Abstract
Background:
Toll-like receptor 9 (TLR9), an important component of the innate immune system, contributes to the pathogenesis of lung injury. However, its role in paraquat (PQ)-induced lung injury has not been studied.
Method:
Mice were divided into PQ group (n = 24) and control group (n = 12). Mice in both groups were killed on either day 7 or day 28 after PQ (40 mg/kg intraperitoneally) or saline administration. TLR9 expression was evaluated through real-time polymerase chain reaction and immunohistochemistry. Concurrently, histopathological examinations of lung tissues were performed. On day 7, the lung weight to body weight ratios (LW/BW), total protein content, and inflammatory cytokine levels in bronchoalveolar lavage fluid (BALF) were measured. On day 28, fibrogenic cytokine expressions in lung tissues were measured. The correlations between TLR9 messenger RNA (mRNA) expression and different indicators of lung injury were then evaluated.
Results:
TLR9 expression was significantly increased over time in lung tissues after PQ poisoning. On day 7, TLR9 expression increased in parallel with LW/BW ratio (r = 0.403, p < 0.05), BALF protein content (r = 0.706, p<0.01), and BALF inflammatory cytokine levels (interleukin 6 (IL-6): r = 0.619, IL-1β: r = 0.930, tumor necrosis factor α: r = 0.589, all p < 0.05). On day 28, elevated TLR9 expression was closely correlated with Ashcroft score (r = 0.726, p < 0.01), mRNA expressions of α-smooth muscle actin, type I collagen and type III collagen (r = 0.926, 0.957, and 0.924, respectively; all p < 0.01).
Conclusions:
The TLR9 expression in lung tissue is markedly elevated during PQ-induced acute lung injury and pulmonary fibrosis and positively correlated with the severity of lung injury in mice.
Introduction
Paraquat (PQ) poisoning due to voluntary or accidental ingestion is a frustrating condition with exceptionally high mortality rates. 1,2 Lung injury is the main cause of death in PQ intoxication. It is characterized by acute pulmonary alveolitis followed by diffused lung fibrosis even after PQ is completely eliminated. 1,3 A mechanistic explanation for such progressive lung damage induced by PQ has not been fully understood.
Accumulating evidences showed that host immune response may contribute importantly to lung injury. Toll-like receptors (TLRs) constitute a crucial part of the innate immune system and have aroused increased interest in recent years. 4 –9 Among all TLRs, TLR9 specially recognizes pathogen DNA. Its role in lung diseases has been gradually uncovered. Recent studies have proved the TLR9 messenger RNA (mRNA) and protein expression in lung tissue. 10 Moreover, increasing TLR9 expression was detected in acute lung injury and chronic inflammatory lung diseases such as hypersensitivity pneumonitis, idiopathic pulmonary fibrosis, and chronic pulmonary obstructive disease. 10 –13 Inflammatory cytokines were induced by TLR9 activation to eliminate pathogens. 14 Neutrophils–endothelial cell interaction, which is critical for the initiation of acute lung injury, was demonstrated to be promoted by TLR9. 15 TLR9 also induces the differentiation of pulmonary fibroblasts in the development of idiopathic pulmonary fibrosis. 16,17 A few studies have showed that it may serve as a potential indicator to monitor the progression of lung fibrosis. 16,18
Given that TLR9 has been suggested to play an important role in the initiation and progression of lung injury, we speculate that TLR9 would involve in PQ-induced lung injury. In the present study, we therefore hypothesized that TLR9 expression in lungs would be elevated in a mouse model of PQ intoxication and its elevation would be closely associated with the severity of lung injury.
Methods
Animals and reagents
C57BL/6J mice (8 weeks) were purchased from Chinese Academy of Sciences (Shanghai, China). All study protocols were approved by the Animal Care Committees of Shanghai Jiaotong University Medical School. PQ was purchased from Sigma (St Louis, Missouri, USA).
Study protocol and sampling
A total of 36 mice were randomly divided into 2 groups: (1) PQ group (n = 24): Mice were intoxicated intraperitoneally (i.p.) with PQ (40 mg/kg) and divided into two subgroups as PQ-7d and PQ-28d group (n = 12 for each subgroup). 19 Mice were either killed on day 7 or day 28 after PQ administration. (2) Control group (n = 12): identical volume of normal saline was applied i.p.. Mice were killed concurrently as the PQ group on day 7 and 28 (n = 6, respectively).
