Abstract
Alcohol consumption and increased estrogen levels are major risk factors for breast cancer, and peroxisome proliferator-activated receptor γ (PPAR-γ) plays an important role in alcohol-induced breast cancer. PPAR-γ activity is inhibited by ethanol, leading to increased aromatase activity and estrogen biosynthesis ultimately leading to breast cancer. If other organic solvents inhibit PPAR-γ activity, they should also lead to increased oestrogen biosynthesis and thus be potential breast carcinogens. Ten commonly used hydrophilic organic solvents were first tested in a cell-based screening assay for inhibitory effects on PPAR-γ transactivation. The chemicals shown to inhibit PPAR-γ were tested with vectors encoding PPAR-γ with deleted AB domains and only the ligand-binding domain to rule out unspecific toxicity. Next, the effects on biosynthesis of estradiol, testosterone and oestrone sulphate were measured in the H295R steroidogenesis assay after incubation with the chemicals. Ethylene glycol, ethyl acetate, and dimethyl sulphoxide inhibited PPAR-γ transactivation in a dose-dependent manner. The inhibitory effect on PPAR-γ was specific for PPAR-γ since the AB domain of PPAR-γ was required for the inhibitory effect. In the second step, ethylene glycol significantly increased production of oestradiol by 19% (p < 0.05) and ethyl acetate inhibited production of testosterone (p < 0.05). We here show that screening of 10 commonly used organic solvents for the ability to inhibit PPAR-γ transactivation followed by a well-established steroidogenesis assay for production of sex hormones in exposed H295 R cells may provide a screening tool for potential breast carcinogens. This initial screening thus identified ethylene glycol and possibly ethyl acetate as potential breast carcinogens.
Keywords
Introduction
Breast cancer is the most common form of cancer among women worldwide, with the highest incidence in the industrialized countries. 1 The incidence of breast cancer has doubled over the last 30 years. 1 Both environmental and genetic factors are known to contribute to the aetiology of breast cancer. 2 The majority of the well-established risk factors are associated with prolonged exposure to increased levels of oestrogens, for example, early menarche, late menopause, few or no full-term pregnancies, short or no post-partum lactation and late childbirth. 2,3 Alcohol consumption is a well-known risk factor for breast cancer, 4 –6 and intake of one drink per day is associated with 7–10% increased risk of breast cancer. 2,7 –11 Controlled human and observational studies have shown that alcohol consumption is associated with increased blood levels of several sex hormones in pre- and postmenopausal women. 12 –18
A single nucleotide polymorphism in the PPARG2 gene has been associated with alcohol-related breast cancer in postmenopausal women. 19 Wild-type allele carriers of PPARG2 Pro 12 Ala had a 20% increased risk of breast cancer per 10 g alcohol (corresponding to one drink) per day, whereas variant allele carriers had no risk of alcohol-related breast cancer. 19 This indicates that the peroxisome proliferator-activated receptor γ (PPAR-γ), which is encoded by PPARG, is an important modulator in alcohol-related breast carcinogenesis. PPAR-γ regulates the expression of many adipose-specific genes via binding of the heterodimer PPAR-γ/retinoic X receptor (RXR) to specific DNA response elements (PPREs) in target gene promoters. 20 PPAR-γ is a negative regulator of aromatase expression in a complex with the co-activator PPAR-γ co-activator (PGC-1α). 21,22 Oestrogen is synthesized from testosterone in the biosynthesis step catalysed by aromatase. 23 We have shown that ethanol inhibits the PPAR-γ-PGC-1α complex and inhibits PPAR-γ activity. 24 Animal studies have demonstrated that ethanol intake leads to increased aromatase transcription, 25 which, in turn, leads to increased oestrogen biosynthesis. In support of this notion, we have confirmed that ethanol increases oestrogen excretion in the human H295R cell line. 24 These experiments collectively suggest that alcohol increases breast cancer risk by inhibition of PPAR-γ activity, resulting in obviation of a PPAR-γ-mediated inhibition of aromatase expression, leading to an alcohol-dependent increase in female sex hormone synthesis and thus increased breast cancer risk. 24 Inhibitors of PPAR-γ activity should therefore be considered as potential breast carcinogens.
