Abstract
The mechanism of doxorubicin (DOX)-induced cardiotoxicity remains controversial. Wistar rats (n = 66) received DOX injections intraperitoneally and were randomly assigned to 2 experimental protocols: (1) rats were killed before (−24 h, n = 8) and 24 h after (+24 h, n = 8) a single dose of DOX (4 mg/kg body weight) to determine the DOX acute effect and (2) rats (n = 58) received 4 injections of DOX (4 mg/kg body weight/week) and were killed before the first injection (M0) and 1 week after each injection (M1, M2, M3, and M4) to determine the chronological effects. Animals used at M0 (n = 8) were also used at moment −24 h of acute study. Cardiac total antioxidant performance (TAP), DNA damage, and morphology analyses were carried out at each time point. Single dose of DOX was associated with increased cardiac disarrangement, necrosis, and DNA damage (strand breaks (SBs) and oxidized pyrimidines) and decreased TAP. The chronological study showed an effect of a cumulative dose on body weight (R = −0.99, p = 0.011), necrosis (R = 1.00, p = 0.004), TAP (R = 0.95, p = 0.049), and DNA SBs (R = −0.95, p = 0.049). DNA SBs damage was negatively associated with TAP (R = −0.98, p = 0.018), and necrosis (R = −0.97, p = 0.027). Our results suggest that oxidative damage is associated with acute cardiotoxicity induced by a single dose of DOX only. Increased resistance to the oxidative stress is plausible for the multiple dose of DOX. Thus, different mechanisms may be involved in acute toxicity versus chronic toxicity.
Introduction
The use of anthracycline against malignant neoplasms is one of the major successful therapies for cancer. 1 Among anthracyclines, doxorubicin (DOX) is a powerful drug used for treating a wide variety of human tumors, including breast cancer, sarcomas, childhood solid tumors, leukemia, Hodgkin’s disease, non-Hodgkin’s lymphomas, and many others. However, its irreversible cardiotoxicity has limited its use. 2 Recent meta-analysis has shown that anthracyclines increase the risk for cardiac death by 4.94-fold as compared to non-anthracycline regimes. 3 Consequently, therapeutical administration of DOX needs to be carefully monitored to minimize the incidence of cardiotoxicity, which may significantly reduce its effectiveness against tumors. While acute cardiotoxicity exhibits temporary clinical signs, chronic events can include irreparable cardiomyopathy that affects approximately 26% of patients receiving a 550 mg/m2 total dose. 4
Although DOX-induced injury appears to be multifactorial, a common denominator to most of the proposed mechanisms is cellular damage mediated by reactive oxygen species (ROS). 5 –8 However, it remains increasingly controversial as to whether oxidative stress is a determinant mechanism. 9 –18 Using an early chronic protocol assessment 1 week after the last injection of multiple DOX doses (cumulative dose, 16 mg/kg body weight), we found that lycopene supplementation does not improve left ventricular dysfunction induced by DOX. 19 Interestingly, we have also discovered that DOX prevented lycopene metabolism and enhanced cardiac total antioxidant performance (TAP) in rats 1 week after ending treatment. These findings suggest DOX antioxidant activity after chronic treatment. This antioxidant activity was confirmed in vitro in a dose-dependent manner. 17 As it is well known that cardiomyopathy induced by DOX is dose dependent, it would be important to determine whether or not the antioxidant role of DOX 17 is also dose dependent. This would be especially important for new therapy strategies in patients undergoing chemotherapy with DOX.
The present study investigated the time course of DOX-induced cardiotoxicity using total antioxidant capacity, DNA damage, and morphological analysis in rat myocardium. First, we evaluated the acute effect of a single dose of DOX. Second, we studied the chronological effect of multiple DOX doses on cardiotoxicity by monitoring over time.
Materials and methods
Chemicals
Doxorubicin hydrochloride was purchased from Pharmacia (Milan, Italy). Sodium pentobarbital (Cristalia, Paulinia, São Paulo, Brazil) was used for anesthesia. Chemicals used in TAP analysis were fatty acid analog 4,4-difluoro-5-(4-phenyl-1,3-butadienyl)-4-bora-3a,4a-diaza-s-indacene-3-undecanoic acid (BODIPY 581/591) purchased from Molecular Probes (Eugene, Oregon, USA); radical initiator 2,2′-azobis(4-methoxy-2,4-dimethylvaleronitrile) (MeO-AMVN) purchased from Wako Chemicals (Richmond, Virginia, USA);
Animals
Male Wistar rats (n = 66, 12-week-old, weighing 350–400 g) were purchased from CEMIB (Unicamp, Campinas, São Paulo, Brazil). Animals were housed for 7 weeks in the animal facility of the Experimental Laboratory at Botucatu Medical School, UNESP. They consumed water and a commercial diet ad libitum, were weighed weekly, and clinically evaluated.