On day 7 and 28, animals in each group were killed with pentobarbital (60 mg/kg, i.p.). A median thoracosternotomy was performed. The mouse lungs were lavaged three times with 1 ml of sterilized phosphate-buffered saline. The retained bronchoalveolar lavage fluid (BALF; about 90% of the instilled volume) was centrifuged at 1200 r/min for 10 min at 4°C. The recovered supernatants were collected and stored at −80°C until ready for assay. Both lungs were then harvested. The lung wet weight/body weight (LW/BW) ratio was determined. The left lung was then cut into two pieces, snap frozen in liquid nitrogen, and stored until mRNA extraction. The right lung was immediately fixed in 10% neutral-buffered formalin for histopathology.
BALF protein and inflammatory cytokine levels
Protein concentration and inflammatory cytokine levels (interleukin 1β (IL-1β), IL-6, and tumor necrosis factor α (TNF-α)) were measured by enzyme-linked immunosorbent assay (ELISA) according to the manufacturer’s instructions (BCA Protein Assay Kit, Pierce Chemical Co., Rockford, Illinois, USA; IL-1β, IL-6, and TNF-α ELISA kit, Shanghai Xitang Biological Technology Ltd, Shanghai, China).
Histopathological and immunohistochemical analysis
Lung tissue samples were embedded in paraffin. Routine hematoxylin and eosin (H&E) staining were performed. Lung fibrosis was identified by using Masson’s trichrome stain for collagen. For the area analysis of fibrotic changes, a quantitative fibrotic scale (Ashcroft scale) was used. 20,21
Immunohistochemical staining of TLR9 and α-smooth muscle actin (α-SMA) was performed on lung tissues. Consecutive slices were dewaxed and rehydrated with xylene or ethanol and subjected to antigen retrieval by citric acid (pH 6.0). To block endogenous peroxidase activity, the slices were incubated in 3% hydrogen peroxide for 10 min. Then, the samples were incubated overnight at 4°C with an anti-TLR9 antibody or anti-α-SMA antibody (Abcam, Cambridge, Massachusetts, USA). After that, the sections were incubated for 15 min at room temperature with horseradish peroxidase-labeled polymer conjugated with secondary antibody (Max-Vision™ kit; MaxVision BioSciences, Washington, USA). 3,3′-Diaminobenzidine was performed to develop color, and Mayer’s hematoxylin was used for counterstaining.
Real-time quantitative PCR analysis
Total RNA was extracted using TRIzol reagent (Invitrogen, Carlsbad, California, USA) according to the manufacturer’s instructions. Total RNA (1 μg) was reverse transcribed using the Prime Script cDNA Synthesis kit (Takara, Japan). Real-time polymerase chain reaction was performed using the SYBR Premix Ex (Takara, Japan) on ABI Prism 7900HT sequence detection system according to the manufacturer’s protocol. mRNA expression was assayed using the following primers: TLR9, (sense) 5′-CAACCTCAGCCACAACATTC-3′ and (antisense) 5′-GCACCTCCAACAGTAAGTCT-3′; α-SMA, (sense) 5′-GCATCCACGAAACCACCTA-3′ and (antisense) 5′-CACGAGTAACAAATCAAAGC-3′; type I collagen, (sense) 5′-GACGCCATCAAGGTCTACTG-3′ and (antisense) 5′-GAATCCATCGGTCATGCTCTC-3′; type III collagen, (sense) 5′-GGTCCACAAGGATTACAAGGCATA-3′ and (antisense) 5′-TCCAGGAGCACCGACTTCAC-3′; β-actin, (sense) 5′-AGATTACTGCTCTGGCTCCTA-3′ and (antisense) 5′-TCGTACTCCTGCTTGCTGAT-3′. Transcript levels were normalized by comparison with β-actin using the
Statistical analysis
Results were expressed as mean ± SD or median (interquartile range) as indicated. The 28-day survival rates were analyzed by the Kaplan–Meier method. The differences of survival were assessed using the log-rank test. All other parameters were evaluated by t test in combination with one-way analysis of variance test. The correlations between the variables were investigated using the Spearman’s correlation coefficient. Statistical analyses were performed using SPSS software (version 19.0, SPSS Inc., Chicago, Illinois, USA). Two-tailed values of p < 0.05 were defined as statistically significant.
Results
Changes in the destructive phase (day 7)
Six mice in the PQ-7d subgroup survived until killing on day 7. Pathological examination by H&E staining revealed diffused alveolar damage including alveolar wall disruption, alveolar hemorrhage, edema, and severe inflammatory cell infiltration in the PQ group compared with the control group. Masson staining showed that partial lung interval was broken which was presented as bulla and fibroblast proliferation in the PQ group (Figure 1).

