Abstract
Illegal alcohol beverages known as bogma raki in our country are consumed widely in our region. The studies investigating the relationship between alcohol consumption and hearing ability report different results. In this study, we aimed to investigate the toxic effects of bogma raki that contains neurotoxic substances on cochlea by electron microscopy. To the best of our knowledge, this study is the first in the literature. A total of 48 Wistar male albino rats (aged 12–16 weeks and weighing 200–240 g) were used in the study. The rats were divided into 4 groups with 12 animals in each group. The groups include control, bogma raki, walnut, and walnut + bogma raki groups. Bogma raki (30% v/v, 9.2 ml kg−1 day−1) is added to drinking water of rats in bogma raki group (n = 12) for 4 weeks. Walnut group rats (n = 12) are fed with standard rat food and walnut without limitation (10 g kg−1 day−1). Bogma raki + walnut group rats (n = 12) are fed with standard rat food and walnut and bogma raki is added to drinking water. The cochleas were dissected and removed en bloc and examined by electron microscopy. Perineuronal oedema around neurons of spiral ganglion and hairy cells of organ of Corti were present in the bogma raki group, walnut group and bogma raki + walnut group under electron microscopic examination. Comparing these three groups, there were no differences in the ultrastructural pathological changes. In the ultrastructural examination of the myelinated axons forming cochlear nerve, no ultrastructural pathology was detected in all the groups.
Introduction
In Turkey, like all other countries in the world, illegal alcohol consumption is an important public health problem. Illegal alcohol consumption is widespread in East Europe, South America and Africa. In Turkey, it is widespread in Cukurova region. Illegal alcohol beverage known as bogma raki in our country is consumed widely in Cukurova region and in the sites of Hatay in the borderland of Syria. 1 The contents of these home-made beverages are different from each other and other legal alcohol beverages because they are produced by traditional methods. They are preferred to legal alcohol beverages produced in commercial and formal factories due to their low cost. 1 Zerenet al. examined bogma raki consumed in our region and reported detectable levels of severe alcohols such as trans-anethole, isoamyl alcohol, butanone 1-propanol in these beverages. 1 The neurotoxic effects of alcohol, methyl alcohol and their metabolites such as acetaldehyde, formaldehyde and formic acid are previously known. 2,3
The studies investigating the relationship between alcohol consumption and hearing ability report different results. 4,5 Some authors reported that alcohol consumption prevents hearing loss by prohibiting the disturbance of cochlear blood flow with an anti-thrombotic activity. 6 But there are also studies reporting hearing loss due to chronic alcohol consumption. 5,6
Walnut is used in the treatment of various diseases because of its anti-inflammatory and antioxidant (contains vitamin E, folic acid, melatonin and flavonoids) features. 5,7 Walnut is a nutrient rich in omega-3 fatty acids and α-linoleic acid. 5 It is previously reported that omega-3 polyunsaturated fatty acid intake may cease or prevent hearing loss due to ageing. 8 Moreover, vitamin E has a protective effect for hearing loss due to noise by reducing oxidative stress and increasing antioxidant enzyme activity by its antioxidant activity. 9
In this study, we aimed to investigate the changes in cochlea induced by bogma raki, which is widely used in this region due to low cost and which contains neurotoxic substances, and the effects of walnut which is an antioxidant nutrient rich in vitamin E by electron microscopy. To the best of our knowledge, this study is the first in the literature.
