Abstract
Human biomonitoring is a well-recognized tool for estimating the exposure of humans to environmental pollutants. However, heavy metals’ pollution from anthropogenic origin is a cause for concern because of its potential accumulation in the environment and living organisms, leading to long-term toxic effects. This study was aimed to assess the concentrations of cadmium (Cd), chromium (Cr), lead (Pb), copper (Cu), nickel (Ni), cobalt (Co), manganese (Mn), iron (Fe), and zinc (Zn) in human biological samples (urine, whole blood, hair, and nails) and antioxidant response in blood samples from 48 individuals exposed to heavy metals and to compare them with different age classes and sites. The results indicated that there were metal-specific differences in concentration in exposure groups among the studied sites. The concentration of heavy metals in blood samples showed the following order : Pb > Cd > Ni > Co > Cr. In urine samples, the order was Cu > Pb > Cr > Ni > Co > Cd; in nails samples, the order was Pb > Ni > Cr > Co > Cd > Mn; and in hair samples, the trend was Pb > Ni > Cr > Mn > Cd > Co. A significant (p > 0.05) decrease in antioxidants enzymes activity was observed with increase in heavy metals concentrations. This is the first study reporting biological evidence of altered toxic metals’ concentration in humans in Lahore, Pakistan, due to environmental exposure. Further research, including risk analysis studies, food chain contamination, and epidemiological and clinical investigations, are needed to assess optimal levels for dietary exposure in the study area and associated adverse health outcomes.
Introduction
Heavy metals of natural source are always present at a background level with their occurrence in soils being related to weathering of parent rocks and pedogenesis. 1 However, the concentration of numerous heavy metals has increased noticeably in certain ecosystems due to anthropogenic activities. 2 Heavy metals often occur as cations, as a result of changing environmental conditions that strongly interact with the soil matrix and become mobile. 3 Concentrations of Heavy metals increased rapidly through waste disposal, atmospheric deposition, fertilizer and pesticide use, and the application of sewage sludge in land. 4 Under certain conditions, either toxic elements, such as lead (Pb), cadmium (Cd), and copper (Cu), or essential micronutrients, such as Cu and zinc (Zn), in the environment might accumulate to a toxic concentration, leading to ecological damages.
Heavy metals are accumulated in marine environments, including ground water, fish, and sediments, and consequently transferred to human beings through the food chain. 5 Heavy metals, such as Cd, Pb, and arsenic (As), deserve special consideration among these pollutants, as they represent a group of highly toxic substances accumulating in the tissues of marine organisms and being transported through the food chain to humans. 6
The increased industrialization, urbanization, population growth, and overall human’s greed to exploit Mother Nature have provoked the pollution level. When discharged into the marine environments, the toxicity, persistence, and accumulative behavior of heavy metals can cause damage to the diversity of marine species, as well as ecosystems, 7,8 and finally consumed by humans resulting in health risks. The concern is growing more serious globally especially in developing countries. 9 Food consumption and drinking water had been identified as the major pathway of human exposure to heavy metals accounting for 90% compared to other ways of exposure. 10
As the trend toward increased environmental pollution of industrial agglomerations continues to go upward, the assessment of the degree of health risk of this pollution is becoming increasingly a major public health concern. 11 In this context, a primary concern of environmental toxicology is the assessment of human exposure. For the assessment of environmental exposure to humans, examination of a human tissue appears to be more suitable than the analysis of plant or animal materials currently used in ecological studies to reveal environmental pollution. 12 The human biological materials that are accessible for sampling include blood and urine, and also hair and nails, 13 –15 and successful attempts have also been made to measure heavy metals’ exposure in humans using biological samples. Hence, the objective of this study was to measure the concentration of Cd, chromium (Cr), Pb, Cu, nickel (Ni), cobalt (Co), manganese (Mn), iron (Fe), and Zn in urine, whole blood, hair, and nails; to check the response of antioxidant in blood samples; and to compare them among different age classes and sites.
