Abstract
Valproic acid (VPA) is a drug used for the treatment of epilepsy, bipolar psychiatric disorders, and migraine. Previous studies have reported an increased generation of reactive oxygen species and oxidative stress in the toxic mechanism of VPA. Edaravone, a free radical scavenger for clinical use, can quench free radical reaction by trapping a variety of free radical species. In this study, effect of edaravone on some small intestine biochemical parameters in VPA-induced toxicity was investigated. Thirty seven Sprague Dawley female rats were randomly divided into four groups. The groups include control group, edaravone (30 mg–1 kg–1 day–1) given group, VPA (0.5 g–1 kg–1 day–1) given group, VPA + edaravone (in same dose) given group. Edaravone and VPA were given intraperitoneally for 7 days. Biochemical parameters such as malondialdehyde, as an index of lipid peroxidation(LPO), sialic acid (SA), glutathione levels and glutathione peroxidase, glutathione-S-transferase, superoxide dismutase, catalase, myeloperoxidase, alkaline phosphatase (ALP), and tissue factor (TF) activities were determined in small intestine samples by colorimetric methods. Decreased small intestine antioxidant enzyme activities, increased LPO and SA levels, and increased activities of ALP and TF were detected in the VPA group. Based on our results edaravone may be suggested to reverse the oxidative stress and inflammation due to VPA-induced small intestine toxicity.
Keywords
Introduction
Epilepsy is a chronic neurodegenerative disease which afflicts more than 50 million people worldwide. 1 Although there are many studies about the progression of epilepsy, it still remains very limited. It was reported that increased levels of free radicals and/or reduced activity of antioxidation defense system of body can cause seizures and increase the risk of seizure recurrence. 2,3
Valproic acid (2-n-propylpentanoic acid; VPA) is a branched-chain carboxylic acid used as an anticonvulsant and for the treatment of generalized epilepsy, bipolar disorder, migraine, and neuropathic pain. 4 Although VPA is used as an antiepileptic drug, it has several therapeutic indications and its use is associated with idiosyncratic hepatotoxicity and may cause fatality. 2 Significant side effects have been reported that can occur during VPA therapy, even in a patient on a prolonged and well-maintained dose. There is still a lack of knowledge about the incidence and occurrence of these side effects of VPA.
In several studies it was reported that VPA elevates the amount of free radicals and triggers oxygen-dependent tissue injury in the body and also has many side effects including pancreatitis, liver toxicity, skin rash, and tremor. 3,5,6 Furthermore, VPA toxicity can cause anorexia, nausea, and vomiting, and diseases such as jaundice, coagulation disorders, and hypoglycemia can also develop. 7 If the production of reactive oxygen species (ROS) is over the removal capacity, the cell membrane will be disturbed by free radicals.
Edaravone is a free radical scavenger which is used in patients with acute ischemic stroke in Japan. 8,9 It was shown that edaravone also has antiapoptotic, anti-necrotic, and anti-inflammatory effects, and it may prevent cell injuries and tissue damage by scavenging free radicals such as peroxy and hydroxyl radicals and by inhibiting lipid peroxidation (LPO). 10–13
The aim of this study is to evaluate the effects of VPA on oxidant and antioxidant status and inflammation markers and to investigate the potential role of edaravone on these effects in the intestine.
Materials and methods
Animal groups and treatment
All experimental protocols were approved by the Marmara University Animal Care and Use Committee. Animals were kept under standard laboratory conditions with free access to tap water and pellet food. Thirty seven Sprague Dawley female rats were randomly divided into four groups. The groups include group 1: control, group II: given only VPA (0.5 g–1 kg–1 day–1), group III: given only edaravone (30 mg–1 kg–1 day–1), and group IV: given VPA + edaravone (in same dose). Edaravone, VPA, and VPA + edaravone were given intraperitoneally for 7 days. In our preliminary experiments, dose–response studies have been performed. Then the experiment was performed with dosing; edaravone (30 mg–1 kg–1 day–1) and VPA (0.5 g–1 kg–1 day–1) were given to the related groups. VPA was given to the animals 1 h after edaravone treatment every day. On the day 8 of the experiment, animals were fasted overnight and then killed under anesthesia. Small intestine tissues were taken and homogenized; 10% (w/v) homogenates were prepared. Total protein, LPO, sialic acid (SA), glutathione (GSH) levels and glutathione peroxidase (GPx), glutathione-S-transferase (GST), superoxide dismutase (SOD), catalase (CAT), myeloperoxidase (MPO), alkaline phosphatase (ALP), and tissue factor (TF) activities were determined in small intestine homogenates.