Histopathological changes in the lungs after 7 days exposure to PQ. Bars = 100 μm (outside of inset) or 50 μm (insets). HE: hematoxylin and eosin stain; Masson: Masson’s trichrome stain; PQ: paraquat (57 x 40 mm2; 600 × 600 dots per inch).
The LW/BW ratio increased significantly for the PQ group compared with the control group (p < 0.01; Figure 2(a)).

LW/BW ratio, BALF protein, and inflammatory cytokine concentrations in control and PQ group (day 7). PQ: paraquat; LW/BW: lung weight-to-body weight; BALF: bronchoalceolar lavage fluid; IL-1β: interleukin-1β; IL-6: interleukin-6; TNF-α: tumor necrosis factor α (50 × 32 mm2; 300 × 300 dots per inch).
The BALF protein concentration was significantly higher in the PQ group than in the control group (p < 0.01; Figure 2(b)). Similarly, IL-1β, IL-6, and TNF-α levels in BALF were significantly elevated in the PQ group compared with the control group (all p < 0.05; Figure 2(c) to (e)).
Changes in the proliferative phase (day 28)
The 28-day survival rates of the PQ and the control group were 50% and 100%, respectively. Six mice in the PQ-28d subgroup survived. Significant difference of 28-day survival was observed between the PQ and the control group (p < 0.05).
Pathological examination of H&E staining showed extensive accumulation of inflammatory cells, thickening of alveolar walls, and fibrotic lesions in the PQ group (Figure 3). In the Masson-stained slides, the PQ group showed a greater amount of green fibers compared with the changes of the control group (Figure 3). The Ashcroft score semiquantitatively demonstrated that the degree of pulmonary fibrosis in the PQ group was significantly greater than that in the control group (p < 0.05; Figure 4(a)).

Histopathological changes of lungs on day 28 after PQ intoxication. Bars = 100 μm (outside of inset) or 50 μm (insets). HE: hematoxylin and eosin stain; Masson: Masson’s trichrome stain; PQ: paraquat (58 × 42 mm2; 600 × 600 dots per inch).
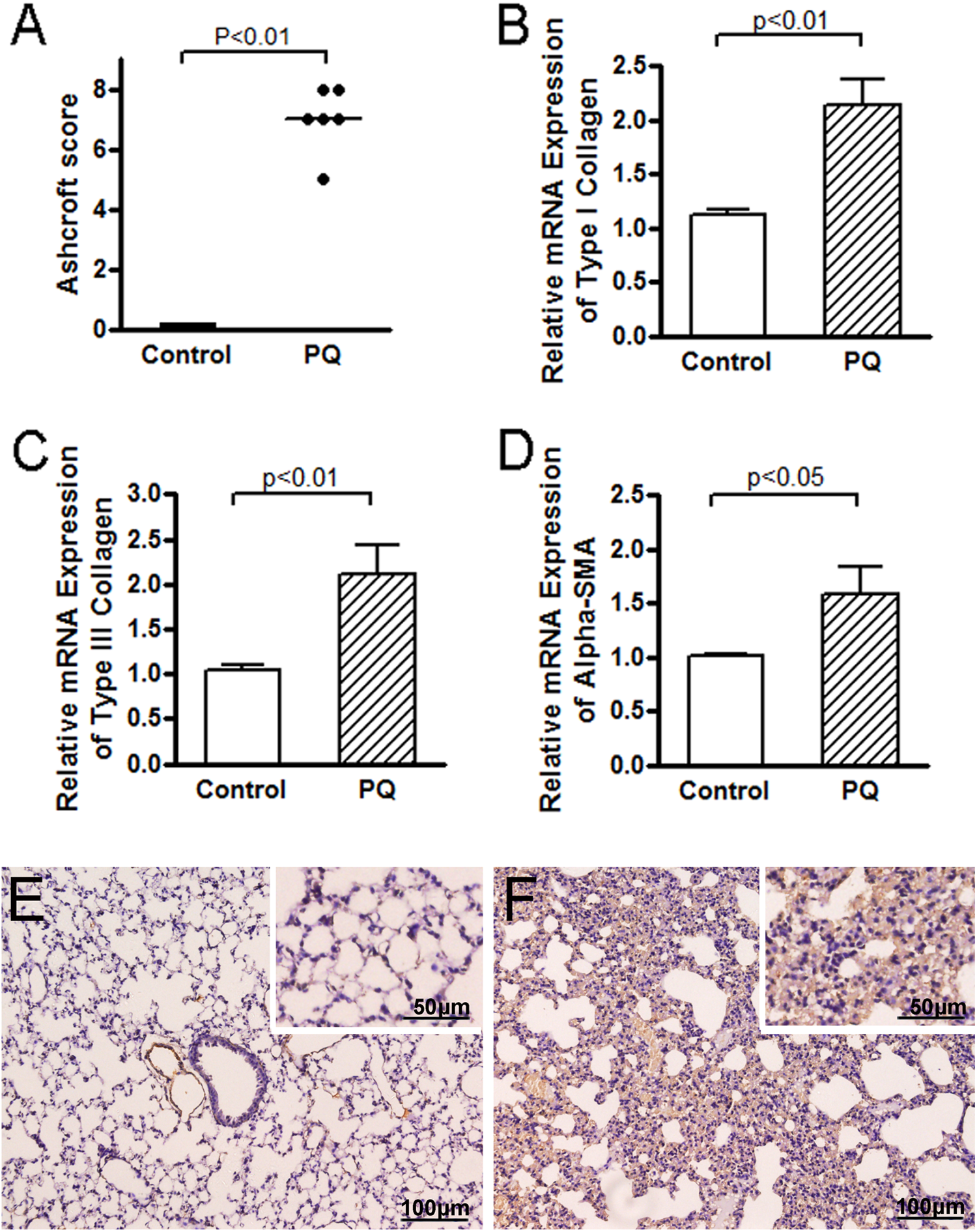
Grade of pulmonary fibrosis and expressions of fibrogenic cytokines in lungs (day 28). (a) Comparison of Ashcroft scores for the control and PQ group. (b, c, d) Comparison of relative mRNA expressions of type I collagen, type III collagen, and α-SMA in lungs for the control and PQ group. (e) Immunohistochemistry analysis of α-SMA in lungs from the control group. (f) Immunohistochemistry analysis of α-SMA in lungs from the PQ group. Bars = 100 μm (outside of inset) or 50 μm (insets). PQ: paraquat; mRNA: messenger RNA; α-SMA: α-smooth muscle actin (101 × 128 mm2; 300 × 300 dots per inch).