We hypothesized that other chemicals that inhibit PPAR-γ activity may cause breast cancer by a mechanism similar to alcohol. Consequently, screening chemicals for the ability to inhibit PPAR-γ activity may provide a tool for identifying breast carcinogens. Indeed, several occupational studies have linked exposure of different chemicals, including organic solvents, to breast cancer. 26 –30 The largest study to date encompassing 15 million people aged 30–64 years with 373,361 cases of incident female breast cancer shows major occupational variation in breast cancer risk. 27 Some of the variations may be explained by differences in birth pattern, education and physical activity; however, as Hansen and Meersohn discuss in their Danish study, the differences in risk of breast cancer point to exposure in the working environment. 26 Consistent with this, working with organic solvents for just 1 year was associated with a 20–70% increased risk of breast cancer in another study by Hansen. 30 The risk estimates tended to increase with increasing duration of employment. All analyses were adjusted for social class, age at first child and number of children emphasizing the significance of work-related exposure to organic solvents as an isolated risk factor for breast cancer. 30
Most mammary carcinogens among organic solvents are lipophilic. 31 –33 They are hypothesized to cause cancer due to their ability to reside in the adipose tissue surrounding the breast parenchyma and lobules where the solvents and their bioactivated metabolites may exert harmful local effects. 31 In this study, we hypothesize that organic solvents may inhibit PPAR-γ in a manner similar to the organic solvent ethanol. Furthermore, we hypothesize that such solvents will also lead to increased oestrogen biosynthesis in the H295R assay. To test this hypothesis, we screened 10 chemicals for their ability to inhibit PPAR-γ transactivation in a widely used HEK293 cell-based screening assay. 34,35 The examined organic solvents were selected based on the following criteria: miscibility with water (>3% in water), widely used in Denmark with ‘very probable exposure’, according to the Danish SPIN2000 database, 36 suspected as being a mammary carcinogen 32,33 and/or found in human breast milk with a milk/plasma ratio above 1. 31 Afterwards, the positive candidates from the initial screening were assessed for oestrogen biosynthesis in an Organisation for Economic Co-operation and Development-validated method for testing of chemicals and their effect on androgen or oestrogen steroid hormone production. 37 We show that exposure to some widely used hydrophilic organic solvents increases female sex hormone levels possibly by a PPAR-γ-dependent mechanism and may thereby increase the risk of female breast cancer.
Methods
Test compounds
The following 12 organic solvents were initially included in the study (CAS number in brackets): methanol (67-56-1), dimethyl sulphoxide (DMSO) (67-68-5), isopropanol (67-63-0), ethylene glycol (107-21 -1), 1-propanol (71-23-8), acetone (67-64 -1), 1,4-dioxane (123-91 -1), benzaldehyde (100-52-7; Sigma-Aldrich®, Copenhagen, Denmark), acetonitrile (75-05-8; Merck®, Darmstadt; Germany), N,N-dimethylformamide (68-12-2; Fluka®, Copenhagen, Denmark), ethyl acetate (141-78-6) and diethyl ether (60-29-7; Rathburn Chemicals Ltd, Højbjerg, Denmark). Diethyl ether, however, cannot be pipetted in small amounts due to its high vapour pressure and benzaldehyde was extremely cytotoxic, even in small doses. Consequently, these two chemicals were excluded, leaving 10 organic solvents in the study.
Transient transfection
Human embryonic kidney (HEK293) cells 38 were grown in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum and antibiotics (62.5 µg/ml penicillin and 100 µg/ml streptomycin). HEK293 cells were transfected using 0.11 µl Metafectene®Easy+ (Biontex, Germany) per well in a 96-well plate. The plasmids used were the PPAR-γ-responsive luciferase reporter plasmid 39 (pcDNA3.1; Invitrogen, Waltham, Massachusetts, USA)-based expression constructs containing the full-length human PPAR-γ2 complementary DNA (wild type) and pRL-CMV normalization vector (Promega, Madison, Wisconsin, USA). All organic solvents tested positive, by giving indication of an inhibitory or inducing effect, were tested with (1) the same assay as above except that the pcDNA3 expression construct contains human PPARγ2 with deleted AB domains (N-terminal region) (pcDNA3hPPARγΔAB containing aa 110-475 (PPAR-γ1 coordinates)) 40 and (2) an assay with the following plasmids: A Gal4-responsive photinus luciferase expressing reporter plasmid (UAS-Gal), a pM1-based expression construct containing the ligand-binding domain (LBD) (aa 174-475 (hPPARγ1 coordinates)) of human PPAR-γ1 (donated by Dr Jan Fleckner, Novo Nordic, Denmark) and the pRL-CMV normalization vector. For each well in a 96-well dish, 30 ng reporter, 15 ng expression vector and 5.0 ng normalization vector were used. Six hours after transfection, the cells were supplemented with fresh media containing vehicle, rosiglitazone (1 µM) and test substance (0.1, 0.3, 1 or 3%). Some of the solvents showed severe sign of cytotoxicity at 3% and the top dose was consequently reduced to 1%. Cells were harvested after 18 h and Photinus and Renilla luciferase activities were measured directly in the plate using EnVision 2104 Multilabel Reader (PerkinElmer, Waltham, Massachusetts, USA). Photinus luciferase values were normalized to Renilla luciferase values to correct for differences in transfection efficiency and cytotoxicity or other cellular stress. All experiments were performed with four replicates and repeated twice. Each experiment represents one data point (mean value) leading to n = 3 in the data analyses.