Determination of proteins
Total protein levels of intestine tissues were determined according to the method described by Lowry et al. 14 Proteins react with copper ions in alkali medium and are reduced by Folin reagent. The absorbance of the blue-colored product was evaluated at a wavelenghth of 500 nm. Bovine serum albumin was used as a standard, and total protein levels of small intestine tissues were used to express the the results of the parameters per protein.
Determination of LPO
Malondialdehyde (MDA) is one of the products of LPO in tissues. The method described by Yagi 15 was determined by thiobarbituric acid (TBA) reactive substances (TBARS). LPO was expressed in terms of MDA equivalents using the extinction coefficient of 1.56 × 105 M–1 cm–1. Results are expressed as nanomoles of MDA per milligram protein.
Determination of GSH
GSH levels were determined in small intestine tissues according to the method described by Beutler. 16 Metaphosphoric acid was used for protein precipitation and 5,5′-dithiobis-2-nitro benzoic acid for color development. The extinction coefficient of 1.36 × 104 M–1 cm–1 was used for calculation of GSH levels. Results are expressed in milligrams of GSH per gram of protein.
Determination of SA
SA levels of small intestine tissues were assayed by the TBA method of Warren. 17 Briefly, samples were incubated with 0.1N sulfuric acid at 80°C for 1 h and hydrolysate was used for SA determination. In this assay, SA is oxidized with sodium periodate in concentrated phosphoric acid. The periodate oxidation product is coupled with TBA, and the resulting chromophore is extracted into cyclohexanone. The absorbances were measured at 549 nm, and the results were expressed in milligrams of SA per gram of protein.
Determination of ALP activity
ALP activity of small intestine tissues were determined by the method described by Walter and Shult. 18 ALP hydrolyzes p-nitrophenyl phosphate to p-nitrophenol depending on the pH of the medium. The absorbances were recorded spectrophotometrically at a wavelength of 405 nm and ALP activity was expressed as units per gram protein.
Determination of TF activity
TF activities of small intestine tissues were determined according to Quick’s one-stage method. 19 Pooled plasma, collected from healthy subjects, was used. TF activity was performed by mixing 0.1 ml of small intestine homogenate with 0.1 ml of plasma; the clotting reaction begins with the addition of 0.1 ml of 0.02 M calcium chloride. All reagents were brought to 37°C, which is the reaction temperature, before admixture. Since the clotting time is inversely proportional to the TF activity, the increase in the clotting time is a manifestation of decreased TF activity.
Determination of GPx activity
GPx activity of small intestine tissues was determined by the method described by Paglia and Valentine. 20 GPx catalyzes the conversion of hydrogen peroxide (H2O2) to water. The generated glutathione disulfide (GSSG) is reduced to GSH with consumption of nicotinamide adenine dinucleotide phosphate (NADPH) by glutathione reductase. During the oxidation of NADPH to NADP, the decrease in absorbance was measured spectrophotometrically at a wavelength of 366 nm. GPx activity was calculated using an extinction coefficient of 6.22 mM−1 cm−1, and the results are expressed as units per gram protein.
