Abstract
The aim of this study was to evaluate the acute effect of high-dose acetylsalicylic acid (ASA) on kidney and testis, and the potential protective and therapeutic effects of melatonin on ASA-related pathology. A total of 40 rats were randomly divided into the following 5 groups (n = 8): group 1: control, not given any drug; group 2: only 200 mg/kg ASA was given; group 3: 5 mg/kg melatonin was given 45 min before administering 200 mg/kg ASA; group 4: 5 mg/kg melatonin was given 45 min after administering 200 mg/kg ASA; and group 5: only 5 mg/kg melatonin was given. The histopathological changes and the biochemical findings; such as malondialdehyde (MDA), superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPX), reduced glutathione (GSH), and blood urea nitrogen (BUN) as well as serum creatinine (Cr) levels were evaluated. ASA significantly increased MDA levels in both kidney and testis, whereas it significantly decreased the values of SOD, CAT, GPX, and GSH in kidney and CAT levels in testis. Melatonin significantly decreased MDA levels in kidney and ameliorated it in testis, whereas it caused elevation in the levels of antioxidants. BUN and Cr levels were higher after ASA, whereas these levels were diminished after melatonin administration. The improvement obtained by melatonin on ASA-induced histological alterations was more prominent when it was used after ASA in kidney and before ASA in testis. In this study, we demonstrated the beneficial effect of melatonin on high-dose ASA-related pathology of kidney and testis for the first time.
Introduction
Acetylsalicylic acid (ASA) is a readily available, non-steroidal anti-inflammatory drug first synthesized in 1897 and has been a worldwide drug. ASA shows the anti-inflammatory, antipyretic, analgesic, and anti-thrombotic effects via blocking the pathway of prostaglandins synthesis from arachidonic acid (AA) by inhibiting cyclooxygenase (COX). 1,2 In addition, several suggested mechanisms besides the COX inhibition have been reported for its beneficial effects. 3 On the other hand, ASA has also been reported to increase oxidative stress and to cause toxicity and apoptosis in some studies. 3–5 The generation of reactive oxygen species (ROS), depletion of cellular scavenger of ROS, AA metabolism, mitochondrial dysfunction, and apoptosis have been shown as the underlying reasons for ASA-induced nephrotoxicity and gonadal dysfunction. 6–9 Early studies clearly demonstrated that ASA even at low doses can cause lipid peroxidation and impairment in the defense mechanisms against oxidative stress. 10,11
Melatonin (N-acetyl-5-methoxytryptamine), which is referred as hormone and secreted from the mammalian pineal body, has immunomodulatory, anti-inflammatory, and antitumor effects. 12,13 It is the major element of the defense system against both ROS and reactive nitrogen species (RNS). 14–16 Melatonin protects cell via supporting the endogenous antioxidant enzymes such as superoxide dismutase (SOD) and glutathione peroxidase (GPX). 17,18 Moreover, it stimulates the production of reduced glutathione (GSH), which is another endogenous antioxidant and substrate for GPX, and prevents inactivation of catalase (CAT). 13,19,20 All these products have significant roles in the defense and the maintenance mechanisms of the cell viability. In addition, the metabolites of melatonin are also very efficient scavengers similar to main molecule in oxidative stress and inflammatory reactions. 21,22
The toxic effects of high-dose ASA taken during therapy, inadvertently or in suicide attempt are well-known. 5,9–11 The current study was designed to evaluate the acute effect of high-dose ASA on kidney and testicular tissues, and to detect the potential protective and therapeutic effects of melatonin on ASA-related pathology. To demonstrate these effects, we investigated the histopathological changes of kidney and testis, and biochemical analyses (including renal and testicular tissue malondialdehyde (MDA), SOD, CAT, GPX, GSH, and blood urea nitrogen (BUN) and serum creatinine (Cr) levels).
Materials and methods
This animal experimental study was designed according to ARRIVE guidelines. 23
Animals and experimental protocol
This study was approved by the Ethical Committee on Animal Research of Inonu University. The experimental procedures were carried out pursuant to the Guidelines for Animal Research from the National Institutes of Health publication. A total of 40 post-pubertal (10–12 weeks) Wistar Albino male rats, weighing 250–300 g, were obtained from Inonu University Laboratory Animals Research Center and maintained in a humidity of 60 ± 5%, temperature of 21 ± 2°C, and at 12:12-h light and dark cycle controlled room on a standard commercial pellet diet and water ad libitum.
The rats were randomly divided into the following 5 groups (n = 8): group 1: control, not given any drug; group 2: only 200 mg/kg ASA was given intraperitoneally (i.p.); group 3: 5 mg/kg melatonin was given i.p. 45 min before administering 200 mg/kg ASA i.p.; group 4: 5 mg/kg melatonin was given i.p. 45 min after administering 200 mg/kg ASA i.p.; and group 5: only 5 mg/kg melatonin was given i.p. All drugs were administered as a single dose. The dosage of ASA and melatonin were chosen according to previous dose-response studies, 24–26 and melatonin and ASA were purchased from Sigma Chemical Co. (St Louis, Missouri, USA).
Surgical procedure
Four hours after ASA administration, the rats were anesthetized with ketamine (70 mg/kg) and xylazine (8 mg/kg). Each animal’s abdominal area was shaved with electric clippers and then prepared with povidone–iodine (Poviiodeks®, Kim-Pa Corporation, Istanbul, Turkey) and aseptic conditions were provided in a local sterile environment. Following preparation of the surgical area, midline laparotomies were carried out and, following of the extraction of blood samples, left nephrectomies and left orchidectomies were immediately performed on all animals in the groups. The kidneys were quickly removed, decapsulated, and divided equally into two longitudinal sections. One of these sections was placed in formaldehyde solution for routine histopathological examination by light microscopy. The other half of the kidney was placed in liquid nitrogen and stored at −70°C until assayed for MDA, SOD, CAT, GPX, and GSH. The same procedure was also carried out for the testes. The blood samples were quickly centrifuged to evaluate the serum levels of BUN and Cr using an Olympus Autoanalyzer (Olympus Instruments, Tokyo, Japan).