Methods
Ethics committee approval is obtained from Mustafa Kemal University Animal Research Native Ethics Committee. All experimental procedures were performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals issued by the Institute of Laboratory Animal Resources, Commission on Life Sciences, National Research Council. 10
Animals and treatment
A total of 48 Wistar male albino rats (aged 12–16 weeks and weighing 200–240 g) were used in the study. They were maintained according to the standard guidelines. All animals were housed reasonably in cages under standard environmental conditions (room temperature between 22°C and 24°C and 50% relative humidity within a 12-h light/12-h dark cycle photo period). The rats were divided into 4 groups. The groups include group 1: control; group 2: bogma raki; group 3: walnut; and group 4: bogma raki + walnut. Control group rats (n = 12) are fed with standard rat food without limitation for 4 weeks. Bogma raki (30% v/v, 9.2 ml kg−1 day−1) is added to drinking water of rats in bogma raki group (n = 12) for 4 weeks. Walnut group rats (n = 12) are fed with standard rat food and walnut without limitation for 4 weeks (10 g kg−1 day−1). Bogma raki + walnut group rats (n = 12) are fed with standard rat food and walnut without limitation for 30 days and bogma raki is added to drinking water of rats for 4 weeks.
Operation procedure
All procedures were performed under clean but non-sterile conditions. The rats were killed by intraperitoneal injection of 10 mg kg−1 xilazin (Rompun, Bayer, Turkey) and 40 mg kg−1 cetamine (Ketalar, Eczacıbaşı, Turkey), and the rats were then placed on the experiment table. The rats were euthanized by taking blood from their hearts. The skullbones of the subjects were cut in the midline, and each temporal bone was dissected using a scalpel and a pair of scissors. The cochlea were dissected and removed en bloc. The biopsy materials are stored in the lower section of refrigerator at +4°C according to cold chain in gluteraldehyde solutions for 72 h. At the end of 72 h, the specimens were taken into buffer solutions.
Tissue preparation
For transmission electron microscopic examination, the temporal bones of the rats were put into decalcification solution. This solution was prepared by mixing 0.5 cc gluteraldehyde (25% electron microscopy grade) and 0.3 g ethylene diamine tetraacetic acid (EDTA), and this mixture was completed to 4 cc by Sorenson’s phosphate buffer solution (pH 7.4). The folds of this solution was used during the preparation of decalcification solution, and every 2 weeks, this solution was changed with a freshly prepared solution. The duration of the decalcification period was 4 months and at the end of this period, the cochlea samples that were located inside the temporal bones were exposed by dissection and were put into 1% osmium tetroxide in phosphate buffer (pH 7.4) for 2 h for post-fixation. Then, the tissues were dehydrated in increasing concentrations of alcohol, washed with propylene oxide and embedded in epoxy resin embedding media. Semi-thin sections about 2 µm in thickness and ultra-thin sections about 60 nm in thickness were cut with a glass knife on a LKB-Nova (LKB-Produkter AB, Bromma, Sweden) ultrotome. The semi-thin sections were stained with methylene blue and examined using a Nikon Optiphot (Nikon Corporation, Tokyo, Japan) light microscope. Following this examination, the tissue blocks were trimmed, their ultra-thin sections were taken using the same ultrotome and were stained with uranyl acetate and lead citrate. Following staining, all the ultra-thin sections were examined using a Jeol JEM 1200 EX (Jeol Ltd, Tokyo, Japan) transmission electron microscope.
The composition of bogma raki
The toxicological analysis is carried out by the method followed by Zeren et al. Ethanol, methanol, 1-propanol, 1-butanol, 2-butanol, 2-methyl-1-propanol (isobutanol) and 3-methyl-1-butanol (isoamyl alcohol) were used as standards for qualitative and quantitative analyses of spirits. All chemicals were of analytical grade and were supplied by Merck (Darmstadt, Germany). The spirits were analyzed on a Hewlett-Packard (Palo Alto, California, USA) Gas chromatography–mass spectrometry (GC/MS) system consisting of an HP 6890 gas chromatograph, an HP 5972 mass selective detector (MSD) and an HP 6890 automatic liquid sampler. Separations of spirits compounds of methanol, ethanol, 2-butanol, 1-propanol, 1-butanol and isoamyl alcohol were accomplished using an HP-FFAP (25 m × 0.2 mm inner diameter, 0.33 µm film thickness) cross-linked capillary column (Hewlett-Packard Co.).