Determination of GST activity
GST is an enzyme that catalyzes the conjugation reaction between GSH and 1-chloro-2,4 dinitrobenzene. The absorbance of mixture at 25°C is monitored by spectrophotometer at 340 nm. GST activities in small intestine tissues were calculated using an extinction coefficient of 9.6 mM−1 × cm−1, and the results were expressed as units per gram protein. 21
Determination of SOD activity
SOD activity was quantified by the method which is based on its ability to increase the effect of riboflavin-sensitized photooxidation of o-dianisidine. 22 The activity of SOD is generated by illuminating the reaction mixture that contains o-dianisidine dihydrochloride and riboflavin by light from a fluorescent lamp. The oxidation of o-dianisidine, which is sensitized by riboflavin, is enhanced by SOD, and the increase is linearly dependent on SOD concentration. The absorbance of the colored product is quantified spectrophotometrically at a wavelength of 460 nm. Absorbances were measured at 0 and 8 min of illumunation, and the net absorbance was calculated. A standard curve was prepared routinely with bovine SOD (Sigma Chemical Co., S-2515-3000 U) as reference, and the results were expressed in units per milligram protein.
Determination of CAT activity
CAT activity of small intestine tissues was determined by the method described by Aebi. 23 The principle of the method was based on the conversion of H2O2 to H2O by the effect of CAT enzyme. This conversion was observed as a decrease of absorbance at 240 nm. CAT activity of samples was expressed as units per milligram protein.
Determination of MPO activity
MPO activity of small intestine tissues was determined by phenol-4-aminoantipyrine (4-AAP) assay containing a solution of tissue homogenate, phenol, H2O2, and 4-AAP as a color-generating substance. 24 The reaction occurs by adding excess amount of phenol, H2O2, and 4-AAP. The initial rate of the reaction is directly proportional to the amount of active enzyme in solution. One unit of MPO activity enzyme in small intestine tissue was defined as the amount of the enzyme present per gram of protein that caused a change in absorbance at 460 nm per minute in 37°C.
Statistics
Statistical analysis was performed using GraphPad Prism 5.0 (GraphPad Software, San Diego, California, USA). All data were expressed as means ± standard deviation. Kruskal–Wallis test was used for the comparison of groups of data followed by Dunn’s multiple comparison tests. A p value <0.05 was regarded as significant.
Results
Levels of LPO, GSH, and SA and activities of ALP and TF
