granulocyte colony-stimulating factor did not significantly enhance the recovery of the process or the restoration of leucocytes count, suggesting a limited utility in this type of agranulocytosis.
Keywords
Introduction
Drug-induced agranulocytosis is a severe, potentially lethal, haematological disorder that is manifested as drug reaction type B, that is, unpredictable, low incidence (rare) and high morbidity. 1 The agranulocytosis induced by antineoplastic drugs is classically excluded from this classifications because it can be explained by the mechanism of action of these drugs. 1 This adverse drug reaction is characterized by a sudden and severe agranulocytosis accompanied by neutrophil count less than 0.5 × 10 L−1 or even lower (reaching 0 × 10 L−1). In some cases, agranulocytosis is asymptomatic, and diagnosis occurs when patients have a blood test for an unrelated reason. The complication of agranulocytosis most commonly observed is an increased risk of infections. The location of these infections occur most often at the level of mucous membranes, such as the inside of the mouth and the skin but it can occur in every corporal district. 2 The main causes of agranulocytosis are alterations in the synthesis and maturation of granulocytes or damage to mature granulocytes. The causes of agranulocytosis most commonly observed in clinical settings are due to the decrease in neutrophils production in the bone marrow, destruction of neutrophils outside the bone marrow, infection and nutritional deficiency. 1,3–4 However, in the case of drug-induced agranulocytosis, the pathophysiological mechanisms are not fully understood and there is a constant need to pharmacological vigilance to study and report these harmful drug reactions and to describe it with different drugs and in case of different pathologies. Recent theories converge into the agranulocytosis induced by drugs is due to a previous sensitization with the drug as manifested as a consequence of individual susceptibility (idiosyncrasy), mediated by a pathological process that is not always dose dependent or strictly related to the dose. Finally, genetic factors seems likely to increase the susceptibility to this disorder. 3,5–6
Patients with severe agranulocytosis are likely to experience life-threatening and sometimes fatal infections. Due to its low incidence, few studies have addressed this issue, and continuous surveillance for new and old drugs is necessary in order to know the drugs able to cause this disorder and the management of this potentially fatal complication.
In this work, we investigated the incidence of drug-induced agranulocytosis in the haematology unit of a tertiary hospital in Spain (Valencia), the drugs involved and the relationship between agranulocytosis with age, gender, neutrophil counts at the beginning and at the end of the process, type of infections, exitus and treatment.
Materials and methods
Design of the study
An observational, transversal and retrospective study of cases of drug-induced agranulocytosis occurred in 14 years in the haematology unit of a tertiary hospital in Valencia (Spain) between 1996 and 2010. Clinical information was retrieved by reviewing the medical histories of the cases of drug-induced agranulocytosis. The study follows the rules of declaration of Helsinki and approved by hospital institutional review board.
Diagnostic and exclusion criteria
Diagnostic criteria of drug-induced agranulocytosis were age >2 years old, neutrophil count <500 cells mm−3 with haemoglobin levels >10 g dL−1 and platelets count >100,000 cells mm−3, with previous drug exposure and without any known cause/disease of agranulocytosis. 7 The causality between drug use and agranulocytosis was previously established using the algorithm proposed by Karch and Lasagna. 8
Other exclusion criteria were the presence of pathologies that can cause agranulocytosis per se such as autoimmune diseases, myelodysplasic syndrome and secondary agranulocytosis induced by radio- and chemotherapy in oncologic patients.
Variables
Drugs that induced agranulocytosis and the time of onset after drug administration were recorded. The neutrophil count was analysed at the beginning and at the end of agranulocytosis. The fold increase in neutrophil count was calculated for each patient by dividing the neutrophil count at the end of agranulocytosis (when neutrophil count reach 500 per microlitre and above) by the neutrophil count when it was diagnosed. The time (days) necessary to recover neutrophil counts, the hospitalization time (days) and the effect of granulocyte colony-stimulating factor (G-CSF) were also studied.