The pressure of the carrier gas helium was set at 6.0 bar and the split value was set in the ratio of 1:100. The injection unit temperature was set to 250°C and MS quadrupole temperature to be 280°C. The MS quadrupole detector ionization energy was set to 70 eV. The initial column temperature was 60°C (for 4.0 min) programmed by 6°C min−1 to final temperature 160°C and kept for 4 min at 160°C.
A comparison between the retention times of the samples with those of authentic standard mixture (Merck, Darmstadt, Germany; 99.9% purity specific for gas–liquid chromatography), run on the same column under the same conditions, is made to facilitate identification. After that the chromatograms of all spirits used in the study were verified by accessing WILEY database.
The composition of bogma raki applied to rats is shown in Table 1.
Chemical composition of illegally produced bogma raki sample (% v/v; the alcoholic strengths of the bogma raki 44%).

Results
Perineuronal oedema around neurons of spiral ganglion and hairy cells of organ of Corti were present in the bogma raki group (Figure 1). In the ultrastructural examination of the myelinated axons forming cochlear nerve, no ultrastructural pathology was detected (Figure 2).
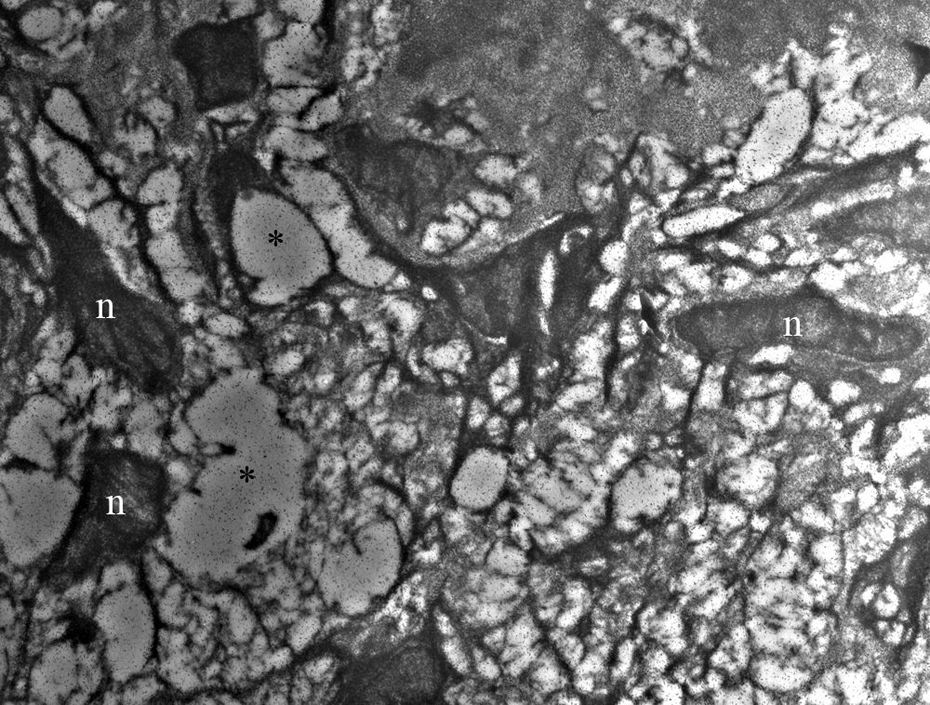
Perineuronal oedema around neurons in spiral ganglion in the bogma raki group (original magnification ×5000). n indicates neuron; * indicates perineuronal oedema.
