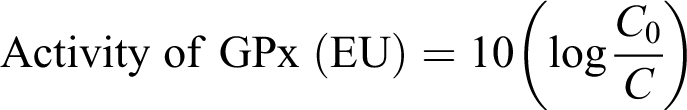Abstract
Gatifloxacin, a fluoroquinolone antibiotic, has been reported to produce several adverse reactions. In the present investigation, gatifloxacin administered at the dose rate of 10 and 20 mg kg−1 body weight per day, respectively, for 21 consecutive days, was evaluated for its potential to induce antioxidant status alterations and retinal damage in rabbits. A significant alteration in the antioxidant status of rabbits particularly in the high-dose group was observed which is indicated by decreased activity of superoxide dismutase and levels of blood glutathione with a concomitant increase in the activity of catalase, glutathione peroxidase, and glutathione S-transferase enzymes. The activity of glutathione reductase differed nonsignificantly between groups throughout the study period. The levels of malondialdehyde were elevated in the high-dose group. The histopathological examination of eyeball tunics revealed clumping of nuclei of the retinal outer nuclear layer in the gatifloxacin-treated groups. The results from this study indicate that repeated gatifloxacin administration produces a dose-dependent oxidative stress and retinopathy.
Introduction
Fluoroquinolones (FQs) are widely used antimicrobials with a broad spectrum of activity. 1 Gatifloxacin, 1-cyclopropyl-6-fluoro- 8-methoxy-7-(3-methylpiperazin-1-yl)-4-oxo-quinoline-3-carboxylic acid, a fourth-generation FQ, inhibits the bacterial enzymes DNA gyrase and topoisomerase IV. Although relatively safe, FQs have been reported to produce certain adverse reactions like gastrointestinal discomfort, hepatotoxic reactions, central nervous system effects, juvenile joint defects, and phototoxicty. 2 –4 Serious side effects have also been reported with gatifloxacin including hallucinations, liver damage, purpura, and dysglycemia. 5 –8 Several FQs have also been reported to produce retinopathy. 2,3,9 FQs produce reactive oxygen species (ROS) that supplements their antibacterial activity. 10 –12 The side effects of these drugs such as phototoxicity 13 and cartilage damage have been attributed to the production of ROS. 14 In this perspective, it is pertinent to evaluate the effect of dose and duration on gatifloxacin-induced oxidative imbalance and retinal degeneration that could give an insight of its toxicological profile.
Materials and methods
Animal experimentation
A total of 15 male Soviet Chinchilla rabbits aged 6 months were kept under normal ambient conditions, with an average day length of 11.5 h, in a well-lighted experimental house. The animals were maintained on feed, green fodder of the season, and water ad libitum. The use of animals was approved by the University Animal Ethics Committee under order No. PS/874-855 dated 14 May 2009. Rabbits were randomly allocated to three groups: control, group I, and group II with five animals in each group. Rabbits of groups I and II were orally administered gatifloxacin (TEQUIN®; gatifloxacin in 5% dextrose), at the dose rate of 10 and 20 mg kg−1 body weight day−1, respectively, for 21 consecutive days. 10 mg kg−1 dose is the human extrapolated therapeutic dose of gatifloxacin to rabbits, and 20 mg kg−1 dose was used as double of this dose to determine the dose dependency of the toxicity. Dextrose (5%), equaling the highest volume (4 ml) of drug used in the high-dose group, was administered to the rabbits of control group. The animals were restrained manually, and the blood samples were collected from the ear vein of these animals in heparinized vials on 0, 7, 14, and 21 days of treatment.
Blood GSH
The blood glutathione (GSH) was determined by the method described by Beutler. 15 To 0.2 ml of whole blood in 1.8 ml of distilled water, 3 ml of the precipitating solution (glacial metaphosphoric acid) was added, and the mixture was allowed to stand for approximately 5 min and then centrifuged at 3000 r min−1 for 15 min. To 2 ml of the supernatant, 8 ml of 0.3 M phosphate solution (di-sodium hydrogen phosphate dihydrate; Na2HPO4·2H2O) and 1 ml of 5,5′-dithiobis-(2-nitrobenzoic acid) (DTNB) reagent were added. The absorbance was recorded at 412 nm and the concentration of GSH was determined by the standard curve of GSH in distilled water.