Biochemical analyses
Determination of MDA
The MDA of the homogenates was defined spectrophotometrically by measuring the existence of thiobarbituric acid reactive substances (TBARS). 27 Phosphoric acid (1%) of 3 ml and 1 ml 0.6% thiobarbituric acid solution were added to 0.5 ml of homogenate pipetted into a tube. The mixture was heated in boiling water for 45 min. After the mixture was cooled, the colored part was extracted into 4 ml of n-butanol. The absorbance was measured by a spectrophotometer (UV-1601; Shimadzu, Kyoto, Japan) at 532 and 520 nm. The amount of lipid peroxides was calculated as TBARS of lipid peroxidation. The results were given in nanomoles per gram of tissue, according to a prepared standard graph.
Determination of SOD activity
Total SOD activity was defined according to the method of Sun et al. 28 The principle of the method is the prevention of nitrobluetetrazolium (NBT) reduction by the xanthine–xanthine oxidase system as a superoxide generator. One unit of SOD was defined as the amount of enzyme to cause 50% inhibition in the NBT reduction rate. The results were given in Units per gram of protein.
Determination of CAT activity
CAT activity was defined according to a method developed by Aebi. 29 The principle of the assay is the determination of the rate constant (k, per second) or the hydrogen peroxide (H2O2) decomposition rate at 240 nm. CAT activities were given as k per gram of protein.
Determination of GPX activity
GPX activity was measured by the method of Paglia and Valentine. 30 An enzymatic reaction in a tube containing NADPH, GSH, sodium azide, and glutathione reductase (GR) was initiated by adding H2O2; the change in absorbance at 340 nm was observed using a spectrophotometer. The results were given as Units per milligram of protein.
Determination of GSH
The concentration of GSH in homogenate was determined spectrophotometrically according to the method of Ellman. 31 Each homogenate sample was mixed with 10 mM 5,5′-dithiobis(2-nitrobenzoic acid) in 100 mM potassium phosphate buffer (pH 7.5) and 17.5 M ethylenediaminetetraacetic acid. The reaction was started by the addition of 0.5 units of GR and 0.4 mM NADPH. The absorbance was measured at 410 nm after 5 min. The concentration of GSH was calculated against a standard curve. The results were given in micromoles per gram of tissue.
Histological analyses and semi-quantitative evaluation
The kidney and testicular tissues were fixed in 10% formalin, dehydrated, and then embedded in paraffin. Sections of 5 μm thickness were cut and stained with hematoxylin and eosin. The kidney tissue sections were evaluated for the presence of the tubular cell injury, desquamation, interstitial congestion, glomerular shrinkage and enlargement of the Bowman’s space. The histological slides of the kidney tissue were evaluated for semi-quantitative analysis according to the presence of tubular epithelial cell desquamation, glomerular capillary congestion, tubulointerstitial inflammation, and congestion. The kidney changes were graded as follows: 0, normal; I, the damage involving <25% of the total area; II, the damage involving 25–50% of the total area; III, the damage involving 50–75% of the total area; and IV, the damage involving >75% of the total area. The histological changes were measured in 10 different fields for each specimen (×20 objective).
The testicular tissue sections were evaluated according to Sayım’s 32 method with minor revision. Histological changes were detected by counting 100 tubules in slides and classified as normal, sloughing (immature spermatogenic cells in the lumen), atrophy (very few or no germ cells), spermatogenic arrest (interrupted spermatogenic cells at various stage of mitotic phase), disintegration in the spermatogenic layer (disrupted cell association), and multinucleated giant cell formation (the cells with more than one nucleus) according to the degree of damage in seminiferous tubules. The sections were examined with a Leica DFC 280 light microscope by an experienced observer unaware of the animal treatment groups.
Statistical analysis
For detecting even minor effects, the required sample sizes used in this experiment were identified using statistical power analysis. The sample sizes necessary for a power of 0.80 were estimated using NCSS software. Data were analyzed using the Statistical Package for the Social Sciences (SPSS) software program for Windows, version 20.0 (SPSS Inc., Chicago, Illinois, USA). The normality of the distribution was confirmed using the Kolmogorov–Smirnov test. According to the results obtained from the normality test, one-way analysis of variance (ANOVA) and the Kruskal–Wallis H test were used for the statistical analysis, as appropriate. Multiple comparisons were carried out by Tamhane’s test after the ANOVA test. The results are expressed as mean ± SD. After a significant Kruskal–Wallis H test, a Conover test was also carried out for giant cell. The results were considered statistically significant at p < 0.05. The values were given as median (min–max).
Results
Body, kidney, and testis weights
No animals died during or after the injections or surgical procedures. There were no significant differences between either the body weights before and after the experiments or kidney and testis weights among the groups (data not shown).
Effect of melatonin on serum parameters
As shown in Table 1, the serum levels of BUN and Cr were higher in ASA alone group (27.9 ± 6.5 and 0.41 ± 0.07 mg/dL, respectively) when compared with control group (22.7 ± 2.6 and 0.35 ± 0.04 mg/dL, respectively); however, these elevations were not statistically significant (p > 0.05). Melatonin, especially in group 4, reduced BUN and Cr levels almost to control levels; however, these reductions were not meaningful (p > 0.05).
The serum levels of BUN and Cr.a