Animals were subjected to two experimental protocols (Figure 1). To determine the DOX acute effect, animals were killed 24 h before (−24 h, n = 8) and 24 h after (+24 h, n = 8) an intraperitoneal (IP) single dose (4 mg/kg body weight). No deaths occurred during the study. To determine the chronological effect of multiple DOX doses, rats (n = 58) received IP injections of DOX (4 mg/kg body weight), once a week for 4 weeks (a cumulative dose of 16 mg/kg body weight). The cumulative dose was selected according to previous studies. 20,21 Animals were randomly selected and killed before the first injection (M0) and one week after each injection. The times were designated as M0, M1, M2, M3, and M4. A total of 58 animals were used for the chronological study (from 58, 8 animals were also used in the acute study). However, non-programmed deaths occurred between M1 and M2 (n = 6), M2 and M3 (n = 5), and M3 and M4 (n = 7). Therefore, those animals (n= 18 were excluded and the remaining 40 animals were used for the chronological study (M0 (n = 8; they were the same animals used at −24 h of acute study); M1 (n = 8); M2 (n = 8); M3 (n = 8); and M4 (n = 8)).

Experimental Protocols. ⋆: animal killing;↑: DOX injection (4 mg/kg body weight); acute study: −24 h, that is, 24 h before DOX injection; +24 h, that is, 24 h after DOX injection; chronological study: M0, 24 h before (−24 h) before DOX treatment; M1, M2, M3, and M4: 1 week after 1st, 2nd, 3rd, and 4th DOX injection, respectively. DOX: doxorubicin.
Rats were fasted overnight, followed by euthanasia under sodium pentobarbital (50 mg/kg body weight) IP injection, and were immediately necropsied. Hearts (left ventricle) were collected and appropriately stored for the following analyses: TAP, oxidative DNA damage (comet assay), and morphology.
The protocol used was in accordance with the Ethical Principles for Animal Research and was approved by the local ethical committee for animal research (protocol # 591).
Histology
A 3-mm thick coronal section of each left ventricle was fixed in 10% buffered formalin for 48 h and embedded in paraffin. Sections of 5 mm thick were cut from the blocked tissue and stained with hematoxylin–eosin (H&E). The severity of pathologic changes identified by H&E was blindly graded by two pathologists on a scale from one (none or minor alterations) to three (marked abnormality) using semiquantitative analysis. 19,22 The analyses considered the myofibrillar disarrangement, myofibrillar necrosis, myocardial structure, and infiltrative changes. Disagreements between the pathologists were resolved by case-to-case discussion until a consensus decision was reached. These investigations were performed using an equipment from Carl Zeiss (Germany) and consisted of a light microscope (Axio Imager A1; magnification 400×) attached to a digital video camera (Zeiss Vision) and connected to a personal computer equipped with image analyzer software (Axio Vision Rel version 4.3, Carl Zeiss).
Total antioxidant performance
Liposome preparation
Liposomes were prepared as previously reported by Ferreira et al. 17 Phospholipids phosphatidylcholine (PC) was dissolved in chloroform and evaporated very slowly under a gentle stream of nitrogen while rolling the flask to deposit a thin lipid film. The flask was then maintained under nitrogen stream for an additional 20 min to eliminate residual solvent. Then, the lipid film was rehydrated with phosphate-buffered saline (PBS, 40 mM, pH 7.4) to a concentration of 2.5 mg of phospholipids per milliliter, frozen, and thawed three times and followed by sonication (Branson Sonifer 450, Danbury, Connecticut, USA) under ice for 30 s and repeated three times.
Cardiac TAP
The TAP of the heart (left ventricle wall) was quantified by comparing the area under the curve relative to the oxidation kinetics of PC liposome suspension used as the reference biological matrix. 23 The left ventricle, stored at −80°C, was weighed (0.1 g), minced, and homogenized for 20 s on ice with 2 mL of phosphate buffer (100 mM, pH 7.4) using an IKA, Ultra-Turrax T8 homogenizer (Wilmington, North Carolina, USA). Aliquots of the supernatant were collected after centrifugation of the cardiac tissue homogenate at 800g for 15 min in a Sorvall RT 6000 refrigerated centrifuge (Du Pont Co., Newtown, Connecticut, USA) and used as a sample for the TAP procedures. Protein concentration in samples was determined using a bicinchoninic acid kit according to the manufacturer’s protocol (Sigma-Aldrich, St Louis, Missouri, USA). After adding 100 µL of sample and 300 µL of PBS, BODIPY 581/591 (100 µL) was incorporated into the lipid compartment of the tissue at a final concentration of 2 µmol/L by vortexing gently for 10 s at low speed followed by a 10-min incubation at 37°C. After incubation, 485 µL of cold phosphate buffer (50 mM, pH 7.4) was added, and it was vortexed gently for 10 s at low speed. The radical initiator, MeO-AMVN (15 µL, final concentration 2 mM), was added into the reaction mixture with gentle mixing using a magnetic stirring bar. External standard curves were obtained by the oxidation kinetics of a control solution in the presence of PC (100 µL). The oxidation of tissue samples was monitored by the green fluorescent oxidation product of BODIPY 581/591 (excitation wavelength = 500 nm, emission wavelength = 520 nm, slit 10 nm) using a microplate reader (Wallac Victor 2, Perkin-Elmer, Boston, Massachusetts, USA). All sample analyses were carried out in triplicate. The results are expressed as TAP values, representing the percentage of inhibition of BODIPY oxidation in the rat left ventricle as compared to that occurring in the external standard samples. 23,24
DNA damage
Cardiomyocyte isolation
DNA damage was assessed in cardiomyocytes isolated from the heart (left ventricle wall) fragments according to the methodology previously described by Ferreira et al. 25 Briefly, the fragment was incubated with 5.5 mg of proteinase K and 3 mg of collagenase I in 3 mL of HBSS for 30 min at 37°C, and then the cells were suspended into 10 mL of HBSS. The resulting suspension was centrifuged at 93g for 5 min, and the supernatant was discarded.