Superoxide dismutase
The activity of superoxide dismutase (SOD) was determined by the method followed by Marklund and Marklund. 16 To a cuvette, 1.5 ml of 100 mM Tris–hydrochloric acid (HCl) buffer, 0.5 ml of 6 mM ethylenediaminetetraacetic acid (EDTA), and 1 ml of 0.6 mM pyrogallol solution were added. The rate of autoxidation of pyrogallol was taken from the increase in absorbance at 420 nm for 4 min after every 30 s lag. This constitutes the reading for blank. For the test, an appropriate amount of enzyme (in 20 μl of erythrocyte lysate) was added to inhibit the autoxidation of pyrogallol to about 50%. A unit of enzyme activity is defined as the amount of enzyme producing 50% inhibition of the autoxidation of pyrogallol observed in blank.
Catalase
The activity of catalase (CAT) was determined according to the method described by Aebi. 17 To 2 ml of phosphate buffer (50 mM, pH 7.0) in a cuvette, 20 μl of erythrocyte lysate was added and mixed well. The reaction was started by the addition of 1 ml of 30 mM hydrogen peroxide (H2O2) and the decrease in absorbance was recorded at every 10 s interval for 1 min at 240 nm. The results were expressed as micromoles of H2O2 decomposed per minute per gram protein using 36 as molar extinction coefficient of H2O2.
Glutathione peroxidase
The activity of glutathione peroxidase (GPx) was assayed by the method described by Hafeman et al.
18
To 0.1 ml erythrocyte lysate, 1 ml of 20 mM GSH, 1 ml of sodium phosphate buffer (0.4 M, pH 7), and 0.5 ml of sodium azide were added, and the volume was made to 4 ml by adding distilled water. After 5 min of preincubation, 1 ml of H2O2 (prewarmed to 37°C) was added. Then, 1 ml aliquots of incubation mixture were removed after 1 min interval and added to 4 ml precipitation solution (m-phosphoric acid). GSH in protein-free filtrate was determined by mixing 2 ml of filtrate with 2 ml of 0.4 M Na2HPO4 and 1 ml of DTNB reagent and the optical density (OD) was recorded at 412 nm within 2 min after mixing. The initial GSH concentration was determined in the same manner using an aliquot from a sample treated similarly except H2O2 is replaced with water.
Where C 0 is the concentration of GSH at zero time and C is the concentration of GSH after 1 min incubation.
The activity of GPx is expressed in enzyme units per milligram hemoglobin (Hb).
Glutathione S-transferase
The activity of glutathione S-transferase (GST) was determined by the method followed by Habig et al. 19 To 2.8 ml phosphate buffer, 0.1 ml GSH solution (30 mM) and 20 μl of erythrocyte lysate were added and mixed in a cuvette. The reaction was started by addition of 0.1 ml of 30 mM 1-chloro-2, 4-dinitrobenzene (CDNB) in 95% ethyl alcohol. The increase in OD at 340 nm in a spectrophotometer was recorded for 3 min, after a lag of 30 s. The extinction coefficient of CDNB conjugate at 340 nm is 9.6 mM−1 cm−1. One unit of enzyme activity is defined as the amount of enzyme that catalyzes the formation of 1 μmol conjugate of GSH and CDNB per minute.
Glutathione reductase
The assay of glutathione reductase (GR) was performed according to the method described by Carlberg and Mannervik. 20 To 3 ml cuvette, 2.6 ml phosphate buffer (0.2 M, pH 7.0, containing 2 mM EDTA), 0.15 ml nicotinamide adenine dinucleotide phosphate (NADPH; 2 mM in 10 mM Tris–HCl, pH 7.0), and 0.15 ml glutathione disulfide (GSSG; 20 mM in water) was added. The reaction was initiated by the addition of 0.1 ml of erythrocyte lysate and the decrease in absorbance at 340 nm was recorded at 30 s interval.
One unit of GR activity is defined as the amount of enzyme that catalyzes the oxidation of 1 μmol of NADPH per minute using 6.22 × 103 as molar extinction coefficient of NADPH.
Lipid peroxidation
Lipid peroxidation (LPO) was assayed by the method described by Stocks and Dormandy. 21 To 2 ml of 10% packed erythrocytes in phosphate-buffered saline (PBS; 0.1 M, pH 7.4), 1 ml of 40 mM H2O2 and 0.1 ml sodium azide were added, and the reaction mixture was incubated at 37°C for 1 h. After incubation, the total volume was made to 4 ml with PBS followed by addition of 2 ml of 28% ice-chilled trichloroacetic acid and centrifugation at 3000 r min−1 for 15 min. To 4 ml of supernatant, 1 ml of 1% thiobarbituric acid (TBA) was added, and the tubes were kept in boiling water bath for 15 min. Finally, the OD was measured at 532 nm against a blank (no H2O2 was added) after cooling the contents of tubes to room temperature. The values were expressed in nanomoles of malondialdehyde (MDA) produced per gram Hb per hour using 1.56 × 105 M−1 cm−1 as the molar extinction coefficient of pure MDA.