Materials and methods
Study area and site description
The study area was from Lahore district located in the Punjab Province, Pakistan (Figure 1(a)). The study area is bounded by 73.22°–74.45°E longitude and 31.20°–31.60°N latitude, covering an area of approximately 4000 km2. Three sites Chung, Manga Mandi, and Kalalanwala were selected, along Lahore–Multan road (Figure 1(c)). Site Chung is located at 74.17°–74.22°E longitude and 31.40°–31.48°N latitude and is adjacent to Sunder Industrial Estate with major industrial units of pharmaceuticals, food processing and engineering, textile/garments, carpet, paper and board plastics, electronics, chemicals, paints, steel, auto parts, and wood products industries. The Manga Mandi and Kalalanwala are close to each other and are located at 74.00°–74.06°E longitude, 31.28°–31.35°N latitude and 74.04°–74.10°E longitude, 31.28°–31.35°N latitude, respectively. The Manga Mandi is situated on Multan road opposite to Honda car manufacturing plant, around 50 km from Lahore. Most of the land use in both areas is agricultural. Fertilizers such as diammonium phosphate and urea are extensively used in agricultural sites. There is also an extensive use of pesticides and insecticides in this agricultural area.

Map showing the study area.
Study design
Questionnaires-based survey
A survey based on questionnaire was used for collection of demographic status, source of drinking water, medical history, dietary habits, and health information from the human subjects. A total of 48 questionnaires were carried out in the study area, including adults, children, and old age people, 12 from each site having age classes of 25–35, 10–15, and 40–50 years, respectively. The self-administrated questionnaires were filled from randomly recruited subjects about their personal characteristics, source of drinking water, and dietary habits. The information included in the questionnaire was age, sex, socioeconomic background, health condition of the individual, education, smoking habits, and possible sources of As exposure. The dietary habits considered aspects such as consumption of sea food, vegetables, and meat, and so on, and for drinking water either individual is entirely dependent on dwellings or use improved water supply by Water and Sanitation Authority. One of the important observations was that the brickkilns are concentrated in and around the selected study areas. Studied subjects have similar lifestyle, socioeconomic conditions, and dietary habits. They engaged in agriculture work, and intake was wheat flour-based food, locally grown seasonal vegetables, and meat.
Recruitment of human subjects and selection criteria
A total of 48 human subjects, 12 from each site consisting of adults (aged 25–35 years), children (aged 10–15 years), and old age people (aged 40–50 years), were selected for collection of blood, urine, nails, and hair samples. Control samples were also collected from a group of people who never drank As-contaminated water and lived at a place far from As-contaminated regions of Lahore.
Ethical statement
All the ethical and legal standards approved by the Ethics Committee, Faculty of biological Sciences, Quaid-i-Azam University, Islamabad were fulfilled and complied with all applicable requirements of international regulations. The subjects in the survey were informed all the protocols, procedures, and aims of the study. After the local approval, only volunteers were selected for sampling.
Collection of samples and storage
Biological samples (blood, urine, hair, and nails), n = 48 each, from the selected human groups were collected from study sites. As reported in previous work, 16 hair samples from nape of the neck were collected using stainless steel scissors, and fingernail and toenail samples were collected by ceramic blade. Hair and nail samples were stored at room temperature individually in clean polyethylene bags and sealed tightly. Then, 5 ml of fresh venous whole blood samples were collected by sterile syringes and stored in 5 ml ethylenediaminetetraacetic acid tubes at 4°C till further analysis. Spot urine samples were collected in prewashed (1 nitric acid (HNO3):1 hydrogen peroxide (H2O2)) polyethylene bottles. After collection, the urine samples stored in salt ice mixture immediately were transported to the laboratory and kept at −20°C prior analysis.
Pretreatment/washing of nail and hair samples
Nail samples were scrubbed using nylon brush to remove the surface dust. The hair and nail samples were cleaned following the procedure outlined by Samanta et al. 15 Firstly, hair and nail samples were washed with distilled water on a stirrer for 15 min in a beaker, then with acetone and three times with distilled water, and again with acetone. The washed samples were placed in glass beakers and allowed to dry at 50°C overnight in a drying oven.