The levels of LPO, GSH, and SA and activities of ALP and TF in small intestine tissues of each groups are shown in Table 1. Significant increase in small intestine LPO and SA levels were detected in VPA group compared with the control group and significant decrease in SA level was detected in VPA + edaravone group compared with VPA group. No significant difference was found in GSH levels between the groups. ALP activity significantly increased in VPA group compared with the control group and significantly decreased in VPA + edaravone group compared with VPA group. A significant increase was detected in small intestine TF activity in VPA group compared with control group and a significant decrease was detected in VPA + edaravone group compared with VPA group. Lengthening in seconds of TF activity is a manifestation of decreased activity.
Small intestine LPO, GSH, SA levels and ALP and TF activities in control and experimental groups.a
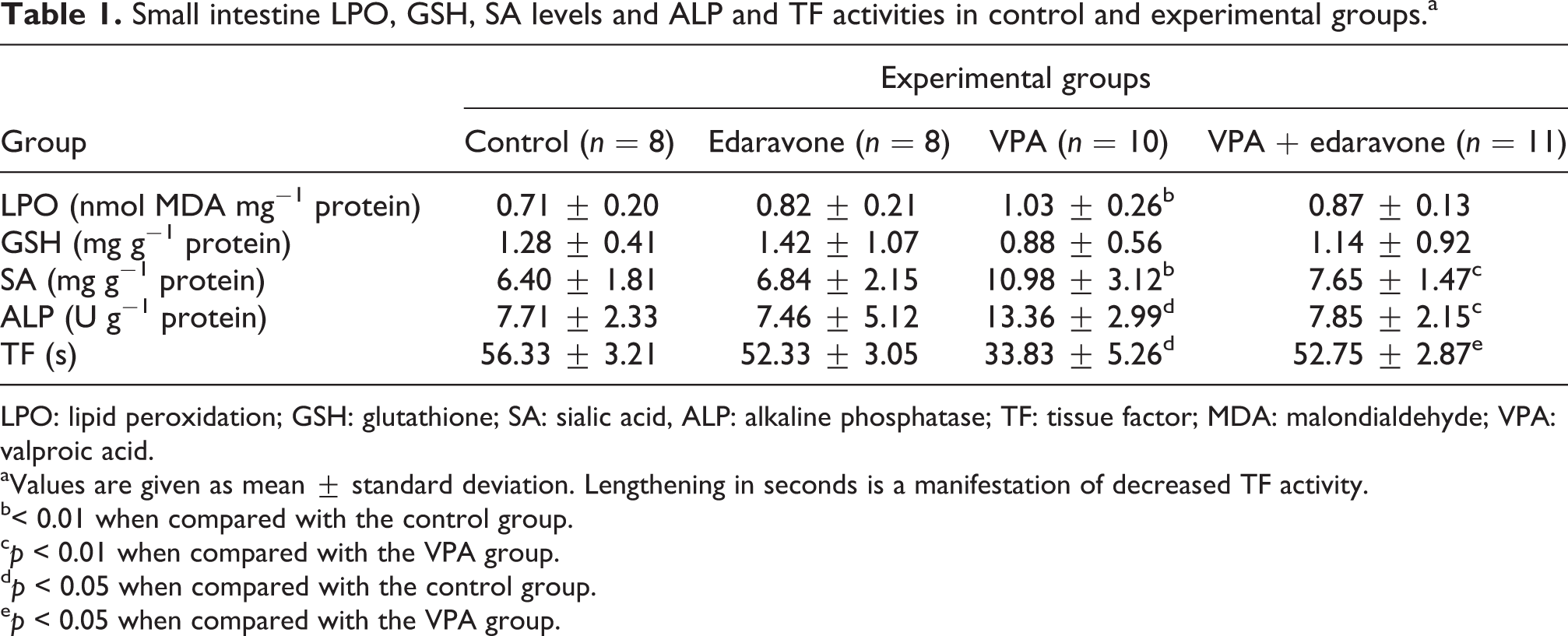
LPO: lipid peroxidation; GSH: glutathione; SA: sialic acid, ALP: alkaline phosphatase; TF: tissue factor; MDA: malondialdehyde; VPA: valproic acid.
aValues are given as mean ± standard deviation. Lengthening in seconds is a manifestation of decreased TF activity.
b< 0.01 when compared with the control group.
c p < 0.01 when compared with the VPA group.
d p < 0.05 when compared with the control group.
e p < 0.05 when compared with the VPA group.
Enzymatic activities of GPx, GST, SOD, CAT, and MPO
Activities of GPx, GST, SOD, CAT, and MPO in small intestine tissue of each groups are shown in Table 2. No significant differences were detected in GPx, SOD, and also CAT activities between the groups. A significant decrease was detected in the small intestine GST activity of VPA group compared with the control group and significant increase was detected in the GST activity of VPA + edaravone group compared with the VPA group. Small intestine MPO activity significantly increased in VPA group compared with the control group.
Activities of GPx, GST, SOD, CAT, and MPO in small intestine in control and experimental groups.a

GPx: glutathione peroxidase, GST: glutathione-S-transferase, SOD: superoxide dismutase, CAT: catalase; MPO: myeloperoxidase; VPA: valproic acid.
aValues are given as mean ± standard deviation.
b p < 0.01 when compared with the control group.
c p < 0.05 when compared with the VPA group.
d p < 0.05 when compared with the control group.
Discussion
VPA has several side effects accompanied by affecting antioxidant system and causing tissue damage such as hepatotoxicity and pancreatitis. It is metabolized by the liver but due to absorption from the gastrointestinal tract in therapeutic dosing of chronic use, gastrointestinal system can also be affected by toxicity of VPA.