Comet assay
The alkaline comet assay was performed in order to detect DNA single- and double-strand breaks (SBs), labile sites, and oxidized purines and pyrimidines. 26 All procedures were conducted in the dark to minimize spurious sources of DNA damage. Briefly, 15 µL of the cardiomyocyte suspension were embedded into 0.5% low-melting point agarose (Sigma) and spread on agarose-precoated microscope slides. Six slides were prepared for each sample. Slides were immersed overnight in freshly prepared cold lysing solution (2.5 M NaCl, 100 mM EDTA, 10 mM Tris, 1% sodium salt N-lauryl sarcosine, pH 10.0, with 1% Triton X-100, and 10% freshly added DMSO) at 4°C. After lysing, the slides were washed 3 times in enzyme buffer and then 2 slides for each treatment were incubated at 37°C for 45 min with 100 µL of endo III (1:1000), 100 µL of FPG (1:1000), or enzyme buffer only. Endo III recognizes oxidized pyrimidines, while FPG identifies oxidized purines, especially 8-oxo-guanine. Enzyme buffer only is used to identify SBs. Subsequently, the cells were exposed to alkali buffer (1 mM EDTA and 300 mM sodium hydroxide, pH approximately equal to 13.4), at 4°C, for 40 min to allow DNA unwinding and expression of alkali-labile sites. Electrophoresis was conducted on the same solution at 4°C for 30 min at 25 V (1 V/cm) and 300 mA. After electrophoresis, the slides were neutralized (0.4 M Tris, pH 7.5), fixed in absolute ethanol, stained with 70 µL Sybr Gold (2:10000), and analyzed in a fluorescent microscope at 40× magnification. Images from 50 nucleoids/animal/type of DNA lesion (from two replicate slides; SBs, oxidized pyrimidines, and oxidized purines) were scored using the comet assay II image system (Perceptive Instruments, Haverhill, Suffolk, UK). Tail intensity (percentage of DNA in the tail) was used to estimate the extent of DNA damage. Human lymphocytes treated with hydrogen peroxide (100 µM) were used as the electrophoresis positive control.
Statistical analysis
Results are expressed as mean ± SE and median (25-–75 percentile) for data with normal and nonnormal distributions, respectively. In the acute study, the significance of differences was calculated by t test or Mann–Whitney rank sum test. While in the chronological study, one-way analysis of variance complemented by all pairwise multiple comparison procedures was used. Myofibrillar necrosis and disarrangement were treated with Kruskal–Wallis one-way analysis of variance on ranks complemented by all pairwise multiple comparison procedures. Linear regression procedures were used to study the associations between variables in the chronological study. The data were analyzed using SigmaStat version 3.5 software (Systat Software, Point Richmond, California, USA). Differences were considered significant when p < 0.05.
Results
Acute effect of a single dose of DOX on cardiac histology, DNA damage, TAP, and body weight
A single dose of DOX caused impairment in several variables at +24 h when compared with −24 h. In detail, increase in myofibrillar necrosis (p < 0.001) and in myofibrillar disarrangement (p = 0.002) were verified (Table 1). Increase in DNA damage (DNA SBs, p < 0.001 and oxidized pyrimidines, p = 0.002) and decrease in TAP (p = 0.010) were identified (Figure 2). No relevant inflammatory infiltrate was seen in myocardium, and no significant changes were observed in body weight (in grams; 412.00 (378.00–436.50) vs. 401.50 (342.50–428.50), p > 0.05), as well as in clinical and necropsy findings. None of the animals died during this experimental protocol.
Effect of a single dose of DOX ((4 mg/kg body weight) on the rat heart) on rat myocardium histological changes.a

DOX: doxorubicin; n ): sample number.
aValues are medians (25–75 percentiles); moments: −24 h (before) and +24 h (after) injection; Mann–Whitney rank sum test was used for comparisons between moments.
b p < 0.001: different from −24 h.
c p = 0.002: different from −24 h.