Samples digestion and As analysis
Hair, nails, and blood
Hair and nail samples were digested by the procedure outlined by Samanta et al. 15 A microwave digestion system MARS 5 (Version 194A02, CEM Corporation, Matthews, North Carolina, USA) was used for digesting hair and nail samples. Five hundred milligram of dry hair and nail and 0.5 ml of blood samples were weighed and put into a dry, clean Teflon digestion vessel. HNO3 and H2O2 were added to the samples, and power with 100% efficiency (600 W) was applied in a three-step process. Each step had the holding time for 10 min. After cooling, digested solution was transferred to a volumetric flask, filtered, and stored in polyethylene bottles for As analysis. The reagent blanks were also digested and diluted in the same manner. All digested samples were kept at 4°C and analyzed within 1 week.
Urine
The frozen urine samples were thawed and prepared according to the method described by Gault et al. 17 Urine samples were filtered and diluted five times with 2% HNO3 for As determination. All the blanks were prepared in similar procedure using distilled water instead of sample. As in all biological samples was analyzed using an atomic absorption spectrometer with a vapor generation accessory (AAS–HG; Vario 6, Analytik Jena, Germany).
Total protein estimation
Protein estimation was determined by the method described previously. 18 About 90 μl blood samples were homogenized in phosphate buffer and centrifuged at 10,000 r/min for 20 min at 4°C. Alkaline solution was mixed with 0.1 ml blood sample and incubated for 10 min, and 1:1 Folin–Ciocalteu reagent was added to each tube and vortexed to mix it thoroughly. After 30 min of incubation, optical density was noted at 595 nm.
Estimation of antioxidant enzymes
Glutathione (GSH) activity was determined by the method described by Ellman. 19 The method reported by Flohé and Günzler 20 was used to measure the gulatathione peroxidase (GPx). Reduced glutathione (GR) concentration in red blood cell suspension was estimated by the method outlined by Lehmann. 21 Catalase (CAT) and superoxide dismutase (SOD) were carried out using the methods reported by Bergmayer 22 and Elstner and Heupel, 23 respectively.
Quality control and assurance
All the analytical methods were assured by the analysis of triplicate samples and reagent blanks. Quality control was ensured by analyzing reference materials, for blood provided by the INSP, Canada (INSP-external quality assessment scheme, EQAS), human urine freeze-dried certified reference material (CRM) from National Institute of Environmental Studies (CRM No. 18, NIES, Japan), and human hair reference material CRM (GBW 09101) from National Research Centre for CRM, Beijing, China, and to the best of our knowledge, no CRM for fingernail is currently available. The recovered concentrations of the analyzed metals were within 90 ± 10% confidence levels.
Statistical analysis
Descriptive statistical calculations were computed by statistical software IBM SPSS Version 20 for Windows. The values were log transformed because the Kolmogorov–Smirnov Z test revealed a non-normal distribution of heavy metals. Analysis of variance (ANOVA) was applied using Statistix (Version 8.1) software to examine the variation of heavy metals among risk sites and age classes. Spearman’s correlation analysis was conducted between heavy metals concentrations and antioxidants enzymes.
Results
Demographic characteristics of exposure groups
The demographic characteristics such as age, gender, smoking habit, working site and work duration, medical history, residential period in study area, and source of drinking water and dietary habits of study participants are presented in Tables 1 and 2. Most of the participants were unemployed and had lived in their respective areas for more than 10 years. Whereas deep wells and shallow water hand pumps were also the sources of drinking water in the study area. A higher percentage of males from rural areas of the community participated in the study. Similar demographic characteristics were shown by low-, medium-, and high exposure groups.
Data represent mean ± SE of heavy metals concentrations in human blood for different age-groups.a

Cd: cadmium; Co: cobalt; Cr: chromium; Ni: nickel; Pb: lead; Fe: iron; Zn: zinc; LSD: least significant difference.
aMean followed by same letters within columns is not significantly different by LSD at 5% level.
bBold values indicate significant variation of metals in studied sites.