Statistical analysis
Descriptive analysis was done. Differences between groups were done by Mann–Whitney test for independent samples. Correlations between variables were analysed by linear correlation and Spearman coefficient. Statistical analysis was performed with SPSS v.20 software.
Results
Drugs, patients and incidence
We detected 85 cases of non-chemotherapeutic agranulocytosis, of which 21 had drug-induced agranulocytosis and 64 had chronic neutropenia unrelated to the use of drugs. Fifty-two patients had an underlying hematological malignancy. The agranulocytosis in 12 patients was due to other diseases (mainly autoimmune diseases). Patients with agranulocytosis not induced by drugs more frequently had hepatomegaly, splenomegaly, enlarged lymph glands or thrombocytopenia together with severe anemia. In contrast, drug-induced agranulocytosis was more severe. Taking into account the time recorded is 14 years and the number of inhabitants of the city area referring to the hospital, 400,000 people, the incidence of drug-induced agranulocytosis was 4.03 cases/million people. Distribution between genders was similar (9 men and 12 women), median age of patients was 66 years (range 27–85), with mean age for men 63.6 years and for women 60.0 years for women. No statistical difference was observed between male and female patients for any of the variables. Based on the algorithm proposed by Karch and Lasagna, 8 seven patients fulfilled the criterium of ‘definite’ and 14 that of ‘probable’.
The drugs inducing agranulocytosis belong to several pharmacological classes, metamizole (analgesic drug also known as dypirone), carbimazol (anti-thyroid drug that in vivo converted to methimazol), sulphasalazine (aminosalicylate known also as salazopirine, sulphasalazopyridine, used as an anti-inflammatory drug in rheumatic diseases, Crohn’s disease and ulcerative colitis), bisoprolol (β-adrenergic blocker), itraconazole (antifungal), amitriptyline (antidepressant drug used also for neuropathic pain and fibromyalgia), ketorolac (non-steroidal inflammatory drug) and claritomicine + cefuroxime (antibiotics). The metamizole (dypirone) was responsible for 76.2% of cases of drug-induced agranulocytosis when it was administered alone (47.6%, 10 patients) or in combination with other drugs (28.6%, 6 patients; Table 1). None of the patients was drug abuser (information retrieved from databases of medical records for each subject). The doses of each drug inducing agranulocytosis are shown in Table 2.