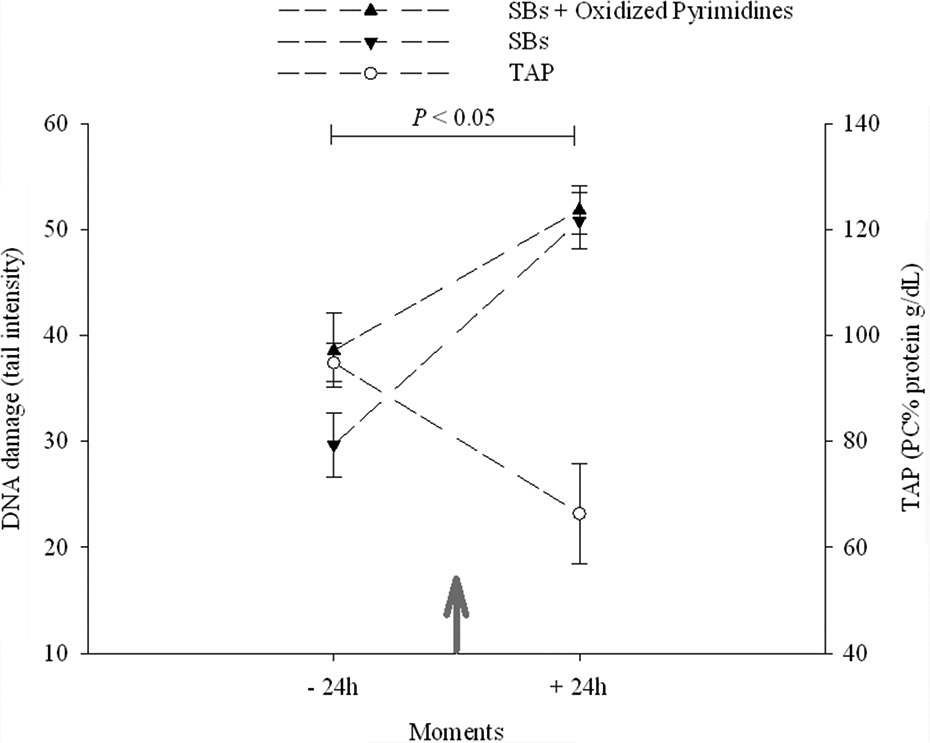
Effect of a single dose of DOX (4 mg/kg body weight) on the rat heart. Data are expressed as means ± SE; ↑: DOX IP injection; moments: −24 h (before) and +24 h (after) injection; data obtained at −24 h (n = 8) and at +24 h (n = 8) were analyzed for DNA SBs and TAP with PC as the standard. Mann–Whitney rank sum test (for DNA damage) or t test (for TAP) were used to compare the moments, * p < 0.05: difference between moments. DOX: doxorubicin; SBs: strand breaks; TAP: total antioxidant performance; IP: intraperitoneal; PC: phosphatidylcholine.
Chronological effect of multiple DOX doses on general changes
Figure 3 shows the relationship between variables and moments after treatment with multiple doses of DOX. Progressive deleterious clinical signs (general status, physical activity, hair brightness, and hair losses) were associated with the increasing doses of DOX therapy. Most animals (>86%) exhibited those clinical changes from M3. Animal weight (mean ± SE sample number, in grams) significantly decreased (Tukey’s test, p < 0.05) over time (M4 (300.91 ± 11.088) < M0 (409.88 ± 16.428), M1 (395.38 ± 20.448), M2 (377.13 ± 18.098) and M3 (337.88 ± 18.888) < M0, M1). Linear regression showed that body weight was inversely associated with time (R = −0.99, p = 0.011), suggesting a progressive weight loss as additional doses of DOX were administered (Figure 3(a)). At necropsy, all the animals displayed evident and progressive cardiac softness, pericardium/myocardium adherence, and accumulation of serous fluid in the pericardial, pleural, and peritoneal cavities from M2 (1 week after second DOX dose). Besides bilateral kidney pale, adherence among hepatic lobes and peritoneal organs (liver, stomach, and spleen) at M4 were noted. The mortality rate was 31% (n = 18/58) in the chronological protocol. Deaths occurred between M1 and M2 (n = 6), M2 and M3 (n = 5), and M3 and M4 (n = 7).

Associations between variables and moments after treatment with multiple doses of DOX. Data are expressed as means ± SE; moments: M1(1 week after first injection); M2 (1 week after second injection); M3 (1 week after third injection); and M4 (1 week after fourth injection). Data were analyzed for DNA SBs and TAP. Linear regression was used to examine the association between variables (body weight (a), myofibrillar necrosis (b), TAP (c), and DNA damage SBs (d)) and moments in treated animals. DOX: doxorubicin; TAP: total antioxidant performance; SBs: strand breaks.
Chronological effect of multiple doses of DOX on cardiac histology
The treatment with DOX was associated with a progressive histological cardiomyocyte injury (Table 2). The lesions were multifocal and were characterized by myofibrils with progressive hyalinization, disarrangement, fragmentation, rarefaction, morphology alteration (increased volume followed by decreased volume, retraction, and culminating with nuclei absence), and staining tone variation of the nuclei (hyperchromatic followed by pale). In addition, it was verified that myofibrillar paired nuclei in M3 and M4, suggesting atrophy. Necrosis was positively associated with time (M1, M2, M3, and M4) in treated animals (R = 1.00, p = 0.004), suggesting a progressive increase as additional doses of DOX were administered (Figure 3(b)). No relevant inflammatory infiltrate was identified in myocardium.