Data represent mean ± SE of heavy metals concentrations in human blood for the studied sites using ANOVA.a
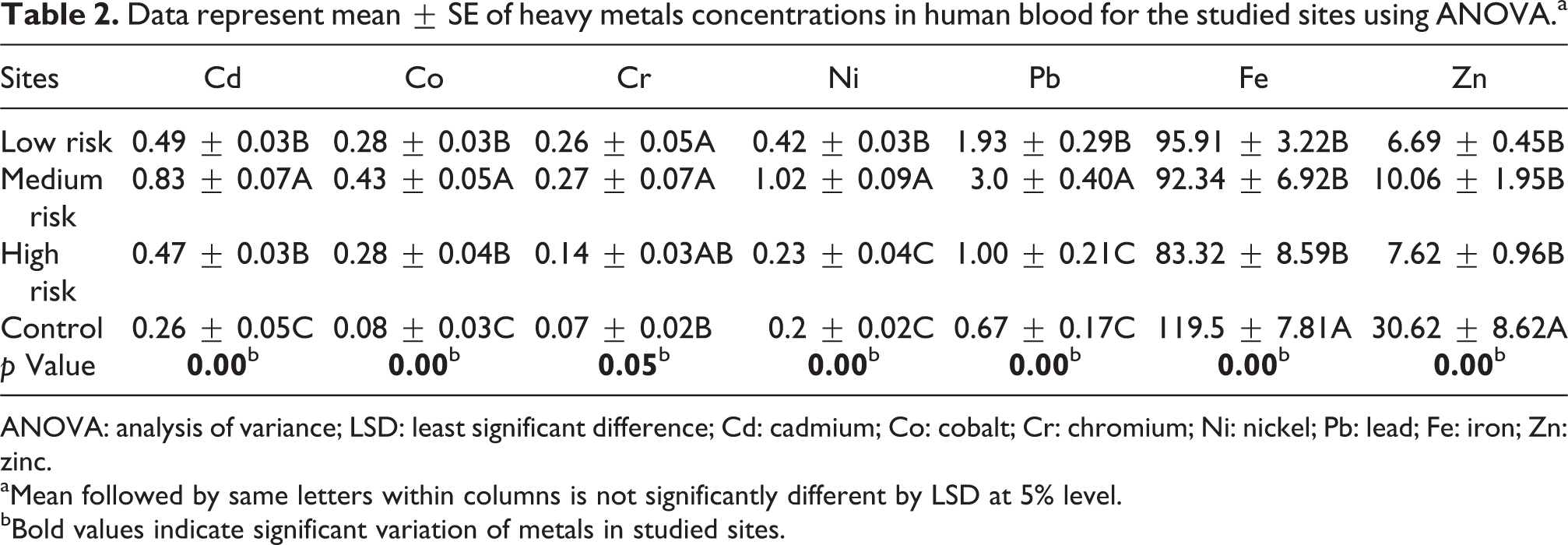
ANOVA: analysis of variance; LSD: least significant difference; Cd: cadmium; Co: cobalt; Cr: chromium; Ni: nickel; Pb: lead; Fe: iron; Zn: zinc.
aMean followed by same letters within columns is not significantly different by LSD at 5% level.
bBold values indicate significant variation of metals in studied sites.
General trends of heavy metals in biological samples
Multielement analysis was validated for nine metals in biological samples. Pattern of heavy metals accumulation varied among the different studied groups. There were metal-specific differences in elemental concentration in exposure groups among the studied sites. Generally, the concentrations of heavy metals in blood were in the following order: Pb > Cd > Ni > Co > Cr. In groups, the concentrations of metals were in the following order: children > patients > adults. One-way ANOVA results of heavy metals’ concentrations in blood samples for age-groups are given in Table 1. The results revealed that Pb concentration varied (p = 0.05) with different age-groups. Whereas all metals strongly varied (p = 0.05) with all studied sites.
In urine samples, concentrations of heavy metals followed the following trend : Cu > Pb > Cr > Ni > Co > Cd. The increased levels of Cd, Ni, Co, Cu, and Pb concentrations were recorded in children, while Cr was higher in adults. In general, the concentrations of heavy metals in groups followed the following order: children > patients > adults. One-way ANOVA of heavy metals’ distribution in age-groups showed significantly variation (p = 0.04) of Cr in the urine samples. In addition, all the analyzed metals showed significant variation (p = 0.05) in exposure sites (Tables 3 and 4). Similarly, in the samples containing human nails, the concentrations of heavy metals were of the following order: Pb > Ni > Cr > Co > Cd > Mn. Generally, the concentrations of all heavy metals in groups were observed in the order of old age > adults > children. Overall highest concentrations of Pb and Ni were observed in all sites, and all elements were found at higher levels than those of control site. One-way ANOVA (Tables 5 and 6) did not show any significant variation of heavy metals between groups. Whereas one-way ANOVA revealed that all analyzed metals significantly varied (p > 0.05) in the sites studied except Cr and Mn in samples of human nails.
