Abstract
The ameliorative effect of calcium channel blockers (CCBs) and a phospholipase-A2 inhibitor in drug-/chemical-induced nephrotoxicity was investigated. Rats were divided into 7 groups of 5 rats in each group. In the gentamicin model, group I rats were pretreated with normal saline (10 ml kg−1), while groups II–VII rats were pretreated with normal saline (10 ml kg−1), ascorbic acid (10 mg kg−1), nifedipine (0.86 mg kg−1), verapamil (4.3 mg kg−1), diltiazem (3.43 mg kg−1), and prednisolone (0.57 mg kg−1), respectively, perorally 1 h before intraperitoneal (i.p.) injection of gentamicin (40 mg kg−1) for 14 days. In the carbon tetrachloride (CCl4) model, rats were pretreated with CCBs and prednisolone for 7 days before inducing nephrotoxicity with 20% CCl4 (1.5 ml kg−1). Rats were thereafter killed and blood and tissue samples were collected for assessments. I.p. injections of gentamicin and CCl4 caused significant hypernatremia, hypokalemia, hypocalcemia, hypophosphatemia, and hyperchloremic alkalosis and reduced renal tissue levels of antioxidants. Also, significant reductions in the hemoglobin, packed cell volume, red blood cells, and platelet indices were observed. Pretreatments with nifedipine (0.86 mg kg−1), verapamil (4.3 mg kg−1), diltiazem (3.43 mg kg−1), and prednisolone (0.57 mg kg−1) significantly ameliorated the deleterious effects of gentamicin and CCl4 possibly via antioxidant and anti-lipoperoxidation mechanisms. The results obtained in this study suggest potential clinical usefulness of tested CCBs and prednisolone in drug-/chemical-induced nephrotoxicity.
Introduction
Drugs, including aminoglycoside antibiotics, nonsteroidal anti-inflammatory drugs, amphotericin B, and angiotensin-converting enzyme inhibitors, have been implicated as causes of nephrotoxicity. 1 Factors such as age, presence of comorbidities, and exposure to scores of drugs and other therapeutic agents could trigger and culminate into drug-induced nephrotoxicity. 2 The primary function of the renal system is the elimination of waste products derived either from endogenous metabolism or from the metabolism of xenobiotics. The kidneys also play a major role in the process of erythropoiesis. 3 The kidney is a common site of chemical toxicity as a result of its constant exposure to chemicals, solutes, and other detrimental materials, toxicants, which it needs to eliminate from the host body. Although the kidneys comprise <1% of the body mass, they receive around 25% of the cardiac output, thus significant amounts of exogenous chemicals and their metabolites are delivered to the kidney. 4 Biotransformation of chemicals to reactive and toxic metabolites is a key feature of nephrotoxicity and many toxicants, including acetaminophen, bromobenzene, chloroform, and carbon tetrachloride (CCl4), can be activated. Drug-induced nephrotoxicity can be seen in both inpatient and outpatient settings with variable presentations ranging from mild to reversible injury to advanced kidney disease. Manifestations of drug-induced nephrotoxicity include acid–base abnormalities, electrolyte imbalances, urine sediment abnormalities, proteinuria, pyuria, hematuria, and decline in the glomerular filtration rate. 1 The effects used clinically to diagnose and assess kidney damage include increases in urinary glucose, amino acids, or protein, changes in urine volume, osmolarity or pH, blood urea nitrogen (BUN), plasma creatinine, and serum enzymes. 5
Drug-induced nephrotoxicity is a relatively common complication, and its incidence has been on the increase with the ever-increasing plethora of drugs and the ease of availability of over-the-counter drugs with detrimental effect on the renal function. Drugs cause approximately 20% of community- and hospital-acquired episodes of acute renal failure. 6,7 Although renal impairment is often reversible if the offending drug is discontinued, the condition can be costly and may require multiple interventions including hospitalization. 8 In the acute care hospital setting, drug-induced nephrotoxicity has been implicated in 8–60% of all cases of inhospital acute kidney injury and is a recognized source of significant morbidity and mortality. 9 Inhospital drug use may contribute to 35% of all cases of acute tubular necrosis, most cases of allergic interstitial nephritis, as well as nephrotoxicity due to alterations in renal hemodynamics and postrenal obstruction. 10 The incidence of antibiotic-induced nephrotoxicity alone may be as high as 36%. 11 Thus, there is no gainsaying that drug-induced nephrotoxicity in a clinical setting is prominent and has profound negative impact on the patient, the health-care service provider, and the community at large. One important impact of this is an increase in the hospitalization rate in the hospitals due to drug-induced nephrotoxicity and renal damage in both inpatients and the community.
