Abstract
There is a large body of evidence that nitric oxide (NO) formation is implicated in mediating silica-induced pulmonary fibrosis. As a reactive free radical, NO may not only contribute to lung parenchymal tissue injury but also has the ability to combine with superoxide and form a highly reactive toxic species peroxynitrite that can induce extensive cellular toxicity in the lung tissues. This study aimed to explore the effect of agmatine, a known NO synthase inhibitor, on silica-induced pulmonary fibrosis in rats. Male Sprague Dawley rats were treated with agmatine for 60 days following a single intranasal instillation of silica suspension (50 mg in 0.1 ml saline/rat). The results revealed that agmatine attenuated silica-induced lung inflammation as it decreased the lung wet/dry weight ratio, protein concentration, and the accumulation of the inflammatory cells in the bronchoalveolar lavage fluid. Agmatine showed antifibrotic activity as it decreased total hydroxyproline content of the lung and reduced silica-mediated lung inflammation and fibrosis in lung histopathological specimen. In addition, agmatine significantly increased superoxide dismutase (p < 0.001) and reduced glutathione (p < 0.05) activities with significant decrease in the lung malondialdehyde (p < 0.001) content as compared to the silica group. Agmatine also reduced silica-induced overproduction of pulmonary nitrite/nitrate as well as tumor necrosis factor α. Collectively, these results demonstrate the protective effects of agmatine against the silica-induced lung fibrosis that may be attributed to its ability to counteract the NO production, lipid peroxidation, and regulate cytokine effects.
Introduction
Pulmonary silicosis is a deadly respiratory disease that kills thousands of people every year worldwide. It results from inhalation of silica-containing dusts, which is very common to individuals employed in the mining and construction industries. 1 It is mainly characterized by the loss of alveolar structure through the apoptosis of epithelial and endothelial cells, proliferation of fibroblasts, and excessive deposition of extracellular matrix. 2 Several mechanistic pathways can participate in the development of silica-induced pulmonary diseases that include direct cytotoxicity of silica particles, stimulation of oxidant generation by exposed pulmonary phagocytes, activation of production of chemokines and inflammatory cytokines, and production of fibrogenic factors by lung cells. 3 Other studies have suggested that nitric oxide (NO) may also participate in silica-induced pulmonary inflammation, damage, and fibrosis. 4 The messenger RNA levels for inducible NO synthase (iNOS), an inducible enzyme that produces NO, are increased in bronchoalveolar lavage (BAL) cells after intratracheal instillation of silica in rats. 5 Furthermore, NO production by alveolar macrophages is increased after intratracheal instillation 6 or inhalation of silica. 7 It has also been demonstrated that inflammatory mediators as γ-interferon and tumor necrosis factor α (TNF-α), which are produced in silica-induced response can induce synthesis of NO. 8 Agents that interfere with the production of NO may provide protection against silica-induced fibrosis.
Agmatine, (4-(aminobutyl) guanidine) an amine dication at physiological pH, is formed by decarboxylation of
Materials and methods
Drugs and chemicals
Crystalline silica was generously provided by U.S. Silica Company (Berkeley Springs, Morgantown, West Virginia, USA). Agmatine sulfate was purchased from Sigma-Aldrich (St Louis, MO, USA) and was dissolved in normal saline. All other chemicals and biochemicals used in this study are of high analytical grade.
Experimental animals
Male Sprague Dawley rats (weighing 150–200 g) were purchased from Egyptian Organization for Biological Products and Vaccines, Giza, Egypt. The animals were housed (5 per cage) in an air-conditioned room maintained at 25 ± 2°C with a regular 12-h light/12-h dark cycle. All procedures involving the animals were conducted in accordance with the protocol approved by the committee on animal experimentation of the Faculty of Pharmacy, Mansoura University, Egypt.
Induction of silicosis and experimental design
Silica preparation and injection
The silica dust was suspended in saline (50 mg in 0.1 ml saline/rat), then 20,000 IU penicillium (Sandoz, Egypt) were added to the silica suspension. The suspension was shaken vigorously by a vortex shaker, prior to administration. The animals were lightly anesthetized with ether and were then given silica by intranasal instillation, 21 while normal animals received an equal volume of sterilized saline instead of silica suspension.