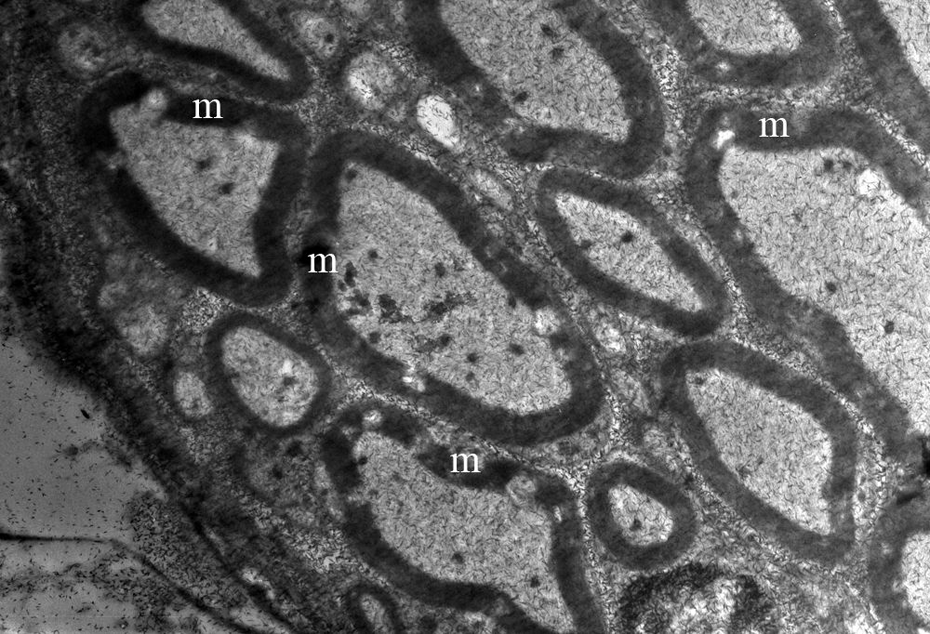
The ultrastructural appearance of myelinated axons forming nervus cochlearis in bogma raki group (m; original magnification ×10,000).
Perineuronal oedema around neurons of spiral ganglion and hairy cells of organ of Corti were present in the walnut group (Figure 3). In the ultrastructural examination of the myelinated axons forming cochlear nerve, no ultrastructural pathology was detected (Figure 4).
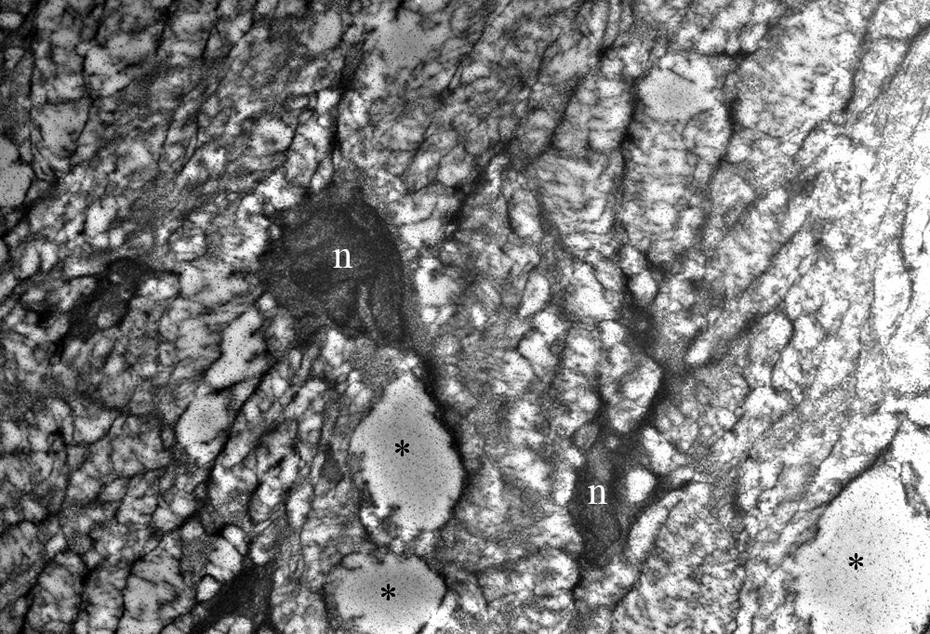
Perineuronal oedema around neurons in spiral ganglion in the walnut group (original magnification ×5000). n indicates neuron; * indicates perineuronal oedema.