Despite the high prevalence of drug-induced and drug-related renal diseases among the human population, there still appears not to be effective clinical drugs available to curtail this public human nightmare. This has necessitated the urgent need to find or develop new drugs in this respect. Various strategies have been known and used to ameliorate or counter the effects of nephrotoxicity whether drug induced or otherwise. Also, studies have been conducted to investigate the nephroprotective properties of different medicinal plants, including Orthosiphon stamineus and Pimpinella tirupatiensis, and pharmaceutical drugs with nephroprotective potentials. The overall objective of these studies is to alleviate nephrotoxic effects by preventing the onset of acute renal failure in the host patient by inhibiting one or more of the several biochemical mechanisms through which renal cell injury occurs. These mechanisms include cell death through apoptosis and oncosis, disruption of cell volume/ion homeostasis, changes in cell membrane integrity/polarity, mitochondrial adenosine triphosphate dysfunction, and disruption in cellular calcium (Ca2+) homeostasis. 5 Other biochemical mechanisms of renal cell injury are mediated through the enzyme systems that include phospholipases, endonucleases, proteinases, and signaling kinases. 5
Ca2+ is a second messenger that plays a critical role in a variety of cellular functions. Its distribution within renal cells is known to be complex and involves binding to anionic sites on macromolecules and compartmentalization within subcellular organelles. However, because the proximal renal tubular cells reabsorb approximately 50–60% of the filtered load of Ca2+, they must maintain low cytosolic Ca2+ concentrations during a large Ca2+ flux. Sustained elevations or abnormally large increase in free cytosolic Ca2+ can exert a number of detrimental effects on the renal cells. An increase in free cytosolic Ca2+ can activate a number of degradative Ca2+-dependent enzymes such as phospholipases and proteinases, which can produce an aberration in the structure and function of cytoskeletal elements. 5 While the precise role of Ca2+ in toxicant-induced injury remains unclear, release of endoplasmic reticulum (ER) Ca2+ stores may be a key step in initiating the injury process and increasing cytosolic-free Ca2+ concentrations. 12 However, prior depletion of ER Ca2+ stores protect renal proximal tubules from extracellular Ca2+ influx and cell death produced by mitochondrial inhibition and hypoxia. 13 Ca2+ channel blockers (CCBs) are known pharmacologically to inhibit the influx of Ca2+ into cells thereby depleting the Ca2+ stores in the renal cells, thus potentially exerting nephroprotective effect.
Phopholipase A2 (PLA2) consists of a family of enzymes that hydrolyze the acyl bond of phospholipids resulting in the release of arachidonic acid and lysophospholipid upon activation during inflammation. PLA2 activation has been suggested to play a role in various forms of cell injury through a variety of mechanisms. 14 The enzyme phospholipase is involved in the biochemical mechanism through which renal cell injury is manifested. A supraphysiologic increase in PLA2 activity could cause an increase in the loss of membrane phospholipids and consequently impair membrane function. It is known that during inflammatory processes, the activity of PLA2 is increased and thus plays a fundamental role in causing damage to cells during inflammation or cell injury. Inhibition of PLA2 activity is thought to alleviate or ameliorate renal cell injury in nephrotoxicity.
This study was designed to investigate the ameliorative effect of CCBs (nifedipine, verapamil, and diltiazem), and PLA2 inhibitor (prednisolone) in gentamicin- and CCl4-induced renal injury in rats.
Materials and methods
Chemicals and drugs
Chemicals used in the study were of analytical grade and include ascorbic acid, potassium chloride (KCl; 1.15%), 10% formalin, 20% CCl4 solution in olive oil, and normal saline (Unique Pharmaceuticals, Sango-Ota, Nigeria).
Drugs used include gentamicin (Lek Pharmaceutical and Chemical Company, Veroskova, Slovenia), nifedipine (Salutas Pharma GmBH for Lek Pharmaceutical and Chemical Company, Veroskova, Slovenia), verapamil (Teva UK Limited, West Yorkshire, UK), diltiazem (Generics UK Limited, UK), and prednisolone (Hovid Berhad, Malaysia).
Animals
A total of 84 young adult male Wistar rats aged 4–6 weeks (weighing 150–200 g) were obtained from the Laboratory Animal Center, College of Medicine, University of Lagos, Lagos, Nigeria. The animals were housed in standard metal cages, maintained under standard laboratory conditions (23–25°C, 12 h/12 h light/dark cycle), and fed with standard rat pellets (Livestock Feeds PLC, Ikeja, Lagos, Nigeria) and water ad libitum. The protocol for this study was in compliance with the requirements of the Experimentation Ethics Committee on Animal Use of the College of Medicine, University of Lagos, Lagos, Nigeria, and the United States National Academy of Sciences Guide for the Care and Use of Laboratory Animals. 15 The rats were acclimatized for 2 weeks before the commencement of the experimental procedures. Seventy-five rats from the stock procured were used for the experiment after acclimatization.
Experimental design
Gentamicin-induced nephrotoxicity
Rats were randomly divided into 7 groups with 5 rats in each group such that the weight differences within and between groups did not exceed ±20%. Group I rats were pretreated with 10 ml kg−1 of normal saline perorally (p.o.) 1 h before intraperitoneal (i.p.) injection of 1 ml kg−1 of 0.9% normal saline. The rats in groups II–VII were pretreated with 10 ml kg−1 of normal saline, 10 mg kg−1 of ascorbic acid, 0.86 mg kg−1 of nifedipine, 4.3 mg kg−1 of verapamil, 3.43 mg kg−1 of diltiazem, and 0.57 mg kg−1 of prednisolone p.o., respectively, 1 h before i.p. injection of 40 mg kg−1 gentamicin. 16 Treatments were carried out once daily between 07:00 h and 09:00 h for a period of 14 days. Nifedipine, verapamil, diltiazem, and prednisolone were administered at their respective therapeutic doses.
CCl4-induced nephrotoxicity
Animals were randomly grouped and treated as was the case in the gentamicin-induced nephrotoxicity model. 17 Treatments were conducted once daily between 07:00 h and 09:00 h for a period of 6 days. After 1 h of peroral administration on day 6, the rats in group I were injected with 1 ml kg−1 of normal saline, while groups II–VII rats were injected i.p. with 1.5 ml kg−1 of 20% CCl4 dissolved in olive oil. After 24 h of post-CCl4 injection, the rats were killed under inhaled diethyl ether.