Summary of recorded cases of drug-induced agranulocytosis.
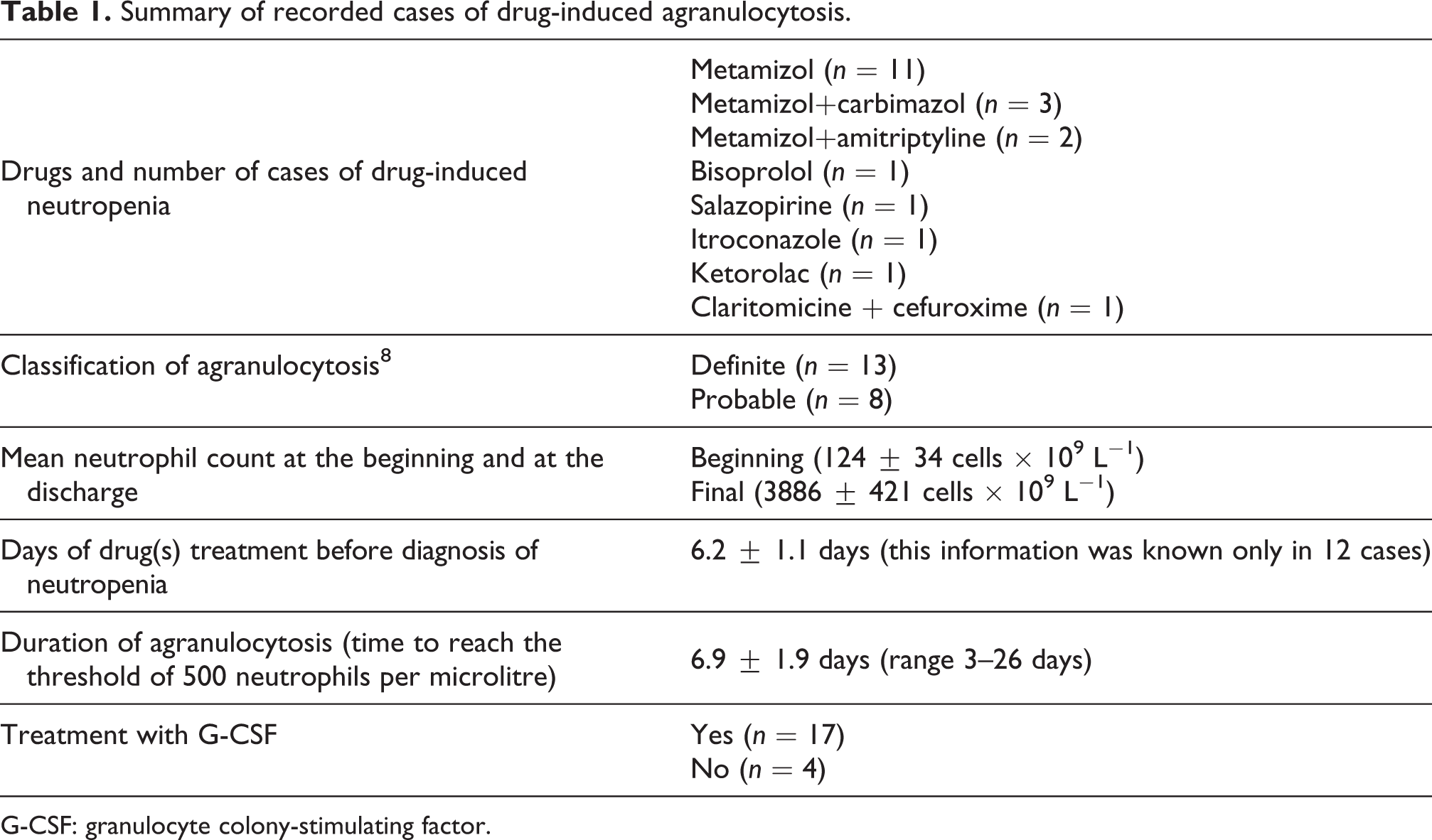
G-CSF: granulocyte colony-stimulating factor.
Doses and route of administration of the drugs that induced agranulocytosis.

Description of cases
Nineteen patients went to the emergency unit of the hospital because of fever and infection that did not decrease after repeated administration of antipyretic or non-steroidal anti-inflammatory drugs with or without antibiotics. Blood analysis performed whilst patients were in the emergency unit revealed agranulocytosis, and then they were hospitalized. Two patients developed agranulocytosis during hospitalization and after administration of metamizole (dypirone): one of them was hospitalized for a retro-pharyngeal abscess (woman, 33 years) and the other for a nephritic colic (man, 27 years). The mean number of co-morbidities evaluated by the Charlson index was 3.3 (range 0–5.3).
Agranulocytosis was diagnosed 6.3 ± 3.8 after drug treatment (range 2–15 days). Twelve patients had clinically defined infection as respiratory (n = 5), urinary (n = 3), oropharyngeal (n = 3), repeated infection in a surgical wounds (n = 1; Table 1). Persistent fever with no sign of infection was reported in nine cases.
All patients showed at the time of diagnosis of agranulocytosis low neutrophil count ranging between 0–400 cells mm−3 (mean 124 cells mm−3). In 11 patients, neutrophil count was 0 cells mm−3. No significant difference was observed in neutrophil count between metamizole-induced agranulocytosis and the other drugs (p = 0.1, Mann–Whitney test). No significant correlation was observed between the Charlson index and neutrophil count (p = 0.09, Spearman correlation test). Data represented in Figure 1 were also analysed by adjusting for the number of comorbidities which gave similar results. One patient (woman 78 years, agranulocytosis induced by metamizole + carbimazol treatment) died 7 days after agranulocytosis diagnosis due to sepsis secondary to a respiratory infection.

