Abstract
The efficacy of methotrexate (MTX), a widely used chemotherapeutic drug, is limited by its gastrointestinal toxicity and the mechanism of which is not clear. The present study investigates the possible role of mitochondrial damage in MTX-induced enteritis. Small intestinal injury was induced in Wistar rats by the administration of 7 mg kg−1 body wt. MTX intraperitoneally for 3 consecutive days. MTX administration resulted in severe small intestinal injury and extensive damage to enterocyte mitochondria. Respiratory control ratio, the single most useful and reliable test of mitochondrial function, and 3-(4,5-dimethylthiazol-2-yll)-2,5-diphenyltetrazolium bromide reduction, a measure of cell viability were significantly reduced in all the fractions of MTX-treated rat enterocytes. A massive decrease (nearly 70%) in the activities of complexes II and IV was also observed. The results of the present study suggest that MTX-induced damage to enterocyte mitochondria may play a critical role in enteritis. MTX-induced alteration in mitochondrial structure may cause its dysfunction and decreases the activities of the electron chain complexes. MTX-induced mitochondrial damage can result in reduced adenosine triphosphate synthesis, thereby interfering with nutrient absorption and enterocyte renewal. This derangement may contribute to malabsorption of nutrients, diarrhea, and weight loss seen in patients on MTX chemotherapy.
Introduction
Methotrexate (MTX), a structural analogue of folic acid, is widely used as a chemotherapeutic agent for malignancies as well as rheumatic disorders. 1 MTX is recently proven to be effective in the treatment of refractory inflammatory bowel disease. 2 However, the efficacy of MTX is often limited by severe side effects and toxic sequelae. Hematopoietic, oral, and gastrointestinal (GI) toxicities are the most frequently observed side effects that may lead to a reduction in the dose or even discontinuation of MTX. 3 Chemotherapy commonly produces structural and functional damage to the intestinal mucosa in cancer patients and in patients with rheumatoid arthritis. 4,5
The small intestinal damage induced by MTX treatment results in malabsorption and diarrhea. 6 This malabsorption results in weight loss and disturbs the chemotherapy in cancer patients. MTX administration deteriorates barrier function of the mucosa, which leads to invasion of gut flora into the circulation. These sometimes progress to colitis or ulcer. Therefore, intestinal mucositis remains a major concern during cancer chemotherapy in >40% of cancer patients after low-dose treatment and in almost 100% of patients treated with high doses. 7,8 Minimizing the GI toxicity of MTX can improve its therapeutic efficacy.
Several attempts have been made to investigate the mechanism of GI toxicity of MTX, but the mechanism is still not clear. Since the mechanism of these side effects has not been completely clarified, cancer chemotherapy has to be accompanied by symptomatic therapy such as antibiotics and antidiarrheal drugs. It is important to clarify the mechanism of the MTX-induced intestinal damage in order to perform the cancer chemotherapy effectively by preventing the side effects (Figure 1). The present study is an attempt to unravel the mechanism of MTX GI toxicity.
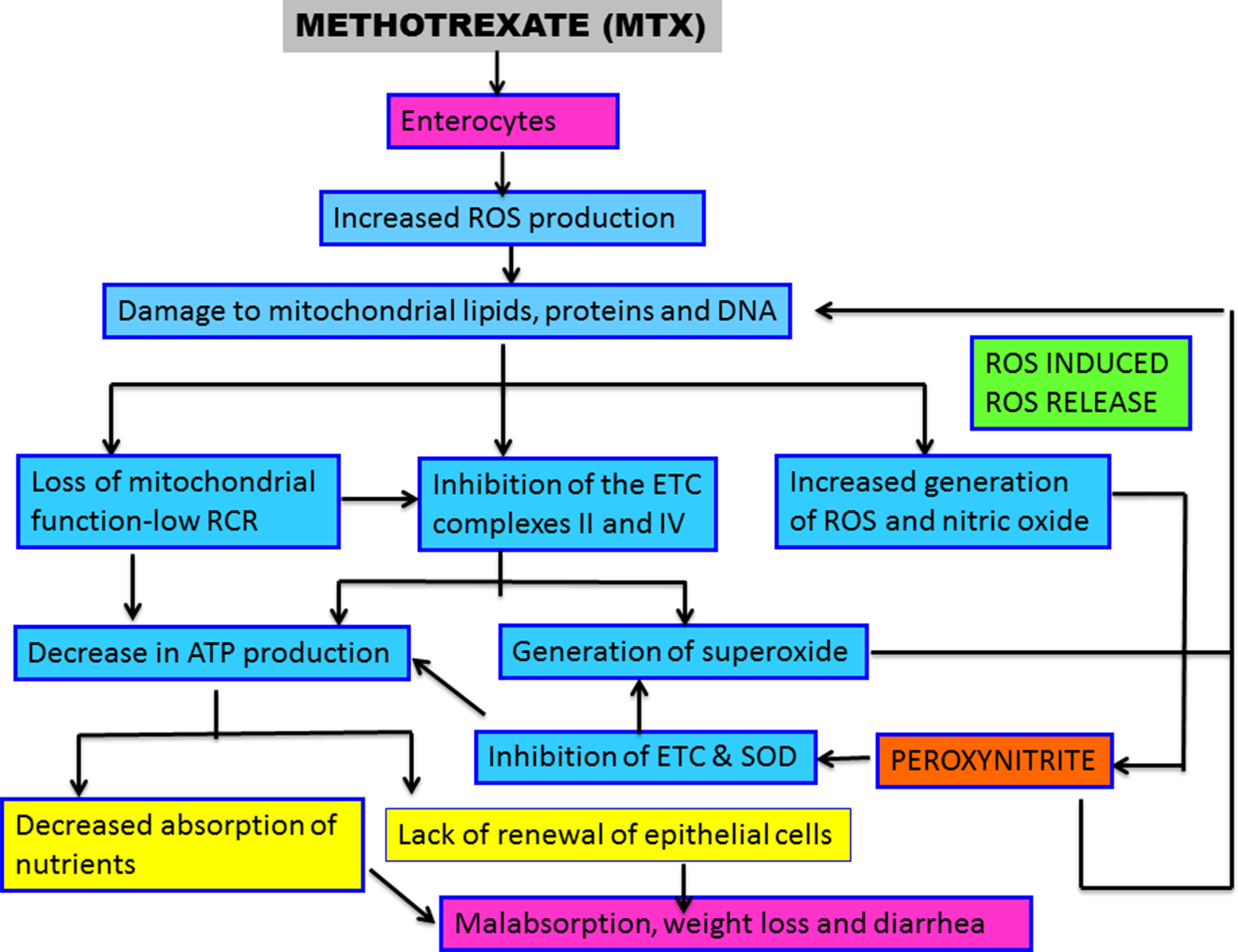