BUN: blood urea nitrogen; Cr: creatinine; ASA: acetylsalicylic acid; MEL: melatonin.
a p < 0.05, statistically significant.
Effect of melatonin on ASA-induced changes in kidney and testicular tissue enzymes and lipid peroxides
As shown in Table 2, kidney tissue MDA levels were found to be significantly higher in ASA alone group (109.06 ± 19.02 vs. 73.42 ± 11.47 nmol/g tissue), whereas the values of SOD, CAT, GPX, and GSH were significantly lower when compared with control group (p < 0.05). On the other hand, melatonin caused a meaningful decrease in the level of MDA in both treatment groups (groups 3 and 4) when compared with the ASA alone group (71.22 ± 14.99 and 65.21 ± 10.39 vs. 109.06 ± 19.02 nmol/g tissue, respectively). Melatonin (groups 3 and 4) ameliorated the levels of SOD and GSH when compared with ASA alone group; moreover, it induced a significant increase in CAT levels when compared with ASA alone group (105.51 ± 12.47 and 114.86 ± 14.88 vs. 71.99 ± 15.52 k/g protein, respectively).
The levels of MDA, SOD, CAT, GPX, and GSH in the renal tissue.

MDA: malondialdehyde; SOD: superoxide dismutase; CAT: catalase; GPX: glutathione peroxidase GSH: reduced glutathione; ASA: acetylsalicylic acid; MEL: melatonin.
a p < 0.05 versus group 1.
b p < 0.05 versus group 2.
As shown in Table 3, MDA levels of testis tissue were significantly higher in ASA alone group (38.22 ± 3.30 vs. 28.04 ± 5.50 nmol/g tissue, respectively), whereas CAT levels were significantly lower when compared with the control group (13.78 ± 5.56 vs. 26.86 ± 8.02 k/g protein, respectively).Although melatonin administration did not cause a significant decrease in MDA levels, it ameliorated the MDA values. Melatonin, when used before ASA administration, significantly increased CAT levels when compared with ASA alone group (24.21 ± 6.33 vs. 13.78 ± 5.56 k/g protein, respectively). Moreover, melatonin induced an increase in SOD and GPX activities in groups 3 and 4 even higher than those in the control group (p < 0.05).
The levels of MDA, SOD, CAT, GPX, and GSH in the testicular tissue.