Animal groups
Rats were divided into four groups (n = 8): (1) control (saline); (2) agmatine (saline + agmatine); (3) silica; and (4) silica + agmatine. Agmatine was administered intraperitoneally (10 mg/kg, body weight) once daily, while animals of the control and silica groups received saline intraperitoneally as vehicle once daily during the entire experimental period of 60 days. The dose of agmatine was selected based on previous investigation which reported that this dosage regimen can produce marked inhibition of iNOS in other models of inflammatory disorders. 22 At the end of the treatment period, the rats were anesthetized by inhalation of diethyl ether, and the chest was opened; the trachea with the heart–lung package was excised from the thorax and then the left main bronchi were clamped. A cannula was inserted into the trachea in situ, and the right lung was lavaged with sterile 0.9% saline to collect the BAL fluid (BALF) for subsequent analysis.
After BALF collection, the right lung tissues were rapidly removed, snap frozen in liquid nitrogen, and stored at −80°C until subsequent analysis. Before the experiment, the lung tissue samples of lung were weighed and homogenized (100 mg/ml) in 0.1 M phosphate buffer (pH 7.4) in an ice bath using a variable speed homogenizer (OMNI International, Kennesaw, Georgia, USA). The homogenate was centrifuged at 3000g for 20 min at 4°C, using cooling centrifuge (Damon /IEC Division, CRU-5000 model, Needham, Massachusetts, USA). Biochemical assays were performed on the supernatant of the lung homogenate.
Measurement of lung W/D weight ratio
The lung wet/dry W/D ratio was calculated to evaluate tissue edema. The wet left upper lung was excised, blotted dry, and weighed to calculate the “wet” weight and then placed in an oven at 80°C for 24 h to calculate the “dry” weight.
Bronchoalveolar lavage fluid
BALF fractions were centrifuged (4000 r/min for 10 min at 4°C) using cooling centrifuge to collect the cell pellet for the total cell count determination. The supernatants of the BALF were stored at −80°C until required for determination of protein content.
Measurement of cell counts and total protein activity in BALF
Total and differential cell counts
The cell pellets obtained after centrifugation of the BALF were resuspended in 100 μl of saline, centrifuged onto slides, and stained for 8 min with Wright–Giemsa staining. The total cell count was carried out using a hemocytometer. Differential cell count was carried out through quantification of the slides for neutrophils and lymphocytes following standard morphological criteria by counting a total of 200 cells/slide at 40× magnification.
Protein content
The total protein concentration was measured using a commercial kit (Thermo Scientific, Rockford, Illinois, USA) in the supernatant of BALF.
Histopathological examination of lung
Part of the left lung was harvested and rinsed with phosphate-buffered saline, then fixed in 10% neutral-buffered formalin for 24 h, embedded in paraffin wax, sectioned (6 μm) and stained with hematoxylin–eosin and Masson’s trichrome. The trichrome stain was used to identify the increase in collagenous tissue or indicate fibrotic change in lung. The tissues were examined under a microscope in a random order and without the knowledge of animal or group. The structural alterations of tissue were assessed based on the degree of cellular proliferation, alveolar wall thickening, inflammatory lesions, and collagen deposition or fibrosis. Such changes were graded in terms of severity and distribution. The grading system adopted is as follows and was utilized for each group of animals. 23
For severity of lesion: 0 = nothing/zero, 1 = marginal, 2 = slight, 3 = moderate, 4 = severe, and 5 = very severe. For distribution of lesion over the tissue: 0 = absent, 1 = rare/occasional (10% of the lung area), 2 = sparse/limited (10–25% of the lung area), 3 = moderate (25–50% of the lung area), 4 = extensive/widespread (50–75% of the lung area), and 5 = very extensive/predominant (over 75% of the lung area).