Potential molecular mechanisms of the role of mitochondrial damage in MTX-induced small intestinal injury. MTX causes increased ROS production and NO production and hence oxidative stress and nitrosative stress in the enterocytes. These reactive species damage the mitochondrial lipids, proteins, and DNA. Mitochondrial damage results in structural alteration (swelling and disruption of cristae), loss of function, decreased activities of complexes II and IV as well as overproduction of superoxide anions at these sites. Superoxide anion reacts with nitric oxide to form PON, a potent oxidising agent. PON nitrates the, ETC complexes II and IV and SOD, resulting in their loss of activity. Inhibition of the, ETC complexes decreases ATP synthesis as well increases the generation of superoxide anions, which accumulate due to decreased activity of SOD. This superoxide anion can combine with mitochondrial nitric oxide to form PON which further damages the mitochondria (ROS induced ROS release). Decrease in ATP synthesis in the enterocytes can decrease the rates of nutrient absorption leading to malabsorption, weight loss and diarrhea. MTX: methotrexate; ROS: reactive oxygen species; NO: nitric oxide; PON: peroxynitrite; ETC: electron transport chain; SOD: superoxide dismutase; ATP: adenosine triphosphate.
Studies including the present study have demonstrated that MTX-induced intestinal injury is both reactive oxygen species (ROS), and reactive nitrogen species (RNS) dependent but the source of the reactive species is not clearly known. 9 – 14 Oxidative stress and nitrosative stress are inseparably linked to mitochondrial dysfunction, as mitochondria are both main generators and targets of both ROS and RNS. 15 The mitochondrial, electron transport chain (ETC) has been recognized as one of the major site of ROS production, with superoxide anion (O2−) being the first ROS produced and therefore a major source of oxidative stress. 16 Additionally, nitric oxide (NO) is produced within the mitochondria by mitochondrial nitric oxide synthase. 17 It reacts with O2− to produce another radical, peroxynitrite (ONOO−). 18 Together, these radicals can do great damage to mitochondria and other cellular components.
Mitochondria, in addition to generating ROS and RNS, are themselves susceptible to oxidant damage, which can decrease respiratory enzyme activities and mitochondrial membrane potential and lead to greater ROS production of ROS-induced ROS release. 19 Within the mitochondria, elements that are particularly vulnerable to free radicals include lipids, proteins, oxidative phosphorylation enzymes, and mitochondrial DNA (mtDNA). 20 NO and its metabolite ONOO– have been implicated as key mediators of mitochondrial dysfunction. 21 Persistent generation of significant levels of ONOO− can lead to the induction of cell death, either apoptosis or necrosis. 22
We hypothesize that mitochondrial damage may play a role in MTX-induced small intestinal damage. The hypothesis is proposed based on the findings that
ROS and RNS play important role in MTX-induced enteritis. 9 – 14
NO and ONOO− contribute to intestinal inflammation and gut barrier failure. 23
Mitochondria are main generators as well as targets of ROS and RNS. 15
The main mitochondrial targets of nitro-oxidative stress are ETC complexes I, II, IV, and V, aconitase, superoxide dismutase, mitochondrial membranes, and mtDNA. 20 and
Enterocytes have a high requirement for adenosine triphosphate (ATP) for their continuous renewal and active transport of nutrients.
Because mitochondria are responsible for generating cellular energy (ATP) that fuels nutrient digestion, absorption, and cell renewal, inefficiencies in intestinal mitochondrial function caused by MTX may reduce the efficiency of nutrient absorption resulting in malabsorption, diarrhea, and weight loss.
Materials and methods
Materials
Adenosine diphosphate (ADP), ethylene glycol tetra acetic acid (EGTA), MTX, ubiquinone–22-theonyl trifluoroacetone (TTFA), 4–(2–hydroxyethyl) piperazine (2–ethanesulfonic acid) (HEPES), 3-(4,5-dimethylthiazol-2-yll)-2,5-diphenyltetrazolium bromide (MTT), sodium succinate, dichlorophenol-indophenol (DCPIP), bovine serum albumin, pyruvate kinase, lactate dehydrogenase, phosphoenol pyruvate, nicotinamide adenine dinucleotide (NADH), ATP, and oligomycin, were obtained from Sigma-Aldrich, India. All other chemicals used were of highest analytical grade available and were purchased from Qualigens Fine Chemicals, Mumbai, India.