Previous studies have reported that as a consequence of enhanced production of ROS, VPA treatment increases the LPO levels. Yuksel et al. 25 reported elevated serum LPO levels in epileptic children who had VPA therapy when compared to pretreatment group. Also, Hamed et al. 26 reported an increase in plasma LPO levels in epileptic adults who were treated with VPA. Tong et al. 27 found elevated LPO levels in liver in the VPA-treated rats, and Raza et al. 28 found increased levels of MDA in the liver and kidney of mice which had sodium valproate treatment. In agreement with these studies, we reported significant increases in LPO levels in VPA group. Although LPO level decreased in VPA + edaravone group, the decrease was insignificant.
SA is a generic term for derivatives of neuraminic acid, which is a monosaccharide with a nine-carbon backbone. Increased levels of SA has been reported during inflammatory processes, likely resulting from increased levels of sialylated acute-phase glycoproteins 29 and also in myocardial infarction, 30 hypertriglyceridemia 31 , and atherosclerosis. 32 SA has been suggested to be used as a marker for several inflammatory diseases and accordingly elevated SA reflects the self protection of the organism. 29,33 In this study, VPA led to increased SA levels in small intestine tissue compared with the control group, and edaravone treatment in VPA group decreased SA levels compared with VPA group.
ALP expression has been associated with the inflammatory process. Previous studies have reported elevated serum ALP activity due to the use of antiepileptic drug that causes an increase in osteoblastic activity. 34,35 Voudris et al. 36 reported increased ALP levels in bone due to VPA treatment and they found increased ALP activity in liver and bowel due to other antiepileptic drugs treatment. Sokmen et al. 37 also found increased ALP activity in rat liver treated with VPA for 15 days. In this study, a significant increase was detected in ALP activity of the VPA group compared with the control group and a significant decrease was detected in VPA + edaravone group compared with the VPA group. Increased activity of ALP shows the inflammation and neutrophil activation caused by VPA in small intestine tissue.
TF is a transmembrane receptor and cellular initiator of blood coagulation. 38 TF plays a critical role in thrombosis and thrombogenesis. 39 Various tissues and body fluids have been known to have TF activity. 40–43 Serdaroglu et al. 44 reported decreased levels of coagulation parameters in patients treated with VPA. We found a significant increase in small intestine TF activity in VPA group compared with the control group and significant decrease in VPA + edaravone group compared with VPA group. The increase in the clotting time is a manifestation of decreased TF activity. Increased TF activity may increase the risk of thrombosis; therefore, we may suggest that edaravone has acted as a protective agent regulating the small intestine TF activity by decreasing it. On the other hand, hematologic toxicity is commonly observed in VPA treatment. In several studies leukopenia and thrombocytopenia have been reported to occur due to VPA. 45
GPx and GSH play an important role in detoxification of peroxides and radicals in cells. GPx is a selenium-dependent enzyme, and in the presence of GSH, GPx catalyzes the conversion of H2O2 to water. The reports on GSH levels in VPA toxicity are different in experimental studies. Ubeda et al. 46 reported increased levels of liver GSH in rat injected with VPA intraperitonally, although Seckin et al. 47 reported reduced levels of GSH in rat liver which had a single dose of VPA intraperitonally. Also Raza et al. 28 found decreased GSH levels in the liver and kidney of VPA-treated mice. Ono et al. 48 found no difference in plasma GSH levels in epileptic group with VPA treatment compared with the control group. In our study, although GSH level decreased in VPA group compared with the control group and increased in VPA + edaravone group compared with VPA group, the difference was insignificant. Conflicting results also exist in GPx activities. Pippenger et al. 49 reported decreased GPx activity in erythrocyte of patients treated with VPA. Also Cotariu et al. 50 reported decreased GPx activity in rats that had VPA intraperitoenally. In contrast to these results, Hamed et al., 26 Cengiz et al. 51 , and Kurekci et al. 52 found increased GPx activity in patients who had VPA therapy. In our study, as GSH results, the decrease in GPx activity in VPA group and increase in VPA + edaravone group were insignificant. Decreased levels of GPx and GSH may present a reduced capacity of antioxidant status to prevent various injuries that occur through VPA metabolism and its side effects. Tong et al. 27 found elevated levels of α-GST, which is a marker of hepatocyte injury, in serum 4 days after VPA treatment in rats, whereas Chaudhary and Parvez 53 found reduced GST activity in cerebellum and cerebral cortex in rats. We detected significant decrease in small intestine GST activity in the VPA group compared with the control group and significant increase in VPA + edaravone group compared with VPA group.