Chronological effect of multiple doses of DOX (4 mg/kg body-weight/week/4 weeks) on rat myocardium histological changes.a

DOX: doxorubicin; M0: before first injection; M1: 1 week after first injection; M2: 1 week after second injection; M3: 1 week after third injection; M4: 1 week after fourth injection; (n): sample number; ANOVA: analysis of variance.
aValues are medians (25–75 percentiles). One-way ANOVA on ranks (Kruskal–Wallis one-way ANOVA on ranks) complemented by Duncan’s method was used for comparisons among moments.
b p < 0.001: different from M0.
Chronological effect of multiple doses of DOX on cardiac TAP
Figure 4 shows the results of the TAP assay in the myocardium from animals treated with (M1, M2, M3, and M4) or without (M0) DOX. No significant decrease in the mean TAP values (adjusted for cardiac tissue protein concentrations and not for tissue weight) was detected between M0 and M1 (data not shown), but significant increase (p = 0.006) was observed from M1 (M0 < M4, M1 < M2, M3, M4). Linear regression in treated animals showed that TAP was positively (R = 0.95, p = 0.049) associated with time (M1, M2, M3, and M4), suggesting a progressive increase as additional doses of DOX were administered (Figure 3(c)).

Chronological effect of multiple doses of DOX (4 mg/kg body weight/week/4 weeks) on rat heart TAP. Data are expressed as means ± SE; ↑: DOX injection (4 mg/kg body weight). Moments: M0 (before first injection; n = 7); M1 (1 week after first injection; n = 7); M2 (1 week after second injection; n = 8); M3 (1 week after third injection; n = 8); and M4 (1 week after fourth injection; n = 8). Data were analyzed for TAP and PC was used as the standard. One-way ANOVA complemented by Duncan’s method was used to compare moments, *p = 0.006: different from M0; # p = 0.006: different from M1. TAP: total antioxidant performance; PC: phosphatidylcholine; DOX: doxorubicin; ANOVA: analysis of variance.
Chronological effect of multiple doses of DOX on DNA
Overall, decreased DNA SBs and oxidized purines and pyrimidines were detected from the second and third DOX doses, respectively. A marked increase in DNA SBs was identified between M0 and M1 (M1 > M0), which was followed by significant decreases (M2, M3, M4 < M1, M3, M4 < M2) after additional doses of DOX (Figure 5(a)). These findings may partially suggest a progressive attenuation of DNA damage from the first DOX dose. When SBs and oxidized purines were considered together, decreased DNA damage was observed at M1 (M1 < M0), M3 (M3 < M0, M2), and M4 (M4 < M0, M1, M2). Additionally, an increase was also detected between M1 and M2 (Figure 5(b)). When DNA damage was assessed by SBs and oxidized pyrimidines, similar behavior was verified, although comparisons among moments have shown differences (M4 < M1, M2, M3; Figure 5(c)). Linearity in regression of treated animals demonstrated significant inverse association between time (M1, M2, M3, and M4) and DNA damage only when SBs were evaluated (R = −0.95, p = 0.049; Figure 3(d)). No significant association was detected for the other type of DNA damage (oxidized purines and pyrimidines).

Chronological effect of multiple doses of DOX (4 mg/kg body weight/week/4 weeks) on DNA damage (tail intensity). Data are expressed as means ± SE; ↑: DOX injection (4 mg /kg body weight); moments: M0 (before first injection; 250 nucleoids, n = 2); M1 (1 week after first injection; 450 nucleoids, n = 3); M2 (1 week after second injection; 375 nucleoids, n = 3); M3 (1 week after third injection; 425 nucleoids, n = 3); and M4 (1 week after fourth injection; 384 nucleoids, n = 3). Data were analyzed for DNA SBs. One-way ANOVA complemented by Duncan’s method was used to compare moments, *p < 0.001: different from M0; # p < 0.001: different from M1; ψp < 0.001: different from M2; θp < 0.001: different from M4. DNA damage SBs (a), DNA damage SBs + oxidized purines (b), DNA damage SBs + oxidized pyrimidines (c). DOX: doxorubicin; SBs: strand breaks; ANOVA: analysis of variance.
Associations between variables
In DOX-treated animals, necrosis was negatively associated with body weight (R = −0.98, p = 0.024; Figure 6(a)), and DNA SBs (R = −0.97, p = 0.027) was also identified (Figure 6(b)). Necrosis was positively associated with TAP (R = +0.96, p = 0.039; Figure 6(c)). TAP was negatively associated with DNA damage (SBs; R = −0.98, p = 0.018; Figure 6(d)). Oxidized purine was positively associated with oxidized pyrimidine (R = 0.97, p = 0.027). No associations were identified between necrosis versus DNA-oxidized purine (R = −0.80, p > 0.05) and pyrimidine (R = −0.78, p > 0.05) and between TAP versus oxidized purine (R = −0.561, p > 0.05) and pyrimidine (R = −0.618, p > 0.05).
