Abstract
Background:
Tiagabine is a novel antiepileptic that acts by increasing synaptic and extracellular gamma-aminobutyric acid concentrations. Information concerning overdose of tiagabine is limited. After introduction, an increasing number of off-label uses suggested that tiagabine use would increase. However in 2005 and 2008, warnings from the Food and Drug Administration (FDA) were issued on the risk of seizures in non-epileptic and increased suicide ideation. We evaluated the temporal trends associated with these two warnings as well as clinical outcomes from tiagabine overdose.
Method:
A retrospective review of all single substance tiagabine exposures in National Poison Data System (NPDS) from 2000 to 2012.
Results:
A total of 2147 patients had ingested tiagabine, with a mean of 165 year−1. This was disproportionally distributed, with a steep rise leading up to 2004 (max 559 year−1) and then a significant decline (p < 0.05) between 2005 and 2006. The number of cases reported to NPDS mirrored the sales of tiagabine. Clinical effects were predominantly neurological, with the most commonly reported effects being drowsiness (27%), agitation (19%), confusion (12%), seizures (11%), and tachycardia (10%). In all, 758 patients (35%) showed a major or moderate medical outcome, with no deaths reported. A disproportionate share of the major outcomes was in the suicide attempt group (73%). The majority of patients (75%) were treated in a health-care facility (HCF).
Conclusions:
The HCF usage is likely due to high rate of symptomatic patients (59%) and the large proportion of suicide attempt cases. The frequency of tiagabine cases in NPDS mirrored pharmaceutical sales, with steep declines temporally related to the 2005 FDA warning.
Keywords
Introduction
Tiagabine is a novel antiepileptic that acts by binding to the gamma-aminobutyric acid (GABA) uptake transporter in presynaptic neurons and glial cells resulting in increased synaptic and extracellular GABA concentrations. The increased concentration of the inhibitory neurotransmitter GABA is believed to be responsible for the antiepileptic effects. Information concerning overdose of tiagabine is limited to case reports and a single case series. 1 –9 These reports suggest the effects in supratherapeutic doses and overdoses that follow the GABA neurological mechanism, with lethargy, confusion, and coma. Seizures, convulsive, and nonconvulsive status epilepticus have also been reported. The epileptogenic mechanism for tiagabine is not known but suggestions have included possible mediation via GABA receptors in the thalamus and stimulation of dopamine, serotonin, or glycine receptors. 9,10 An atypical presentation has been reported with profuse vomiting, hypersalivation, bradycardia, and hypertension. 4
Tiagabine was approved by the Food and Drug Administration (FDA) in 1997 for use in the United States for adjunctive treatment of partial seizures in adults and children 12 years and older. Since its introduction, additional off-label uses have included posttraumatic stress disorder, neuropathic pain, generalized anxiety disorder, cocaine dependence, and rage. 11 –15 Initially, these wider uses suggested that tiagabine use would continue to increase. However, in February 2005, the US FDA announced that a bolded warning would be added to the labeling for tiagabine to warn prescribers of the risk of seizures in patients without epilepsy being treated with this drug. Although tiagabine has been shown to reduce the frequency of seizures in patients with epilepsy, paradoxically tiagabine’s use has been associated with the occurrence of seizures in patients without epilepsy. In 2008, the FDA notified health-care professionals that tiagabine was 1 of 11 drugs used to treat epilepsy as well as psychiatric disorders and other conditions that may cause increased risk of suicidal thoughts or behavior.
We examined National Poison Data System (NPDS) data and compared rate trends in tiagabine exposures reported to poison control centers (PCCs) before and after the issuance of the FDA warnings. We believe that there would be a decrease in overdose/adverse reaction events occurring concomitantly with the warnings due to decreased prescribing and concern for negative effects. While other studies have established a decrease in prescribing following FDA safety warnings, this situation is unusual in that we are able to examine changes in exposure rates following the issuance of multiple FDA warnings and are looking at adverse effects related to exposure to the medication in addition to amount prescribed. 16 Additionally, we sought to describe the clinical picture with tiagabine overdose from a large national database.
Methods
This was a retrospective review of all human tiagabine patients reported to the NPDS of the American Association of Poison Control Centers (AAPCC) from 2000 through 2012. Inclusion criteria included single substance ingestion of tiagabine in a human. Exclusion criteria were polysubstance ingestion, information calls, animal exposures, and confirmed nonexposure. Additionally, annual sales volume for tiagabine was obtained as a measure of use of tiagabine. Data for tiagabine sales were only available from 2000 through 2010. Year to year means were compared using a Student’s t-test, and monthly trend lines (R 2) between 2005 and 2008 were compared.