On day 28, mRNA expressions of fibrogenic cytokines (type-I collagen, type-III collagen, and α-SMA) in the PQ group were significantly increased as compared to the control group (Figure 4(b) to (d)).
Lung tissue immunostaining exhibited a similar greater expression of α-SMA in the PQ group than the control group (Figure 4(e) and (f)).
mRNA and immunohistochemical expression of TLR9
TLR9 mRNA expressions increased over time after PQ treatment. The relative mRNA levels in the PQ group were respectively 1.3 ± 0.3 and 2.4 ± 0.8 on day 7 and day 28 (both p < 0.05 compared with the control group; Figure 5(d)). Concurrently, immunohistochemical analysis of TLR9 expression revealed similar changes that TLR9 was highly expressed in the PQ group (Figure 5(a) to (c)).

Expressions of TLR9 in lungs. (a) Immunohistochemistry analysis of TLR9 in lungs from the control group. (b) Immunohistochemical expression of TLR9 on day 7 after PQ intoxication. (c) Immunohistochemical expression of TLR9 on day 28 after PQ intoxication. (d) Comparison of relative mRNA expression of TLR9 in lungs between groups. Bars = 500 μm (outside of inset) or 100 μm (insets). PQ: paraquat; mRNA: messenger RNA; TLR9: toll-like receptor 9 (93 × 70 mm2; 300 × 300 dots per inch).
Correlation between the TLR9 expression and different indices of lung injury
In the destructive phase (day 7), there were significant positive correlations between the TLR9 mRNA expression and the severity of acute lung injury as accessed by LW/BW ratio, BALF protein, and inflammatory cytokine levels.
Likewise, in the proliferative phase (day 28), significant correlation was shown between the TLR9 mRNA expression and the grades of lung fibrosis as measured by Ashcroft score. The expressions of fibrogenic cytokines (α-SMA, type I collagen, and type III collagen) were also closely correlated with the TLR9 expression (Table 1).
Correlations between the TLR9 mRNA expression and different indices of lung injury.

Discussion
In the present study, we investigated TLR9 mRNA and protein expression in PQ-induced lung injury. PQ (40 mg/kg i.p.) successfully induced two phases of lung injury: a destructive phase and a subsequent proliferative phase characterized by diffuse pulmonary fibrosis. TLR9 expression was significantly increased during PQ-induced lung injury and correlated with the severity of lung injury in both phases.