Data represent mean ± SE of heavy metals concentrations in human urine for different age-groups.a
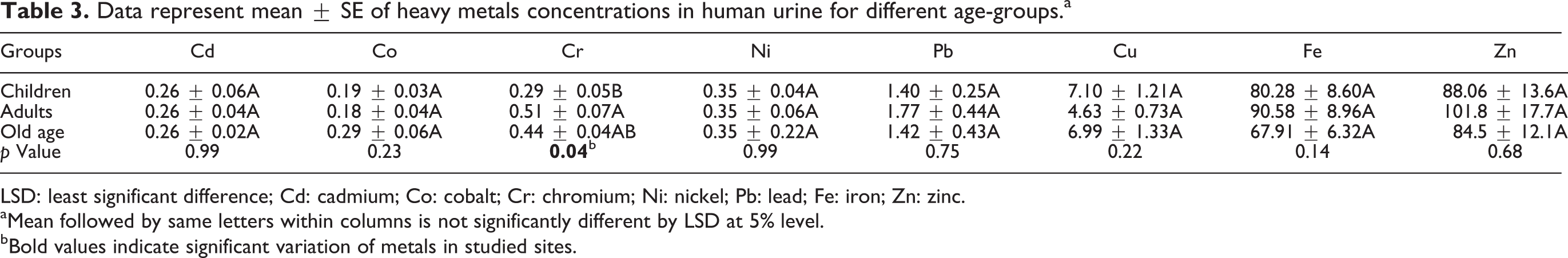
LSD: least significant difference; Cd: cadmium; Co: cobalt; Cr: chromium; Ni: nickel; Pb: lead; Fe: iron; Zn: zinc.
aMean followed by same letters within columns is not significantly different by LSD at 5% level.
bBold values indicate significant variation of metals in studied sites.
Data represent mean ± SE of heavy metals concentrations in human urine for the studied sites using ANOVA.a

Cd: cadmium; Co: cobalt; Cr: chromium; Ni: nickel; Cu: copper; Pb: lead; Fe: iron; Zn: zinc; LSD: least significant difference.
aMean followed by same letters within columns is not significantly different by LSD at 5% level.
bBold values indicate significant variation of metals in studied sites.
Data represent mean ± SE of heavy metals concentrations in human nails for different age-groups.a

Cd: cadmium; Co: cobalt; Cr: chromium; Ni: nickel; Pb: lead; Fe: iron; Zn: zinc; LSD: least significant difference.
aMean followed by same letters within columns are not significantly different by LSD at 5% level.
Data represent mean ± SE of heavy metals concentrations in human nails for the studied sites using ANOVA.a

ANOVA: analysis of variance; LSD: least significant difference; Cd: cadmium; Co: cobalt; Cr: chromium; Ni: nickel; Pb: lead; Fe: iron; Zn: zinc.
aMean followed by same letters within columns is not significantly different by LSD at 5% level.
bBold values indicate significant variation of metals in different age-groups.
In hair samples, the concentrations of all analyzed metals varied. Generally, the mean concentration of heavy metals in hair samples was of the following order: Pb > Ni > Cr > Mn > Cd > Co. The results revealed that highest concentrations of Cr, Cd, and Co were shown in adults, whereas lowest levels were recorded in children. While maximum Ni and Mn concentrations were found in the children, the minimum levels were recorded in adults. The rank order of metals’ levels in groups was of the following order: adults > old age > children. In general, the increased levels of Pb and Ni concentrations in hair samples were recorded in all sites compared to other metals. The one-way ANOVA showed that Cd, Mn, and Zn varied significantly (p < 0.05) with age-groups. Whereas all the other metals significantly (p < 0.05) varied in the sites studied except Cd and Pb (Tables 7 and 8).
Data represent mean ± SE of heavy metals concentrations in human hair for different age-groups.a

LSD: least significant difference; Cd: cadmium; Co: cobalt; Cr: chromium; Ni: nickel; Pb: lead; Fe: iron; Zn: zinc.
aMean followed by same letters within columns is not significantly different by LSD at 5% level.
bBold values indicate significant variation of metals in age-groups.