Collection of blood samples and kidneys
Feed was withdrawn from the evening of the 14th day, but rats still had access to drinking water in respect of the gentamicin-induced nephrotoxicity model. On the 15th day, the rats were weighed and then anesthetized using inhalational diethyl ether. In the CCl4 model, animals were anesthetized 24 h post-CCl4 injection. Blood samples for biochemical assays (renal function tests) and hematological assays (full blood counts) were obtained by cardiac puncture and collected into plain sample bottles and ethylenediaminetetraacetic acid-treated bottles, respectively.
After blood collection, the kidneys were harvested from each animal and weighed. One of the kidneys was rinsed in 1.15% KCl solution in order to preserve the oxidative enzyme activities of the kidney before being placed in a clean sample bottle which itself was in an ice-pack filled cooler. This is to prevent the breakdown of the enzymes for kidney function enzyme biomarkers. Thereafter, the oxidative stress markers activity was determined.
Kidney enzymatic and nonenzymatic antioxidant parameters
Superoxide dismutase (SOD) activity was determined by its ability to inhibit the auto-oxidation of epinephrine by the increase in absorbance at 480 nm as described by Sun and Zigman. 18 Catalase (CAT) activity was determined by measuring the decrease in absorbance at 240 nm due to the decomposition of hydrogen peroxide (H2O2) in ultraviolet-recording spectrophotometer. 19 Reduced glutathione (GSH) content was estimated according to the method described by Sedlak and Lindsay, 20 using 10% trichloroacetic acid, 0.5 ml of Ellman’s reagent (5,5′-dithiobis-(2-nitrobenzoic acid)), and 3.0 ml phosphate buffer (0.2 M, pH 8.0), and the absorbance was read at 412 nm.
Serum biochemical parameters
Creatinine was determined by adding an aliquot of 0.5 ml of serum sample to 3.5 ml of picric acid. The mixture was centrifuged for 5 min. Then, 3 ml of the supernatant was taken, and to this, 0.2 ml of 4 N sodium hydroxide was added. The mixture was incubated for 1 min, and the absorbance was read at 520 nm. The concentration of creatinine was determined.
21
In respect of urea determination, 0.1 ml of serum sample was added into a universal bottle containing 19.9 ml of distilled water and the suspension was shaken very well. Then, 1 ml of the suspension was transferred into a test tube and 1 ml of color reagent was added followed by 1 ml of acid reagent. The mixture was heated in boiling water for 20 min. It was then cooled and the absorbance was read at 520 nm against blank.
22
Serum levels of sodium (Na+), potassium (K+), chloride (Cl−), Ca2+, bicarbonate
Hematological assessment
Full blood count analysis of blood samples was carried out using the SYSMEX KX-21N auto analyzer (Sysmex Corporation, Kobe, Japan).
Histopathology of the kidneys
The remaining pair of kidneys harvested is gently but briskly rinsed in 0.9% normal saline and fixed in 10% formal saline. The method described by Baker and Silverton 23 was employed for processing the kidney histology. The tissues were fixed in 10% formalin for a minimum of 10 days to prevent microbial invasion and keep the tissue in original form. After fixation, the tissues were dehydrated by passage through different ascending concentrations of absolute alcohol (50, 70, 90, and 95%) for 30 min each time. The tissues were left in xylene overnight to allow for wax impregnation. After impregnation, the tissues were embedded in melted wax and allowed to harden. Sections of the tissues were cut from the blocks using a microtome. The cut sections were floated on warm water bath to smoothen out creases. The sections were then lifted onto a clean glass microscope slide and allowed to dry on a hot plate with temperature set below melting point. The tissues were stained with hematoxylin and eosin technique. The stained slides were mounted and examined using a photomicroscope (Model N-400ME, CEL-TECH Diagnostics, Hamburg, Germany). Photographs were taken and examined.
Statistical analysis
Statistical analysis was performed using GraphPad Prism (Graph Pad Software; version 5.0, Graph Pad Software Inc., La Jolla, California, USA.). Data were expressed as mean ± SD for body weight and relative kidney weight and as mean ± SEM for biochemical and hematological assays. Data were analyzed using one-way analysis of variance for comparison between the control and treated groups, and post hoc test was conducted using Newman–Keuls–Student’s t test. Level of statistical significance was determined at p < 0.05.
Results
Effect of nifedipine, verapamil, diltiazem, and prednisolone on the average body and kidney weight of rats
As shown in Table 1, significant increases (p < 0.05) were observed in the body weight of rats across the treatment groups except the prednisolone-treated set of rats. However, there was no significant change in weight of kidneys in comparison of all other treatment groups with the normal and gentamicin control groups.
Effect of nifedipine, verapamil, diltiazem, and prednisolone on body and kidney weights of gentamicin-treated rats.

I: normal saline + normal saline; II: normal saline + gentamicin; III: ascorbic acid + gentamicin; IV: nifedipine + gentamicin; V: verapamil + gentamicin; VI: diltiazem + gentamicin; VII: prednisolone + gentamicin.
aRepresents significant increase at p < 0.05 between each group weight before and after the start of treatment with test drugs.
In the CCl4 model, significant increases (p < 0.05) were observed in the body weight of rats in the normal control, CCl4 control, diltiazem- and prednisolone-treated groups only. In respect of weight of kidneys, no significant changes were observed in comparison of all other treatment groups with the normal and CCl4 control groups (Table 2).