The ultrastructural appearance of myelinated axons forming nervus cochlearis in walnut group (m; original magnification ×10,000).
Perineuronal oedema around neurons of spiral ganglion and hairy cells of organ of Corti were present in the bogma raki + walnut group (Figure 5). In the ultrastructural examination of the myelinated axons forming cochlear nerve, no ultrastructural pathology was detected (Figure 6).

Perineuronal oedema around neurons in spiral ganglion in the bogma raki + walnut group (original magnification ×5000). n indicates neuron; * indicates perineuronal oedema.

The ultrastructural appearance of myelinated axons forming nervus cochlearis in bogma raki + walnut group (m; original magnification ×10,000).
No ultrastructural differences were detected in neurons of spiral ganglion, hairy cells of organ of Corti and axons forming cochlear nerve by the electron microscopic examination of control group (Figure 7).
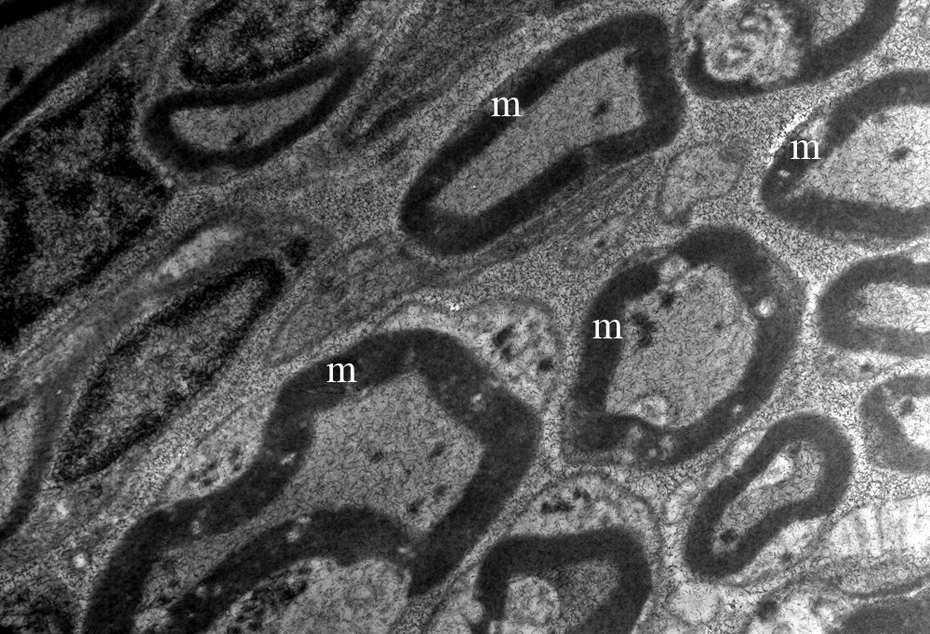
The ultrastructural appearance of myelinated axons forming nervus cochlearis in control group (m; original magnification ×10,000).
Discussion
In this study, we aimed to investigate the effect of bogma raki and walnut consumption on hearing process by examining the cochlea of the rats by electron microscopy. It has been shown that there were no differences between groups; therefore, it is inferred from the results that this consumption did not affect hearing clinically for this time period. The effects of alcohol consumption on the hearing thresholds in early and late periods are unknown. To the best of our knowledge, there is no previous study investigating the cellular effects of alcohol and walnut consumption in the cochlea. However, there are numerous studies investigating the audiological effects of alcohol consumption on hearing.