Hydroxyproline assay
To assess the degree of collagen deposition, hydroxyproline content of the lung was determined by the colorimetric method as described previously 24 using base rather than acid hydrolysis for the dissolution of tissue. 25 Briefly, the lung tissue was incubated with 5% potassium hydroxide solution overnight at 37°C, then hydrolyzed with 10 N sodium hydroxide, then incubated with chloramine-T solution for 3 h at room temperature, then Ehrlich’s aldehyde reagent was added, the sample was incubated at 60°C in a water bath for 20 min, and finally absorbance was measured at 550 nm using a color wave colorimeter (model CO7500, WPA, Cambridge, UK).
Measurement of oxidative stress in lung
Estimation of MDA
The malondialdehyde (MDA) concentration was determined as an indicator of lipid peroxidation in the lung tissue. The MDA content in the supernatant of the lung homogenate was measured according to the method described by Satoh 26 using a commercial kit purchased from Bio-Diagnostic Company (Giza, Egypt). MDA was expressed in nanomoles per gram tissue.
Estimation of SOD
The superoxide dismutase (SOD) activity was estimated according to the method described by Nishikimi et al. 27 using a commercial kit purchased from Bio-Diagnostic Company (Egypt). The SOD activity was expressed in units per milligram protein.
Estimation of reduced GSH
The concentration of reduced glutathione (GSH) in the lung homogenate was measured according to the method described by Beutler et al. 28 using a commercial kit purchased from Bio-Diagnostic Company (Egypt). The GSH was expressed in micromoles per gram tissue.
Measurement of NO2 −/NO3 − concentration
Nitrite/nitrate (NO2 −/NO3 −) production, an indicator of NO synthesis, was measured in the supernatant of the lung homogenate using a commercially available NO assay kit (R&D Systems, Minneapolis, USA) following the manufacturer’s instruction.
Measurement of TNF-α level
The level of TNF-α in the supernatant of the lung homogenate was determined using commercial enzyme-linked immunosorbent assay kit, according to the manufacturer’s instructions (Bender Med Systems GmbH, Vienna, Austria). TNF-α was determined from a standard curve.
Statistical analysis
Statistical analysis was performed using one-way analysis of variance followed by Benferroni’s multiple comparison test. Fibrosis grade was analyzed by the nonparametric Kruskal–Wallis test followed by Dunn’s test. The values were presented as means ± SEM, where n equaled the number of rats. The value of p < 0.05 was considered significant.
Results
Effects of agmatine on the lung W/D ratio and total protein content in the BALF
As shown in Figure 1(a), the lung W/D ratio increased significantly in silica group as compared to the control group (p < 0.001). Agmatine pretreatment significantly decreased the lung W/D ratio (p < 0.001) as compared to silica group. As shown in Figure 1(b), silica produced a significant increase in the BALF protein content as compared to control rats (p < 0.001). The protein content in the silica + agmatine group was significantly lower than the silica group (p < 0.001).

Effects of agmatine on the lung W/D ratio and total protein content in the BALF. Silica (50 mg in 0.1 ml saline/rat, intranasally) was given once to rats. Agmatine (10 mg/kg, intraperitoneally, once daily) was administered for 60 days. Values represent the mean ± SEM of eight rats/group. ***p < 0.001: significantly different compared with the control group (one-way ANOVA followed by Benferroni’s multiple comparison test). ###p < 0.001: significantly different compared with the silica group (one-way ANOVA followed by Benferroni’s multiple comparison test). W/D: wet/dry; BALF: bronchoalveolar lavage fluid; ANOVA: analysis of variance.
Effects of agmatine on the total and differential cell counts in the BALF
Rats of silica group showed a significant increase (p < 0.001) in the total and differential cell counts in the BALF as compared to the control group (Figure 2(a) to (c)). Agmatine treatment led to a significant lowering of total cells, lymphocytes, and neutrophils counts (p < 0.001) as compared to silica group.

Effect of agmatine on the total and differential cell counts in the BALF. Silica (50 mg in 0.1 ml saline/rat, intranasally) was given once to rats. Agmatine (10 mg/kg, intraperitoneally, once daily) was administered for 60 days. Values represent the mean ± SEM of eight rats/group. ***p < 0.001: significantly different compared with the control group (one-way ANOVA followed by Benferroni’s multiple comparison test). ###p < 0.001: significantly different compared with the silica group (one-way ANOVA followed by Benferroni’s multiple comparison test). BALF: bronchoalveolar lavage fluid; ANOVA: analysis of variance.