MDA: malondialdehyde; SOD: superoxide dismutase; CAT: catalase; GPX: glutathione peroxidase GSH: reduced glutathione; ASA: acetylsalicylic acid; MEL: melatonin.
a p < 0.05 versus group 1.
b p < 0.05 versus group 2.
c p < 0.05 versus group 3.
d p < 0.05 versus group 4.
Histological results
In the kidney, there were no or slight histological alterations, such as epithelial desquamation, in groups 1 and 5 (Figure 1(a) and (b)). Moreover, the glomeruli in these groups were observed as intact (Figure 1(c) and (d)). On the other hand, ASA alone group (group 2) had remarkable congestion of glomerular capillaries and tubular epithelial desquamation (Figure 2(a) and (b)). Furthermore, extensive tubulointerstitial damage, such as interstitial congestion and inflammatory cell infiltration, were obvious in this group (Figure 3(a) and (b)). Melatonin administration decreased the renal damage in groups 3 and 4. The slight tubular desquamation was present in these groups and the histological appearances of tubules were almost similar (Figure 4(a) and (b)). However, congestion of glomerular capillaries was more evident in group 3 than in group 4 (Figure 5(a) and (b)). Despite the congestion and the inflammatory cell infiltration in the interstitial area were not as extensive as in group 2, melatonin, given before and after ASA administrations, did not cause a significant reduction in terms of interstitial injury when compared with ASA alone group (Figure 6(a) and (b)). The results of semiquantitative histological examination were shown in Table 4.

Histological photographs of the renal tissues from the following groups: (a) group 1 and (b) group 5; normal tubules except slight tubular epithelial desquamation (H&E, X66). (c) group 1 and (d) group 5; intact glomeruli (stars) (H&E, ×132). H&E: hematoxylin and eosin.

Histological photographs of the renal tissue from group 2: (a) remarkable congestion of glomerular capillaries (arrows) (H&E, ×132); (b) marked and diffuse tubular epithelial desquamation (arrows) (H&E, ×66). H&E: hematoxylin and eosin.

Histological photographs of the renal tissue from group 2: (a) marked interstitial congestion (arrows) and (b) inflammatory cell infiltration in tubules (arrows) (H&E, ×33). H&E: hematoxylin and eosin.

Histological photographs of the renal tissues from the following groups: (a) group 3 and B group 4; slight epithelial desquamation into tubular lumens (arrows) (H&E, ×66). H&E: hematoxylin and eosin.