Hemoglobin
Hb content was determined by the method described by Benjamin. 22
Statistical analysis
The data were subjected to statistical analysis with unpaired t test between the groups at 0, 7, 14, and 21 days of treatment. 23
Histopathology
After completion of the experiment, the rabbits were humanely killed by bleeding under ether anesthesia. The eyes were removed, fixed in 10% neutral-buffered formalin and processed for paraffin sectioning by acetone–benzene schedule. Paraffin sections of 5 μm thickness were obtained and stained with hematoxylin and eosin as per method followed by Luna. 24
Results
Clinical signs
The administration of gatifloxacin to rabbits did not produce any apparent clinical signs of toxicity like tremors, convulsions, or death. There was not any significant effect on feed intake or body weight.
Oxidative stress parameters
The indices indicative of antioxidant status were altered in the gatifloxacin-treated groups (Table 1). In the higher dose gatifloxacin group, the activity of SOD significantly (p < 0.05) decreased on day 7, whereas the activity of CAT increased, significantly (p < 0.05) on day 21 of treatment. The gatifloxacin treatment, at both doses, resulted in a significant (p < 0.01) decrease in blood GSH levels on days 14 and 21 of treatment. The activity of GPx increased significantly in both treatment groups on day 14 (p < 0.05) and day 21 (p < 0.01) of treatment. GST activity was significantly (p < 0.01) increased in the higher dose group from day 7 till the end of treatment. The activity of GR differed nonsignificantly between the three groups throughout the study period. There was an increase in the extent of erythrocyte LPO in the high-dose group on day 21 of treatment.
Effect of repeated oral administration of gatifloxacin on LPO, blood GSH, and related enzymes in rabbits.a

LPO: lipid peroxidation; SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase; GST: glutathione S-transferase; GR: glutathione reductase; EU: enzyme unit; Hb: hemoglobin; GSH: reduced glutathione; CDNB: 1-chloro 2,4-di-nitrobenzene; MDA: malondialdehyde; H2O2: hydrogen peroxide.
aValues are represented as mean ± SE.
b p < 0.05: significantly different as compared to the control values.
c p < 0.01: significantly different as compared to the control values.
Histopathology
The histopathological examination of the tunics of eyeball revealed changes in both treated groups. There was a clumping of nuclei of retinal outer nuclear layer in both the groups; however, these changes were more apparent in group II (Figures 1 to 3).

Normal histology of retina. Hematoxylin–eosin staining ×40.

Retina, group I; mild and occasional clumping of nuclei of outer nuclear layer (thick arrows). Hematoxylin–eosin staining ×40.

Retina, group II; marked clumping of nuclei of outer nuclear layer (thick arrows). Hematoxylin–eosin staining ×40.
Discussion
The study was aimed to evaluate the effect of dose and duration on gatifloxacin-induced oxidative imbalance and retinal degeneration in rabbits. Fluoroquinolones have been shown to induce free radical production
25
due to their low redox potential.
26
FQs modulate the oxidative metabolism of leukocytes and stimulate ROS production in the neutrophils
27
and bacteria.
12,28
The decreased SOD activity, GSH levels, and total antioxidant status and increased LPO have been reported with FQs in human patients.