Effects of agmatine on lung histology of rats with silica-induced fibrosis
As shown in Figure 3, rats of the control and agmatine groups showed normal lung architecture, while rats of the silica group showed a marked increase in the severity and the distribution of the lesions as compared to the control group (Table 1). The lung specimen of rats of the silica group showed severe inflammatory reaction and fibrosis with evident granuloma formation mainly around the bronchial tree and vascular beds (Figure 3(a)). Perivascular and peribronchilal fibrosis associated with silicotic nodules and distributed hyalinized collagen fibers were observed in the nodules (Figure 3(b)). The spread of the lesions is extensive and widespread between 50 and 75%.

Effects of agmatine on lung histopathological examination in silica-induced fibrotic rats. Silica (50 mg in 0.1 ml saline/rat, intranasally) was given once to rats. Agmatine (10 mg/kg, intraperitoneally, once daily) was administered for 60 days. Control: no signs of neither inflammation nor fibrosis; silica: extensive nodular and peribronchial reaction and fibrosis; silica + agmatine: reduced peribronchial reaction and fibrosis. (a) Hematoxylin–eosin staining (×100) and (b) Masson’s trichrome staining (×100). Black arrows indicate inflammatory cells infiltration and green arrows indicate collagenous tissues.
Effects of agmatine on lung histopathology.a

aSilica (50 mg/kg in 0.1 ml saline/rat, intranasally) was given once to rats. Agmatine (10 mg/kg, intraperitoneally, once daily) was administered for 60 days. One section was taken from each animal. The numbers indicate the total number of animals that fall under particular score in different groups. The average severity and distribution grades are represented as the mean ± SEM of eight rats/group.
b p < 0.001: significantly different compared with the control group (Kruskal–Wallis Test).
c p < 0.01: significantly different compared with the control group (Kruskal–Wallis Test).
d p < 0.001: significantly different compared with the silica group (Kruskal–Wallis Test).
Treatment with agmatine resulted in a marked improvement in the lesion distribution grade as compared to the silica group. The lung specimen of agmatine group (Figure 3) showed minimal inflammatory reaction with peritracheal infiltration and fibrosis, and the distribution of the reaction is moderate of about 25–50% spread.