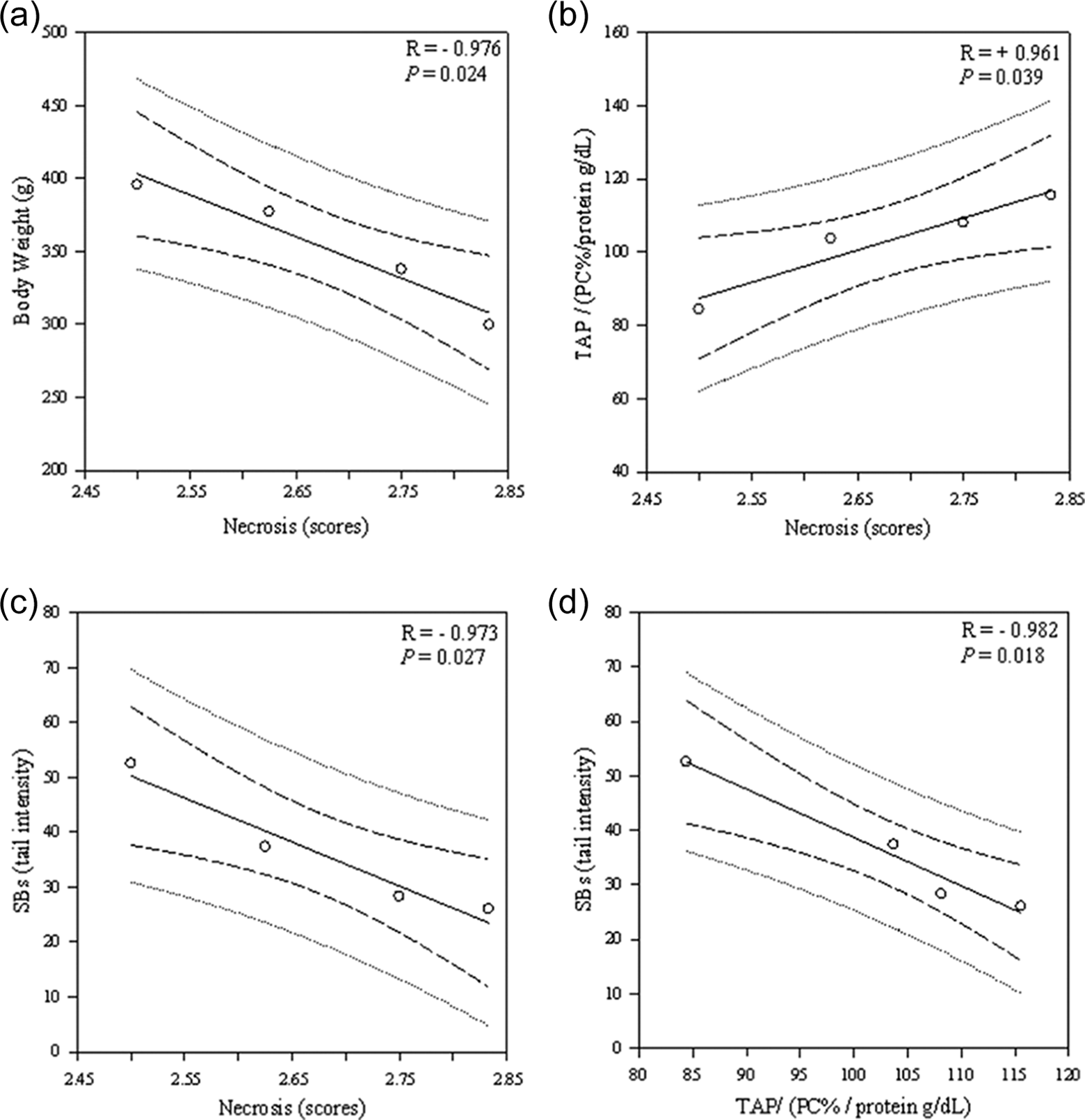
Associations between variables after treatment with multiple doses of doxorubicin. Data are expressed as means ± SE; each plot represents moments: M1 (1 week after first injection); M2 (1 week after second injection); M3 (1 week after third injection); M4 (1 week after fourth injection). Linear regression was used to examine the association between variables (necrosis vs. body weight (a), SBs (b), TAP (c); TAP vs. SBs (d)) in treated animals. DOX: doxorubicin; SBs: strand breaks; TAP: total antioxidant performance.
Discussion
Our results showed that single dose of DOX was related with increased cardiac disarrangement, necrosis, and DNA damage, including oxidized pyrimidines and with decreased TAP. While, the chronological effect of multiple DOX doses was associated with progressive and deleterious general signs (clinical, body weight, and necropsy data). Linear regression analysis also showed progressive worsening of cardiac necrosis, enhanced TAP, and decrease in DNA damage. Associations between several variables also were identified when using multiple DOX doses. DNA damage was inversely associated with TAP, while necrosis was negatively associated with DNA damage and body weight and positively associated with TAP.
The mechanism of DOX-induced cardiotoxicity still remains unclear. Several hypotheses have been postulated, including the generation of ROS. 27 However, ROS-induced cardiotoxicity has not been confirmed and different mechanisms may be involved in acute toxicity versus chronic toxicity. Numerous studies have used DOX regimes in which animals are treated with a single and extremely high anthracycline dose. Therefore, the outcomes from these studies must be cautiously interpreted. Also, depending on the protocol study, there are risks of oversimplification and of inappropriately mixing acute and chronic cardiotoxic effects. 1 In fact, a number of studies using antioxidant supplements, such as vitamin E, 28 –30 N-acetylcysteine, 31 iron chelator, 32 and polyphenol 33 has failed to show a protective effect on chronic cardiomyopathy.