SOD is another antioxidant enzyme to protect the cell against the harmful effects of superoxide anion radicals and H2O2. Pippenger et al. 49 found decreased erythrocyte SOD activities in patients receiving long-term VPA treatment. Yis et al. 54 found elevated levels of erythrocyte SOD activity although their results were insignificant. Also, in their 2-year prospective study, Yuksel et al. 25 found increased erythrocyte SOD activity after 1 year treatment but there was no difference between VPA treatment group and control group after 2 years. In agreement with previous studies, 52,55 in this study, SOD activity did not change in VPA group compared with the control group. Content of antioxidant enzymes varies for different samples, and this may be a reason for different results in enzymatic activity in VPA treatment.
CAT plays a role in detoxification of H2O2 in cells. There are limited studies about CAT activities in VPA treatment. Peker et al. 55 found no difference in serum CAT activities in children receiving VPA. In this study, CAT activities also did not change in the VPA group.
MPO is an enzyme that generates hypochlorous acid and other ROS, and it is present at high levels in circulating neutrophils and monocytes. Zhang et al. 56 found an increase in MPO activity in epileptic children, and also Sokmen et al. 37 found an increase in MPO activity in liver tissue of rats treated with VPA. In this study, we found increased small intestine MPO activity in VPA group. MPO is a marker for tissue neutrophil consent. Increased MPO activity shows an activated state of neutrophils against tissue injury.
In previous studies, VPA treatment has been shown to increase free radical formation and cell injury due to oxidative damage in various tissues. This study shows that VPA treatment does not only affect the target organs but also has some side effects on other tissues. Accordingly, we may suggest that in addition to known hepatotoxic and nephrotoxic effects, VPA also has toxic effects on gastrointestinal system.
VPA treatment may trigger oxygen-dependent cell injury and increase LPO levels that can cause deleterious effects due to increased H2O2. The risk of toxicity in cells increases due to increased ROS showing the impaired oxidant and antioxidant status, and consequently, oxygen-connected tissue damage occurs. Deficiency in detoxification and free radical elimination have been postulated as a cause of VPA toxicity, and long-term ROS increase may decrease antioxidant enzymes which prevent free radical formation.
Increased levels of SA and ALP and increased TF activity show that VPA treatment can have a role in development of inflammation caused by oxidative injury. Decreased levels of antioxidant enzymes show that detoxification system does not work due to changes in antioxidant enzyme activity. The decrease in these enzymes may increase the vulnerability of tissues to oxidative stress due to the imbalance between antioxidants and oxidants.
Edaravone, a potent free radical scavenger, can eliminate free radicals and helps to tolerate changes in antioxidant enzymes status. In previous studies, it was reported that edaravone has therapeutic effects on gastrointestinal system such as attenuation of intestinal system injury after ischemia reperfusion 57 and decrease of intestinal LPO. 58 We found decreased SA levels as an inflammation marker and increased GST activity in edaravone-treated VPA group. Edaravone inhibited free radical generation and ameliorated oxidative stress-induced inflammation. These results suggest that edaravone may be helpful to compensate the toxic effects of VPA during treatment and also edaravone may be used as a free radical scavenger in the treatment of ROS-related disease.
To our knowledge, this is the first study that evaluated the toxic effects of VPA treatment in small intestine tissue of rat. Besides the known side effects of VPA, we found hematologic and inflammatory effects on small intestine tissue. In patients receiving VPA therapy, our findings may be helpful for considering these side effects of VPA before treatment or other conditions. Edaravone may be able to reverse VPA-induced damage in tissues.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