Histological photographs of the renal tissues from the following groups: (a) group 3 and (b) group 4; similar histological appearance except more obvious congestion of glomerular capillaries in group 3 (H&E, ×132). H&E: hematoxylin and eosin.
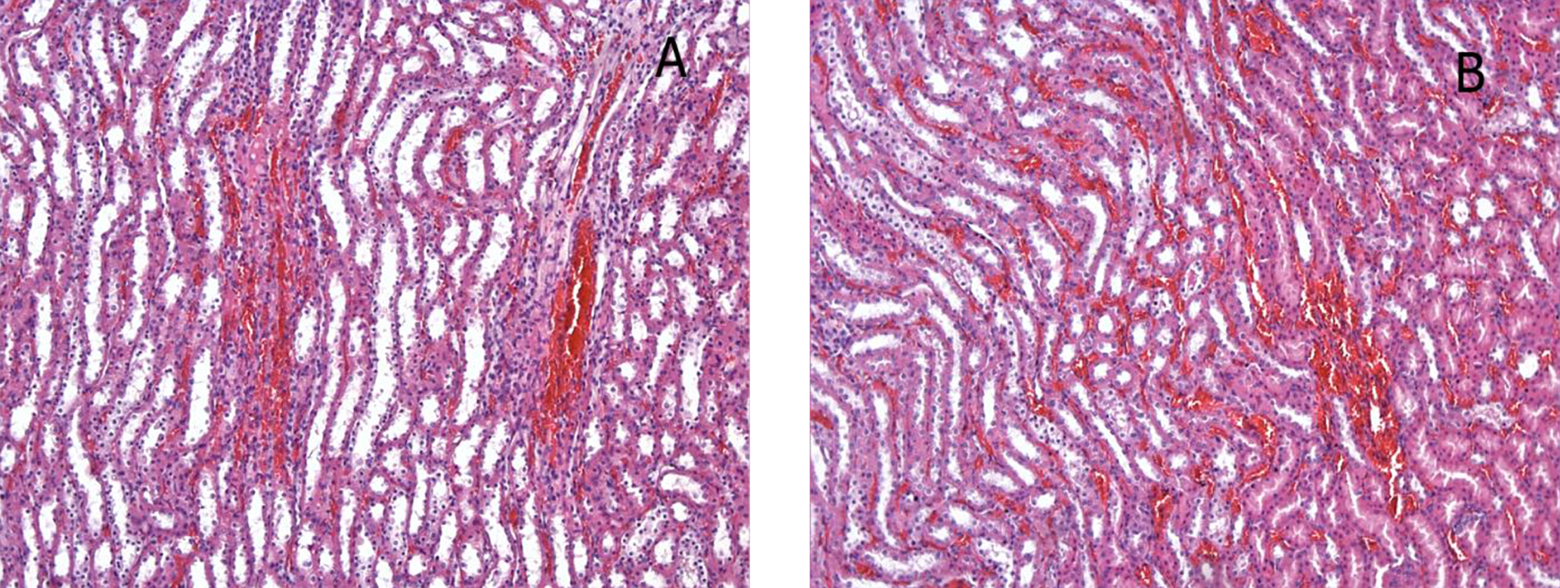
Histological photographs of the renal tissues from the following groups: (a) group 3 and (b) group 4; slight tubulointerstitial congestion (H&E, ×33). H&E: hematoxylin and eosin.
Comparison of severity of the renal damage among the groups.