Data represent mean ± SE of heavy metals concentrations in human hair for the studied sites using ANOVA.a

ANOVA: analysis of variance; LSD: least significant difference; Cd: cadmium; Co: cobalt; Cr: chromium; Ni: nickel; Pb: lead; Fe: iron; Zn: zinc.
aMean followed by same letters within columns are not significantly different by LSD at 5% level.
bBold values indicate significant variation of metals in age-groups.
Status of antioxidants in human blood
Analyzed metals (Cr, Cd, Pb, Ni, Co, Mn, Zn, and Fe) and antioxidant enzymes activity in human blood samples varied in volunteers from the study areas. The higher concentrations of toxic metals were recorded in blood samples of volunteers from polluted area as compared to those from control area. Lower concentrations of Fe were found in blood samples from exposed areas than the control area. The results showed the highest concentration of SOD, GSH, GPx, GR, and CAT in control site, while lowest activities of GSH, GPx, and CAT were found in all exposed sites (Figure 2). All the sites showed the lowest SOD and GSH activity in children, followed by adults and old age. GPx and GR activities were lower in the adults and old age than the children. Similarly, lowest CAT activity was observed in adults, followed by old age and children. Generally, in control and exposed sites, the SOD and GPx status were in the following order: medium risk > low risk > high risk > control. On the other hand, GSH and GR activities were obtained in the following rank order: high risk > low risk > medium risk > control. The contents of CAT in blood were in the following order: low risk > medium risk > high risk > control.
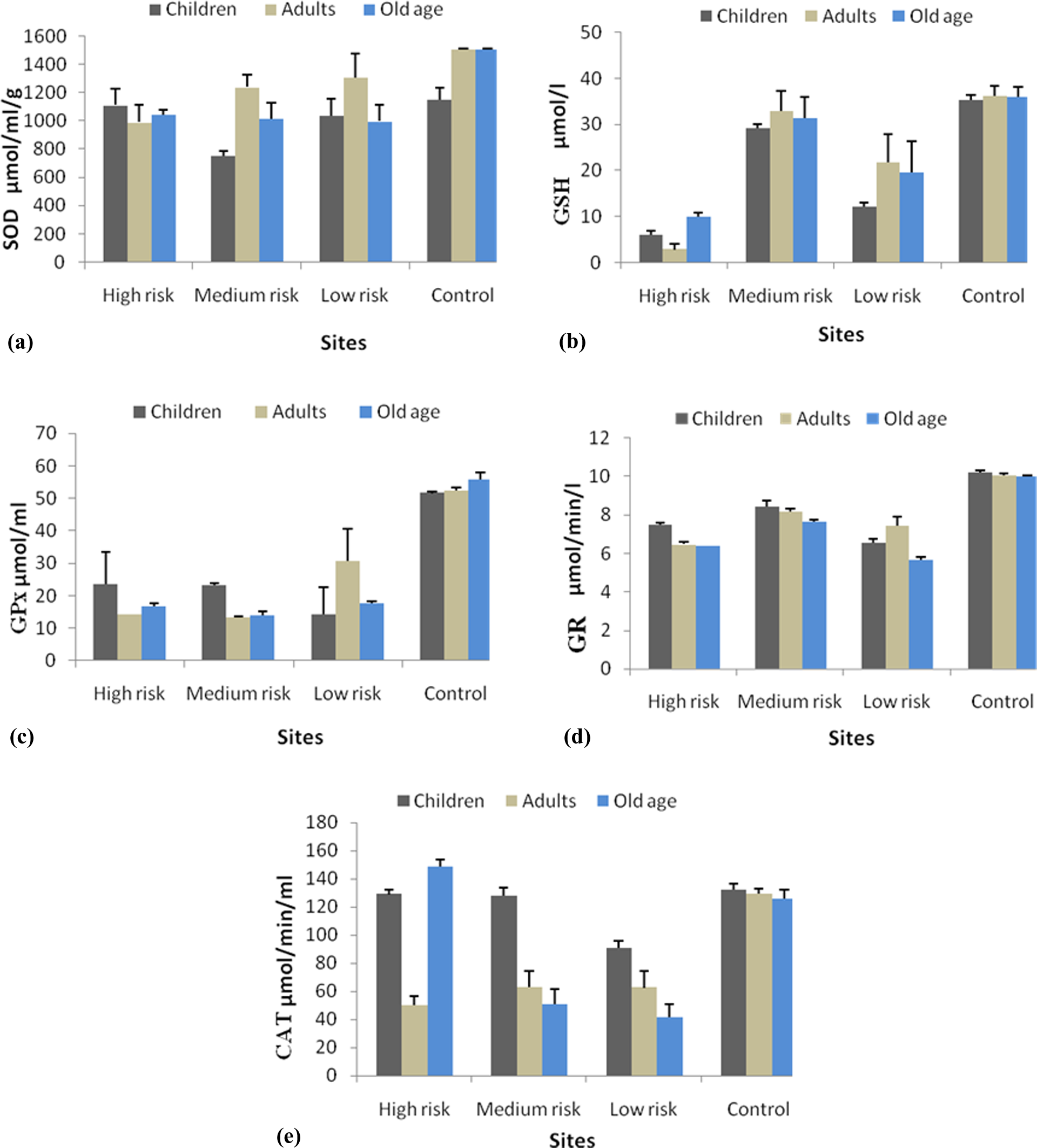
The activity of antioxidant enzymes in human blood samples.
Pearson’s correlation analysis of heavy metals with antioxidants in blood
The Pearson’s correlation was performed to understand the antioxidants’ response with heavy metals in human blood (Table 9). In blood, Cr, Cd, and Co showed highly significant positive correlation (p = 0.01) but negative correlation with GPx. Similarly, Cd exhibited significantly negative (p = 0.01) correlation with SOD. Ni showed significant negative correlation (p = 0.01) with SOD, GSH, and GPx in blood. On the other hand, Zn and Fe showed highly significant correlation (p = 0.05) with GPx and GR. While SOD and CAT were positively correlated (p = 0.01) with Fe. Similarly, GSH showed significantly positive correlation with Zn.