Effect of administration of G-CSF. (a) Duration of agranulocytosis in patients that did (with G-CSF) or did not (without G-CSF) receive g-CSF and (b) fold increase in neutrophil count in patients that did (with G-CSF) or did not (without G-CSF) receive G-CSF. The fold increase in neutrophil count was calculated for each patient by dividing neutrophil count at the end of agranulocytosis by neutrophil count when it was diagnosed. G-CSF: granulocyte colony-stimulating factor.
Effect of administration of G-CSF
After diagnosis, the drug(s) treatment was discontinued andintravenous antibiotic therapy was started in order to treat the underlying infections. In order to stimulate the production of neutrophils, administration of G-CSF was carried out in 17 patients (81% of cases). However, the time needed to recover agranulocytosis was not significantly different between patients who received G-CSF (n = 17) from those (n = 4) who did not receive the drug (p = 0.91, Mann–Whitney test; Figure 1(a)). The increase achieved in neutrophil count at the discharge was not significantly different between patients that received G-CSF (n = 17) from those (n = 4) who did not receive the drug (p = 0.05, Mann–Whitney test), although a tendency towards a better effect of G-CSF ensues (Figure 1(b)). In order to detect any relation between variables, correlational analysis was performed between age, sex, severity of agranulocytosis, duration of the process and the percentage of the increase in neutrophil counts. The only significant association that emerged is the inverse relationship between age and the increase in neutrophil count at the discharge (resolution of agranulocytosis; p < 0.05).
No significant associations were found between age, sex, the type of drug (metamizole versus the rest of compounds) or the duration of the process.
Discussion
Non-chemotherapy drug-induced agranulocytosis belongs to potentially life-threatening blood idiosyncrasies, thought to result from a partly elucidated immune and/or toxic damage on granulocytopoiesis due to a multitude of drugs. 1,3–4
The incidence of drug-induced agranulocytosis has not changed in the western hemisphere over the last 30 years. Yet the drug panorama has changed considerably. This implies that host factors may play an intriguing role for this idiosyncratic reaction. The knowledge as to mechanisms for the reaction has advanced with emerging understanding of granulocytopoiesis and immune regulation. Nonetheless, it is still remarkably difficult to pinpoint why and how a drug causes this unexpected, severe adverse event in a patient and there is constant need to update the list of drugs that cause this severe adverse reaction and to analyse clinical features and therapeutic management of these patients. In our study, we observed that the annual incidence of drug-induced agranulocytosis in the study population is 4.03 cases per million inhabitants. The sample appears somewhat biased in inclusion since it deals with only one hospital; however, our results are similar to those obtained previously in Spain showing that the incidence is maintained over time, despite the increase knowledge in the field. 9 The International Study on Aplastic Anemia and Agranulocytosis, a multicentre case-control study conducted over 4 years (1980–1986) in 7 European countries and Israel, showed an incidence of drug-induced agranulocytosis around 1.7 in Milan (Italy) and 9 cases per million population per year in Budapest (Hungary), with an average of 6.2 cases per million inhabitants per year. 2–3,10 The issues concerning the distribution of the process by gender and age are consistent with all studies consulted, being slightly (although no significant) higher in women, and increasing with age. The effect of age can be explained by the greater consumption of drugs in the elderly (due to higher comorbidity index) as well as an increased sensitivity to the adverse effects of certain drugs and a decrease in the body’s homeostatic mechanisms associated with ageing. 3,11 Our study shows that metamizole is the drug most involved in this adverse drug reaction. These results agree with those obtained in other studies concerning drug-induced agranulocytosis. According to data from the observatory of drugs, metamizole (dipyrone) was the second most commonly consumed drug in Spain in 2011, 12 which could explain its frequency in a great number of cases of agranulocytosis. The proposed molecular mechanism by which metamizole produces agranulocytosis may be the synthesis of cytotoxic lymphocytes that generate killer cells against drug-coupled bone marrow granulocytic cells. 13 The results from Garcia-Martinez et al. 14 demonstrated that in vitro metamizole neither alters the granulocyte differentiation process nor induces relevant apoptosis on terminally differentiated granulocytes. These results indirectly suggest that the mechanism of metamizole-induced agranulocytosis should be of immunoallergic origin since a toxic effect of this drug is excluded.