Effect of nifedipine, verapamil, diltiazem and prednisolone on body and kidney weight of CCl4-treated rats.

CCl4: carbon tetrachloride; I: normal saline + normal saline; II: normal saline + CCl4; III: ascorbic acid + CCl4; IV: nifedipine + CCl4; V: verapamil + CCl4; VI: diltiazem + CCl4; VII: prednisolone + CCl4.
aRepresents significant increase at p < 0.05 between each group weight before and after the start of treatment with test drugs.
Effect of nifedipine, verapamil, diltiazem, and prednisolone on the serum electrolytes, urea, and creatinine
Repeated single daily i.p. injection with 40 mg kg−1 of gentamicin was associated with significant (p < 0.05) increases in the serum level of Na+, Cl−,
Effect of nifedipine, verapamil, diltiazem, and prednisolone on serum electrolytes, urea, and creatinine in gentamicin-treated rats.

I: normal saline + normal saline; II: normal saline + gentamicin; III: ascorbic acid + gentamicin; IV: nifedipine + gentamicin; V: verapamil + gentamicin; VI: diltiazem + gentamicin; VII: prednisolone + gentamicin.
aRepresents significant increases at p < 0.05 when compared with group I.
bRepresents significant decreases at p < 0.05 when compared with group II.
cRepresents significant decreases at p < 0.05 when compared with group I.
dRepresents significant increases at p < 0.05 when compared with group II.
Single i.p. injection with 1.5 ml kg−1 of CCl4 was associated with significant (p < 0.05) increases in the serum levels of Na+, Cl−,
Effect of nifedipine, verapamil, diltiazem, and prednisolone on serum electrolytes, urea, and creatinine in CCl4-treated rats.
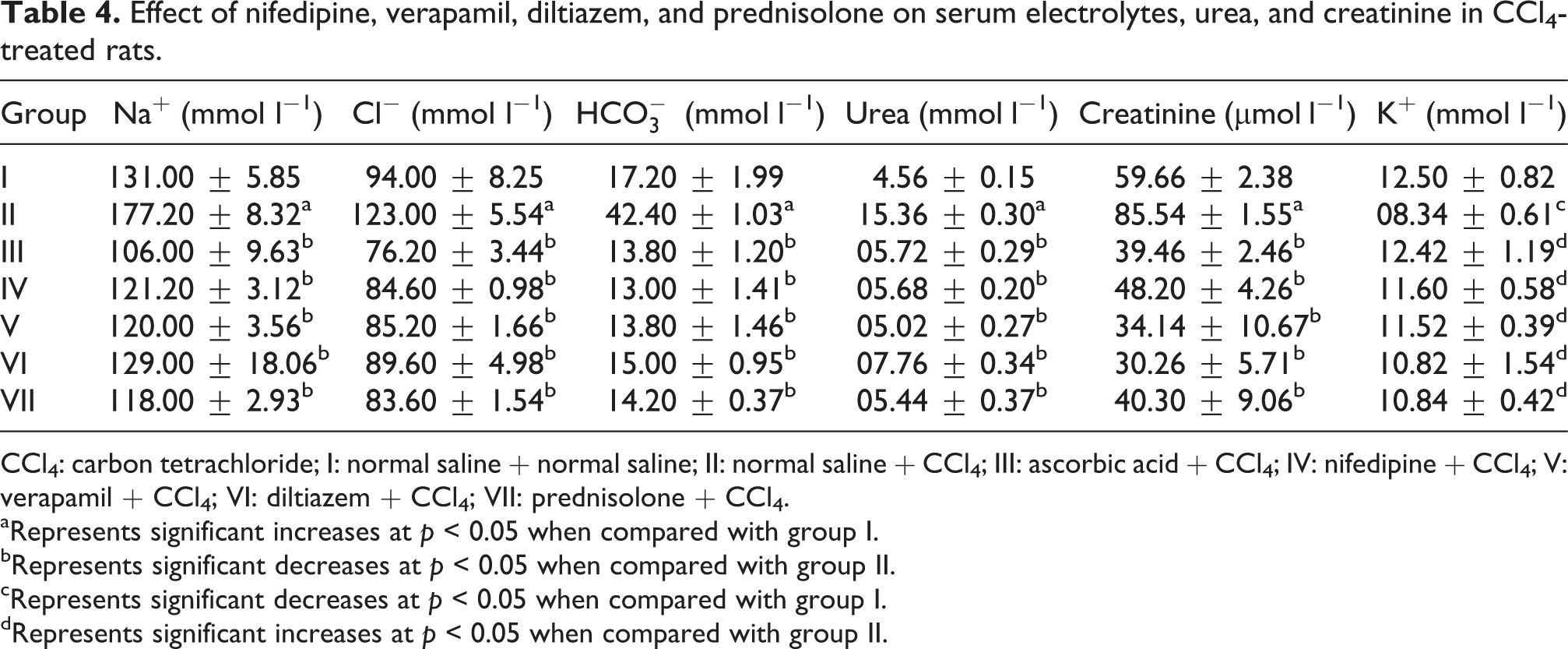
CCl4: carbon tetrachloride; I: normal saline + normal saline; II: normal saline + CCl4; III: ascorbic acid + CCl4; IV: nifedipine + CCl4; V: verapamil + CCl4; VI: diltiazem + CCl4; VII: prednisolone + CCl4.
aRepresents significant increases at p < 0.05 when compared with group I.
bRepresents significant decreases at p < 0.05 when compared with group II.
cRepresents significant decreases at p < 0.05 when compared with group I.
dRepresents significant increases at p < 0.05 when compared with group II.