Effects of agmatine on hydroxyproline content in the lung
Silica instillation caused a significant increase (p < 0.01) in the lung hydroxyproline content as compared to the control group. However, agmatine treatment significantly decreased (p < 0.05) the hydroxyproline content of lung as compared to that of silica group (Figure 4).
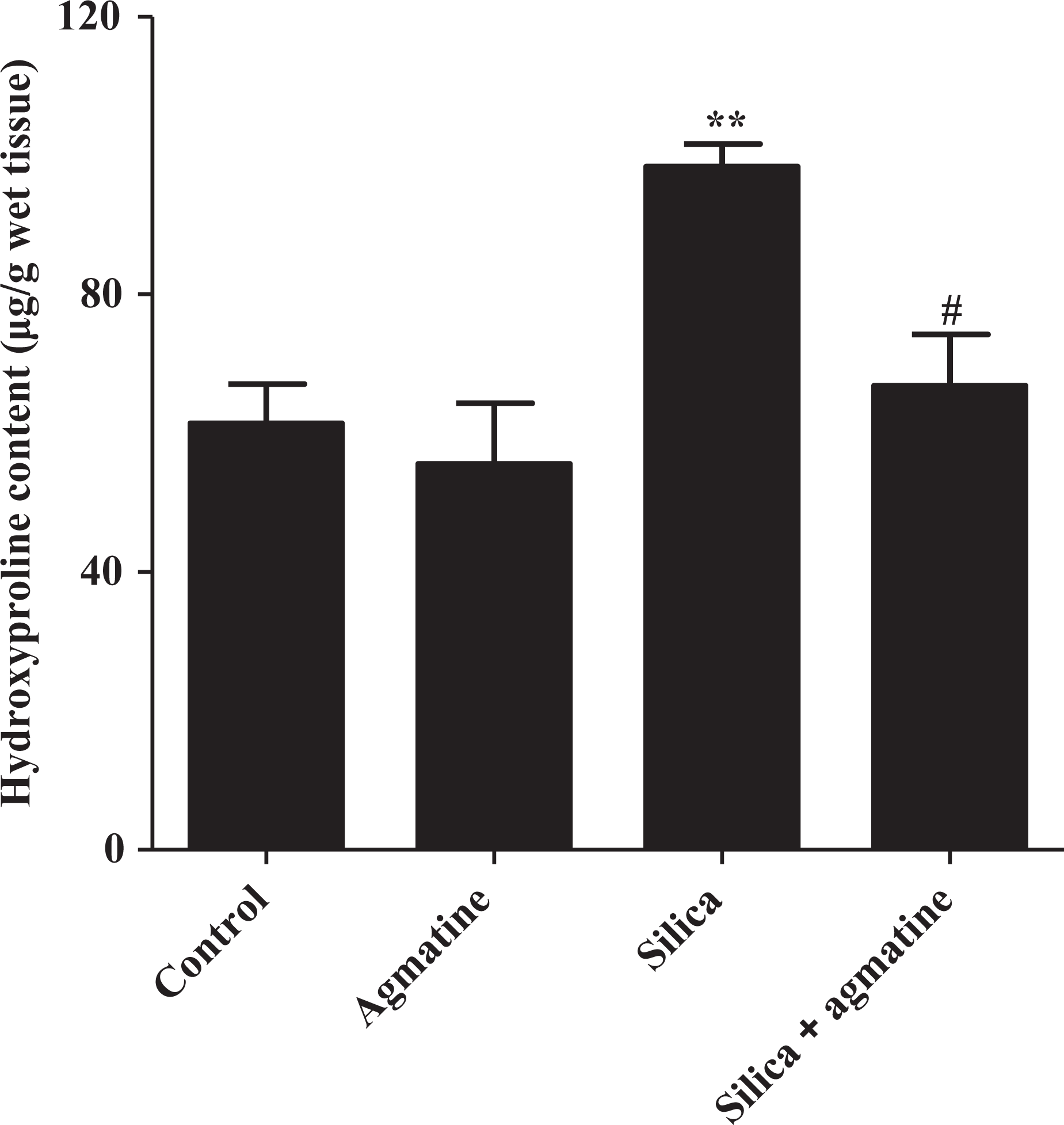
Effect of agmatine on hydroxyproline content in lung tissue. Silica (50 mg in 0.1 ml saline/rat, intranasally) was given once to rats. Agmatine (10 mg/kg, intraperitoneally, once daily) was administered for 60 days. Values represent the mean ± SEM of eight rats/group. **p < 0.01: significantly different compared with the control group (one-way ANOVA followed by Benferroni’s multiple comparison test). #p < 0.05: significantly different compared with the silica group (one-way ANOVA followed by Benferroni’s multiple comparison test). ANOVA: analysis of variance.
Effects of agmatine on oxidative stress in the lung
Silica instillation resulted in a significant increase in MDA level in lungs, while GSH and SOD activities were markedly decreased (p < 0.001) as compared to the control group. These changes induced by silica were all ameliorated significantly by agmatine treatment (Table 2).
Effect of agmatine on oxidative stress in the lung.a

MDA: malondialdehyde; SOD: superoxide dismutase; GSH: glutathione; ANOVA: analysis of variance.
aSilica (50 mg in 0.1 ml saline/rat, intranasally) was given once to rats. Agmatine (10 mg/kg, intraperitoneally, once daily) was administered for 60 days. The MDA, SOD, and GSH levels were measured in the supernatant of the lung homogenate. Values represent the mean ± SEM of eight rats/group.
b p < 0.001: significantly different compared with the control group (one-way ANOVA followed by Benferroni’s multiple comparison test).
c p < 0.01: significantly different compared with the silica group (one-way ANOVA followed by Benferroni’s multiple comparison test).
d p < 0.001: significantly different compared with the silica group (one-way ANOVA followed by Benferroni’s multiple comparison test).
e p < 0.05: significantly different compared with the silica group (one-way ANOVA followed by Benferroni’s multiple comparison test).