Alcohol is absorbed in gastrointestinal tract rapidly after intake and spreads by blood circulation to whole body and later metabolize in liver. It effects virtually all the organs in the body. 2,4,11 Various factors such as age, sex, drug use and familial history of alcohol addiction contribute to the effects of alcohol in human body. 4 There are various opinions in the literature about the effect of alcohol on hearing. Kahkonen et al. reported that due to ageing hearing is affected and in addition, due to alcohol consumption temporary effects on hearing during ageing become permanent in prolonged period. 12 Upile et al. examined acoustic reflex thresholds in narrow and wide bands in young population before and after alcohol intake and reported that a significant shift in reflex thresholds in wide bands after alcohol intake was present. 4 Moreover, they reported that low frequencies were affected more than high frequencies. Similarly, Popelka et al. reported that alcohol consumption reduced the capability of hearing. 13 In our study, the early effects of bogma raki and walnut are investigated. As the present study was conducted for a period of 4 weeks, the study period was inadequate to indicate the damage outcome of chronic alcohol consumption. Similar studies conducted for longer periods will be further beneficial. Hwang et al. reported that acute alcohol intake as a level of intoxication caused no change in auditory thresholds but reduced the amplitude of high frequencies on otoacoustic emissions. 14 The authors also reported that moderate acute alcohol intake caused a significant decrease in the amplitude of high frequencies on otoacoustic emissions. 14 Uhles et al. reported that moderate alcohol intake caused significant decrease in acoustic reflex threshold in chinchillas. 15 Audiological tests were not carried out because of technical inadequacy. However, without audiological tests the effects of bogma raki and walnut on hearing thresholds were not adequate.
Some authors defend that there is no relationship between alcohol consumption and hearing. Curhan et al. reported that they detected no relationship between moderate alcohol consumption and hearing in 26,809 healthy people. 6 But they reported that higher doses of alcohol consumption (especially liquor) and low vitamin B12 levels are related with hearing loss. The author reported that alcohol protected the cochlear blood flow in optimal level by reducing coagulation and increasing high-density lipoprotein cholesterol levels and did not cause hearing loss. 6 Brant et al. did not detect a significant relationship between moderate alcohol consumption and hearing loss. 16 Gopinath et al. reported that alcohol consumption has protective effects on hearing in low frequencies. The author reported no relationship between moderate alcohol consumption and hearing loss. 17 Fransen et al. reported that hearing loss is affected by multiple factors, but moderate alcohol consumption has no effect on hearing loss. 18
Walnut is an antioxidant and anti-inflammatory nutrient that contains omega-3 fatty acids, polyunsaturated fatty acids (α-linoleic acid), vitamin E, folate, melathonin, flavonoids and polyphenolics. 5 Shabani et al. reported that walnut prevented from neurotoxicity due to cisplatin without reducing its anti-cancer effect. 5 Although no study intended the relationship between walnut and hearing, there are studies indicating the positive relationship between antioxidants and anti-inflammatory agents and hearing. 8,9 Gopinath et al. reported that long-chain polyunsaturated fatty acid intake in diet reduced hearing loss risk with a ratio of 24%. 8 They reported that they did this by supplying cochlear blood flow in an adequate level due to their cardiovascular effects. Generally, antioxidants such as vitamin E contribute to non-enzymatic cellular defence against lipid peroxidation. As a result, antioxidants show positive effects by preventing hearing loss. 9 Kalkanis et al. investigated the cochlear damage in rats by causing ototoxicity with cisplatin intake and simultaneously provided vitamin E as a protector. The author detected that cochlear outer hairy cells were protected in the vitamin E-applied group. 19 However, in the present study, no significant differences were detected in outer hairy cells in electron microscopic examination between the groups fed with walnut, bogma raki and bogma raki + walnut. This may bedue to shorter application time for the cellular effects to begin their action in cochlea of severe alcohol metabolites.
Although with some limitations, the present study provides adequate information on the effects of bogma raki and walnut on cochlea in acute period under electron microscopic examination. Furthermore, more detailed studies investigating the effects of bogma raki and walnut with their pathways will be beneficial.
Conclusion
Perineuronal oedema around neurons of spiral ganglion and hairy cells of organ of Corti were present in the bogma raki group, walnut group and bogma raki + walnut group in electron microscopic examination. In the comparison of these three groups, there were no differences in the ultrastructural pathological changes. In the ultrastructural examination of the myelinated axons forming cochlear nerve, no ultrastructural pathology was detected in all the groups.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