Animals
Adult male Wistar rats (200–250 g) were used for the studies. They were housed in standard rat cages (421 × 290 × 190 mm). All animals were exposed to 12-h light–dark cycles and allowed access ad libitum to water and rat chow. The experiments done were approved by the institutional animal ethics committee and were in accordance with the guidelines of the Committee for the Purpose of Control and Supervision of Experimentation on Animals (CPCSEA), Government of India.
Animal treatment
The rat model of MTX-induced enteritis which is described in the literature causing consistent intestinal injury in normal rats was used for the study. 24
MTX was administered to the rats at a concentration of 7 mg kg−1 body weight (corresponding to intermediate MTX dose in humans) intraperitoneally (i.p.) for 3 consecutive days. The rats were sacrificed 12 and 24 h after the final dose of MTX. Vehicle-treated rats served as control.
For electron microscopic studies high-dose MTX was used, that is 25 mg kg−1 body wt. MTX was given i.p. and the rats were euthanized after 6, 12, 24, and 48 h.
Tissue procurement
The rats were anesthetized with halothane and sacrificed by cervical dislocation. The entire length of the small intestine was removed, washed with ice-cold phosphate-buffered saline (PBS) and used for analysis.
Light microscopy
The sections were fixed in 10% buffered formalin then embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E) for examination of mucosal injury by light microscopy. Mucosal injury, inflammation, and hyperemia/hemorrhage was assessed and graded in a blinded manner using the histological injury scale previously defined by Chiu et al. 25
Electron microscopy
For ultrastructural study, a portion of the tissue were fixed in 2.5% glutaraldehyde, post-fixed in osmium tetroxide, and embedded in araldite (epoxy resin). Ultrathin sections were cut from selected blocks with a Leica ultracut UCT (Wien, Austria) and a diamond knife (Diatome, Switzerland). Sections were mounted on copper grids and stained with uranyl acetate and lead citrate, and examined with a Philips EM 201C electron microscope (Eindhoven, Netherlands).
Mitochondrial injury gradation
Mitochondrial injury scoring system based upon the stages of cellular injury was used to grade the injury Trump et al. 26 Mitochondrial ultrastructure was evaluated by electron microscopy at three different locations within each duodenum, jejunum, and ileal samples. Specifically, mitochondrial ultrastructure was analyzed within the villus tips, villus crypts, and the circumferential muscle layer. These sites were selected because of their relative susceptibilities to injury (e.g., the villus tips are more prone to various forms of injury than other mucosal structures) and their unique morphological and functional characteristics (i.e., mucosal tissue has different characteristics than does smooth muscle). Examinations were made in a blinded fashion by two reviewers, and the severity of ultrastructural injury was quantified by determining a composite score (based on the scale of 0–5 as shown in Table 1) that represented all of the mitochondria visualized within the microscopy field. It was derived from a scoring system based on the progressive stages of cellular injury, as described by Trump and colleagues. 26 This staging system enumerates the characteristic progression of ultrastructural changes that occurs within cells in various models of inflammatory injury (e.g., ischemia–reperfusion). The association between the degree of mitochondrial injury and the defined stages of cellular injury (Table 1) was employed to quantify and standardize the severity of ultrastructural injury to the small intestinal mitochondria.
Mitochondrial injury scoring system based upon the stages of cellular injury. 26

ER: endoplasmic reticulum. Scores greater than 3.0 are predictive of ultimate cell death.
Isolation of different enterocyte populations (villus, middle, and crypt cells)
The whole length of the small intestine was washed gently with cold saline containing 1 mM dithiothreitol (DTT). Enterocytes of various stages of maturation (villus to crypt cells) was isolated by the metal chelation method as described. 27 Briefly, the intestine was filled with solution A [1.5 mM potassium chloride, 96 mM sodium chloride, 27 mM sodium citrate, 8 mM monopotassium phosphate, and 5.6 mM disodium phosphate pH 7.3], clamped at both ends and incubated at 37°C for 15 min. Following incubation, the luminal contents of the intestine were discarded and the intestine was filled with solution B (25 mM PBS pH 7.2 containing 1.5 mM ethylenediaminetetraacetic acid (EDTA), and 0.5 mM DTT), and incubated at 37°C for different time intervals of 4, 2, 2, 3, 4, 6, 7, 10, and 15 min. At the end of each time period, the incubated solution containing cells was collected in separate tubes. These fractions were numbered 1–9, and these 9 fractions were pooled as villus (1–3 fractions), middle (4–6 fractions), and the crypt (7–9 fractions) cells. 27 The cell fractions were then centrifuged at 900g for 5 min, suspended in PBS, homogenized, and the homogenate was used for experiments. Separated villus and crypt cells were identified by the enrichment of the marker enzyme, alkaline phosphatase in the villus cells.
Isolation of enterocyte mitochondria
Enterocyte cell fractions prepared as described above were homogenized using homogenizing buffer consisting of 250 mM sucrose/5 mM HEPES/1 mM EGTA; pH 7.4. 28 The homogenates were centrifuged at 1000g for 10 min and the nuclear pellet was discarded. Crude mitochondrial fraction was obtained by centrifuging at 12000g for 20 min and the pellet was washed thrice with wash buffer containing 250 mM sucrose/5 mM HEPES; pH 7.4. Purity of the mitochondria was established by enrichment of marker enzyme, succinate dehydrogenase. The isolated mitochondria were used for assessing their function and also for measuring the activities of mitochondrial complexes.