Effect of nifedipine, verapamil, diltiazem, and prednisolone on the renal tissue antioxidant indices
Repeated single daily injection of 40 mg kg−1 of gentamicin to treated rats was associated with significant (p < 0.05) decreases in the tissue concentration of GSH and activities of SOD and CAT when compared with untreated control. However, SOD and CAT activities were significantly (p < 0.05) restored in kidneys of rats pretreated with ascorbic acid, nifedipine, verapamil, diltiazem, and prednisolone when compared with gentamicin-only-treated rat kidneys, while GSH levels were significantly (p < 0.05) increased in diltiazem- and prednisolone-treated kidneys when compared with gentamicin-only-treated rat kidneys (Table 5).
Effect of nifedipine, verapamil, diltiazem, and prednisolone on renal tissue antioxidant indices in gentamicin-treated rats.

GSH: glutathione; SOD: superoxide dismutase; CAT: catalase; I: normal saline + normal saline; II: normal saline + gentamicin; III: ascorbic acid + gentamicin; IV: nifedipine + gentamicin; V: verapamil + gentamicin; VI: diltiazem + gentamicin; VII: prednisolone + gentamicin.
aRepresents significant decreases at p < 0.05 when compared with group I.
bRepresents significant increases at p < 0.05 when compared with group II.
Single i.p. injection of 1.5 ml kg−1 of CCl4 to treated rats was associated with significant (p < 0.05) decreases in the tissue concentration of GSH and activities of SOD and CAT when compared with untreated control. However, GSH concentration and SOD and CAT activities were significantly (p < 0.05) restored in kidneys of rats pretreated with ascorbic acid, nifedipine, verapamil, diltiazem, and prednisolone with diltiazem producing the most significant restorative effect when compared with CCl4-only-treated rat kidneys (Table 6).
Effect of nifedipine, verapamil, diltiazem, and prednisolone on renal tissue antioxidant indices in CCl4-treated rats.

GSH: glutathione; SOD: superoxide dismutase; CAT: catalase; CCl4: carbon tetrachloride; I: normal saline + normal saline; II: normal saline + CCl4; III: ascorbic acid + CCl4; IV: nifedipine + CCl4; V: verapamil + CCl4; VI: diltiazem + CCl4; VII: prednisolone + CCl4.
aRepresents significant decreases and increases at p < 0.05 when compared with group I.
bRepresents significant increases and decreases at p < 0.05 when compared with group II.
Effect of nifedipine, verapamil, diltiazem, and prednisolone on the full blood count
Repeated i.p. injection of gentamicin at a single daily dose of 40 mg kg−1 body weight of rats for 14 days was associated with significant (p < 0.05) decreases in hemoglobin (Hb), packed cell volume (PCV), and red blood cell (RBC), while causing significant (p < 0.05) increases in the mean corpuscular volume (MCV) and mean corpuscular Hb (MCH) when compared with gentamicin-only-treated values (group II). However, this repeated treatment caused nonsignificant (p > 0.05) increases in total white blood cell count (TWBC) and mixed (monocytes, eosinophils, and basophils) percentage (%MXD) and nonsignificant (p > 0.05) decreases in platelet (PLT), lymphocyte percentage (%LYM), and neutrophils percentage (%NEU) when compared with gentamicin-only-treated (group II) values. Pretreatments with ascorbic acid, nifedipine, verapamil, diltiazem, and prednisolone significantly (p < 0.05) reversed the deleterious effect of gentamicin treatment on the Hb, PCV, and RBC returning their values to those of untreated control values (group I). However, pretreatment with the aforementioned drugs significantly (p < 0.05) decreased the MCV and MCH values when compared with gentamicin-only-treated (group II) values. Neither gentamicin treatment nor oral pretreatment with ascorbic acid, nifedipine, verapamil, diltiazem, and prednisolone produced significant (p > 0.05) alterations in the TWBC, %LYM, %NEU, and %MXD values (Table 7).
Effect of nifedipine, verapamil, diltiazem, and prednisolone on hematological parameters in gentamicin-treated rats.

Hb: hemoglobin; PCV: packed cell volume; RBC: red blood cell; MCV: mean corpuscular volume; PLT: platelet; MCH: mean corpuscular hemoglobin; TWBC: total white blood cell count; LYM: lumphocyte; NEU: neutrophils; MXD: mixed (monocytes, eosinophils, and basophils); I: normal saline + normal saline; II: normal saline + gentamicin; III: ascorbic acid + gentamicin; IV: nifedipine + gentamicin; V: verapamil + gentamicin; VI: diltiazem + gentamicin; VII: prednisolone + gentamicin.
aRepresents significant increases at p < 0.05 when compared with group I.
bRepresents significant decreases at p < 0.05 when compared with group II.
cRepresents significant decreases at p < 0.05 when compared with group I.
dRepresents significant increases at p < 0.05 when compared with group II.
Effect of nifedipine, verapamil, diltiazem, and prednisolone on the renal histopathology
Figures 1 and 2 depict the renal histopathological changes associated with repeated high dose of gentamicin treatment and the effect of oral pretreatment with nifedipine, verapamil, diltiazem, and prednisolone over 14 days. Repeated i.p. treatment with high-dose gentamicin was associated with patchy glomerular atrophy and mild tubulonephritis (Figure 1(b)) when compared with the control kidney (Figure 1(a)). However, in kidneys of rats pretreated with ascorbic acid, nifedipine, verapamil, diltiazem, and prednisolone, there were no remarkable histological changes in the renal architecture except for scanty tubular necrosis (Figures 1(c) and (d) and 2(b) to (d)).