We have previously observed that lycopene did not improve left ventricular dysfunction induced by DOX treatment in an early chronic protocol (16 mg/kg body weight, fractionated doses). 19 Interestingly, we have identified that DOX prevented the lycopene metabolism and enhanced total antioxidant capacity in the rat heart 1 week after the end of treatment with multiple DOX doses, suggesting an antioxidant function of DOX under chronic treatment. 17 As it is well known that the cardiomyopathy induced by DOX is exponentially dose dependent, 34,35 it would be important to figure out whether the antioxidant role of DOX suggested in the past 17 is also dose dependent. This answer would be especially contributory for new therapy strategies in patients undergoing chemotherapy with DOX.
In the current study, animals received a cumulative dose of 16 mg/kg body weight (4 doses of 4 mg/kg body weight), which was equivalent to 500 mg/m2 of DOX in a 50-kg human being. 36 The study design mimics clinical situations in which multiple doses of anthracycline are given to patients. It is important to underline that this protocol was chosen to evaluate the acute effect of a single dose of DOX and also the kinetic behavior of multiple doses on the heart.
The occurrence of cardiac disorders caused by DOX was monitored by several toxic end points (clinical features, necropsy findings, histology, TAP, and DNA damage). To determine the DOX acute effect on heart, animals were killed before (−24 h) and after (+24 h) a single dose (4 mg/kg body weight). To determine the chronological effect of multiple doses of DOX, rats received the drug dose (4 mg/kg body weight) once a week for 4 weeks and were killed before the 1st injection (M0) and 1 week after each injection (M1, M2, M3, and M4).
Acute effect of DOX
After 24 h of a DOX single dose, decrease in TAP values and increase in myofibrillar necrosis, myofibrillar disarrangement, and DNA damage (DNA SBs and DNA SBs + oxidized pyrimidines) were identified. Nevertheless, no obits and no changes in body weight, or clinical signs and necropsy findings were observed. Similar results have been found in previous studies using a single dose of DOX.
Earlier cardiac myocytes necrosis has been detected in cell cultures 7 h after DOX exposure (0.5 µg/mL) 37 and in Wistar rats, 48 h after DOX treatment (25 mg/kg body weight). 38 Other studies have shown myofibrillar disarrangement 38 and decreased cardiac hydrophilic antioxidant defense (glutathione, 39 catalase, 40 superoxide dismutase, and glutathione peroxidase 40,41 ), after the first, 39 second, 41 or third 40 day of DOX injection in rodents. Clinical study identified significant reduction of plasma total hydrophilic antioxidant defense (TRAP, peroxyl radical-trapping antioxidant) at 8 h after the first adriamycin infusion in patients. 42
Increased DNA damage, including oxidized pyrimidines and purines, has been detected in H9c2 cardiac myocytes treated with DOX (0.5 μg/mL) for 4 h, suggesting that DNA damage occurs early in the DOX cardiotoxicity. 43 Additionally, it has been reported that the overexpression of glutathione transferase (α4-isoform) in H9c2 cardiac myocytes eliminated oxidative stress induced by DOX (0.5 μg/mL for 7 h) but did not abolish the total and apoptotic cell death. Taken together, all these data reinforce oxidative stress as an important mechanism but not the exclusive mediator of cardiac myocyte death induced by acute DOX treatment. 37
Chronological effect of multiple DOX doses
It is well known that the dose dependency of cardiotoxicity is induced by anthracyclines. 34,35 However, few studies have chronologically followed the drug’s effect on cardiac damage. The damage was monitored over time for body weight, 44,45 echocardiogram, 46,47 cardiomyocyte histology, 48 and oxidative stress markers (cardiac malondialdehyde, superoxide anion, 44 and hydrophilic individual antioxidant components 49 ). On the other hand, to our knowledge, no study has ever chronologically monitored the in vivo effect of multiple DOX doses on cardiac DNA damage (using comet assay) or total antioxidant defense (using TAP).
Here, we detected progressive deleterious clinical signs (general status, physical activity, hair brightness, and hair losses) associated with increased DOX therapy. Most animals (>86%) exhibited those clinical changes from M3. Similarly, lethargy 20,50 and progressive decrease in body weight 19,25,44,45 were observed in rats treated with multiple doses of DOX. Our findings of necropsy are also in accordance with those observed in rabbits treated with adriamycin (2.2 mg/kg body weight/week/10 weeks). 48 The progressive rise in cardiac myofibrillar necrosis observed with increasing DOX dosage is in accordance with a previous study that examined the effect of adriamycin (1.1 mg/kg body weight twice weekly for 10 weeks) on the hearts of rabbits at three different time points. 48 The mortality rate (31%) is consistent with other reports. 21,51,52
With regard to TAP, our data demonstrated that, in vivo, cardiac defense rises with increasing DOX dose. This finding is confirmed by our previous in vitro study that showed antioxidant activity of DOX increases in a dose-dependent manner (1, 2, 4, and 8 µM). 17 Various studies have evaluated the effect of multiple doses of DOX on individual components of the cardiac antioxidant system at only one time moment, and no changes in rodent superoxide dismutase 35,49 and catalase 35,53 activities have been found. However, increased catalase activity has also been described. 54
An important aspect to be mentioned is the fact that few studies have evaluated the chronological effect of DOX multiple doses on the antioxidant system. In addition, those few studies have reported antioxidant system hydrophilic components only. Cardiac glutathione peroxidase and manganese-superoxide dismutase activities were decreased at 1 and 2 h after the last injection in rats that received fractionated adriamycin doses (cumulative dose, 15 mg/kg body weight). 49 However, these results should be viewed cautiously since the moments (1 and 2 h) analyzed may represent a transient acute effect of the last drug injection. No linear relationship between hydrophilic antioxidant and the number of adriamycin injections have been found in rabbits (cardiac total glutathione, reduced glutathione, and gluthatione peroxidase) 48 and in humans (plasma TRAP). 42 The differences between this and previous studies may be due to the methodology used. We adopted the TAP assay that measures antioxidant capacity in both the hydrophilic and lipophilic compartments. Additionally, the TAP assay allows evaluation of the interaction between the antioxidants in the two compartments 55 against oxidative stress.