The definitions for medical outcome were the standard definitions used by NPDS as follows: no effect (no signs or symptoms as a result of the exposure), minor effect (signs or symptoms were minimally bothersome and resolved rapidly; e.g. self-limited gastrointestinal symptoms, drowsiness, and sinus tachycardia without hypotension), moderate effect (signs or symptoms were more pronounced, more prolonged, or more systemic in nature, usually requiring treatment but not life threatening; e.g. acid–base disturbances, hypotension rapidly responsive to treatment, and isolated brief seizures), major effect (signs or symptoms were life threatening or resulted in significant residual disability; e.g. repeated seizures or status epilepticus, respiratory compromise requiring intubation, and ventricular dysrhythmias), or death (death resulted from the exposure or direct complication of the exposure). A serious outcome was defined as combining moderate outcome, major outcome, and death.
Results
There were 2147 patients with a single substance ingestion of tiagabine. There was a mean of 165 patients/year, but this was disproportionally distributed with a steep rise leading up to 2004 (max 559 year−1) and then a significant decline (p < 0.05) between 2005 and 2006 (Figure 1). A decline in exposures was also noted between 2008 and 2009, but this was found to be nonsignificant. We observed a significant downtrend (R 2 = 0.789) in 2005 following the month of February. We did not see a downtrend change throughout the year of 2008 (R 2 = 0.0056). The number of cases reported to NPDS mirrored sales reported of tiagabine (Figure 1).

Annual number of patients reported to NPDS with tiagabine poisoning and sales of tiagabine by year. NPDS: National Poison Data System.
Demographically, the mean age of patients was 30.7 years, with 1267 females (59%). The patients were predominantly adults (72%), with 239 young children <6 years of age (11%).
The clinical effects were predominantly neurological, with the most commonly reported effects being drowsiness, agitation, confusion, seizures, and tachycardia (see Table 1). There were a minority of cases that presented with the “atypical” findings of vomiting, hypertension, muscle rigidity, and bradycardia. There were 758 patients (35%) with either a major or moderate medical outcome. There were no deaths in the single substance group. A majority of included exposures (67%) occurred due to one of the three reasons: a suicide attempt, an adverse drug reaction, or a therapeutic error (Figure 3). Among individuals in the moderate outcome group, the reason for exposure was evenly distributed among the top three reasons for exposure. Major outcomes were disproportionate due to intentional suicide attempt (73%) (Figures 2 and 3). The reason for exposure changed over the study period with a decrease in the percentage of patients with a suicide attempt as a reason and an increase in patients with a therapeutic error or an adverse drug reaction (Figure 4). However, major outcomes were disproportionate due to intentional suicide attempt. The majority of patients (n = 1612, 75%) were treated in a health-care facility (HCF). The duration of clinical effects was short and similar to previous reports: 6% with duration <2 h, 32% with duration 2 to ≤8 h, 42% with duration 8 to ≤24 h, 13% with duration >24 h, and 7% not recorded.
Reported clinical effects with single substance ingestion of tiagabine.

SZ: seizures.

Percentages of reason for tiagabine ingestion in all patients and those patients with serious (major of moderate) medical outcome.
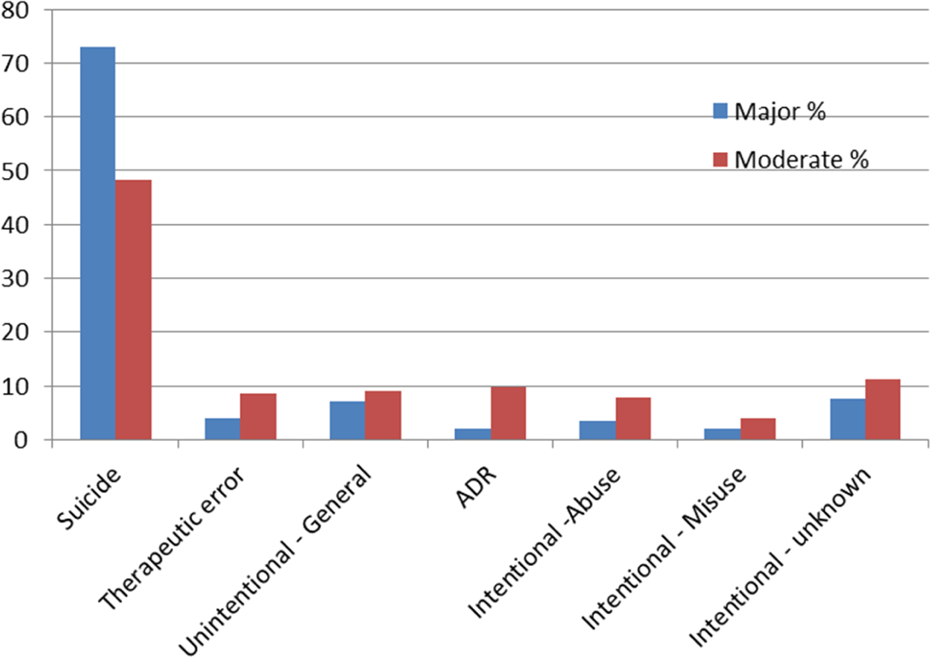
Distribution of major and moderate medical outcomes by reason for tiagabine ingestion.