Representative sections: (a) normal kidney showing normal tubular brush borders and intact glomerulus surrounding Bowman’s capsule; (b) 40 mg kg−1 day−1 of intraperitoneal gentamicin-treated rat kidney showing glomerular atrophy and significant patchy tubular necrosis indicating mild tubulonephritis; (c) 10 mg kg−1 day−1 of oral ascorbic acid-treated rat kidney showing an intact glomerulus and tubules; (d) 0.86 mg kg−1 day−1 of oral nifedipine-treated rat kidney showing an intact glomerulus and mild patchy tubular necrosis (×400 magnification, hematoxylin and eosin staining).

Representative sections: (a) normal kidney showing normal tubular brush borders and intact glomerulus surrounding Bowman’s capsule; (b) 4.3 mg kg−1 day−1 of oral verapamil-treated rat kidney showing an intact glomerulus and mild few early tubular necrosis that may not be significant; (c) 3.43 mg kg−1 day−1 of oral diltiazem-treated rat kidney showing an intact and normal glomerulus and significant patchy necrosis of the proximal tubules; and (d) 0.57 mg kg−1 day−1 of oral prednisolone-treated rat kidney showing an intact and normal glomerulus and very scanty necrosis of the proximal tubules (×400 magnification, hematoxylin and eosin staining).
Induction of drug-induced nephrotoxicity with 1.5 ml kg−1 of 20% CCl4 in olive oil manifested as hemorrhagic areas with congested vascular channels in renal stroma and glomeruli (Figure 3(b)) when compared with normal renal architecture found in untreated control kidney (Figure 3(a)). Oral pretreatments with ascorbic acid, nifedipine, and verapamil manifested as improvements of some of the histopathological lesions induced by subsequent CCl4 treatment (Figures 3(c) and (d) and 4(b)). However, pretreatments with diltiazem and prednisolone did not offer protection against the deleterious effect of CCl4 as there were both renal cortical and medullary hemorrhages in the treated kidneys (Figure 4(c) and (d)).
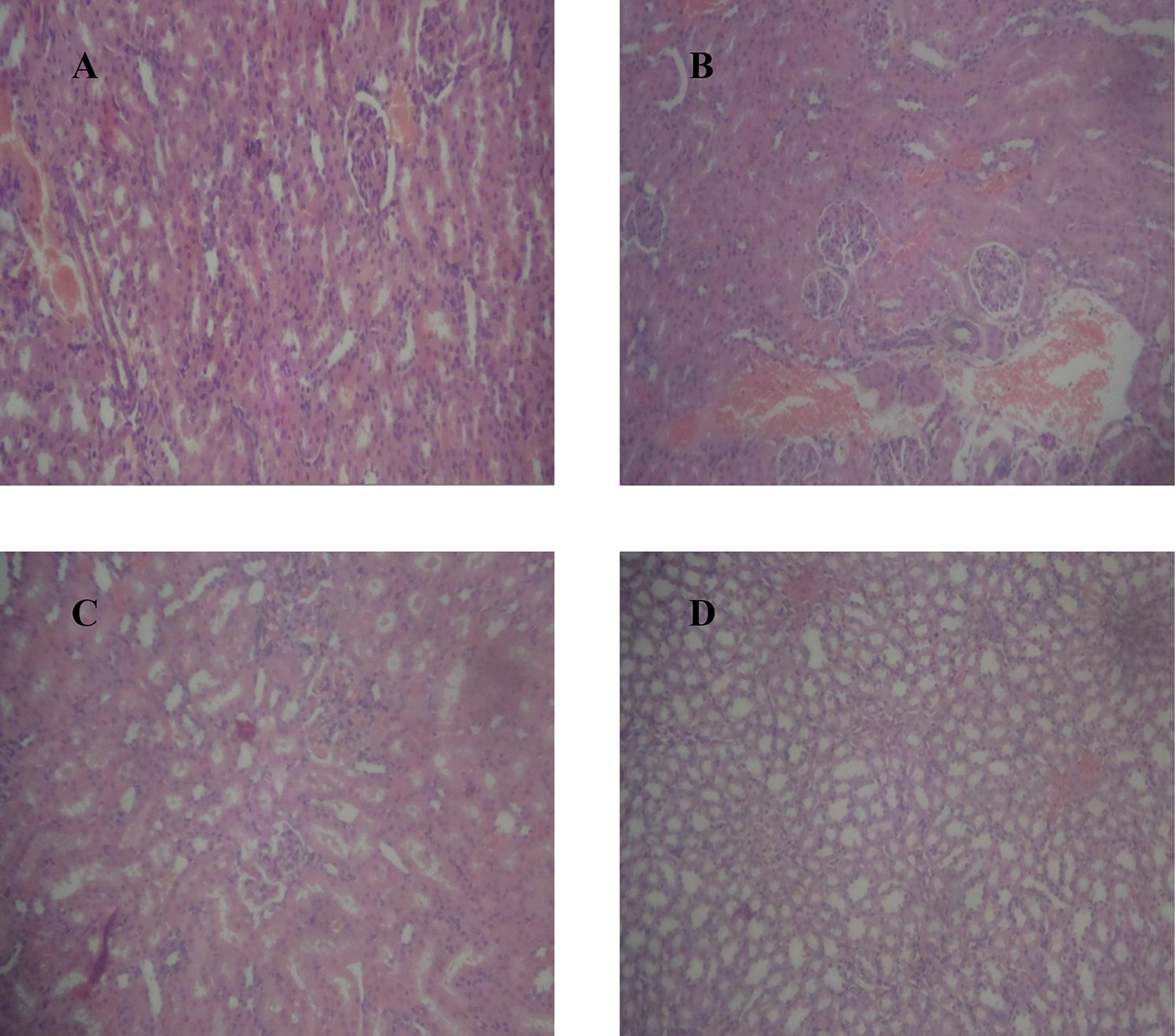
Representative sections: (a) normal rat kidney with regular glomerular and tubular architecture; (b) 1.5 ml kg−1 of 20% CCl4 in olive oil-treated rat kidney showing diffused cortical and medullary hemorrhages with congested vascular channels and tubular necrosis; (c) ascorbic acid-pretreated, CCl4-treated rat kidney showing areas of focal hemorrhages and mild glomerular capillary congestion; and (d) nifedipine-pretreated, CCl4-treated rat kidney showing scattered hemorrhages at the medulla and sparing the cortical areas (×100 magnification, hematoxylin and eosin staining). CCl4: carbon tetrachloride.