A decrease in cardiomyocyte DNA SBs and oxidized purines and pyrimidines were currently detected after the second and 3third DOX injections, respectively. Using the same DOX protocol (fractionated doses), we have previously reported similar levels of DNA damage (SBs, SBs + oxidized purines, and SBs + oxidized pyrimidines) in rat heart cells at M4. 25 The progressive attenuation of damage verified in cardiomyocytes in vivo was also observed in peripheral blood lymphocytes in healthy adult humans after in vitro treatments with DOX at 0.25, 0.5, 0.75, and 1 µM. 56 Similar data were also found in human breast adenocarcinoma cell lines treated with adryamicin. According to the authors, this effect might be due to the induction of both DNA SBs and cross-links. 57 On the other hand, marked increase of DNA damage (SBs) was presently identified between M0 and M1 (M1 > M0), after a single dose of DOX. This result is in agreement with other studies that have shown increased DNA damage in vitro (normal cell incubation) 56 and in vivo (cancer patients) 58 after DOX treatment.
Our study also showed identifying associations between some variables in animals treated with multiple doses of DOX. Necrosis was progressive and negatively associated with body weight and DNA damage, and positively with TAP, indicating that oxidative stress is not a determinant mechanism for the necrosis induced by multiple doses of DOX. Furthermore, TAP was negatively associated with DNA damage, suggesting that the progressive DNA damage attenuation is related with the total antioxidant defense improvement. This last result could be explained in part by the antioxidant effect of multiple doses of DOX, as previously shown by our group. 17 These results suggest that the progressive cardiomyofibrillar necrosis and weight loss induced by multiple DOX doses occur despite the gradual DNA damage attenuation and the total antioxidant defense increase.
In conclusion, our study showed that (1) single dose of DOX was associated with augmented cardiac disarrangement, necrosis, and DNA damage, including oxidized pyrimidines, and with diminished TAP; (2) the chronological effect of multiple DOX doses was associated with progressive and deleterious general signs (clinical, body weight, and necropsy data). Linear regression analysis also showed gradual worsening of cardiac necrosis, gradual increase in TAP, and decrease in DNA damage. Moreover, DNA damage was inversely associated with TAP, while necrosis was negatively associated with DNA damage and body weight and positively associated with TAP.
Taken together, our data indicated that the oxidative stress mechanism is an early event in the DOX-induced cardiotoxicity. Additionally, the results demonstrated that the cardiomyocyte injury induced by multiple DOX doses occurred despite DNA damage attenuation and the total antioxidant defense increase. Therefore, it is highly probable that cardiotoxicity induced by multiple DOX doses is not related to an oxidative stress mechanism. Although the experimental design using single and multiple doses of DOX mimics clinical situations, it would be imprudent to extrapolate the current results to humans. However, the present study represents a contribution to our understanding of the myocardium damage mechanism in acute and chronic DOX treatment. Further studies are needed to confirm that different mechanisms may be involved in acute versus chronic cardiotoxicity induced by DOX. Moreover, chronological evaluations should be considered while studying chronic cardiotoxicity.
Footnotes
Acknowledgments
We appreciate Barbara B Golner’s editing of the English grammar. We thank Alexandre L Loureiro, Corina Correa, Elenize Jamas Pereira, Jose A Souza, Jose C Georgete, Jian Qin, Mario A Dallaqua, Mario B Bruno, Rogerio A Monteiro, Sandra A Fábio, Sueli Clara, and Vitor M Souza for their technical help during this study.
Funding
The present study was financially supported in part by Fundação de Amparo à Pesquisa do Estado de São Paulo, São Paulo, Brazil (FAPESP # 2007/07455-2), Conselho Nacional de Pesquisa (CNPq PQ-II # 302293/2012-4), by Pro-Reitoria de Pesquisa UNESP, São Paulo, Brazil, and by a grant from the U.S. Department of Agriculture, Agricultural Research Service (# 58-1950-7-0707). Any opinions, findings, conclusion, or recommendations expressed in this publication are those of the authors and do not necessarily reflect the view of the funders.