Cell culture
Human adrenocortical carcinoma NCI-H295 R cells (ATCC CRL-2128)
37
were cultured in DMEM/nutrient mixture F-12 Ham without phenol red (Sigma) supplemented with 1% ITS Plus premix (BD Biosciences, Franklin Lakes, New Jersey, USA), 2.5% NuSerum (VWR, Radnor, Pennsylvania, USA), 1%
Analysis of hormone production
NCI-H295R cells were cultured as described above and used to investigate whether treatment with ethylene glycol, ethyl acetate, acetone or DMSO altered the production of oestradiol, testosterone and/or oestrone sulphate. The cytotoxic effect of each compound was assayed using the CellTiter 96 AQueous One kit (Promega). The hormone production assay was then performed with the highest concentration of each compound, which gave rise to a cell viability decrease of maximum 10% in four independent determinations leading to n = 4.
NCI-H295R cells were plated in 24 well plates and cultured for 48 h. The cell culture medium was then renewed, and the cells were treated with ethylene glycol, ethyl acetate, acetone or DMSO or left untreated (control) for 40 h. The culture medium was then collected and the hormone levels analysed. Production of oestradiol, testosterone, and oestrone sulphate was measured by enzyme-linked immunosorbent assay (ELISA). The ELISA kits (DE4399, DE1559, and DEV9933) were purchased from Demeditec Diagnostics GmbH, Germany. The analyses were performed in accordance with the manufacturer’s recommendations.
Statistical methods
Transient transfection: one-way analysis of variance (ANOVA) was performed, followed by a Dunnett’s posttest to examine differences between exposed groups and controls using GraphPad Prism 5 (San Diego, California, USA).
Hormone production: Kruskal–Wallis test was used to compare mean concentrations of the hormones. When results of the Kruskal–Wallis test were statistically significant, the groups were reanalysed using the Mann–Whitney test. These tests were conducted in Minitab 16.
Data were assessed for normal distribution and homogeneity of variances. The criteria for statistical significance were p = 0.05, p = 0.01, and p = 0.001 leading to the marking *, **, and ***, respectively.
Results
A transient transfection screening assay using a PPAR-γ2 expression construct 24 was performed in order to measure a possible inhibitory effect on PPAR-γ transactivation of 10 frequently used hydrophilic organic solvents.
Four of the tested solvents (ethyl acetate, ethylene glycol, DMSO and N,N-dimethylformamide) inhibited PPAR-γ-mediated transcriptional activation statistically significantly in a dose-dependent manner (Figure 1). However, the apparent inhibitory effect of N,N-dimethylformamide was caused by an increased expression of the pRL-CMV normalization vector, rather than by inhibition of PPAR-γ induced transcription. Consequently, N,N-dimethylformamide was excluded from further analyses. Ethyl acetate, ethylene glycol, DMSO and 1,4-dioxane were also able to inhibit rosiglitazone-induced PPAR-γ transactivation (Figure 2). Rosiglitazone is a selective PPAR-γ ligand, which was included in the assays to assess potential inhibition during ligand-dependent activation of PPAR-γ.

Transient transcription activation of PPAR-γ in a luciferase reporter gene in the presence of 10 different organic solvents. The full-length PPAR-γ2 cDNA was transiently transfected into HEK293 cells in combination with a PPRE luciferase reporter construct and PPAR-γ-mediated transcription activation was measured. Reporter activity was normalized to pRL-CMV values. Some of the experiments showed cytotoxicity at 3% and the top dose was consequently reduced to 1%. All transfections were performed independently three times in four replicates (i.e. n = 3). Results are presented as mean ± SD. *p < 0.05; **p < 0.01; ***p < 0.001: significant difference between treated and non-treated samples. PPAR-γ: peroxisome proliferator-activated receptor γ; cDNA: complementary DNA; PPRE: peroxisome proliferator response element.