Correlation analysis between heavy metals in blood and antioxidant enzymes.

Cd: cadmium; Co: cobalt; Cr: chromium; Ni: nickel; Pb: lead; Fe: iron; Zn: zinc; GPx: gulatathione peroxidase; GSH: glutathione; GR: reduced glutathione; CAT: catalase; SOD: superoxide dismutase.
aCorrelation is significant at the 0.05 level (two tailed).
bCorrelation is significant at the 0.01 level (two tailed).
Discussion
Altered heavy metals’ concentrations in blood and urine
Results showed that of the nine metals studied, seven metals (Cr, Cd, Ni, Co, Pb, Zn, and Fe) were present in blood samples in exposed groups in comparison with the control. We observed higher concentrations of Pb, Cd, Ni, and Co in children than adults. For Cr, our results were persistent with thr results reported by Fátima et al. 24 who suggested that these concentrations were in relation with source and concluded that the concentrations of metals were related with environmental contamination. Our findings were concordant with Yousef et al. 25 who have recorded that mean concentrations of Pb, Cd, and Co were significantly higher in children than other groups and controls. Similar trend for Pb was found by Rahman et al. 26 who had shown a relationship between high levels of Pb and adverse behavior as well as bad performance on psychometric and educational tests.
For the concentrations of Cr and Ni in blood samples, our results were similar with Yuan et al. 27 who suggested that the levels of Cr and Ni in blood samples seemed to be associated with smoking or betel quid chewing. Setcos et al. 28 confirmed that dental alloys containing Ni might not expose the oral cavity to metals. Similar trend was recorded by Yuan et al. 27 who also checked the use of dental appliances and found no significant relation between cases and control. Therefore, increased levels of Ni and Co concentrations could not be attributed to occupational exposure, and the higher concentrations of Ni and Co might be caused by environmental factors, such as exposure rate, inhalation, and food intake. The presence of higher concentrations of metals in children than adults may be due to the exposure of children to contaminated soil by playing, dietary patterns, and gastrointestinal absorption.