Effects of agmatine on NO2 −/NO3 − concentration in the lung
As shown in Figure 5, the silica group showed significantly higher production of NO2 −/NO3 − as compared to the control group (p < 0.001). However, agmatine significantly attenuated pulmonary NO production as compared to the silica group (p < 0.001).

Effect of agmatine on total NO2−/NO3− level in the lung. Silica (50 mg in 0.1 ml saline/rat, intranasally) was given once to rats. Agmatine (10 mg/kg, intraperitoneally, once daily) was administered for 60 days. The total NO2−/NO3− level was measured in the supernatant of the lung homogenate. Values represent the mean ± SEM of eight rats/group. ***p < 0.001: significantly different compared with the control group (one-way ANOVA followed by Benferroni’s multiple comparison test). ###p < 0.001: significantly different compared with the silica group (one-way ANOVA followed by Benferroni’s multiple comparison test). NO2−/NO3−: nitrite/nitrate; ANOVA: analysis of variance.
Effects of agmatine on TNF-α content in the lung
Silica administration caused a significant increase (p < 0.001) in TNF-α level as compared to the control group. Agmatine treatment resulted in a significant decrease (p < 0.001) in TNF-α level as compared to the silica group (Figure 6).
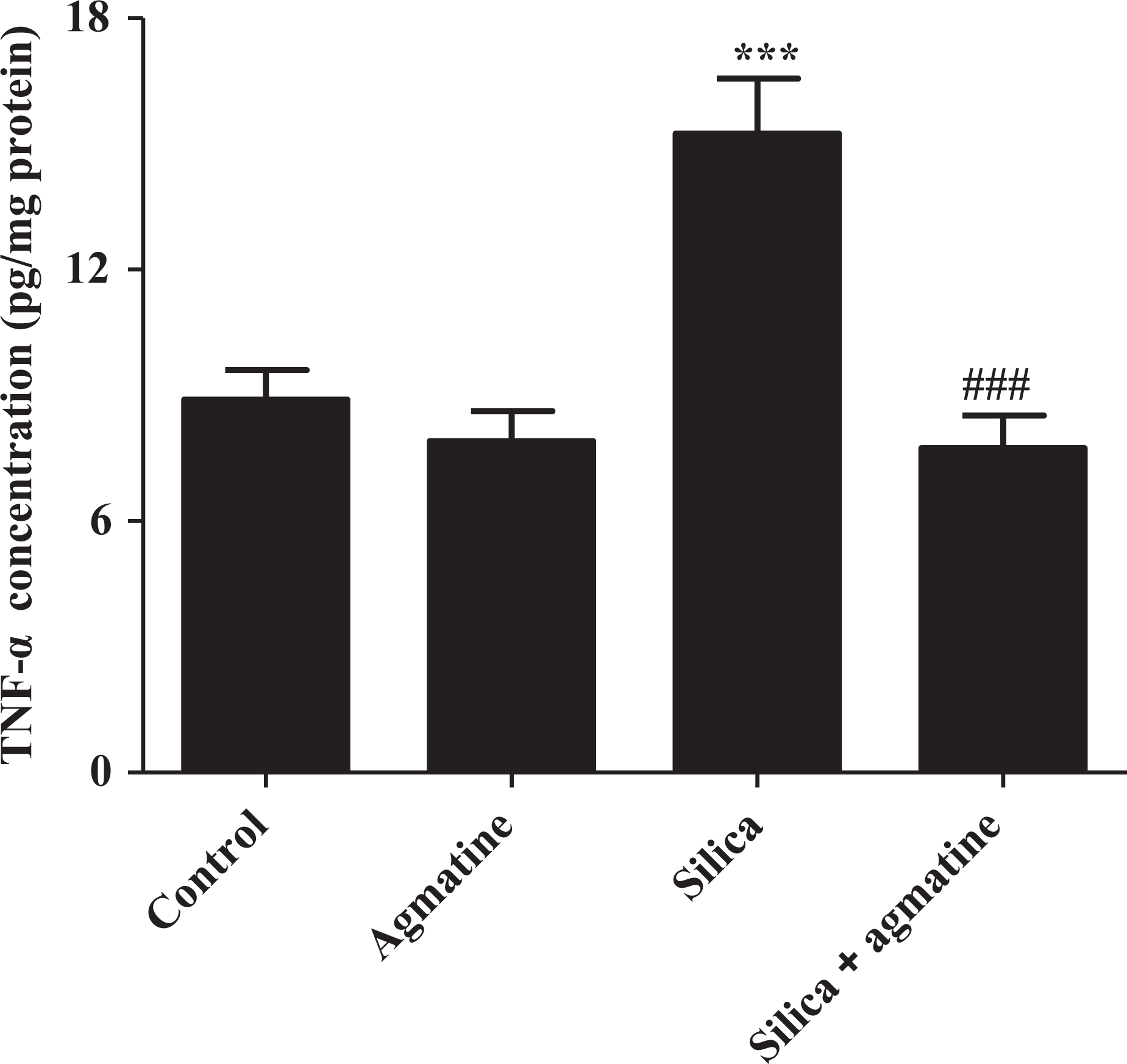
Effect of agmatine on TNF-α level in the lung. Silica (50 mg in 0.1 ml saline/rat, intranasally) was given once to rats. Agmatine (10 mg/kg, intraperitoneally, once daily) was administered for 60 days. The TNF-α concentration was measured in the supernatant of the lung homogenate. Values represent the mean ± SEM of eight rats/group. *** p < 0.001: significantly different compared with the control group (one-way ANOVA followed by Benferroni’s multiple comparison test). ###p < 0.001: significantly different compared with the silica group (one-way ANOVA followed by Benferroni’s multiple comparison test). TNF-α: tumor necrosis factor α; ANOVA: analysis of variance.