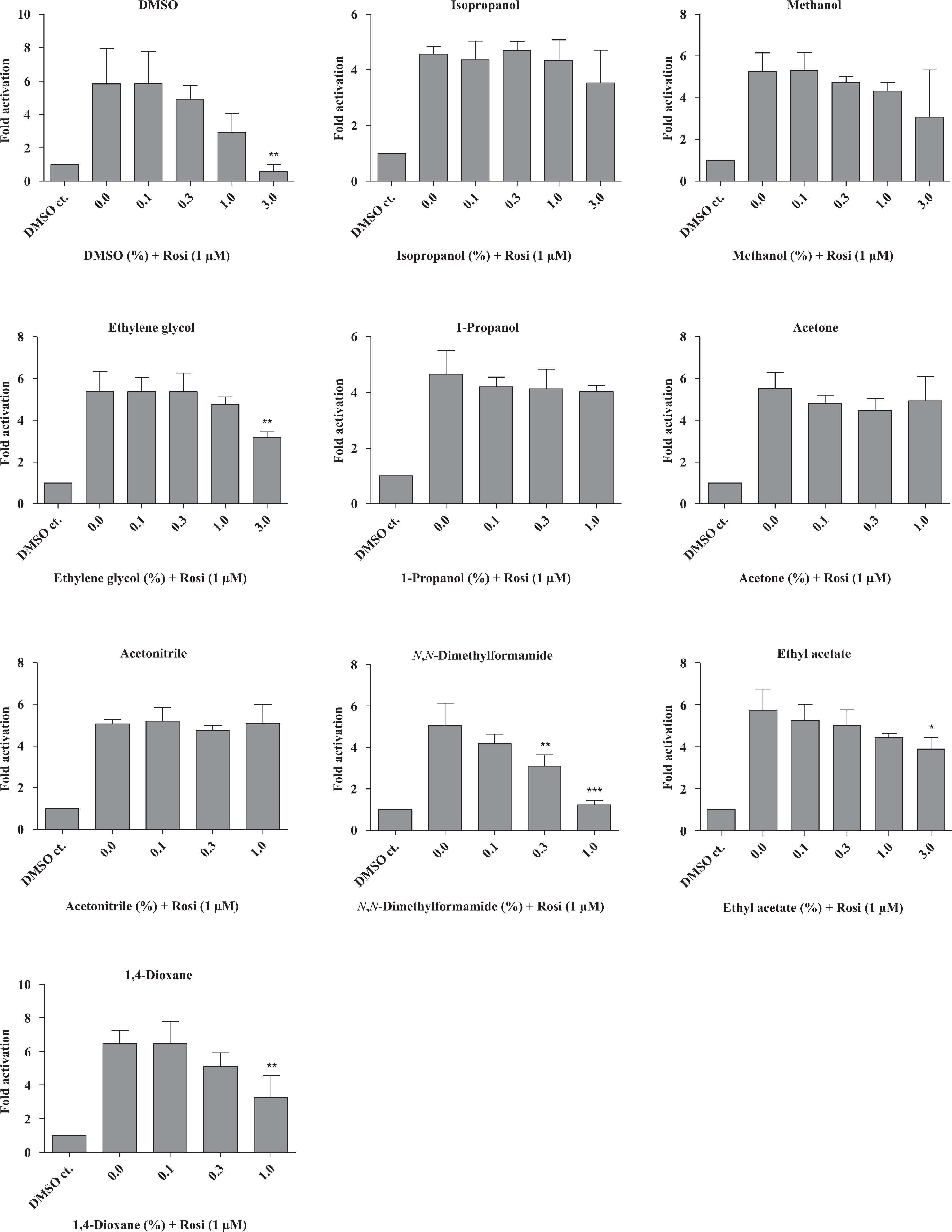
The same experiment as in figure 1, but with 1 µM rosiglitazone added in combination with the test compound. Rosiglitazone contains 0.1 % DMSO; therefore, a negative DMSO containing control is added to each experiment in order to isolate the DMSO effect. All transfections were performed independently three times in four replicates (i.e. n = 3). Results are presented as mean ± SD. *p < 0.05; **p < 0.01; ***p < 0.001: significant difference between non-treated samples (i.e. with rosiglitazone, but no test compound) and treated samples. DMSO: dimethyl sulphoxide.
In order to clarify whether inhibition of PPAR-γ was related to the region encompassing the PPAR-γ Pro 12 Ala substitution (i.e. the N terminal part of the AB domain) and to ensure that the observed inhibitory effect was not caused by general toxicity, we performed additional transactivation assays with vectors encoding PPAR-γ with deleted AB domains and PPAR-γ encompassing only the LBD domain, respectively.
Ethylene glycol and ethyl acetate did not inhibit transcriptional activation by hPPAR-γ(ΔAB) nor hPPAR-γ(LBD) (Figures 3 and 4). However, they did inhibit the rosiglitazone-induced transcription mediated by both constructs at 3% (Figures 3 and 4) and ethyl acetate also inhibited the rosiglitazone-induced transcription by the hPPAR-γ(ΔAB) at 1% (Figure 4). DMSO inhibited hPPAR-γ(ΔAB)-mediated expression at 1%, but not hPPAR-γ(LBD) expression (Figure 5). DMSO was able to inhibit the rosiglitazone-induced expression via both the hPPAR-γ(ΔAB) and the hPPAR-γ(LBD) construct but only at the highest dose (3%) (Figure 5). This indicates that the effect is PPAR-γ specific and that the AB domain was required for the inhibitory effects of ethylene glycol and ethyl acetate and to some extent also of DMSO. Furthermore, in concordance with previous observations, 40 the ligand rosiglitazone only enhanced transactivation by full-length hPPAR-γ or hPPAR-γ(ΔAB) by 4- to 6- and 6- to 8-fold, respectively, whereas ligand-dependent activation of the hPPAR-γ(LBD) construct was up to 30-fold.