ASA: acetylsalicylic acid; MEL: melatonin; GC: glomerular capillary congestion; TD: tubular epithelial cell desquamation, TIC: tubule–interstitial congestion; TII: tubule–interstitial inflammation.
Data are expressed as mean ± SD or median (min–max).
a p < 0.05 versus control group 1.
b p < 0.05 versus ASA group 2.
In the testis, the seminiferous tubules were intact and the germ cells were regular in concentric layers in groups 1 and 5. Therewithal, in these groups, the lumens of the seminiferous tubules had an abundant amount of spermatid and sperm (Figure 7(a) and (b)). However, there were obvious damages, such as sloughing, atrophy, spermatogenic arrest, disorganization of germinal cells, and multinucleated giant cell formation, in ASA alone group (group 2). The numerous round germ cells, which were detected from the spermatogenic layers, were observed in some of the seminiferous tubules in this group (Figure 8(a)). There was a significant decrease in the number of germ cells in several seminiferous tubules (Figure 8(b)). Also, some of the seminiferous tubules had many spermatogenic cells, which were arrested at various stages of the division (Figure 8(c)). Additionally, the multinucleated giant cells, which were formed as a result of fusion of spermatids, were observed in the epithelium of a few seminiferous tubules (Figure 8(d)). On the other hand, the number of intact seminiferous tubules significantly increased by melatonin administration in groups 3 and 4 when compared with ASA alone group (group 2; p < 0.0001). The histological appearance of group 3 was similar to group 4 in respect to sloughing (Figure 9(a) and (b)) and spermatogenic arrest (Figure 9(c) and (d)) in the tubules (p > 0.05). However, the number of atrophic tubules was significantly lower in group 3 than in group 4 (Figure 10(a) and (b); p < 0.0001). The multinucleated giant cells were not observed in these groups. The results of the histological alterations in the seminiferous tubules were classified and shown with percentages in Table 5.

Histological photographs of the testicular tissues from the following groups: (a) group 1 and (b) group 5; normal morphology of seminiferous tubules with regular germinal cells (H&E, ×66). H&E: hematoxylin and eosin.

Histological photographs of the testicular tissues from group 2; (a) remarkable sloughing of immature germ cells into the tubular lumen (arrows) (H&E, ×66), (b) clear loss of germ cells due to the tubular atrophy (H&E, ×66), (c) marked spermatogenic arrest (arrows) (H&E, ×132), (d) marked giant multinucleated cell formation, derivative of round spermatids (arrow) (H&E, ×132). H&E: hematoxylin and eosin.

Histological photographs of the testicular tissues from the following groups: (a) group 3 and (b) group 4; mild sloughing with nearly normal histological appearance of the tubules (H&E, ×66); (c) group 3 and (d) group 4; the rare arrested spermatogenic cells (arrows) at various stages of the division (H&E, ×132). H&E: hematoxylin and eosin.

Histological photographs of the testicular tissues from the following groups: (a) group 3 and (b) group 4; the rare atrophic tubules, loss or decrease in germ cells is more apparent in group 4 than group 3 (H&E, ×33). H&E: hematoxylin and eosin.
The histopathological classification of seminiferous tubules in testis.