29
SOD, which dismutates superoxide radical
The detoxification of H2O2 to H2O is primarily carried out by CAT at high concentrations of H2O2 or GPx at lower concentrations. 34 GPx also catalyzes the detoxification of a wide range of peroxides, including lipid and hydroperoxides, using GSH as reducing equivalent. 36 GST catalyzes the nucleophilic conjugation of reduced GSH via the sulfhydryl group to electrophilic centers of a wide variety of substrates. 37 The primary response of body to ROS is the expression of Nrf-2, a transcription factor that increases the expression of antioxidant response-mediating genes. 38 The induction of CAT, GPx, and GST is a protective response of body to detoxify the increased concentration of the ROS produced. The decline in GSH levels could be due to increased utilization by GPx or GST. 39 The depleted GSH levels have not been restored fully as there was not any significant increase in the activity of GR that is required for the synthesis of GSH and maintains a high level of GSH/GSSG ratio. 3,35
The increased LPO with FQs reported in several studies corroborates with our current study of gatifloxacin. 3,29,40 –42 LPO and hydroperoxide levels, as well as markers of protein oxidation, such as carbonyl proteins, are markers of oxidative damage by ROS. 43,44 However, due to nonspecificity of the MDA assay, 45 other markers of the oxidative stress, like the endogenous antioxidant GSH and various antioxidant enzymes, are also taken into consideration to determine the occurrence of oxidative stress. 35,46 The extent of LPO indirectly reflects the degree to which biomembrane lipids are attacked by free radicals. When the generation of reactive free radicals overwhelms the antioxidant defense, the resultant LPO of the cell membrane causes disturbances in the cell integrity leading to cellular damage or cell death. 29 The increased LPO inactivates enzymes by MDA cross-linking that leads to an increased accumulation of ROS which further exacerbates LPO and other macromolecular damage. 47
Enrofloxacin administration in cats resulted in diffused retinal degeneration primarily affecting outer nuclear layer and photoreceptors along with hypertrophy and proliferation of retinal pigment epithelium. 2 Ofloxacin-induced dose-dependent retinal degeneration in rabbits is characterized by clumping of pigmented epithelial cells in the form of beads with pigment migrating in to the photoreceptor layer resulting in to the disruption of this layer. 3 Compared with the findings of present investigation, severities of retinal changes were more pronounced in cats and rabbits following administration of enrofloxacin and ofloxacin, respectively. 2,3 The difference in the extent of damage by different FQs varies with the substitution at position 8 of basic quinolone nucleus. FQs with methoxy substitution at position 8 have been reported to be least phototoxic. 48 –50 The lesser extent of retinal damage with gatifloxacin could be due to the presence of methoxy group at position 8 of basic quinolone nucleus, whereas both enrofloxacin and ofloxacin have H substitution at position 8. 50
The retinal degeneration associated with quinolone phototoxicity has been reported to be triggered by a cascade of events that include disruption of mitochondria, consequent release of cytochrome c, and apoptosis-inducing factor and the subsequent caspase 3-mediated apoptotic cell death of photoreceptors. 51 Singlet oxygen and/or superoxide anion have been directly detected in FQ solutions under ultraviolet A (UV-A) irradiation, whereas the continuous administration of antioxidants completely prevents the occurrence of retinal degeneration induced by FQ phototoxiciy. 52 Retinal degeneration-associated simultaneous FQ administration and UV-A irradiation has also been linked to the generation of ROS 51 that can be measured directly in the tissues, like vitreous or retina. However, in this study such measurements have not been carried out since it could alter the histopathological examination of the retina due to possible damage to the retina during harvesting of the vitreous or retina for determining the levels of ROS or antioxidants. It was also hypothesized in this study that whether oxidative stress might play a role in the retinopathy induced by gatifloxacin. It can be speculated from the dose-dependent oxidative stress and retinopathy produced by gatifloxacin that oxidative stress plays some role in the gatifloxacin-induced retinopathy.
There are certain limitations in this study. The number of animals in each group should have been more for a better statistical analysis. Since determination of MDA by reaction with TBA is a nonspecific assay, MDA should have been determined by high-performance liquid chromatography assay which is more specific and precise. 44 The ratio of GSH/GSSG and free radicals-like nitric oxide level are also important indicators of oxidative stress which also needs to be determined. 53 To determine the extent of oxidative stress in the retina itself, such parameters should also be determined in the eyes to determine directly the probable role of oxidative stress in the retinopathy.
The results of the present study conclude that the prolonged administration of gatifloxacin produces a dose-dependent oxidative imbalance in blood. Dose-dependent retinal degeneration characterized by histopathological changes in the outer nuclear layer is also produced. The dose-dependent oxidative stress in blood and the retinal histological alterations indicate that the gatifloxacin-induced retinopathy may be mediated by oxidative stress. However, more studies need be carried out in respect to the limitations of this study. In particular, the studies relating to the molecular mechanisms of retinopathy would give insight of the other probable reasons of retinopathy to understand and manage this toxicological manifestation associated with FQs.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
The current research has been carried out at the Department of Pharmacology and Toxicology, Guru Angad Dev Veterinary and Animal Sciences University (GADVASU), Ludhiana, Punjab, India. The study is a part of the research project, “Evaluation of retinopathic potential of fluoroquinolones” assigned by University Grants Commission, India, to the Department of Veterinary Pharmacology and Toxicology, GADVASU, Ludhiana, Punjab, India, under letter no. FNo. 40-279/2011(SR). Financial assistance to the project has been made by University Grants Commission, India.