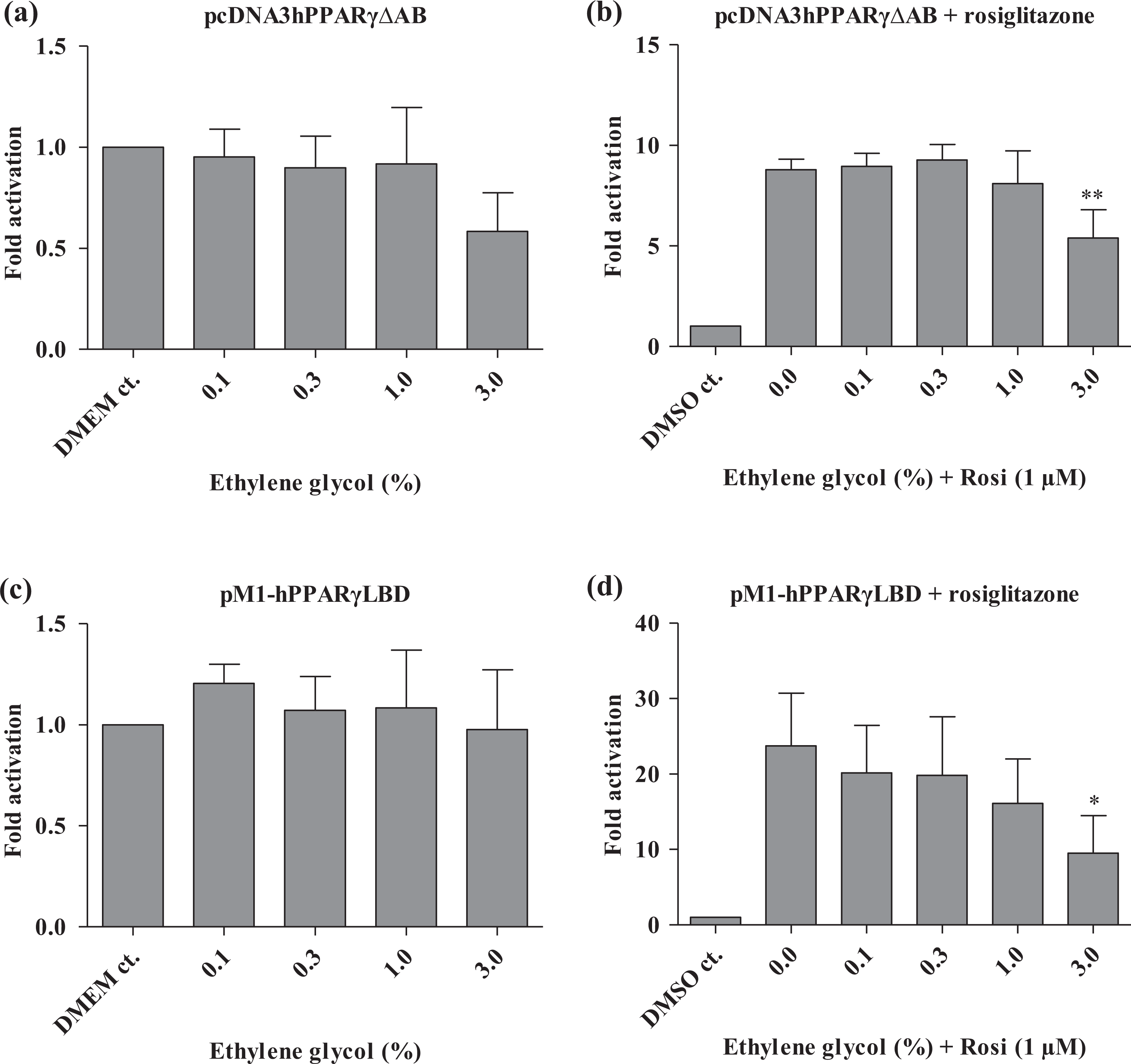
Effect of ethylene glycol on PPAR-γ constructs by transient transfections in HEK293 cells using either vectors encoding PPAR-γ with deleted AB domains (a and b) or pM1-PPAR-γ(LBD) (c and d) fusions with the appropriate luciferase reporter constructs. In (b) and (d), rosiglitazone is added to all wells except for DMSO controls. Reporter activity was normalized to pRL-CMV values. All transfections were performed independently three times in four replicates (i.e. n = 3). Results are presented as mean ± SD. *p < 0.05; **p < 0.01: significant difference between treated and non-treated samples. PPAR-γ: peroxisome proliferator-activated receptor γ; DMSO: dimethyl sulphoxide; LBD: ligand-binding domain.