Measurement of parameters of mitochondrial function
Measurement of oxygen uptake
Oxygen uptake was determined polarographically using a Clark type electrode. 29 Oxygen uptake was stimulated with 0.3 mM ADP and the rate of state 3 and state 4 respirations were measured. Oxygen uptake was calculated from the decrease in percent saturation of the mixture. The ratio of state 3/state 4 respiratory rates was calculated for the respiratory control ratio (RCR).
MTT reduction assay
The MTT assay was done on the mitochondria using a microplate reader as described. 30 The amount of MTT formazan formed was calculated using the molar extinction coefficient of MTT formazan E570 of 17,000 M−1cm−1 at pH 7.4–8.0.
Mitochondrial swelling
Swelling of mitochondria was determined by following the decrease in absorbance at 540 nm for 10 min as described. 30
Assay of the activities of the electron transport chain complexes
The activities of the, ETC complexes: complex I (NADH–ubiquinone oxidoreductase), complex II (succinate–ubiquinone oxidoreductase), complex III (ubiquinol cytochrome c reductase), complex IV (cytochrome c oxidase), and complex V (ATP synthase) were assayed spectrophotometrically. The enzyme activity is expressed as k per minute per milligram of protein. 31,32
Assay of manganese superoxide dismutase
Superoxide dismutase was measured as described by Ohkuma et al. 33 The assay mixture consisted of 100 µl of phosphate buffer, 10 µl of BSA, 50 µl of TritonX-100, 5 µl of EDTA, 5 µl of xanthine oxidase, 50 µl of xanthine. To this finally 150 µl MTT and sample (50–150 µg protein) were added and the volume is made up to 1 ml with water. The mixture was incubated for 5 min at room temperature (30°C) and the reaction was terminated with the addition of 1 ml of stop buffer. This was read at a wavelength of 540 nm. Amount of superoxide formed is calculated using the molar extinction coefficient of MTT formazan E540 of 17,000 M−1cm−1 at pH 7.4–10.5. The percentage of inhibition by the presence of SOD is calculated from the reduction of the MTT color formation as compared to the MTT formazan formed in the absence of SOD, which is taken as 100%. One unit of SOD is defined as the amount of protein required to inhibit MTT reduction by 50%.
Statistical analysis
The data obtained were analyzed by analysis of variance (ANOVA), using a Bonferroni correction for multiple t-test as a post hoc test. Value of p < 0.05 was taken to indicate statistical significance. Data analysis was carried out using Statistical Package for the Social Sciences, version 11 (SPSS Inc., Armonk, New York, USA).
Results
MTX administration results in significant body weight loss and intestinal weights of rats
MTX-treated rats had significant weight loss (6–10%) compared with control. The weights of the intestine were significantly reduced (by 38%) in the MTX-treated rats compared with control (1.25 ± 0.15 vs. 2.00 ± 0.30 g 100 g−1 body wt., p < 0.02). The MTX-treated rats had diarrhea.
MTX induces moderate to severe mucosal injury
The results of the light microscopic examination of the small intestines is shown in Figure 2.The administration of MTX resulted in moderate to severe mucosal injury (grade 3 to 5) in accordance with the criteria suggested by Chui et al. 30,34 On gradation 12 h after the final dose of MTX, the damage was in duodenum and jejunum (Grade 3) > ileum (Grade 2). Exfoliation of the tips of the villi and slight destruction of the crypts of Lieberkuhn was observed in the duodenum 12 h after the final dose of MTX (Figure 2(a)). In the jejunum, the villi were destroyed and the surface epithelium was degenerated (Figure 2(b)). In the ileum, the villi appeared normal, 12 h after the final dose of MTX. However, dilated capillaries were seen (Figure 2(c)).

Light microscopic changes in mucosa of MTX-treated rats (H&E, ×100). (a) Histology of the duodenum 12 h following the final dose of MTX. There is slight exfoliation of the tip of the villus and slight destruction of the crypts of Lieberkuhn. (b) Histology of the jejunum 12 h after the final dose of MTX indicates the degeneration of surface epithelium. (c) Histology of the ileum12 h after the final dose of MTX. The villi appear normal, capillaries were dilated. (d) Histology of the duodenum 24 h after the final dose of MTX. Arrow indicates moderate destruction of the villus. Asterisks indicate the crypts of Lieberkuhn which are reduced in number. (e) Histology of the jejunum 24 h after the final dose of MTX. There is more destruction of the villus. Arrow indicates destruction of the surface epithelium and villus fusion. (f) Histology of the ileum 24 h after the final dose of MTX. There is destruction of the villus (Arrow) and reduction of the crypts of Lieberkuhn (Asterisks). (g), (h), and (i) Histology of the duodenum, jejunum and ileum of control rats showing normal architecture. MTX: methotrexate; H&E: hematoxylin and eosin.
The damage to the small intestine was more severe 24 h after the final dose of MTX compared with 12 h. The damage was more in the ileum (Grade 5), followed by jejunum and duodenum (Grade 4). In the duodenum and jejunum, there was extensive destruction of the villi. The villi were atrophied and focally absent. The crypts of Lieberkuhn were very few in number as compared with 12 h group (Figure 2(d) and (e)). In ileum, the villi were atrophied, blunted, and fused. There was destruction of the crypts of Lieberkuhn (Figure 2(f)). The villus/crypt ratio was decreased and there was transmural acute inflammatory infiltrate in the mucosa, submucosa, and muscularis layers. The overall thickness of the epithelium was decreased accompanied by increased intraepithelial lymphocytes, neutrophil infiltration, and crypt abscess.