Representative sections: (a) normal rat kidney with regular glomerular and tubular architecture; (b) verapamil-pretreated, CCl4-treated rat kidney showing few areas of glomerular hemorrhages; (c) diltiazem-pretreated, CCl4-treated rat kidney showing areas of cortical and medullary hemorrhages; and (d) prednisolone-pretreated, CCl4-treated rat kidney showing diffused areas of cortical and medullary hemorrhages (×100 magnification, hematoxylin and eosin staining). CCl4: carbon tetrachloride.
Discussion
Gentamicin is a prototype of a class of antimicrobials known as aminoglycosides, and it is extensively used in the clinical management of both gram positive and gram negative bacterial infections, with preference for the latter. 24 However, its use either at high dose or prolonged use, even at a clinical dose, has been reported to be associated with organ toxicities such as ototoxicity and nephrotoxicity. 25 –27 Thus, the use of gentamicin as a standard nephrotoxicant in nephrotoxicity studies is well established. 16,28 Also, therapeutic doses of gentamicin and other aminoglycoside antibiotics are known as the most common causes of acute renal failure, 29 possibly due to increased renal uptake of the antibiotic, mainly by the proximal tubules. The effect of gentamicin on biological membranes appears to be important in its toxicity. It has been proposed that the accumulation of aminoglycosides in proximal tubular epithelial cells leads to membrane structural disturbance and cell death by reactive oxygen species (ROS) involvement. 30 ROS produce cellular injury and necrosis via several mechanisms including peroxidation of membrane lipids, protein denaturation, and DNA damage. 29 Hydroxyl radical scavengers such as dimethyl thiourea, SOD, and dietary antioxidants (vitamin E, selenium, vitamin C, taurine, and the carotenoids (β-carotene, lutein, and lycopene)) can decrease the gentamicin-induced reduction in the glomerular filtration rate and the severity of the tubular damage. 31 –34
Nephrotoxicity has been related to a gentamicin trough concentration above 2 μg ml−1, Ca2+ deficiency, Ca2+ channel activation, prostaglandins pathways, free radical generation, pyridoxal phosphate deficiency, and ascorbic acid depletion.
26
Thus, any strategy or intervention that ameliorates these above-mentioned pathways and deficiencies may ameliorate nephrotoxicity. In this present study, nephrotoxicity was successfully induced through repeated daily i.p. injection of 40 mg kg−1 of gentamicin for 14 days, and this was characterized by hypernatremia, hypokalemia, hypocalcemia, hypophosphatemia, and hyperchloremic alkalosis,
35,36
hyperuremia, and hypercreatininemia, which appears to be in agreement with those reported previously.
37
–40
Increased concentration of serum urea and creatinine are considered reliable parameters for assessing the renal functions, particularly, in drug-induced nephrotoxicity in animals and human.
41
BUN is found in liver protein that is derived from diet or tissue sources and is normally excreted in the urine. In renal disease, the serum accumulates urea because the rate of serum urea production exceeds the rate of clearance.
42
Creatinine is mostly derived from endogenous sources by tissue creatinine breakdown.
43
Thus, elevation of urea and creatinine levels the serum can be taken as the index of nephrotoxicity.