Discussion
Silicosis is a progressive pulmonary fibrotic disease that is related to the development of lung cancer, which has no successful treatment available to date. Although silicosis has been studied intensively, the crucial cellular mechanism that initiates and drives the processes of fibrogenesis is not fully understood. 29 Many recent reports have shown the pivotal role of NO in silicosis and that iNOS-derived NO contributes to the pathogenesis of silica-induced lung disease. 5,30 The present study aimed to examine the effect of agmatine, an NO modulator, on silica-induced inflammation and fibrosis. The results presented demonstrate that agmatine has potent anti-inflammatory and antifibrotic effects against silica-induced fibrosis in rats.
Initially, both W/D ratio of lung weight and total protein content were measured to estimate the integrity of the lung air/blood barrier and the extent of lung injury. Silica increased the lung W/D ratio and total protein content in BALF indicating pulmonary edema and leakage of serous fluids into lung tissue. Agmatine treatment significantly reduced silica-induced pulmonary edema indicating that agmatine could prevent the leakage of serous fluid into lung tissue. Total and differential cell counts measured in BALF were used to estimate the amplitude of pulmonary inflammation induced by silica particles in rats. A massive infiltration of inflammatory cells including lymphocytes and neutrophils in the lungs was observed in silica group. All these changes were attenuated by agmatine treatment suggesting the anti-inflammatory effect of agmatine. This was supported by histological analysis of the lungs that revealed marked attenuation of silica-induced inflammation by agmatine treatment.