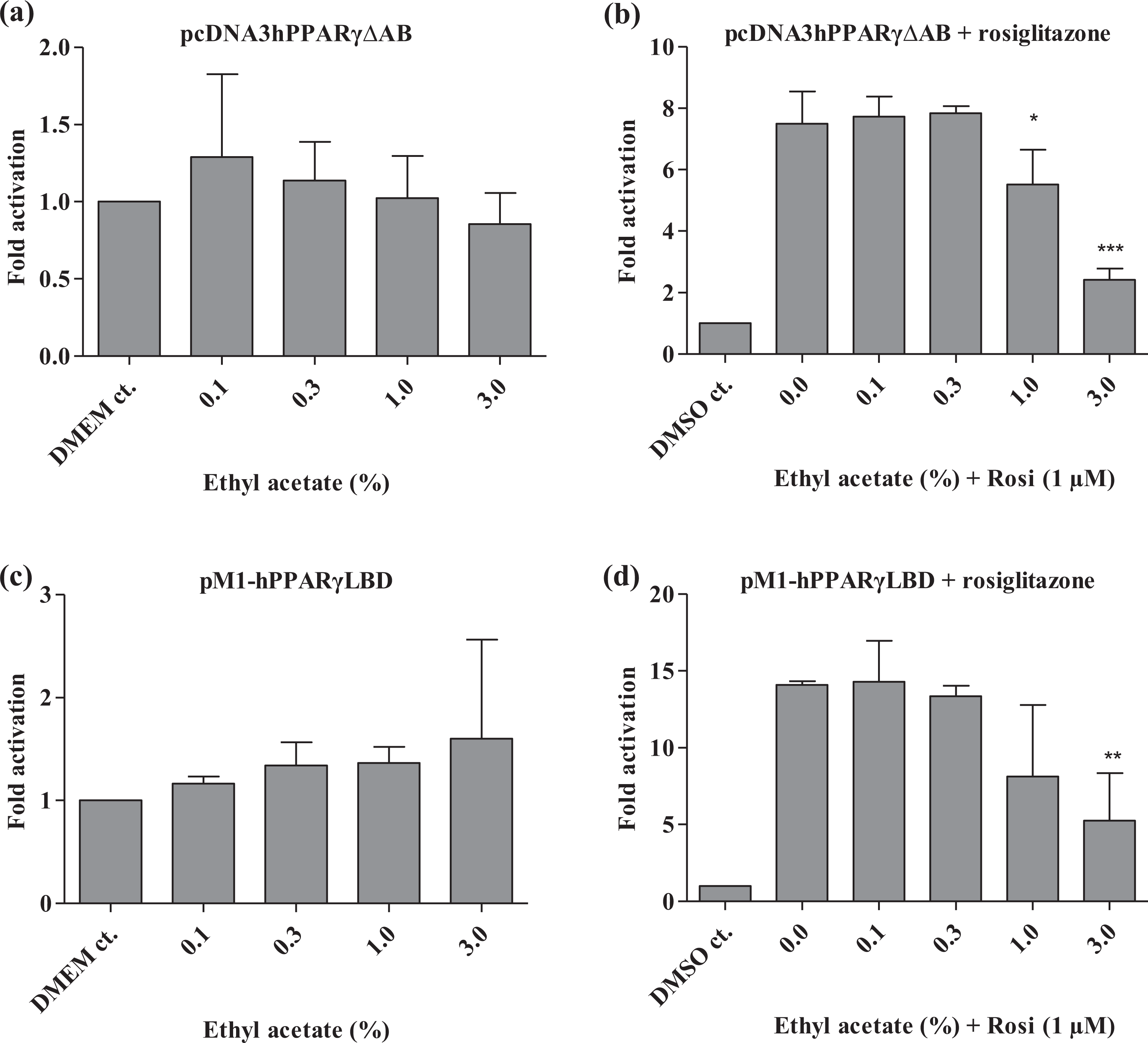
Effect of ethyl acetate on PPAR-γ constructs by transient transfections in HEK293 cells using either vectors encoding PPAR-γ with deleted AB domains (a and b) or pM1-PPAR-γ (LBD) (c and d) fusions with the appropriate luciferase reporter constructs. In (b) and (d), rosiglitazone is added to all wells except for DMSO controls. Reporter activity was normalized to pRL-CMV values. All transfections were performed independently three times in four replicates (i.e. n = 3). Results are presented as mean ± SD. *p < 0.05; **p < 0.01; ***p < 0.001: significant difference between treated and nontreated samples. PPAR-γ: peroxisome proliferator-activated receptor γ; DMSO: dimethyl sulphoxide; LBD: ligand-binding domain.

Effect of DMSO on PPAR-γ constructs by transient transfections in HEK293 cells using either vectors encoding PPAR-γ with deleted AB domains (a and b) or pM1-PPAR-γ (LBD) (c and d) fusions with the appropriate luciferase reporter constructs. In (b) and (d), rosiglitazone is added to all wells except for DMSO controls. Reporter activity was normalized to pRL-CMV values. All transfections were performed independently three times in four replicates (i.e. n = 3). Results are presented as mean ± SD. **p < 0.01; ***p < 0.001: significant difference between treated and non-treated samples. PPAR-γ: peroxisome proliferator-activated receptor γ; DMSO: dimethyl sulphoxide; LBD: ligand-binding domain.