41,43,44
In this study, pretreatments with ascorbic acid, nifedipine, verapamil, diltiazem, and prednisolone significantly (p < 0.05) improved the serum levels of Na+, Cl−, K+,
Cuzzocrea et al. 29 investigated the potential role of the superoxide anion in gentamicin-induced renal toxicity by M40403, a low-molecular-weight synthetic manganese containing a SOD-mimetic that selectively removes superoxide. The authors observed that gentamicin treatment was associated with a significant (p < 0.05) increase in kidney myeloperoxidase activity and lipid peroxidation in gentamicin-treated rats suggesting a pivotal role of oxidative stress in the etiopathogenesis of gentamicin-induced nephrotoxicity. During kidney injury, superoxide radicals are generated at the site of damage and modulate SOD and CAT resulting in the loss of activity and accumulation of superoxide radical that damages the kidney. SOD and CAT are the most important enzymes involved in ameliorating the effects of oxygen metabolism. 45,46 The generation of oxidative ROS has been proposed as a mechanism by which many chemicals can induce nephrotoxicity. 47 Other in vivo antioxidants involved in amelioration of the damaging effects of ROS generation include reduced GSH and GSH peroxidase. In this study, repeated single daily injection of 40 mg kg−1 of gentamicin to treated Wistar rats for 14 days was associated with significant decreases in the renal tissue concentration of GSH and activities of SOD and CAT when compared with untreated control values. With oral pretreatments with nifedipine, verapamil, diltiazem, and prednisolone also for 14 days, SOD and CAT activities were significantly restored to about the values recorded for the standard nephroprotective agent (ascorbic acid) used in this study which mediates its protective effect via antioxidant mechanisms. This strongly suggests that nifedipine, verapamil, diltiazem, and prednisolone were able to significantly prevent decrease in tissue concentration of GSH and activities of SOD and CAT suggesting that protective mechanism of these drugs could possibly be mediated via antioxidant mechanism. 48 –50 It is important to note that the prooxidant effect of CCBs has been reported on the reproductive system. 51 This suggests that depending on the target cells, CCBs may produce anti-oxidative effect 49,50 as the results from this study suggests or a prooxidant effect as on the reproductive system. 51
Various in vitro and in vivo studies have shown CCl4 to enhance lipid peroxidation, reduce renal microsomal nicotinamide adenine dinucleotide phosphate cytochrome P450, and renal reduced/oxidized glutathione ratio (GSH/GSSG) in kidney cortex as well as renal microsomes and mitochondria.
52,53
CCl4 is such a widely used environmental toxicant, which is employed to experimentally induce animal models of acute nephrotoxicity and hepatotoxicity.
53
CCl4 is metabolized by cytochrome P450 2E1 to trichloromethyl radical
In the present study, single i.p. injection of 1.5 ml kg−1 of CCl4 to treated Wistar rats was associated with significant decreases in the tissue activities of SOD and CAT and concentration of GSH. These results are in complete agreement with observations previously reported by Adewole et al. 53 and Bandhopadhy et al. 55 Thus, the decreased activity of SOD in kidney in CCl4-treated rats may be due to the enhanced lipid peroxidation or inactivation of the antioxidative enzymes. This would cause an increased accumulation of superoxide radicals, which could further stimulate lipid peroxidation. However, administration of ascorbic acid, nifedipine, verapamil, diltiazem, and prednisolone prior to CCl4 intoxication protected the antioxidant machineries of the kidneys as revealed from increased levels of SOD, CAT, and GSH content with nifedipine producing the most significant restorative effect of all the CCBs tested.
The vital function that blood cells perform, together with the susceptibility of the highly proliferative tissue to intoxication by xenobiotics, makes the hematopoietic system unique as a target organ. 56 Certain drugs including alkylating cytotoxic agents could affect blood formation rate and normal range of hematological parameters. 56 In this study, repeated i.p. injection of gentamicin at a single daily dose of 40 mg kg−1 body weight of rats for 14 days was associated with significant decreases in Hb, PCV, and RBC, while causing significant increases in the MCV and MCH. Previous studies have shown gentamicin to induce anemia via vitamins B6, 37,57 C, and E deficiencies. 30,32,38 However, pretreatments with ascorbic acid, nifedipine, verapamil, diltiazem, and prednisolone significantly prevented the deleterious effect of gentamicin treatment on the Hb, PCV, and RBC. Also, the decrease in Hb, PCV, and RBC in the gentamicin-only-treated group may be due to the damage to the kidney cells that are involved in the process of erythropoiesis. 3 A reduction in the erythropoiesis process produces less erythropoietin, a polypeptide hormone produced by the macula densa cells of the kidneys, which stimulates the red bone marrow, thereby increasing the rate of RBC production. With more RBCs in circulation, the oxygen-carrying capacity of the blood is greater. 3 Lower RBC means lower Hb in the blood which could eventually lead to hypoxia and then death. All four drugs (nifedipine, verapamil, diltiazem, and prednisolone) were able to prevent the deleterious effect on RBC and Hb lowering associated with kidney damage.
Another interesting finding of this study is the histopathological lesions induced by gentamicin treatment on the treated kidneys. Repeated i.p. treatment with high-dose gentamicin was associated with patchy glomerular atrophy and mild tubulonephritis when compared with the control kidney. Again, rat kidneys pretreated with nifedipine, verapamil, diltiazem, and prednisolone showed no remarkable histological changes in the renal architecture except for scanty tubular necrosis. These findings corroborated the biochemical and hematological findings of this study.
In conclusion, both CCBs (nifedipine, verapamil, and diltiazem) and PLA2 inhibitor (prednisolone) showed significant nephroprotection against drug-/chemical-induced renal toxicities, which was mediated via antioxidant and anti-lipoperoxidation mechanisms. Thus, this study establishes the chemopreventive potentials of CCBs and prednisolone against drug-/chemical-induced nephrotoxicities. However, further research would be required in understanding the molecular mechanisms involved in the protective effect of these CCBs and prednisolone against drug-induced nephrotoxicity.
Footnotes
Acknowledgments
The authors thank Mr Sunday Adenekan, Department of Biochemistry, Faculty of Basic Medical Sciences, College of Medicine, University of Lagos, Lagos, Nigeria, for the technical assistance rendered in respect of tissue antioxidant indices assays.
Conflict of interest
The authors declared no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