TLR9, under disease conditions, recognizes endogenous DNA such as mitochondrial DNA (mtDNA). 22 –24 In the setting of PQ poisoning, we speculate that mtDNA could be a strong activator of TLR9. PQ-induced multifocal cellular injury and necrosis release damaged mitochondria into the extracellular space, where they leak mitochondrial damage-associated molecular patterns (DAMPs) such as mtDNA. 25 Such changes were obviously noted as early as 24 h. 26,27 Our result that TLR9 expression was constantly elevated in the development of lung injury suggested that potential activators might stimulate TLR9 continuously and trigger a subsequent signaling cascade. The fact that PQ selectively accumulates in lung tissue to concentrations 6 to 10 times higher than those in plasma and maintains even when blood levels start to decrease is offered as part of the explanation. 1
Acute lung injury usually occurs within 1 week after PQ poisoning, and the severity can be evaluated by histological and biochemical changes. 1 It is in line with our results that apparent extensive alveolar damage and leukocyte infiltration in lung tissue accompanied by elevated inflammatory cytokines and protein content in BALF were found on day 7. Concurrently, TLR9 levels in lung tissue paralleled with the levels of TNF-α, IL-1β, and IL-6 in BALF which is consistent with previous studies regarding the relationship between TLR9 and inflammatory response. TLR9 is expressed not only predominantly in immune cells such as peripheral blood leukocytes but in various lung cells as well. 10 Although its role in lung cells has been little studied, TLR9 has been proved to promote leukocyte migration and transcription of inflammatory cytokine genes. 25 Knuefermann et al.’s study demonstrated a strong activation of pulmonary nuclear factor κB as well as a significant increase in pulmonary TNF-α and IL-1β mRNA/protein levels caused by mtDNA alike bacteria DNA via TLR9. 28 Moreover, it is noticeable that TLR9-targeted therapies have shown effects in some inflammatory lung diseases. Inhibition of TLR9 significantly attenuated the production of inflammatory cytokines. 13 TLR9-deficient mice displayed reduced inflammation, decreased accumulation, and activation of polymorphonuclear and reduced lung expression of inflammatory cytokines and chemokines. 11,28 Our finding that TLR9 was associated with the severity of acute lung injury implies that TLR9 may play an important role in the development PQ-induced acute lung injury.
A proliferative phase featuring diffuse lung fibrosis was appeared obviously on day 28 in the present study. TLR9 expression continuously went up and was significantly greater than the control group. Although there is no relevant data as for TLR9 expression after PQ exposure, our result was consistent with a previous study demonstrating that TLR9 expression was significantly higher in patients with idiopathic pulmonary fibrosis. 12 TLR9 was demonstrated to promote myofibroblast differentiation in lung fibroblasts. TLR9 activation significantly increased the expression of α-SMA, the main marker of myofibroblast differentiation. 17,18 It is consistent with our results that the elevated TLR9 expression was correlated with the severity of lung fibrosis as accessed by Ashcoff score and fibrogenic cytokine expression. These data suggest that PQ-caused TLR9 upregulation may drive the pathogenesis of progressive lung fibrosis caused by PQ.
To interpret the results of our experimental study, it is necessary to take several limitations into consideration. First, it is a 28-day experimental animal study. Although TLR9 expression was markedly increased during PQ-induced lung injury and associated with the severity of lung injury, whether or not TLR9 could serve as a prognostic marker is still inconclusive at the moment. More powerful studies are necessary to prove this hypothesis in the future. Second, the effect of TLR9 on fibroblast differentiation during the fibrotic process was not investigated. In the development of pulmonary fibrosis, multiple cells including epithelial cells, endothelial cells, and bone marrow progenitor cells have been identified as sources of fibroblasts. 29 –31 Deciphering interactions between TLR9 and these cells would improve our understanding of its role in PQ-induced lung injury. Third, mtDNA levels were not directly assessed in the present study although the effects of mtDNA on TLR9 expression were observed in previous studies. 32
In conclusion, our data demonstrate that TLR9 expression in lung tissue is markedly elevated during PQ-induced acute lung injury and pulmonary fibrosis and correlated with the severity of lung injury in mice. Further studies are needed to reveal the role of TLR9 in the progression of PQ-induced lung injury.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by Shanghai Science and Technology Commission [grant No.10140904000].