The duodenum, jejunum, and ileum of the control group rats showed normal architecture (Figure 2(g)–(i)). The mucosa was lined by villi which are short and leaf-like in duodenum, tall in jejunum, and of intermediate height in ileum. The villus epithelium consisted of an admixture of tall columnar epithelial cells and goblet cells, which are more numerous in ileum. Crypts of Lieberkuhn formed the lower 20% of the epithelium and were surrounded by per crypt fibroblast sheath. Lamina propria was composed of loose connective tissue matrix containing lymphocytes, and plasma cells.
High-dose MTX causes severe mitochondrial injury (25 mg kg−1 body wt .)
The control epithelium contained variable quantities of mitochondria, in the form of granules, short rods, and filaments (Figure 3). The concentration of mitochondria close to the brush border and the basement membrane is indicative of their importance in production of the energy needed for active transport across the plasma membrane. The mitochondria of enterocytes in the control rats appeared normal with dense matrix and regular cristae. Endoplasmic reticulum and dense bodies were seen in well-defined cellular territories (Figure 3 (a)).

Ultrastructural changes in the jejunal mucosa after HDMTX treatment. (a) The control epithelium contained variable quantities of mitochondria in the form of short rods and filaments. (b) 6 h after MTX treatment, the mitochondria were swollen (white arrow), (c) cristae were disrupted at 12 h (white arrow), (d) there was extensive disruption of cristae at 24 h (white arrow), (e) complete dissolution of cristae at 48 h (white arrow), and (f) dilatation of endoplasmic reticulum and golgi apparatus was seen as a degenerative vacuolar appearance at 24 h after MTX treatment. HDMTX: high-dose methotrexate; MTX; methotrexate.
MTX-induced ultrastructural changes in the enterocyte include swollen mitochondria, extensive disruption of cristae, altered mitochondrial shape, dilatation of endoplasmic reticulum and Golgi apparatus cytoplasmic vacuolation, and degeneration and fragmentation of microvilli.
Mitochondrial damage was evident as early as 6 h after MTX treatment, and became more severe with time. The mitochondria were swollen (Figure 3(b)) 6 h after MTX treatment, and cristae were disrupted at 12 h (Figure 3(c)). There was extensive disruption of cristae at 24 h (Figure 3(d)) and complete dissolution of cristae was seen at 48 h (Figure 3(e)). Dilatation of endoplasmic reticulum and golgi apparatus was seen as a degenerative vacuolar appearance at 24 h after MTX treatment (Figure 3(f)). When mitochondrial injury was graded as described by Trump (Table 1), the mitochondrial injury score in the enterocytes of MTX-treated rats was >4, indicating severe mitochondrial injury that can result in enterocyte apoptosis/necrosis. Scores greater than 3.0 are predictive of ultimate cell death as irreversible cell damage is noted to occur during this stage of mitochondrial injury. 26
MTX causes enterocyte mitochondrial dysfunction
RCR indicates the tightness of the coupling between respiration and phosphorylation. It was significantly reduced in the villus, middle, and crypt cells of MTX-treated rats suggesting mitochondrial dysfunction (Figure 4(a)).
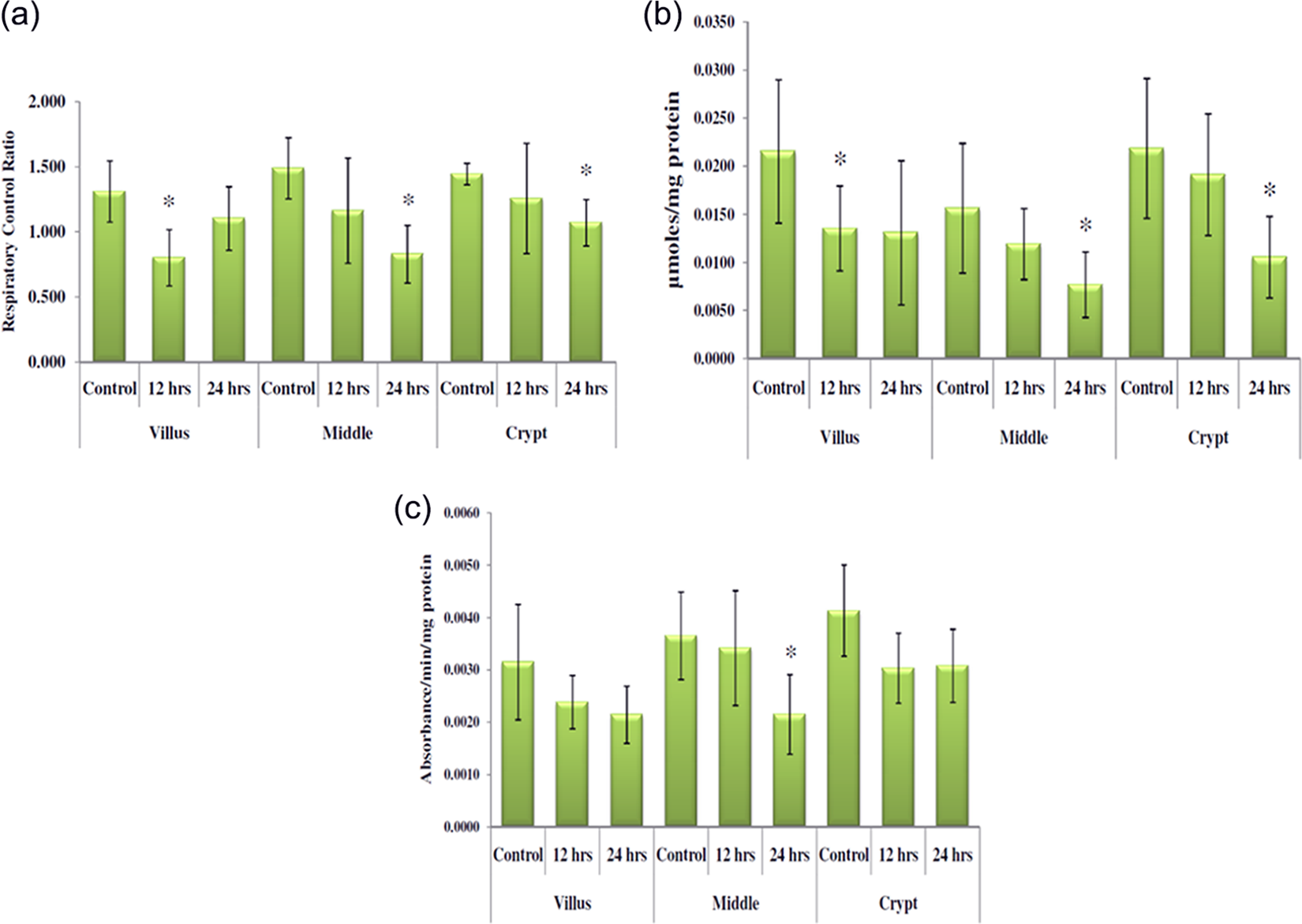
Tests of mitochondrial functions. (a) RCR: Values represent ± S.D., n = 6 rats, *p ≤ 0.01 as compared with the corresponding control. (b) MTT reduction: Values represent ± S.D., n = 6 rats, *p≤ 0.05 as compared with the corresponding control. (c) Mitochondrial swelling: Values represent ± S.D., n = 6 rats, *p ≤ 0.05 as compared with the corresponding control. RCR: respiratory control ratio; MTT: 3-(4,5-dimethylthiazol-2-yll)-2,5-diphenyltetrazolium bromide.
MTT reduction assay is a measure of mitochondrial activity as well as cell viability. MTT reduction was significantly decreased in all the fractions of enterocytes after MTX treatment (Figure 4(b)). This suggests decreased mitochondrial function and enterocyte cell viability in MTX-treated rats.