We hypothesized that inhibition of PPAR-γ transactivation would lead to increased aromatase activity and thus increased oestrogen biosynthesis. In order to examine a direct effect on sex hormone levels, oestradiol, testosterone and oestrone sulphate production were measured in H295R cells after incubation with ethylene glycol, DMSO and ethyl acetate for 40 h. Moreover, acetone was included as a ‘negative control’ since acetone appeared to have no effect on PPAR-γ transcriptional activation. The highest concentration with maximum 10% effect on cell viability was considered as the no observed effect concentration for toxicity and was used in the test. A concentration of 0.3% ethylene glycol significantly increased production of oestradiol with 19% (p < 0.05; Figure 6) and 3% ethyl acetate significantly inhibited production of testosterone (p < 0.05; Figure 7). None of the tested solvents had any effect on oestrone sulphate production (result not shown). All results are provided in Table S1 as numerical values.

NCI-H295R cells were cultured for 48 hours and treated with ethylene glycol, ethyl acetate, acetone or DMSO or left untreated (control) for 40 h. The culture medium was then collected and the oestradiol production was measured by enzyme-linked immunosorbent assay. The hormone production assay was performed with the highest concentration of each compound, which gave rise to a cell viability decrease of maximum 10%. Results are presented as mean ± SD, n = 4. *p < 0.05: significant difference between treated and non-treated samples. DMSO: dimethyl sulphoxide.

NCI-H295R cells were cultured for 48 h and treated with ethylene glycol, ethyl acetate, acetone or DMSO or left untreated (control) for 40 h. The culture medium was then collected and the testosterone production was measured by enzyme-linked immunosorbent assay. The hormone production assay was performed with the highest concentration of each compound, which gave rise to a cell viability decrease of maximum 10%. Results are presented as mean ± SD, n = 4. *p < 0.05: significant difference between treated and non-treated samples. DMSO: dimethyl sulphoxide.
Discussion
Alcohol inhibits PPAR-γ-mediated inhibition of aromatase activity, which leads to increasing levels of circulating female sex hormones potentially increasing the risk of breast cancer. We hypothesized that exposure to other organic solvents may cause breast cancer by a similar mechanism. To test this hypothesis, we designed a two-step screening. We used an in vitro transactivation assay to screen for inhibition of PPAR-γ transactivation and, as a second step, screened candidates for increased oestrogen biosynthesis. Ten different hydrophilic organic solvents were selected for the screening based on their widespread use in Denmark and, hence, exposure potential. Moreover, 1,4 dioxane is a known mammary carcinogen, 32 and methanol has been found in human breast milk 31 indicating exposure.
We show that three out of 10 tested chemicals inhibit PPAR-γ transactivation in a dose-dependent manner using transient transfections with plasmids expressing three different PPAR-γ constructs – full-length PPAR-γ2, PPAR-γ with deleted AB domains and a construct consisting of only the LBD of PPAR-γ. The AB domain that encompasses the PPAR-γ Pro 12 Ala polymorphism seems to be essential for the inhibitory effect, especially for ethylene glycol and ethyl acetate where inhibition was confined to full-length PPAR-γ2. This confirms that the observed inhibition of PPAR-γ is not caused by general toxic mechanisms such as denaturation of all enzymes. In the second screening step, ethylene glycol, ethyl acetate, DMSO and acetone were tested in the H295R steroidogenesis assay for production of oestradiol, testosterone and oestrone sulphate production to determine whether inhibition of PPAR-γ results in increased female sex hormone levels. Acetone was included as a non-PPAR-γ inhibitor. Ethylene glycol increased production of oestradiol in a statistically significant manner at a concentration of only 0.3% and ethyl acetate inhibited production of testosterone with statistical significance at a concentration of 3%. Increased oestradiol levels could be a result of an increased synthesis from testosterone catalysed by aromatase or to inhibition of the subsequent metabolism of oestradiol to sulphate conjugate, catalysed by SULT1E1. Inhibition of SULT1E1 would result in accumulation of both oestradiol and oestrone. None of the tested chemicals affected oestrone sulphate production (results not shown). Thus, the results were consistent with the interpretation that ethylene glycol-inhibited PPAR-γ transactivation and caused increased oestradiol synthesis in H295R cells.
PPAR-γ is an important modulator of alcohol-induced breast cancer, 19,24 and the current study shows that PPAR-γ may be an important regulator of chemical-induced hormone changes. Thus, many chemicals have been shown to lower sex hormone production by stimulating PPAR-γ activity, leading to female infertility and male genitalia malformations. 41 –43 Interestingly, PPAR-γ agonists have been suggested to have therapeutic utility in the treatment of postmenopausal breast cancer due to their anti-oestrogenic properties. 23 Here, we screen for chemicals with the opposite, inhibitory effects on PPAR-γ transactivation activity, leading to increased oestrogen levels and breast cancer risk.