Reason for tiagabine exposure (percentage of total group) by year.
Discussion
Our study noted several interesting findings. There was an obvious mirroring of tiagabine sales data and NPDS reports, including a significant decrease in both exposures and sales, following the FDA warning in 2005. In a substantial number of patients (>10%), tiagabine appeared to have a pro-convulsant effect and the toxicity associated with tiagabine was higher than expected as measured by cases with a serious outcome (35%) and those receiving treatment at a HCF (79%).
While we see both a significant decrease of reported exposures between 2005 (the year of the first FDA safety warning) and 2006 and a marked negative trend line for exposures throughout 2005, we did not appreciate similar effects in 2008 (the year of the second FDA safety warning). The initial FDA warning was in regard to the use of tiagabine in non-epileptics, as the medication was being used for a large number of “off-label” uses. This warning may have curtailed non-indicated uses, leaving only users who were prescribed tiagabine under the approved indication receiving the medication and thus unlikely to be taken off the medication even following an adverse exposure or reaction. The FDA warning in 2008 was less specifically targeted to tiagabine, with tiagabine listed as 1 of 11 drugs involved in the warning concerning the possible increase in suicide ideation. While we did not see a decrease in the number of patients in 2008, we did see a decrease in percentage of patients with suicide attempt suggesting an impact of the FDA warning. The finding of decreased exposures in NPDS after the 2005 warning is consistent with previous studies, noting the impact of the FDA warnings on medication use and prescriptions by providers. 17 –22 A second finding of note in our study is the mirroring of the decrease in sales of tiagabine with the drop in NPDS tiagabine exposures.
Seizures have been previously reported with tiagabine exposures and were specifically identified as a potential adverse event from tiagabine use by the FDA. 1 –9 Our study provides additional support that tiagabine has pro-convulsant properties, especially in overdose. Tiagabine joins a group of anticonvulsants that appear to have pro-convulsant properties in overdose, such as carbamazepine, lamotragine, zonisamide, and phenytoin. The clinical effects reported (Table 1) are similar to those reported in a previous smaller study with 2 years of data from seven US states. 1 While the reported effects are similar, the percentage of patients with these clinical effects in the present larger study is lower. Also of note is the high morbidity and HCF utilization associated with tiagabine; more than 35% of patients experienced either a moderate or major outcome, which is abnormally high for poison center cases, and 79% of patients received treatment in a HCF, again abnormally high for poison center cases. This large proportion of patients managed in a HCF is most likely due to high rate of symptomatic patients (59%) and the large proportion of suicide attempt cases. Not unexpectedly the highest number of serious outcomes was in the intentional groups.
Limitations
The AAPCC maintains the national database of information logged by the country’s PCCs. Case records in this database are from self-reported calls, and they reflect only information provided when the public or health-care professionals report an actual or potential exposure to a substance (e.g. an ingestion, inhalation, or topical exposure) or request information or educational materials. Exposures do not necessarily represent a poisoning or an overdose. The AAPCC is not able to completely verify the accuracy of every report made to member centers. Additional exposures may go unreported to PCCs, and data referenced from the AAPCC should not be construed to represent the complete incidence of national exposures to any substance.
The NPDS does not have fields for coding past medical history of patients, so we are unable to tell how many of the reported seizures occurred in non-epileptic subjects.
Conclusion
The steep decline in tiagabine use appeared temporally related to the 2005 FDA warning, but no significant change in pattern was noted following the 2008 warnings. The initial warning about off-label use specific to this one drug had greater effect than subsequent warnings, which were about many drugs including tiagabine and which were not focused on off-label use. As the pattern of cases seen in NPDS mirrored pharmaceutical sales, we believe NPDS may serve as a sentinel for changes in dosing/prescribing patterns.
Footnotes
Authors’ Note
The information in this manuscript will be presented as a poster at NACCT in New Orleans, October 2014.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