As previously reported, the time course of rat pulmonary responses to inhaled silica is biphasic. During the first 41 days of silica exposure, elevated and relatively constant levels of inflammation and damage, with no fibrosis has been observed. While from 41 to 116 days of exposure, rapidly increasing pulmonary inflammation and damage with concomitant development of fibrosis has occurred. 3 The inflammatory phase is followed by reparative phase in which tissue repair is associated with extracellular matrix deposition mainly collagen I, collagen II, and fibronectin leading to interstitial fibrosis and silicotic nodules formation. 31,32 This is consistent with the results presented that revealed elevated hydroxyproline content in lung homogenates and marked increase in the severity and distribution of fibrosis in lung specimen of rats of silica group. These effects were ameliorated by agmatine treatment revealing the protective effects of agmatine against the development of silica-induced fibrosis. Previous studies have shown the ability of agmatine to attenuate the matrix metalloproteinase -2 and matrix metalloproteinase-9 expressions that are associated with basement membrane and extracellular matrix remodeling. Agmatine effects were mediated by the increase of eNOS after oxygen–glucose deprivation and reperfusion injury in the cerebral endothelial cells. 33,34
The next step was carried out to explore the possible mechanism(s) of the protective action of agmatine against silica-induced fibrosis, oxidative stress, and inflammatory factors were evaluated in the lung tissue. As already demonstrated, the lung response to silica in rats was accompanied by an overproduction of reactive oxygen species (ROS) and reactive nitrogen species. During the inflammatory response, neutrophils undergo a respiratory burst and produce superoxide. Overproduction of ROS is highly toxic to host tissues and their interactions with various cellular macromolecules lead severe pathophysiological consequences. 2,29 Results showed that silica instillation increased lipid peroxidation in the lungs, as it is evident from the significant increase in MDA content which is commonly known as a marker of oxidative stress and antioxidant status. 35 This was accompanied by a significant decrease in SOD and GSH activities. Agmatine treatment significantly reduced the level of MDA and increased the SOD and GSH levels. This is consistent with the previous reports that have shown the ability of agmatine to scavenge free radicals and resultant decrease in oxygen derivative, such as hydrogen peroxide. 36,37 The mechanism of the antioxidant effects of agmatine is still unknown; however, it has been attributed previously to its effect in suppressing NF-κB activation in hyperglycemic mesengial cells 38 or through α-2 adrenergic signaling in retinal ganglion cells. 39 Therefore, the present study showed that agmatine may effectively reduce oxidative burden during the inflammatory response to silica particles.
Previous reports have shown that free radicals are not the only cause of silica-induced disease as crystalline silica regardless of source, purity, and age is capable of inducing various lung pathological reactions. 1 So it was essential to evaluate the effect of agmatine on silica-induced production of inflammatory cytokines that induce, enlarge, and facilitate the entire or focal inflammatory reaction. It has been reported that alveolar macrophages and neutrophils as well as other cell types can cause an overproduction of NO after silica exposure. 4 Although NO seems to be beneficial by induction of apoptosis in inflammatory cells, the NO produced in a silica-induced lung reaction may be harmful. That is because NO can combine with superoxide to form peroxynitrite free radical and this substance can cause severe lung damage. 30,40 The prolonged and induced production of NO in combination with other oxidant gives rise to a range of toxic NO metabolites that potentially induce direct cytotoxicity or initiate unique signaling pathways. Furthermore, NO upregulates inflammatory cytokines, such as TNF-α, production and amplifies the inflammatory response during inflammation. 41 Agmatine is a potent competitive inhibitor of iNOS. 42 Previous studies reported that agmatine significantly suppressed LPS-induced NO production in a concentration-dependent manner in vitro. 43 Besides, agmatine inhibits the expression of iNOS by suppressing the activation of NF-κB. 44 It was reported that agmatine can reduce phosphorylation of NF-κB in cultured retinal neuronal cells. 45 Reduction of NO generation may therefore be the key target for hepatoprotection by agmatine.
Previous reports have shown that macrophages release TNF-α that has an important role in the pathogenesis of silicosis. 46 Elevated TNF-α level correlates with increased collagen deposition. 47 Results have shown that agmatine significantly decreased TNF-α level. This is consistent with the previous study of Hong et al. 45 , which showed that agmatine effectively suppressed TNF-α production of rat retinal ganglion cells in hypoxic condition. This may suggest the ability of agmatine to attenuate TNF-α production.
In conclusion, the present study shows that agmatine can protect against silica-induced lung fibrosis. Agmatine reduces pulmonary edema, inflammatory cells accumulation as well as the histpathological damage. The protective effect of agmatine may be related to its ability to decrease lipid peroxidation, enhance antioxidant status, and suppress NO as well as TNF-α production. Therefore, this study provides an evidence that agmatine may serve as therapeutic strategy in case of lung fibrosis.
Footnotes
Acknowledgments
The authors acknowledge Dr Asem A Shalaby, Department of Pathology, Faculty of Medicine, University of Mansoura, Mansoura, Egypt, for histopathological evaluation.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