Urinary concentrations of studied metals were higher in children as compared to adults and control. Cr levels were slightly higher in other groups. Despite this difference, mean concentrations of all metals were higher except Mn (not detected). The higher concentration of Cu was recorded in all groups with maximum in children and was similar with recent studies, 29,30 which found that higher concentrations of Cu in children might be probably associated with differences in dietary behavior, weaker metabolism, and frequent contact with soil. The concentrations of cadmium in urinary samples were similar with Bjermo et al. 31 who suggested that absorption might take place due to lower levels of Fe in the body. Furthermore, the authors also suggested that urinary Cd is inversely proportional to meat intake. While Bjermo et al. 31 observed no strong dietary factors attributing to blood and urine Cd concentrations. The authors concluded that the dietary habits within the groups were too homogeneous to detect any change in concentration, such as cereals (NFA, 2012); mainly bread and potato have been considered the dietary Cd sources; 32 Since the population in the area of study consume the locally grown vegetables the increased levels of all metals might be due to dietary habits.
Heavy metals in hair and nails
Human hair and nails are good indicators for toxic elements to evaluate long-term exposure (2–18 months). The main limitation of using hair and nails is the exogenous contamination. Despite the external contamination and the lack of standardized methodology, the World Health Organization, Environmental Protection Agency, and the International Atomic Energy Agency recommended the use of hair and nails as reliable biological material for worldwide human biomonitoring. 33
The variation in the concentrations of heavy metals in hair and nails samplesmight be due to different environmental settings. All studied metals showed higher concentration in nail samples except Cu. The mean concentrations of Pb in nail samples were similar with Samanta et al., 15 while the concentration of Cr, Cd, and Ni in nail samples complied with the results reported by Sukumar and Subramanian. 34 The concentration of Cd was higher in adults than children, suggesting that increased level of Cd in adults might be associated with tobacco smoking habits. However, the higher levels of Cr, Cu, and Ni might be due to change in diverse food habits, use of metal cookware, and differences in environmental exposure. Zn reflected lower levels in hair and nails samples, which could be due to drinking water, soil, and foodstuffs. 35 Afridi et al. 16 concluded that the elevated levels of Cd are responsible for the depletion of Zn in humans.
In nails samples, the concentrations of heavy metals were lower than those reported in previous studies. Maximum mean concentrations were found in adults than children. The concentrations of Cd, Cr, and Co were recorded higher in adults. The concentrations of Cd were similar with the results of Oyoo-Okoth et al., 36 while Cr and Pb concentrations were in good concordance with the results reported by Afridi et al. and Sukumar and Subramanian. 16,34 On the other hand, Ni concentrations were persistent with Samanta et al. 15 The other toxic elements like As, Cd, and Co can deposit in body from respiratory exposure. 37 The low levels of Fe and Zn are related to Cd, Pb, Ni, and Cu, 38 and common deficiency of Zn in world attributed mainly to nutritional factors.
Antioxidants’ response with increased concentrations of heavy metals
A metal is thought to cause toxic effect because of its capacity to lose electrons, thus resulting in production of reactive oxygen species (ROS). Therefore, exposure to high concentrations of a single heavy metal might result in its accumulation and potentially oxidative damage. 39 The activites of antioxidant enzymes and levels of endogenous antioxidants in blood are indirect but are reliable methods for assessing the activity of free radicals and oxidative stress. 40 Dimri et al. 41 found decrease in enzymatic activity with exposure to heavy metals. Lower levels of GSH in human blood samples might be due to its enhanced utilization for neutralization of excess free radicals generated. Similar concentration-dependent decrease in GR and GPx levels in human blood samples exposed to heavy metals was observed by Hunaiti and Soud. 42
SOD and CAT are primary antioxidant enzymes present in mammalian cells. SOD catalyzed the formation of O2 from ROS. A coproduct of SOD activity is H2O2, which is converted to water by CAT. 40 Decrease in SOD activity might be attributed to suppression in its synthesis to counteract free radicals.
Conclusion
The present study concluded that there were metal-specific differences in elemental concentrations in exposure groups among the studied sites. Higher concentrations of all heavy metals were found in blood samples compared to urine and hair samples. The activities of antioxidants enzymes decreased with increase in the concentrations of heavy metals. The results suggested that there is a need to assess optimal levels for dietary exposure in the study area and associated adverse health outcomes.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