Mitochondrial swelling is considered as an indicator of altered membrane permeability transition (MPT). MPT is a physiopathological event that results in increased permeability of the inner mitochondrial membrane to solutes with a molecular weight less than 1.5 kDa resulting in mitochondrial swelling. Mitochondrial swelling was observed in all the fractions of enterocytes but was statistically significant only in the middle cells (Figure 4(c)).
MTX causes drastic decrease (more than 70%) in the activities of the ETC complexes II and IV, but has no effect on complexes I and III
A drastic decrease in the activity of complex II was observed in all the 3 fractions of MTX-treated enterocytes (Figure 5(a)). In villus cells, complex II activity was decreased by 81%, in middle cells by 73%, and in crypt cells by 68%. Similarly, a drastic decrease in complex IV activity was observed in all the fractions of enterocytes of MTX-treated rats (Figure 5(b)). In the villus and middle cells it was decreased by 68% and in the crypt cells by 74%. The activities of complexes I and III were not significantly altered after MTX treatment. (Figure 5(c) and (d)). The activity of complex V was increased slightly in villus and middle cells of MTX-treated rats as compared to controls (Figure 5(e)).

Activities of electron chain complexes in the mitochondria of enterocytes isolated from the three fractions in MTX-treated and control rat mucosa. Values represent ± S.D., n = 6 rats. (a) Complex I activity in the three fractions of enterocytes isolated from MTX-treated rats and control rats. (b) Complex II activity in the three fractions of enterocytes isolated from MTX-treated and control rats. *p ≤ 0.001 as compared with control. (c) Complex III activity in the three fractions of enterocytes isolated from MTX-treated and control rats. (d) Complex IV activity in the three fractions of enterocytes isolated from MTX-treated rats and control rats. *p ≤ 0.01 as compared with control. (e) Complex V activity in the three fractions of enterocytes isolated from MTX-treated and control rats. *p ≤ 0.05 as compared with control. MTX; methotrexate.
MTX causes a drastic decrease in SOD activity
MTX treatment resulted in a 45% decrease in the activity of SOD in the small intestines of rats as compared with control (Figure 6).

SOD activity in the MTX-treated and control mucosa. Values represent ± S.D., n = 6 rats, *p ≤ 0.01. MTX; methotrexate; SOD: superoxide dismutase.
Discussion
MTX continues to be a commonly used chemotherapeutic agent despite its GI toxicity because of its superior efficacy and broad-spectrum action. The small intestinal injury induced by MTX treatment results in malabsorption and diarrhea. 6 This malabsorption results in weight loss and diarrhea and disturbs the chemotherapy in cancer patients. During chemotherapy, the balance between epithelial cell proliferation, differentiation, and cell death at the villus tips disrupted by premature death of the dividing epithelial cells, thereby decreasing the surface area for absorption of nutrients.
MTX administration deteriorates barrier function of the mucosa which leads to invasion of gut flora into the circulation.As a result of mucosal barrier injury, a common side effect of MTX chemotherapy is severe enterocolitis. The inflamed intestines demonstrate the presence of oxidant stress. Preclinical studies including our study suggest that increased oxidative/nitrosative stress associated with an impaired antioxidant defense status plays a critical role in MTX-induced small intestinal damage. 9 – 14 Although several studies have shown that ROS and RNS play important roles in MTX enteritis, the source of the reactive species and the consequence of overproduction of these reactive species are not known.