The decreased testosterone production after exposure to ethyl acetate could be caused by interference with other targets located prior to testosterone synthesis in steroidogenesis. However, if the substrate for oestradiol production (i.e. testosterone) is low, then the level of oestradiol could also become low. That indicates that there is indeed an increase in oestradiol production, but due to the low concentration of the substrate, the level of oestradiol is comparable with that of the control. Therefore, ethyl acetate and ethylene glycol may act via a similar mechanism. Our results therefore indicate that ethyl acetate also interferes with other targets in steroidogenesis. Indeed, it is a well-known phenomenon that chemicals affect more than one target in steroidogenesis. 44
It has previously been demonstrated that ethylene glycol is weakly oestrogenic in rainbow trout due to induction of the oestrogen receptor; 45 however, to our knowledge, no other studies have investigated the effect of ethylene glycol or ethyl acetate on steroidogenesis. DMSO, conversely, has proven to induce a variety of effects including numerous effects on lipid metabolism (summarized in the work by Santos et al. 46 ). Our results on DMSO are in agreement with these observations, since PPAR-γ is a major regulator of genes involved in lipid and lipoprotein metabolism. 20
This study was intended as an initial screening of commonly used hydrophilic organic solvents and their ability to (1) inhibit PPARγ in a well-established high-throughput cell-based screening assay and (2) increase endogen oestrogen levels in the H295R steroidogenesis assay. Since the H295R steroidogenesis assay is far more time consuming and expensive, but has been proven to correlate with in vivo mechanisms, we use the initial screening comprising of the HEK293 cells, which are easy to grow and transfect very readily to select appropriate candidates for this assay. 47 However, since organic solvents possess different properties, some of the negative findings in the transient transfection assay may simply be due to the chemicals not being able to enter the cells. Moreover, there may be discrepancies in the effect of absorption and metabolism of the test chemicals in different cell lines. The H295R steroidogenesis assay is designed to identify substances that affect targets in steroidogenesis from cholesterol to the production of the terminal steroid hormones, that is, not substances that affect steroidogenesis via a mechanism that involves the hypothalamic–pituitary–gonadal axis. 37 Therefore, stimulation of the aromatase activity caused by inhibition of PPAR-γ is a plausible mechanism for the effects observed in this study, but we did not assess PPAR-γ activity in the H295R assay, nor did we examine aromatase messenger RNA expression. However, H295R cells are of adrenal gland origin, which indicates that this specific mechanism is not limited to adipocytes. The genotype of the H295R cell line have previously been genotyped to carry the PPARG Pro 12 Ala wild type, 24 proving that the H295R cell assay is capable of PPAR-γ-PGC-1α interaction as the PGC-1α-dependent co-activation of the PPAR-γ-complex is compromised for the PPAR-γ Ala 12 variant.
It is highly important to identify human carcinogens as a mean to prevent future exposures. Mutagenic carcinogens are identified in a number of in vitro 48,49 and in vivo screening tests. 50 However, non-mutagenic carcinogens are more difficult to identify. We have previously provided evidence that ethanol causes breast cancer by a PPAR-γ-dependent increase in aromatase activity. 24 Based on this proposed mechanism of action, we here propose a screening approach for identification of breast carcinogens. We demonstrate that an in vitro screening of inhibition of PPAR-γ transactivation can be used to identify chemicals that may be breast carcinogens. As the second screening step, we used the H295R steroidogenesis assay to confirm an activation of aromatase activity. We show that at least one of three chemicals, ethylene glycol, leads to increased estradiol synthesis in H295R cells. The next screening step would be to show a similar effect in vivo in rodent models or in epidemiological studies. It has previously been demonstrated that ethanol intake increased aromatase activity and transcription levels in adipose tissue in orally dosed rats. 25 Conversely, human studies like register-based epidemiological studies are difficult to interpret since exposure often is a mixture of chemicals and they often lack information of important potential confounders such as cigarette smoking, alcohol intake and hormone replacement therapy. 51
Furthermore, our results indicate that some of the widely used organic solvents may possess endocrine-disrupting properties possibly via a PPAR-γ-dependent mechanism. 0.3% Ethylene glycol caused a 19% increase in oestradiol biosynthesis. It is not clear whether the observed inhibitory effect will cause increased hormone levels at occupationally relevant exposure levels. However, it is possible that mixed exposure to several organic solvents, including alcohol consumption, may contribute to breast cancer risk by the proposed mechanism and, as such, explains the observed association between exposure to organic solvents and breast cancer risk. 30
Conclusion
Based on the knowledge of mechanism of action of ethanol-mediated breast carcinogenesis, we propose a screening method for breast carcinogens. We show that screening of chemicals for the ability to inhibit PPAR-γ transactivation followed by a screening for increased oestradiol biosynthesis in exposed H295R cells may provide a screening for potential breast carcinogens. This initial screening identified ethylene glycol and possibly ethyl acetate as potential breast carcinogens. These candidates should be tested in rodent model systems for their effect on aromatase expression and activity in adipose tissues.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
The study was supported by a Mobility PhD grant (09-06 7572) from the Danish Council for Independent Research.