ASA: Acetylsalicylic acid; MEL: Melatonin.
Data are expressed as mean ± SD or median (min–max).
a p < 0.0001 versus control (group 1).
b p < 0.0001 versus ASA (group 2).
c p < 0.0001 versus MEL + ASA (group 3).
Discussion
Oxidative stress, due to the overproduction of ROS and a decrease in the antioxidant level, causes membrane lipid peroxidation, oxidation of cell proteins, damage within the DNA helix and cell death, and also initiates an inflammatory response cascade. The dead cells, which release ROS, and pro- and anti-inflammatory cytokines, and endothelial and macrophage-like cells activated by ionic reactions, can lead to this inflammatory response that can last a long time and causes irreversible tissue damage. 33,34
ASA, a commonly used pharmaceutical agent, even at therapeutic doses can induce oxidative stress, decreases the levels of SOD, GPX, and GSH, and increases ROS, which occur together with mitochondrial dysfunction. 35,36 In the current study, we demonstrated that ASA induced an increase in the levels of MDA, which is an important sign of oxidative stress and increases due to lipid peroxidation, in the renal tissue. In a similar study, it was shown that ASA caused an elevation in MDA levels even in non-toxic lower doses. 11 In another study related to gastric injury, ASA induced two-fold increase in gastric MDA levels. 37 In this sense, melatonin and its metabolites are the essential components of the defense of organisms against oxidative stress and lipid peroxidation. It is well established that melatonin has scavenging actions against both ROS and RNS. 14–16 In the present study, both the usage types of melatonin (before and after ASA treatment) decreased MDA values when compared with the ASA alone group.
On the other hand, SOD, CAT, and GPX, which are enzymatic antioxidants, and GSH, which is non-enzymatic antioxidant, are the most important components of the defense mechanism of biological systems against ROS activities. 38 In our study, ASA caused significant decrease in the levels of all these antioxidants in the renal tissue. The reduction in the levels of these antioxidants has also been reported in the previous studies. 11,36,39 In the present study, melatonin caused amelioration in the levels of these antioxidants in renal tissue. However, this improvement was significant only in the levels of CAT. These ameliorating effects were found as parallel to the previous studies related to the effect of melatonin on the antioxidants. 13,17–19
Hyperemia in the glomerulus and degeneration and necrosis in renal tubulointerstitial cells, which can be observed in the early period following even in single doses of ASA administration, are usually reversible first renal histopathological changes. 6,40 In our study, ASA induced prominent glomerular and tubulointerstitial damages in the histological evaluations (Figures 2 and 3), whereas melatonin ameliorated ASA-induced histological damage in varying amounts in the treatment groups. In accordance with our biochemical assessment, the improvement was more prominent with the use of melatonin after ASA (Figure 5(a) and (b)). Therewithal, ASA led to an increase in the levels of BUN and Cr, a sign of impairment of glomerular function; however, melatonin, especially when used after ASA administration, achieved improvement in their levels (Table 2). Similarly, it has also been reported that ASA toxicity may not cause an increase in BUN and Cr levels. 6
In the previous studies, in addition to the clearly shown toxic effect of ASA on the kidney, it is well established that AA itself and metabolites have a major role to maintain the function of hypothalamic–pituitary–testicular axis. 41–43 Also, ASA was shown to inhibit luteinizing hormone (LH) response in humans. 41 In our study, in the testicular tissue, ASA caused evident histological alterations; such as sloughing, atrophy, spermatogenic arrest, disorganization of germinal cells, and multinucleated giant cell formation, which were also reported in the previous studies. 9,44 However, as clearly shown in the present study, melatonin, especially given before ASA administration, ameliorated this ASA-induced histological damage in the testicular tissue in the treatment groups.
The role of AA itself and metabolites or the inhibitors of their metabolism in testicular tissue has not clearly explored, yet. 8 Despite the limited studies have been conducted, ASA was shown to cause a dramatic reduction in CAT activity in a study related to the effect of ASA on antioxidant enzyme activities in the testes of rats. 45 In the current study, in the testicular tissue, we found that ASA induced a significant increase in MDA levels, indicating oxidative stress on lipid layers, and significantly decrease in CAT levels. Although melatonin given in order to ameliorate ASA toxicity reduced MDA levels, the reduction was not statistically significant. On the other hand, melatonin induced a significant elevation in the levels of CAT. However, in the testicular tissue, ASA did not cause any significant decrease, which was seen in the renal tissue, in the levels of other antioxidants, such as SOD, GPX, and GSH. On the contrary, ASA induced an increase, which was not significant, in the levels of these antioxidants in the testicular tissue. Besides, its cytotoxic activity occurred by oxidative stress in cells and organs, we already know that ASA protects endothelial cells from oxidative damage by nitric oxide/cyclic guanosine monophosphate pathway in atherosclerosis, 46 and prevents acetaminophen-induced liver toxicity via downregulation of proinflammatory cytokines. 47 Here, the similar paradox existed in the effect of ASA on the antioxidants in the testicular tissue, which was demonstrated, in our study. We need further investigations to understand the real mechanism/mechanisms of this status.
In summary, in the current study, we demonstrated the beneficial effect of melatonin on the high-dose ASA-induced kidney and testis injury for the first time. Moreover, the acute effect of high-dose ASA on testicular tissue was comprehensively evaluated together with the histological and biochemical results for the first time. Although the toxic effect of ASA on kidney is well known, its effect on testis is not clear yet. Melatonin ameliorated the kidney and testicular damages that occurred due to high-dose ASA administration in varying amounts.
The improvement in the histological appearance and the general biochemical determinants of oxidative stress in kidney were more obvious when melatonin used after ASA. However, such amelioration in the testis was found as more prominent when melatonin used before ASA administration. According to our results, melatonin could be used to prevent the kidney and testicular damage caused by the use of high doses of ASA, a commonly used and easily accessible pharmaceutical agent, after further clinical and experimental trials.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