Oxidative stress is inseparably linked to mitochondrial dysfunction, as mitochondria are both main generators as well as targets of ROS and RNS. 16 Once a mitochondrion is damaged, mitochondrial function can be further compromised by increasing the cellular requirements for energy repair processes. Besides, the damaged mitochondria generate ROS which further damages the mitochondria. 19 We hypothesized that mitochondrial damage may play an important role in MTX-induced enteritis and hence contribute to malabsorption, weight loss, and diarrhea seen in patients and animals treated with MTX. Therefore, the present study was designed to study the effects of MTX on enterocyte mitochondrial structure and function.
Firstly we studied the effect of a single dose of 25 mg kg−1 body weight MTX (high-dose MTX) on the ultrastructural changes in the rat enterocytes using TEM. Mitochondrial damage was evident as early as 6 h and the damage became more severe with time. MTX caused severe mitochondrial injury and cellular injury when graded according to the criteria of Trump and colleagues. 26 The findings were comparable with those reported on jejunal biopsy of children with acute lymphoblastic leukemia on MTX treatment. 35
MTX-induced damage to mitochondria can have two consequences. (a) Mitochondrial dysfunction and (b) increased generation of ROS. Mitochondria are key organelles for cellular function and survival and are the major sites of ATP production, so known as powerhouse of the cell. 36 The small intestines are the primary site for nutrient absorption and transport. The concentration of mitochondria close to the brush border and the basement membrane is indicative of their importance in production of the energy needed for active transport across the plasma membrane. Cellular ATP generated in mitochondria via oxidative phosphorylation fuels nutrient absorption and transport in the small intestine. A considerable amount of energy is used by the gut for the maintenance of this tissue because intestinal epithelium is continuously renewed. Mitochondrial damage can thus decrease the availability of ATP for nutrient absorption as well as for enterocyte renewal thereby contributing to malabsorption, and diarrhea. Therefore we assessed mitochondrial function by the measurement of RCR, MTT reduction, and mitochondrial permeability.
Gross changes in mitochondrial shape, distribution, or appearance disrupted cristae might indicate altered function. All of these markers may/may not indicate mitochondrial dysfunction, and so the cell RCR should be measured. 37
RCR measurement is considered the single most useful and reliable test of mitochondrial function in cell populations. 38 A high RCR implies that the mitochondria have a high capacity for substrate oxidation and ATP turnover and a low proton leak. RCR was significantly reduced in the villus, middle, and crypt cells of MTX-treated rats suggesting mitochondrial dysfunction. Decreased RCR indicates uncoupling of oxidative phosphorylation and is diagnostic of extensive mitochondrial damage. 39
MTT reduction assay is a measure of mitochondrial activity as well as cell viability. 40 MTT reduction assay requires intact mitochondrial membrane. Mitochondrial damage will inevitably result in loss of the ability to provide energy for metabolic function and so the extent of reduction of MTT will be decreased. MTT reduction was significantly decreased in all the fractions of enterocytes after MTX treatment suggesting decreased mitochondrial function and enterocyte cell viability upon MTX treatment.
Mitochondrial swelling is considered as an indicator of altered MPT. 34 MPT is a physiopathological event that results in increased permeability of the inner mitochondrial membrane to solutes resulting in mitochondrial swelling. Mitochondrial swelling was observed in enterocytes suggesting increased mitochondrial permeability. All these findings suggest MTX-induced mitochondrial dysfunction.
Mitochondrial damage in addition to causing its dysfunction can result in increased generation of oxygen free radicals, mainly O2−. A defect in the activity of the respiratory complexes mainly complex I and complex III is considered potential sites of superoxide production although specific sites of ROS production are controversial. 40 Therefore, we next assayed the activities of the mitochondrial, ETC. We found a marked decrease (approximately 70%) in the activities of complexes II and IV in MTX-treated rat enterocytes, in contrast to unaltered activities of complexes I and III that are established sources of ROS. 40 Recent studies have shown that reduction in the activities of complexes II and IV add to the total amount of O2− produced by mitochondria. 41,42 The present study therefore suggests that the defect in complex II and IV may be one of the sources of O2− MTX-treated rat intestines. It is apt to mention here that mitochondria are not the only source of ROS in MTX-induced mucositis. Other sources of ROS namely NADPH oxidases, xanthine oxidase, myeloperoxidase, uncoupled NOS have also been reported. 43
Marked decrease in the activities of complexes II and IV have been reported to decrease the rates of mitochondrial respiration and ATP production. 44 Therefore, decreased activities of complexes II and IV observed in the MTX-treated enterocytes may cause reduced ATP production leading to impaired nutrient absorption and enterocyte renewal as both the processes have an absolute requirement for ATP. Lack of adequate ATP can thus result in malabsorption, diarrhea, and weight loss. Further it may hinder the renewal process leaving bare areas in the intestinal epithelium, and gut barrier failure that facilitates bacterial translocation ultimately resulting in enteritis. It is possible that the reduced activities of complexes II and IV observed in the present study may be due to their inactivation by ROS and RNS, as these complexes have been shown to be vulnerable to inactivation by these reactive species. and paraoxonase (PON). 45,46 Our suggestion that decrease in the activities of complexes II and IV may be due to their inactivation by PON is supported by a recent finding that in dextran sodium sulfate (DSS)-induced colitis in mice reduction in activity of complex IV and complex II was inhibited by aminoguanidine, an inducible nitric oxide synthase (iNOS) inhibitor. 47
Having found out that MTX causes mitochondrial damage and inhibition of electron transport chain complexes (ETC) II and IV, we next investigated whether MTX mediated disruption of mitochondrial respiratory complexes II and IV affected ATP generation. The generation of ATP is through complex V in the mitochondria. To determine the levels of ATP, we evaluated complex V ATP synthase activity in the mitochondria isolated from control and MTX-treated rats. A significant increase in the activity of complex V was observed in the MTX-treated rat intestine. This suggests a compensatory increase in ATP synthesis to meet cellular needs. In the MTX-treated rat enterocytes, despite the increased activity of complex V that synthesizes ATP, necrosis was observed. Although there is no clear explanation for this, we suggest that the ATP synthesized by the enterocytes is utilized by poly ADP ribose polymerase, the nuclear repair enzyme activity to repair the damaged epithelia, thereby decreasing the availability of ATP for other vital processes in the enterocyte such as nutrient absorption and renewal, resulting in necrosis.