However, it should bear in mind that the number of cases is not by itself a measure of the real risk attributable to each drug, indeed the rate of the drug prescription must be also considered. Even though there is a long list of drugs involved, there are few publications assessing relative risk attributable to each of these drugs. 2,4 There is a need to promote the adequate notice of these rare but serious adverse reactions to provide information for the development of epidemiological surveillance studies and to know the real risk of suffering this process when prescribing these drugs (many of them are frequently prescribed and with high therapeutic effectiveness) and encourage their rational use. Since the first description of drugs able to induce agranulocytosis, the list has grown (pyrazolones, butazones and indometacine), phenotiazines, sulphamides, antithyroid drugs, 15–16 antihistaminic drugs, aprindin, captopril, mianserin, clozapine, amodiaquine and dapsone. 4
Although there is a wide range of information about most of drugs causing agranulocytosis in the biomedical literature, in the case of a small number of these drugs, the information is limited and ineffective. To our knowledge, this is the first report in the literature showing agranulocytosis induced by bisoprolol and itraconazole, which is a valuable information for clinicians in order to update the list of drugs able to induce agranulocytosis. Our results also provide reassurance regarding the risk associated with a number of newly marketed drugs.
There is a variable elapse of time between the administration of the drug causing agranulocytosis and its appearance. The speed of this process depends on the rate of synthesis of neutrophils and its survival time in the blood stream. If damage occurs to more undifferentiated white cells, the latency of the disorder can be even longer. 3–4
In our work, we found that the increase in the numbers of neutrophils inversely and significantly correlated with the age of the subject. We have not found similar series to make comparisons, but it would be logical to think that without other associated pathology, bone marrow from a younger individual had a greater capacity to recover 17,18
Even there are accepted diagnostic tools to assess the origin of agranulocytosis induced by drugs, it requires high suspicious index and excluding other factor that can cause agranulocytosis. The causal exposure to the drug, haemogram and the resolution or improving agranulocytosis after removing the drugs point to a clinical scenario of drug-induced agranulocytosis. 3 New treatment such as G-CSF or granulocyte–macrophage CSF has improved the treatment of patients with agranulocytosis due to several causes. 1 Both factors stimulate the proliferation and differentiation of progenitor cells, leading to a fast restoration of granulocytes counts by reduction in morbidity and mortality. However, in the case of drug-induced agranulocytosis, there is not a general consensus regarding a clear benefit. 12,19 Administration of G-CSF (filgrastim, 300 μg once a day subcutaneously) was performed in order to increase neutrophils production in 19% of the reported cases. No side effects were observed during filgrastim treatment. These patients were treated with G-CSF to accelerate the recovery of neutrophil count since there was a small increase in neutrophil count (10–30%) in these patients after 48 h from drug withdrawal. These four patients did not have any different features from the rest of patients included in the series (e.g. type of the drugs, age of patients and gender). The administration of G-CSF did not demonstrate faster properties in reversing drug-induced agranulocytosis compared to the removal of the causative drug. Although some authors justify the non-use of G-CSF with the fact that the mere suspension of the drug gets recovery levels of granulocytes, other authors advocate for its use in case of severe agranulocytosis or in those patients with worst general condition. 20 –22 Our report adds important data regarding drug-induced agranulocytosis in our country, suggesting that the list of drugs able to induce agranulocytosis might be constantly updated. Neutrophil count and/or the presence of persistent fever (with or without an underlying infectious process) should be monitored when these drugs are administered.
Footnotes
Authors’ Note
RNM and ECS contributed equally to this work.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by PRECOMP13-115500 from the University of Valencia and GV2014-043 from Generalitat Valenciana.