SOD is the first line of defense against mitochondrial O2− and hence known as the guardian of the powerhouse. 48 SOD has several protective roles within the mitochondria. SOD is important in preventing ROS-induced inactivation of, ETC complexes by degrading the O2−. 49 It is also important for removing excess superoxide that may contribute to ONOO− formation. Therefore the activity of SOD was determined in the intestines. We found 45% decrease in SOD activity in the small intestines of MTX-treated rats compared with control. SOD is reported to be susceptible to inactivation by tyrosine nitration. 50 We have previously demonstrated that MTX-induced intestinal injury is associated with increased immunoreactivity to 3-nitrotyrosine, a molecular footprint of PON. 12 Decreased activity of SOD observed in the present study may be due to nitration of SOD by PON.
The small intestine is the primary site for nutrient absorption and transport. Cellular ATP generated in mitochondria via oxidative phosphorylation fuels nutrient absorption and transport in the small intestine. A considerable amount of energy is used by the gut for the maintenance of this tissue because intestinal epithelium is continuously renewed. New cells are produced in the crypts, migrate to villus tip, and are sloughed off in 3–5 days. It has been estimated that 11–18% of the whole energy expenditure in ruminants is used by the gut, and the majority of this energy is spent for Na+K+ ATPase activity (6–12%), and protein synthesis (4.0–4.6%). 51 Thus, the absorptive capacity of the small intestine is dependent on the availability of ATP to fuel Na+K+ ATPase and to renew epithelial cells. Inefficiencies in mitochondrial function caused by MTX may limit nutrient absorption resulting in weight loss and diarrhea. In agreement with other studies, our study showed significant loss of body weight of rats in response to MTX when compared with the control. The weights of the small intestine were significantly reduced (by 38%) in the MTX-treated rats as compared with controls. The MTX rats had diarrhea also.
Several studies have shown that absorptive function of the intestine injured by MTX is reduced.
4
–6
In humans, the standard
Mucositis is a serious, common, and debilitating consequence of cancer chemotherapy. 52 GI mucositis, which represents injury of the rest of the alimentary tract beyond oral mucositis is most prominent in the small intestine, but it also occurs in the esophagus, stomach, and large intestine. 53 The antimetabolites, MTX, and 5-Fluorouracil (5-FU), are known to damage the proximal small intestine, preferentially. 54,55 Cytotoxic drugs impair the turn-over of intestinal epithelia and induce flattening of the villi and an increased exposure of luminal contents to crypts. 56 These alterations could be involved in the frequent recurrence of GI symptoms, such as abdominal pain and diarrhea, in patients undergoing chemotherapy. 57
Overproduction of ROS and proinflammatory cytokines such as tumor necrosis factor alpha (TNF-α) and interleukin-1 beta (IL-1β) 58,59 have been reported to play important roles in chemotherapeutic drug induced mucositis. Recent findings suggest a role for iNOS and cyclooxygenase-2 in the onset of mucositis. 60,61 It is widely accepted that ROS initiate the cascade of events–including increased levels of inflammatory cytokines–that leads to mucosal damage. 62 – 64 Cisplatin-induced jejunal injury in Wistar rats is associated with increased oxidative stress, and apoptosis via up-regulating the expression of caspases-6 and -3. Cisplatin-induced lipid peroxidation, increased xanthine oxidase activity, glutathione depletion, decrease in antioxidant enzymes namely catalase, glutathione reductase, and glutathione-S-transferase in the rat jejunum. 63 In 5 FU-induced mucositis, upregulation NADPH oxidase 1(NOX1) and the production of ROS, TNF-α, and IL-1β, are reported to play important roles. 64 In MTX-induced mucositis, neutrophil-derived ROS, TNF-α, IL-1β, macrophage inflammatory protein-2, cytokine-induced neutrophil chemoattractant-2, inflammatory cytokines, and chemokines contribute to MTX mucositis and intestinal barrier dysfunction. 9,65 None of these studies have examined the plausible role of mitochondria in chemotherapy-induced mucositis, although mitochondria are major cellular sources of ROS.
Our study shows that MTX-induced mucositis is associated with distortion of the mitochondrial structure, mitochondrial dysfunction, and decreased activities of the respiratory chain complexes II and IV in the enterocytes. MTX administration caused functional impairment of mitochondria as evidenced by reduced RCR and MTT reduction, and increased mitochondrial swelling. The decreased activities of complexes II and IV may contribute to decreased ATP production as well as increased generation of ROS. We suggest that the inefficiencies in mitochondrial function caused by MTX may limit nutrient absorption and cell renewal resulting in weight loss, malabsorption and diarrhea. This mechanism may be applicable to the gastrointestinal side effects induced by other chemotherapeutic drugs.
Footnotes
Acknowledgments
The authors would like to thank the Centre for Scientific and Industrial Research (CSIR), New Delhi for the financial support and Mr. Viswa Kalyan Kolli senior research fellow for this project.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
